Abstract
Background
The differential diagnosis between postneurosurgical bacterial meningitis and aseptic meningitis remains challenging both for the clinician and the laboratory. Combinations of markers, as opposed to single ones, may improve diagnosis and thereby survival.
Methods
This prospective cohort study included patients with suspected bacterial meningitis after neurosurgery. The patients were divided into two groups according to the diagnostic criteria of meningitis involving a postneurosurgical bacterial meningitis group and a postneurosurgical aseptic meningitis group. Four biomarkers, including cerebrospinal fluid procalcitonin, lactate, interleukin-8 and interleukin-10 were assayed separately, and three algorithms were constructed using a linear combination. The area under the receiver operating characteristic curve was used to compare their performances.
Results
A cohort of 112 patients was enrolled in our study. Forty-three patients were diagnosed with postneurosurgical bacterial meningitis, and the cerebrospinal fluid values of their biomarkers were higher in patients with postneurosurgical bacterial meningitis than with postneurosurgical aseptic meningitis. The area under the receiver operating characteristic curves for the detection of postneurosurgical bacterial meningitis were 0.803 (95% confidence interval [CI], 0.724–0.883) for procalcitonin; 0.936 (95% CI, 0.895–0.977) for lactate; 0.771 (95% CI, 0.683–0.860) for interleukin-8; 0.860 (95% CI, 0.797–0.929) for interleukin-10; 0.937 (95% CI, 0.897–0.977) for the composite two-marker test; 0.945 (95% CI, 0.908–0.982) for the composite three-marker test and 0.954 (95% CI, 0.922–0.989) for the composite of all tests. The area under the receiver operating characteristic curves of the combination tests were greater than those of the single markers.
Conclusions
Combining information from several markers improved the diagnostic accuracy in detecting postneurosurgical bacterial meningitis.
Keywords
Introduction
Neurosurgery may run the risk of developing nosocomial meningitis caused by infection with hospital pathogens, which usually is termed as postneurosurgical bacterial meningitis (PNBM), with an incidence from 1% to 10%.1,2 PNBM is the main reason for an increased hospital stay and an increased overall cost of hospital care. Although the incidence of bacterial meningitis has declined over the last 10 years because of the reasonable use of antibiotics, the mortality and neurological outcomes of bacterial meningitis have not improved. 3 An accurate and rapid diagnosis is therefore needed for better prognoses of PNBM patients.
In neurosurgery, postneurosurgical aseptic meningitis (PNAM) is another common disease that was triggered by an aseptic inflammatory response to haemolysis products, sloughing tissue, tumour antigens and bone dust, 4 which are usually produced during neurosurgery. PNBM and PNAM share some clinical symptoms and physical signs, including headache, fever, neck stiffness, changes in mental status and vomiting, which make the clinical manifestations of these diseases not sufficiently specific to diagnose PNBM. The values of cerebrospinal fluid (CSF) biochemical parameters, such as erythrocytes, leukocytes, protein and glucose are limited because of low sensitivity and specificity,5,6 especially in distinguishing PNBM from PNAM. The CSF bacterial culture remains the gold standard in PNBM diagnosis. However, the per cent of positive results is only 6–8% because of numerous reasons, including the low amount and contamination of the CSF sample, time constraints and antibiotic drug administration.7,8 Another consideration involves the limitations of polymerase chain reaction (PCR) testing, including a high false-positive percentage because of sample contamination during the lumbar puncture, plus interactions between the gene probes of other unrelated bacteria, resulting in low specificity. 9 Moreover, the high cost and complexity of the procedure also limit the wide clinical application of PCR testing. 10 Accordingly, clinicians always use the patient’s clinical and laboratory-related biochemical parameters to diagnose a disease which tends to be more subjective and less sensitive. Many factors can affect the diagnostic accuracy for PNBM, such as interobserver variation, poor repeatability and specificity and problematic standardization. Given all these influencing factors, novel diagnostic markers are needed to provide more diagnostic options and evaluate the efficacy of antibiotic drugs.
The preliminary results of our study showed that using a serial test and a parallel test combining CSF procalcitonin (PCT) and lactate could reliably distinguish between PNBM and non-PNBM and could be included in the design of a novel diagnostic approach to circumvent the shortcomings of conventional methods. 11 Interleukin-8 is a proinflammatory cytokine produced during the acute phase response to stimuli, such as infection, surgery or trauma. The sensitivity and specificity of the CSF interleukin-8 concentration have been reported as a diagnostic marker for bacterial meningitis.12–14 Furthermore, excessive inflammation in the central nervous system (CNS) can cause damage and seriously impair its function, and hence the association between CSF interleukin-8 concentrations and neurological outcomes has been previously studied.15,16 Serum interleukin-10 may serve as a potential early diagnostic marker of infection and could improve the sensitivity and specificity of the early diagnosis of sepsis, and a significant increase of interleukin-10 gene expression has already been reported in patients with meningitis. 17
CSF PCT, lactate, interleukin-8 and interleukin-10 have been separately shown to correlate with bacterial infection. Whether the combination of CSF PCT, lactate, interleukin-8 and interleukin-10 improves the diagnostic efficacy is still unknown. In this report, we therefore determined the discriminative power of combining multiple markers to diagnose PNBM.
