Abstract
Background
Invasion of the central nervous system by haematological malignancies is diagnosed by cytological analyses of cerebrospinal fluid or diagnostic imaging, while quantitative biomarkers for central nervous system invasion are not available and needed to be developed.
Methods
In this study, we measured the concentrations of autotaxin and soluble IL-2 receptor in cerebrospinal fluid and evaluated their usefulness as biomarkers for central nervous system invasion.
Results
We observed that both the autotaxin and soluble IL-2 receptor concentrations in cerebrospinal fluid were higher in subjects with central nervous system invasion than in those without, and the cerebrospinal fluid concentrations were independent from the serum concentrations of these biomarkers. ROC analyses revealed that the soluble IL-2 receptor concentration in cerebrospinal fluid was a strong discriminator of central nervous system invasion in subjects with haematological malignancies, while the autotaxin concentration in cerebrospinal fluid also had a strong ability to discriminate central nervous system invasion when the subjects were limited to those with lymphoma. The combined measurement of autotaxin and soluble IL-2 receptor in cerebrospinal fluid improved the sensitivity without notably reducing the specificity for central nervous system invasion in subjects with lymphoma when central nervous system invasion was diagnosed in cases where either value was beyond the respective cut-off value.
Conclusion
These results suggest the possible usefulness of soluble IL-2 receptor and autotaxin concentrations in cerebrospinal fluid for the diagnosis of central nervous system invasion.
Introduction
Invasion of the central nervous system (CNS) by haematological malignancies is diagnosed by cytological analyses of cerebrospinal fluid (CSF) or diagnostic imaging, such as magnetic resonance imaging (MRI). In some clinical situations, however, the use of laboratory testing to help physicians evaluate CNS invasion is desirable, such as when small CNS invasion is barely detectable by pathological testing or imaging or when the efficacy of chemotherapy will be examined using subsequently obtained CSF samples.
At present, soluble IL-2 receptor (sIL-2R) has been reported to be useful for the diagnosis of CNS invasion by acute lymphoblastic leukaemia, 1 adult T-cell leukaemia 2 and acute lymphocytic leukaemia and non-Hodgkin lymphoma.3–5 However, sIL-2R is known to be elevated in several diseases in which lymphocytes are activated, suggesting that sIL-2R could be higher in immunological diseases. Actually, even in CSF samples, sIL-2R has been reported to be elevated in other diseases such as cerebral lupus, 6 multiple sclerosis 7 and neurosarcoidosis. 8 Therefore, the development of novel biomarkers for clinical laboratory testing for the diagnosis of CNS invasion by haematological malignancies is needed.
Autotaxin (ATX) is an enzyme that produces lysophosphatidic acid (LPA) mainly from lysophosphatidylcholine via its lysoPLD activity. 9 Recently, ATX was introduced in Japan as a clinical laboratory test for evaluating the extent of hepatic fibrosis. 10 Since ATX was first identified in conditioned medium from a melanoma cell line, 11 the involvement of ATX in the pathogenesis of cancer has been investigated, and ATX has actually been shown to be associated with several human cancers.12–15 ATX reportedly stimulates proliferation, migration, invasion and tumour angiogenesis through the production of LPA,16–19 suggesting that ATX might be involved in the invasion of the CNS by cancer. Moreover, serum ATX concentrations are higher in subjects with B-cell lymphoma, especially follicular lymphoma,20,21 although the mechanism by which ATX concentrations increase in lymphoma remains to be elucidated.
With this background in mind, we measured both the ATX and sIL-2R concentrations in CSF samples using flow cytometry analyses and investigated whether the ATX and sIL-2R concentrations might be useful for the diagnosis of CNS invasion by haematological malignancies.
Materials and methods
Subjects
We enrolled 49 patients with haematological malignancies whose CSF samples were subsequently tested using flow cytometry analyses between December 2016 and July 2018 at the University of Tokyo Hospital (Tokyo, Japan). The clinical characteristics of the patients are described in Table 1. The clinical parameters were not statistically different between the subjects with CNS invasion and those without CNS invasion. The CSF and serum samples used in this study were residual samples from after the completion of the requested clinical laboratory tests. The presence of CNS invasion was determined by cytological analyses of CSF samples or diagnostic imaging. This study was approved by the Institutional Research Ethics Committee of the Faculty of Medicine, The University of Tokyo (2602 and 3333–141).
Characteristics of the subjects.
