Abstract
Background
The spectrophotometric examination of cerebrospinal fluid for bilirubin is an established investigation in patients with suspected subarachnoid haemorrhage. This study assesses the diagnostic specificity of an elevated cerebrospinal fluid bilirubin and how this may be influenced by the presence of oxyhaemoglobin and the concentration of cerebrospinal fluid total protein.
Methods
One thousand cerebrospinal fluid spectroscopy reports were reviewed. Electronic patient records were examined to determine the clinical outcome in patients with an elevated cerebrospinal fluid bilirubin.
Results
Forty-four out of 1000 cerebrospinal fluid scans showed an increase in cerebrospinal fluid bilirubin unrelated to elevated serum bilirubin concentrations. This was associated with subarachnoid haemorrhage in 16 (36%) cases. Subarachnoid haemorrhage was confirmed in 5/17 (29%) patients positive for cerebrospinal fluid bilirubin alone and in 11/27 (41%) patients positive for both cerebrospinal fluid bilirubin and oxyhaemoglobin. At cerebrospinal fluid total protein concentrations <1 g/L, the specificity for subarachnoid haemorrhage improved: 4/9 (44%) vs. 1/8 (13%) with an increase in cerebrospinal fluid bilirubin alone; 6/10 (60%) vs. 5/17 (29%) in patients with increases in both bilirubin and oxyhaemoglobin.
Conclusion
While an increase in cerebrospinal fluid bilirubin may be considered consistent with subarachnoid haemorrhage, the likelihood that a subarachnoid haemorrhage has occurred is influenced by the presence of oxyhaemoglobin and the concentration of total protein in the cerebrospinal fluid.
Keywords
Introduction
The spectrophotometric examination of cerebrospinal fluid (CSF) for bilirubin is performed in the investigation of patients with a history suggestive of subarachnoid haemorrhage (SAH) but without demonstrable blood on a computed tomography (CT) scan. While expert guidelines have been published for performing and reporting CSF examinations, 1 there is often uncertainty about the diagnostic specificity of the results considered to be consistent with SAH. For example, a false-positive increase in CSF bilirubin may also accompany another source of CSF blood, an increase in CSF total protein or an increase in serum bilirubin.
The present study attempts to address this issue by reviewing the clinical outcomes of patients with an increase in CSF bilirubin as defined in the expert guidelines.
Methods
One thousand consecutive CSF spectroscopy reports from November 2008 to September 2013 were examined and where the CSF bilirubin was elevated, electronic patient records scrutinized to determine clinical outcome.
The CSF scans were performed using a UVIKON XS double beam UV/Vis spectrophotometer with UVIKON Bio-C fully automated interpretive software which follows the decision-tree of the guidelines. 1 All CSF scans and interpretive comments were reviewed prior to reporting.
Results and discussion
Fifty-two out of 1000 CSF scans were positive for bilirubin and reported as recommended in national guidelines. The pattern of results and associated clinical findings are indicated in Figure 1. In 8/52 cases, the increased CSF bilirubin was attributable to an increase in serum bilirubin and adjusted accordingly. Of the remaining 44 cases, SAH was confirmed by CT angiography in 16 (36%) of which 10/16 and 5/16 patients, respectively, had negative or equivocal CT scans. One patient did not have a CT scan prior to spectrophotometry. Previous studies have reported true positive rates of 43% and 21%.2,3 In the present study, non-specific increases in CSF bilirubin were associated with an increase in CSF total protein in patients with traumatic taps, central nervous system (CNS) infections and CNS malignancies (Figure 1). In these patients, SAH was excluded following CT angiography (all patients with traumatic tap and one patient with CSF infection) or on clinical grounds (remaining patients with CSF infection or malignancy). In the six patients where no alternative explanation for the increase in CSF bilirubin was identified, SAH was excluded by CT angiography.