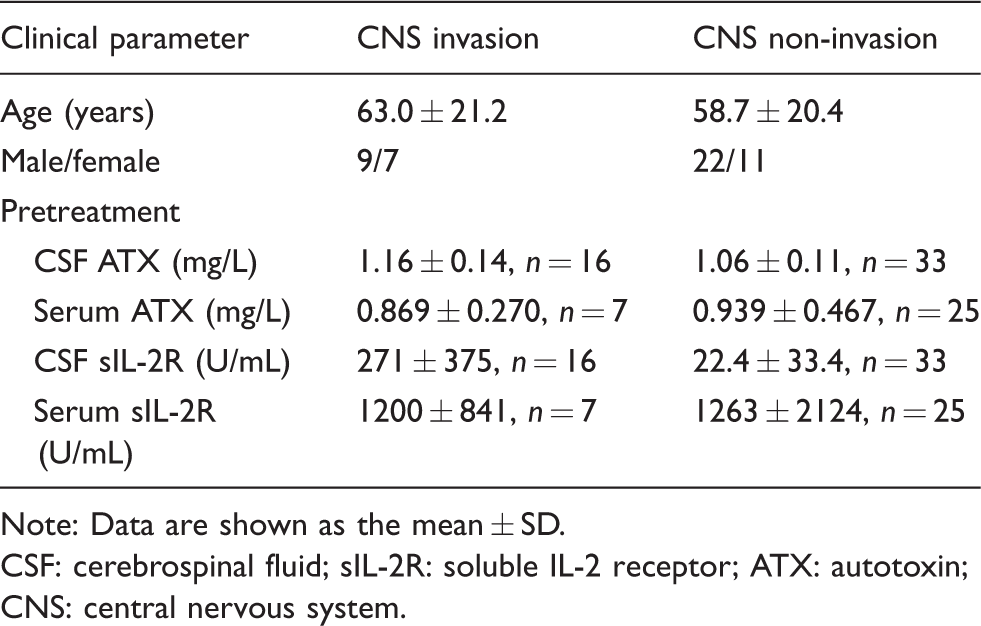
Note: Data are shown as the mean ± SD.
CSF: cerebrospinal fluid; sIL-2R: soluble IL-2 receptor; ATX: autotoxin; CNS: central nervous system.
Measurement of ATX
The ATX antigen concentrations in CSF and serum samples were determined using a two-site immunoenzymetric assay with the TOSOH AIA system (TOSOH, Tokyo, Japan). 22
Measurement of sIL-2R
The sIL-2R antigen concentrations in CSF and serum samples were determined using a chemiluminescent enzyme immunoassay (STACIA CLEIA IL-2R) with the STACIA system (LSI Medience, Tokyo, Japan).
Statistical analysis
All the data were statistically analysed using SPSS (Chicago, IL, USA). Data were presented as the 95% confidence interval (CI) for the median. We used the Mann-Whitney U test for comparisons between two groups and the Spearman correlation test for correlations, since normality or equality of variance was rejected by the Kolmogorov-Smirnov test or the Levene test for most of the parameters. A value of P < 0.05 was regarded as denoting statistical significance in all the analyses.
Results
ATX and sIL-2R concentrations in CSF samples were higher in subjects with CNS invasion
In the previous reports, serum ATX concentrations were higher in female than in male,15,22 while those in CSF samples were not influenced by gender. 23 In the present cohort, we observed no difference in the ATX or sIL-2R concentrations in serum samples or CSF (Supplementary Figures 1 and 2). Therefore, we analysed the data of male and female together in the present study. First, we compared the ATX and sIL-2R concentrations in CSF samples and found that both the ATX and sIL-2R concentrations were higher in the subjects with CNS invasion (Figure 1(a) and (b)), while the serum concentrations were not different (Figure 1(e) and (f)). When only the subjects with lymphoma were analysed and since the serum ATX concentrations were increased in the subjects with lymphoma, similar results were observed (Figure 1(c), (d), (g) and (h)).
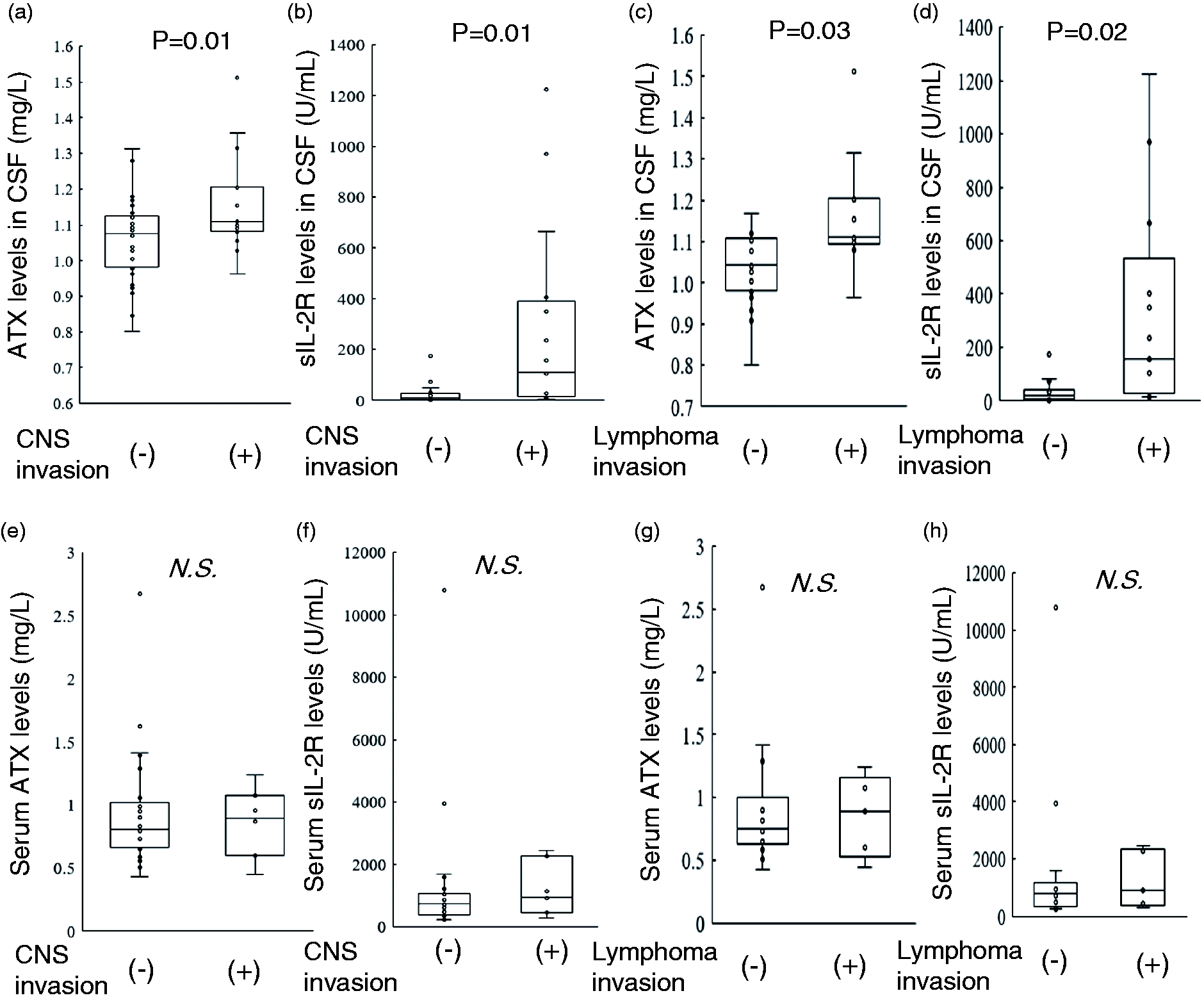
ATX and sIL-2R concentrations in CSF from subjects with or without CNS invasion caused by haematological malignancies. The ATX and sIL-2R concentrations in both CSF and serum were measured in subjects with or without CNS invasion caused by haematological malignancies (a, b, e, f) or lymphoma (c, d, g, h). (a, c) ATX concentrations in CSF (a: n = 49, c: n = 33), (b, d) sIL-2R concentrations in CSF (b: n = 49, d: n = 33), (e, g) serum ATX concentrations (E: n = 32, G: n = 19), (f, h) serum sIL-2R concentrations (f: n = 32, h: n = 19). Data are shown as the 95% CI for the median.
Next, we performed ROC analyses to investigate the ability of the ATX and sIL-2R concentrations in CSF samples to diagnose CNS invasion. As a result, a larger area under the curve (AUC) was obtained for sIL-2R than for ATX (Figure 2(a) and (b)). When the subjects were limited to those with lymphoma, however, the difference in AUCs became smaller (Figure 2(c) and (d)).