Categorization of CSF samples with increased bilirubin according to the National Guidelines. Fifty-two CSF samples had an increased CSF bilirubin (> 0.007 AU). NBA: net bilirubin absorbance, NOA: net oxyhaemoglobin absorbance, TP: total protein, OxyHb: oxyhaemoglobin, FP: false positive, TT: traumatic tap, CM: CNS malignancy. CI: CNS infection, UK: unknown.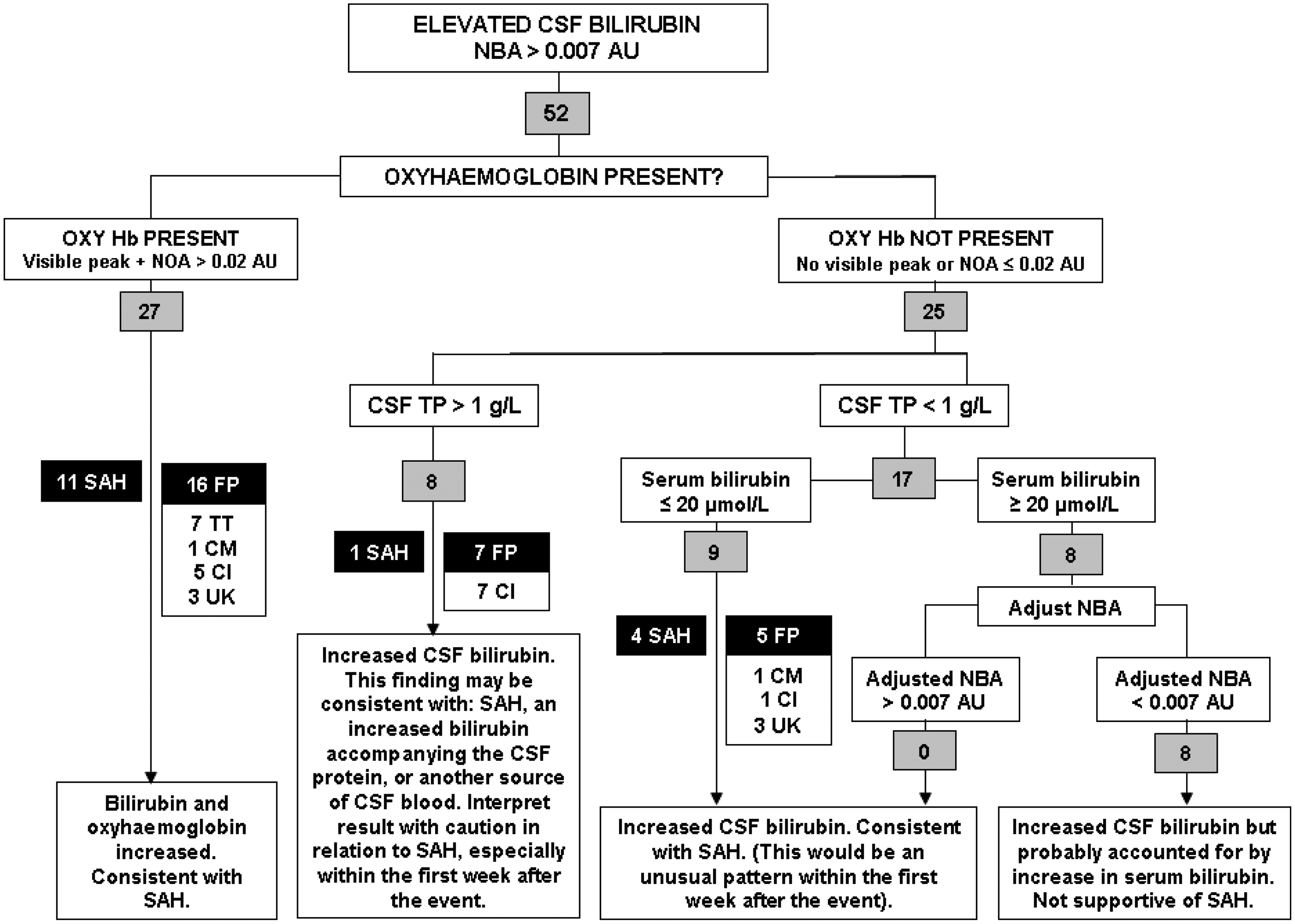
Patients with SAH typically present within one week of the event and as such are more likely to show an increase in CSF oxyhaemoglobin in addition to the presence of bilirubin than those who present later. In the present study, 6/7 patients presenting within seven days showed an increase in both oxyhaemoglobin and bilirubin. However, 3/6 patients presenting later also showed an increase in both oxyhaemoglobin and bilirubin. The time of presentation was not established in the further three patients with confirmed SAH.