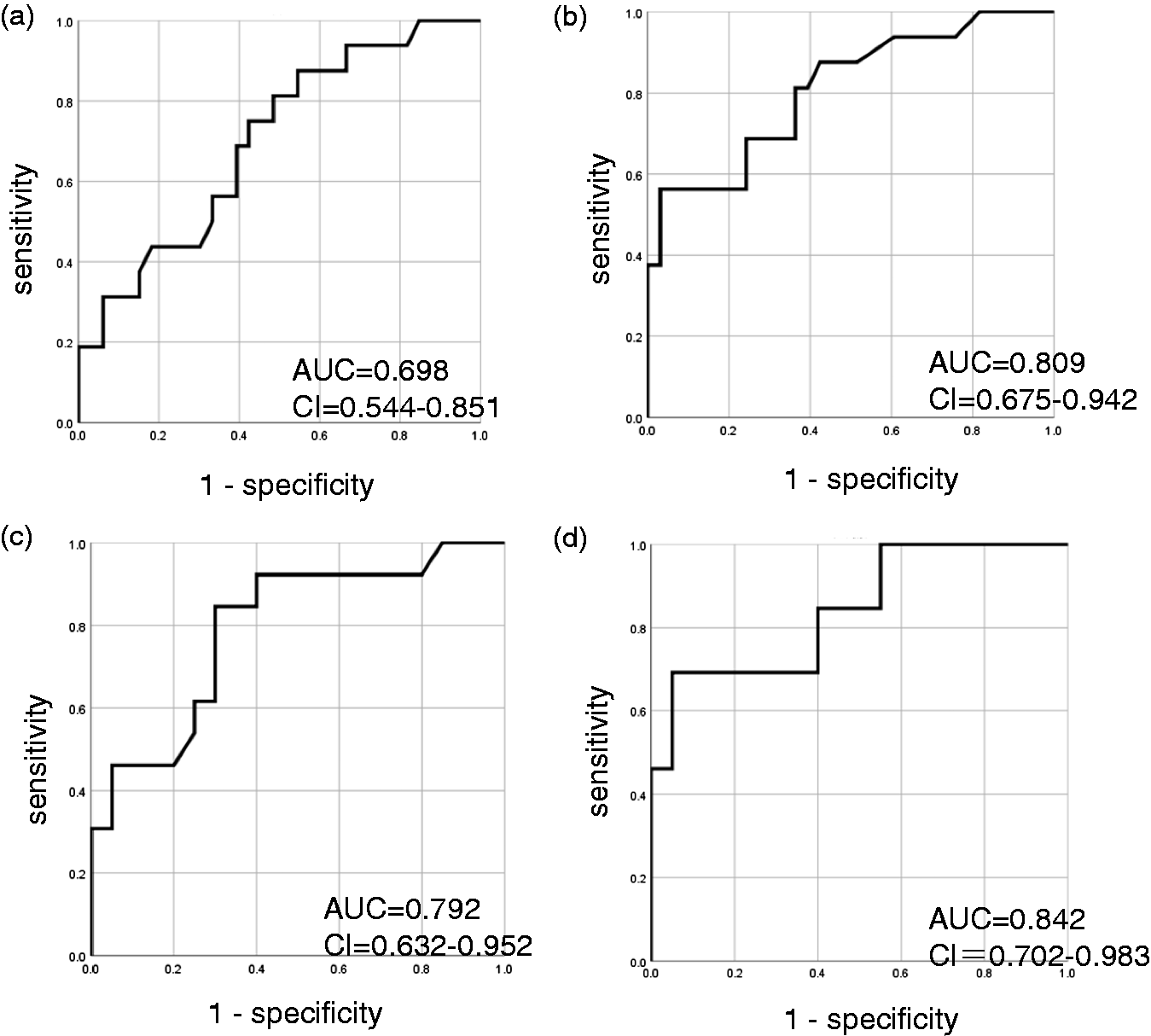
ROC analyses examining the ability of ATX and sIL-2R in CSF to diagnose CNS invasion. ROC analyses were performed to investigate the ability of ATX (a, c) and sIL-2R (b, d) in CSF to discriminate CNS invasion by haematological malignancies (a, b) or lymphoma (c, d).
ATX and sIL-2R concentrations in CSF samples were not significantly correlated with those in serum samples
Next, we investigated the correlations between the serum ATX or sIL-2R concentrations and those in CSF samples to investigate whether the concentrations in CSF samples are independent from those in serum samples. As shown in Figure 3, no significant correlations were observed between the concentrations in CSF and those in serum samples, suggesting that the ATX and sIL-2R concentrations in CSF samples might not be influenced by serum concentrations.
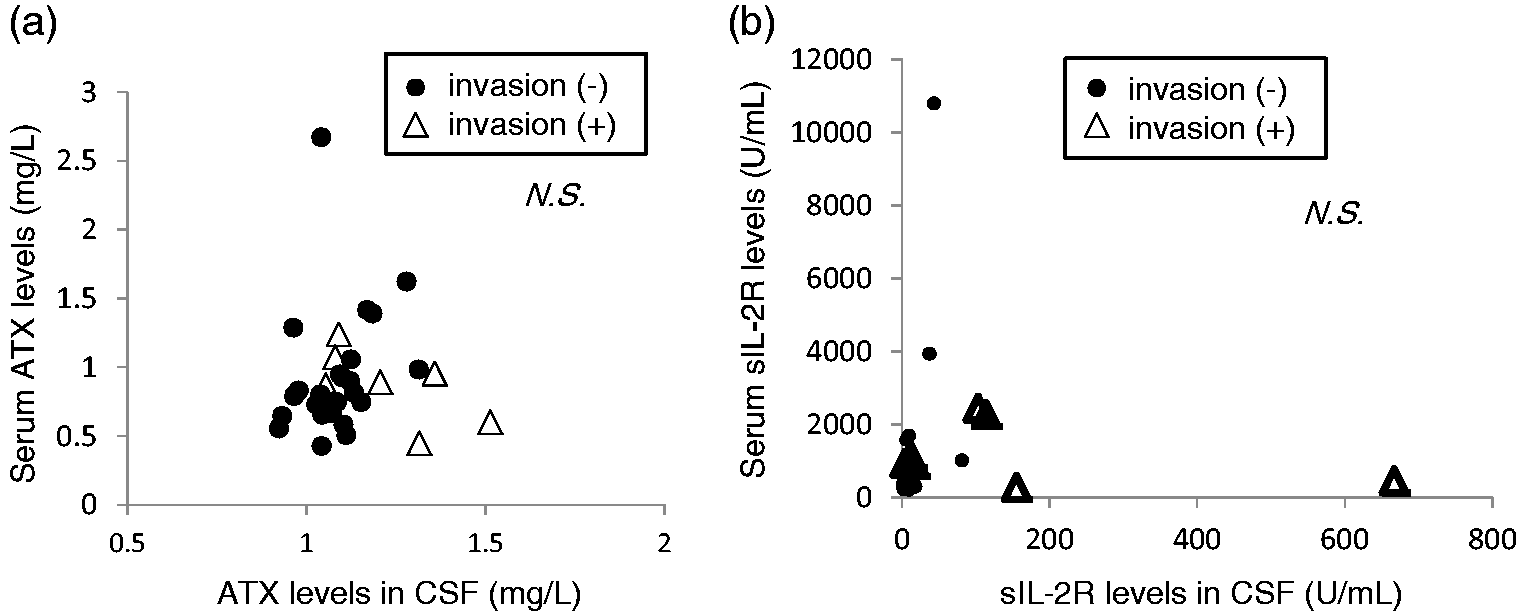
Correlations between ATX or sIL-2R concentrations in CSF and those in serum. The correlations between the ATX (a) or sIL-2R (b) concentrations in CSF and those in serum are shown (n = 32).
Combination of ATX and sIL-2R concentrations in CSF was useful for the diagnosis of CNS invasion
So far, the present findings suggest that the ATX concentration in CSF might possess a similar or lesser capacity to diagnose CNS invasion by haematological malignancies, compared with the sIL-2R concentration in CSF. To investigate the clinical usefulness of ATX, we investigated whether the combination of ATX and sIL-2R concentrations measured in CSF might improve the diagnostic ability of sIL-2R measurements alone. As shown in Figure 4(a) and Table 2A, when the cut-off values for CNS invasion were set as 1.150 for ATX and 100 U/mL for sIL-2R, we observed that the sensitivity improved from 0.56 (when only sIL-2R concentrations were measured) to 0.81 (when sIL-2R and ATX concentrations were measured), while the specificity decreased from 0.97 to 0.79 when CNS invasion was diagnosed in cases where either of these biomarkers was beyond the respective cut-off value. When the above analysis was restricted to subjects with lymphoma, the sensitivity improved from 0.69 to 0.92, while the specificity remained almost unchanged (0.95 to 0.90) when the combination of biomarkers was used (Figure 4(b) and Table 2B).