CSF findings for 44 CSF reports with raised CSF bilirubin.
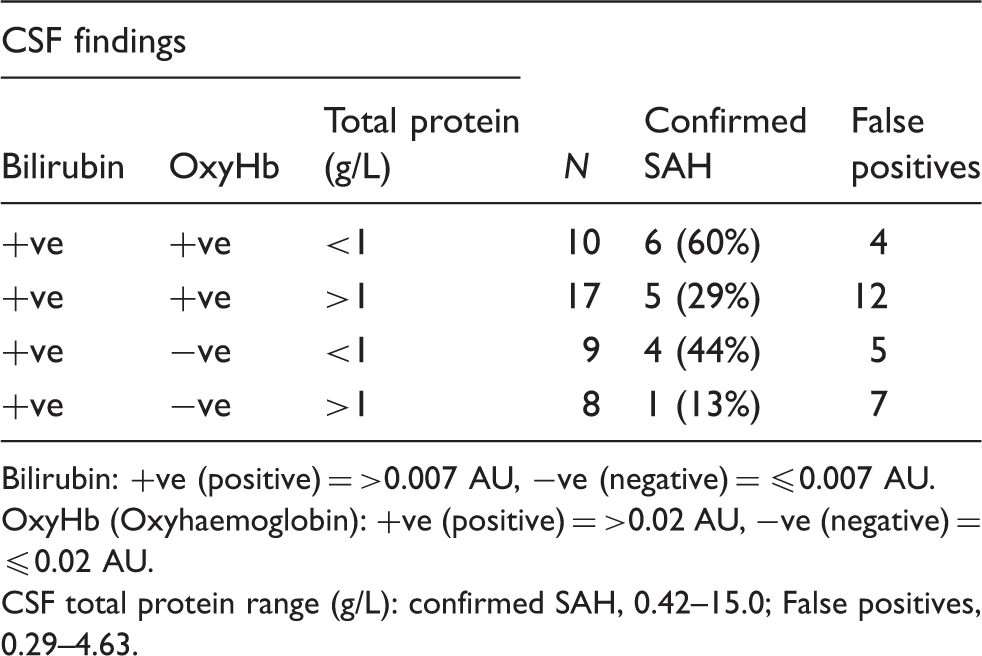
Bilirubin: +ve (positive) = >0.007 AU, −ve (negative) = ≤0.007 AU.
OxyHb (Oxyhaemoglobin): +ve (positive) = >0.02 AU, −ve (negative) = ≤0.02 AU.
CSF total protein range (g/L): confirmed SAH, 0.42–15.0; False positives, 0.29–4.63.
The laboratory may be asked to comment on the likelihood that a patient with an elevated CSF bilirubin has experienced an SAH. Whilst the numbers are small, and must therefore be interpreted with caution, the pattern of data summarized in Table 1 suggests that consideration of the CSF total protein concentration in addition to the presence/absence of oxyhaemoglobin may be helpful in answering this question. The likelihood of SAH tends to be increased if oxyhaemoglobin is present. The likelihood of SAH decreases when the concentration of CSF total protein is >1 g/L, regardless of whether oxyhaemoglobin is present. The national guidelines recognize this by recommending a cautionary comment when the CSF total protein is >1 g/L in the absence of oxyhaemoglobin. However, they do not specifically consider the CSF total protein when both bilirubin and oxyhaemoglobin are present but recommend reporting all such results as ‘consistent with SAH’. While this approach is undoubtedly safe and appropriate, consideration of the CSF protein concentration may be helpful when discussing the likelihood of SAH in individual patients, particularly if there is a more likely alternative explanation for the raised CSF total protein.
The expert guidelines for the examination of CSF advise that ‘the final interpretation should take into account all available clinical information’. The present data endorse the view that specificity will improve if the results are interpreted by laboratory staff who are aware of the clinical circumstances of each case, including the CSF total protein concentration.
Footnotes
Acknowledgements
NA.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
SAW.
Contributorship
HLF reviewed the CSF spectroscopy data. HLF and SAW reviewed the electronic patient records. SAW and JPA conceived the study. HLF drafted the manuscript and all authors were involved in reviewing and editing the manuscript.