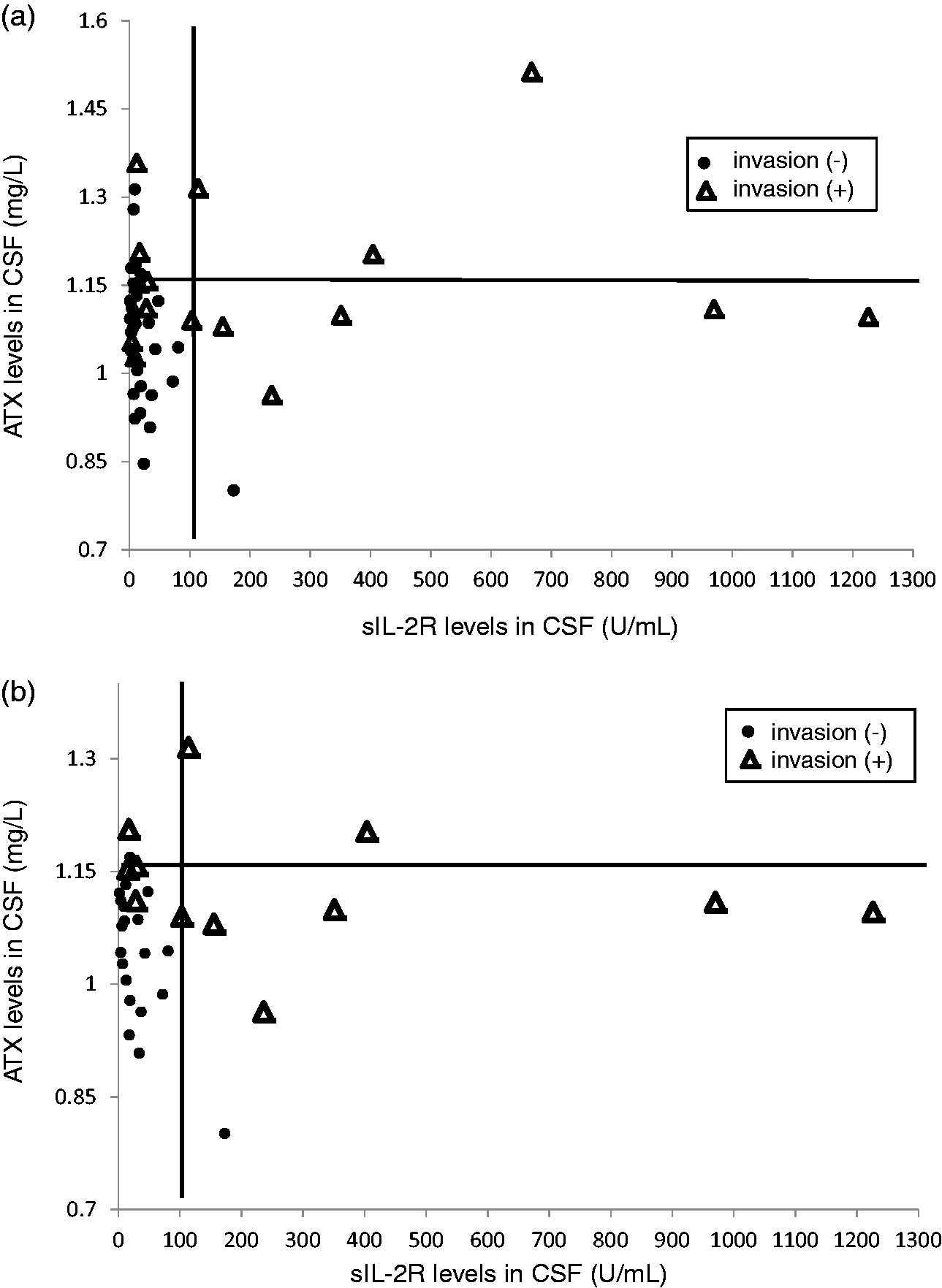
Distribution of subjects according to ATX and sIL-2R concentrations in CSF. The distributions of subjects with haematological malignancies (a, n = 49) or lymphoma (b, n = 33) according to their ATX and sIL-2R concentrations in CSF are shown.
Sensitivity and specificity of sIL-2R, ATX and the combination of sIL-2R and ATX for the diagnosis of CNS invasion.
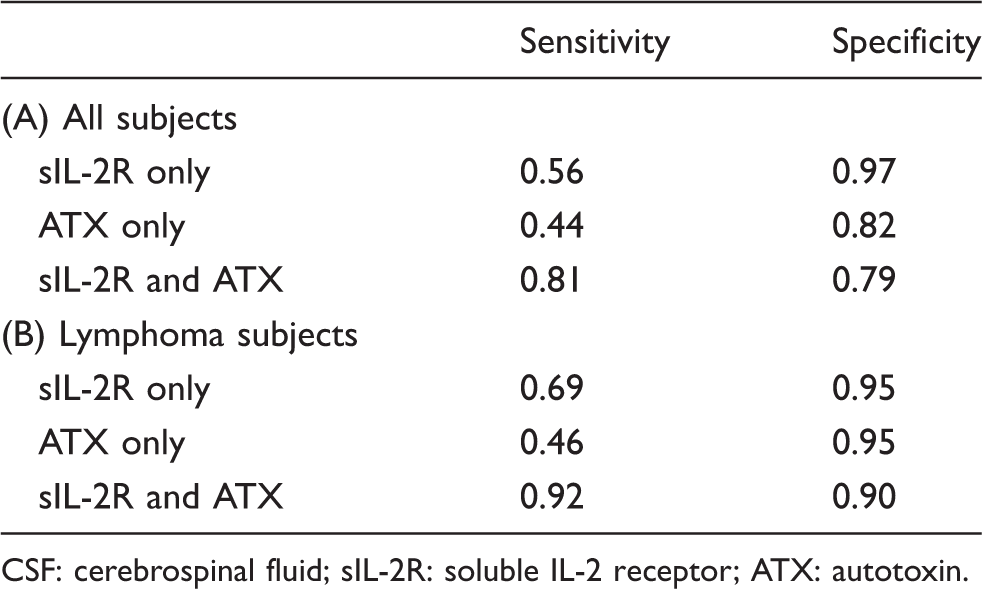
CSF: cerebrospinal fluid; sIL-2R: soluble IL-2 receptor; ATX: autotoxin.
Discussion
In this study, we aimed to evaluate the ability of sIL-2R and ATX concentrations in CSF to diagnose CNS invasion by haematological malignancies and revealed that the concentrations of these biomarkers in CSF were higher in subjects with CNS invasion; furthermore, the combination of sIL-2R and ATX measurements might provide a high sensitivity and specificity, especially in subjects with lymphoma.
Regarding sIL-2R, several studies have demonstrated the usefulness of its concentration in CSF for the diagnosis of CNS invasion1–5; these findings were replicated in the present study (Figures 1(b), (d) and (2b), (d). Importantly, as shown in Figure 3(b), the sIL-2R concentrations in CSF were not significantly correlated with those in serum samples, suggesting that the sIL-2R in CSF might not originate from the circulation and might instead be secreted from malignant cells that have invaded the CNS. However, as shown in Figure 1(b) and (d), some subjects without CNS invasion also had high sIL-2R concentrations in their CSF samples. The reason for this non-specific elevation in CSF remains unknown; however, since sIL-2R reflects the activation of lymphocytes, its concentration can be elevated by factors other than haematological malignancies, such as inflammation. Therefore, another biomarker might be necessary for the diagnosis of CSN invasion.
For this purpose, we hypothesized that the ATX concentration in CSF might be useful for the diagnosis of CNS invasion, since we reported that the serum ATX concentrations were elevated in subjects with lymphoma, 20 and many elegant basic studies have demonstrated that the ATX/LPA pathway is involved in metastasis.16–19 Actually, the ATX concentrations in CSF were higher in subjects with CNS invasion (Figure 1(a) and (c)), and the ATX concentrations in CSF were independent from those in serum samples (Figure 3(a)); however, the diagnostic ability of sIL-2R might be higher than that of ATX (Figure 2). The reason for this difference might be that the sIL-2R concentrations in CSF were much lower than those in serum, while the ATX concentrations in CSF were similar or slightly higher than those in serum 23 (Table 1), since ATX is expressed in glial cells and secretory epithelial cells. 24 Regarding the reason why the ATX concentrations in CSF were elevated in subjects with CNS invasion, ATX might be secreted by cancer cells in the CSF and/or neural cells might secrete ATX in response to malignant invasion. Since the AUC of the ROC became larger when we limited the subjects to those with lymphoma, the former possibility might be the main reason.
Lastly, we investigated the clinical usefulness of ATX measurements in CSF samples. As shown in Figure 4(a) and Table 2A, in all the subjects, the combined measurement of ATX and sIL-2R increased the sensitivity but decreased the specificity; however, when the subjects were limited to those with lymphoma, the combined measurement of ATX and sIL-2R yielded a notably improved sensitivity and only a slight decrease in specificity. Although the number of subjects in this study was not very large, these results suggest that the combination of sIL-2R and ATX in CSF might be useful for detecting CNS invasion, for which no quantitative markers are presently available in clinical practice. Since the biomarker characteristics of sIL-2R and ATX in CSF differ, as described above, they might be able to complement each other; the sIL-2R concentrations in healthy subjects are much lower in CSF than in serum samples, suggesting that sIL-2R in CSF might possess a relatively high sensitivity, whereas it might be easily affected by sIL-2R in blood if the blood–brain barrier has been disrupted. Conversely, the ATX concentrations in CSF are relatively high, suggesting that ATX might have a relatively low sensitivity, but the concentration might be minimally influenced by ATX in blood leaking into the CSF through the blood–brain barrier. Regarding the diseases other than CNS invasion, in which the ATX concentrations in CSF are reportedly high, ATX concentrations in CSF have been reported to be higher in Alzheimer’s disease 25 and multiple sclerosis. 26 Although ATX concentrations in CSF are high in multiple sclerosis as well as sIL-2R, we believe the mechanism for the elevation might be different; sIL-2R might be increased due to immunological activation or disruption of blood–brain barrier, while ATX might be increased as a result of the injuries to neural cells. This speculation was concordant with the idea that the combination of sIL-2R and ATX might be useful for the diagnosis of CNS invasion. Further prospective studies involving a larger number of subjects are needed to demonstrate the usefulness of these markers.
The limitation of the present study is that we compared ATX only with sIL-2R, as well as that we included a rather small number of the subjects. According to a systemic review recently published on diagnostic biomarkers in CSF for CNS lymphoma, 18 markers including sIL-2R has been reported as possible diagnostic biomarkers for CNS lymphoma. 27 Comparing the diagnostic values of ATX and sIL2-R (Table 2) with those described in the article, the sensitivity and specificity of the combination of ATX and sIL2-R are almost similar to those of biomarkers with rather high diagnostic abilities such as miR-21, miR-19b, miR-92a, IL-10, Osteopontin, Neopterin, B2M and sCD27, although either ATX or sIL2-R alone has lower diagnostic values. Another limitation is that we could not collect serum samples from all the subjects, which might result in a potential bias. When we analysed the subjects from whom both serum samples and CSF samples were collected and those from whom only CSF samples were collected separately, we observed that both ATX and sIL-2R concentrations in CSF samples were higher in the subjects with CNS invasion or lymphoma invasion in the subjects from whom both serum samples and CSF samples were collected (Supplementary Figure 3), while only ATX concentrations in CSF samples were higher in the subjects from whom only CSF samples were collected (Supplementary Figure 4), although the number of the latter group was very small.
In summary, sIL-2R in CSF is a useful biomarker for the diagnosis of CNS invasion by haematological malignancies, and ATX in CSF can complement the capacity of sIL-2R to diagnose CNS invasion, especially in subjects with lymphoma.
Supplemental Material
Supplemental material for Autotaxin and soluble IL-2 receptor concentrations in cerebrospinal fluids are useful for the diagnosis of central nervous system invasion caused by haematological malignancies
Supplemental Material for Autotaxin and soluble IL-2 receptor concentrations in cerebrospinal fluids are useful for the diagnosis of central nervous system invasion caused by haematological malignancies by Takuya Shimura, Makoto Kurano, Yoshifumi Morita, Naoyuki Yoshikawa, Masako Nishikawa, Koji Igarashi, Satoshi Shimamoto, Junken Aoki and Yutaka Yatomi in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K. Igarashi and S. Shimamoto are employees of TOSOH Corporation. This study was conducted based on a cooperative research agreement between The University of Tokyo Hospital and TOSOH Corporation and between The University of Tokyo Hospital and LSI Medience.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CREST from JST/AMED, Leading Advanced Projects for Medical Innovation (LEAP) from AMED, practical research for innovative cancer control from AMED, a Grant-in-Aid for Scientific Research on Innovative Areas 15H05906 (YY) and JSPS KAKENHI Grant Number 16H06236 (M.K.).
Ethical approval
This study was approved by the Institutional Research Ethics Committee of the Faculty of Medicine, The University of Tokyo (2602 and 3333–141).
Guarantor
MK.
Contributorship
T. Shimura and M. Kurano designed research; T. Shimura, N. Yoshikawa and M. Nishikawa analysed data; T. Shimura, Y. Morita, S. Shimamoto and K. Igarashi performed research; T. Shimura, M. Kurano, K. Igarashi and Y. Yatomi wrote the paper. All authors read and approved the final version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