Materials and methods
Participants
This prospective study was conducted from August 2015 to March 2016 at the Beijing Tiantan Hospital. Written informed consent was obtained from all the patients with age ≥18 years. All patients who showed clinical symptoms of bacterial meningitis, such as fever, headache, neck stiffness and a disturbance of consciousness within 48–72 h after surgery, were recruited to participate in this study. The investigator and the physician recruited patients and performed lumbar puncture at a fixed time, and the surgery was performed by a single skilled surgeon to minimize the differences between sampling times and specimen qualities. Moreover, none of the patients was treated with antibiotics. The patients were subjected to lumbar puncture to collect CSF samples for diagnostic analyses, which included protein content, glucose, chloride and white blood cell counts (WBCs). The residual CSF samples were used to assay levels of PCT, lactate, interleukin-8 and interleukin-10.
Reference standard
The diagnosis of PNBM was based on the following criteria 5 : (1) clinical symptoms, including headache, fever (temperature ≥38.5°C), meningeal irritation signs and a disturbance of consciousness and (2) CSF bacterial cultures were positive or routine CSF examinations were positive, including: (a) a CSF WBC count ≥ 1000/μL and a polykaryocyte percentage ≥75% and (b) CSF glucose below 2.5 mmol/L or a ratio of CSF glucose to blood glucose < 0.4.
The diagnosis of PNAM: Patients who had the foregoing clinical symptoms, but routine CSF tests that did not meet the aforementioned criteria with CSF WBC count ≤ 500/μL were classified into the PNAM group.
Exclusion criteria: (1) those who have serious complications, multiple organ failure, deep coma, the state of survival of plants; (2)those who have intracranial infection or other parts of infection in admission; (3) there is a clear lumbar puncture taboo; (4) patients have used antibiotics for other reasons; (5) non-intracranial space-occupying lesions (such as ventriculo-abdominal shunt); (6) pregnancy or lactation women; (7) those who are known to have poor compliance.
Test methods
CSF analyses, such as cytology (leukocyte counts and polykaryocyte percentages), bacteriology, PCT, lactate, interleukin-8, interleukin-10 concentrations and related CSF biochemical tests (protein, glucose and chloride), were performed before the start of any antibiotic treatment. The CSF protein, chloride and glucose concentrations were quantitated using a VITROS 5.1 FS automatic chemistry analyzer (Diamond Diagnostics, Citrus Heights, CA, USA) using the velocity method. The other biomarkers, including polykaryocyte percentages and WBC counts were measured on a Sysmex 4000i cell analyzer (Sysmex, Kobe, Japan). The PCT concentration was measured with a commercially available enzyme-linked fluorescent assay (VIDAS® BRAHMS; bioMérieux, Marcy-l’Etoile, France), which had a lower detection limit of 0.05 μg/L. When the CSF PCT concentration of the patients was below this detection limit, it was regarded as 0 μg/L. The lactate concentration in the CSF was measured using VITROS lactate slides (Ortho-Clinical Diagnostics, NY, USA), which had a lower detection limit of 0.50 mmol/L. The interleukin-8 and interleukin-10 concentrations were measured using a commercially available chemiluminescence kit (IMMULITE 1000; Siemens, Berlin, Germany), which have lower detection limits of < 2 ng/L and < 1 ng/L, respectively. We validated the use of these assays using serial dilutions of CSF.
Before the study, we chose cut-off values of 0.075 μg/L and 3.45 mmol/L for PCT and lactate, respectively, which were based on our retrospective study. 11 The best cut-off values of interleukin-8, interleukin-10, two-marker, three-marker and four-marker assays were determined according to the Youden index, because of a lack of reference studies on PNBM.
Statistical analysis
Because all PCT, lactate, interleukin-8 and interleukin-10 data followed an abnormal distribution (Kolmogorov-Smirnov, P < 0.05), a non-parametric test (Mann-Whitney U test) was used to compare the median levels to ascertain whether there was an association between the PNBM and PNAM groups. The sensitivities and specificities were calculated for all single and composite markers. The results of two single best performing markers, three single best performing markers and all four markers were combined using the method reported by Xiong et al. 18 This method was based on the principle of identifying the linear combination of markers that resulted in the greatest area under the receiver operating characteristic (ROC) curve (AUC). The diagnostic performances of the composite markers were compared with the performances of all single markers using the AUC, according to the method of Hanley and McNeil. 19 The diagnostic accuracy was defined as follows: AUC value 0.90–1.00, excellent; 0.80–0.89, good; 0.70–0.79, fair; 0.60–0.69, poor and 0.50–0.59, failure. 20 All analyses were performed using SPSS statistical software for Windows, version 19.0 (IBM, Armonk, NY, USA). The figures were drawn with GraphPad Prism, version 5.01 (GraphPad, San Diego, CA, USA).
Results
In a total of 112 patients who underwent neurosurgery, 43 patients were diagnosed with PNBM according to predefined criteria and 69 patients were diagnosed with PNAM. The incidences of PNBM and PNAM were 38.4% and 61.1%, respectively. A pathogenic bacterium was isolated from the CSF of four patients (9%) in PNBM. There were no statistically significant differences in these biomarkers between the case of positive bacterial cultures and other patients in PNBM. A total of 92 patients complained of headaches after neurosurgery and five patients had seizures. All clinical features and laboratory parameters were compared between the two groups. Age, sex distribution and body temperature did not show a significant difference between the two groups. Similarly, there was no significant difference between the presence of headaches, nuchal rigidity and seizures between the two groups (Table 1).
Baseline characteristics.
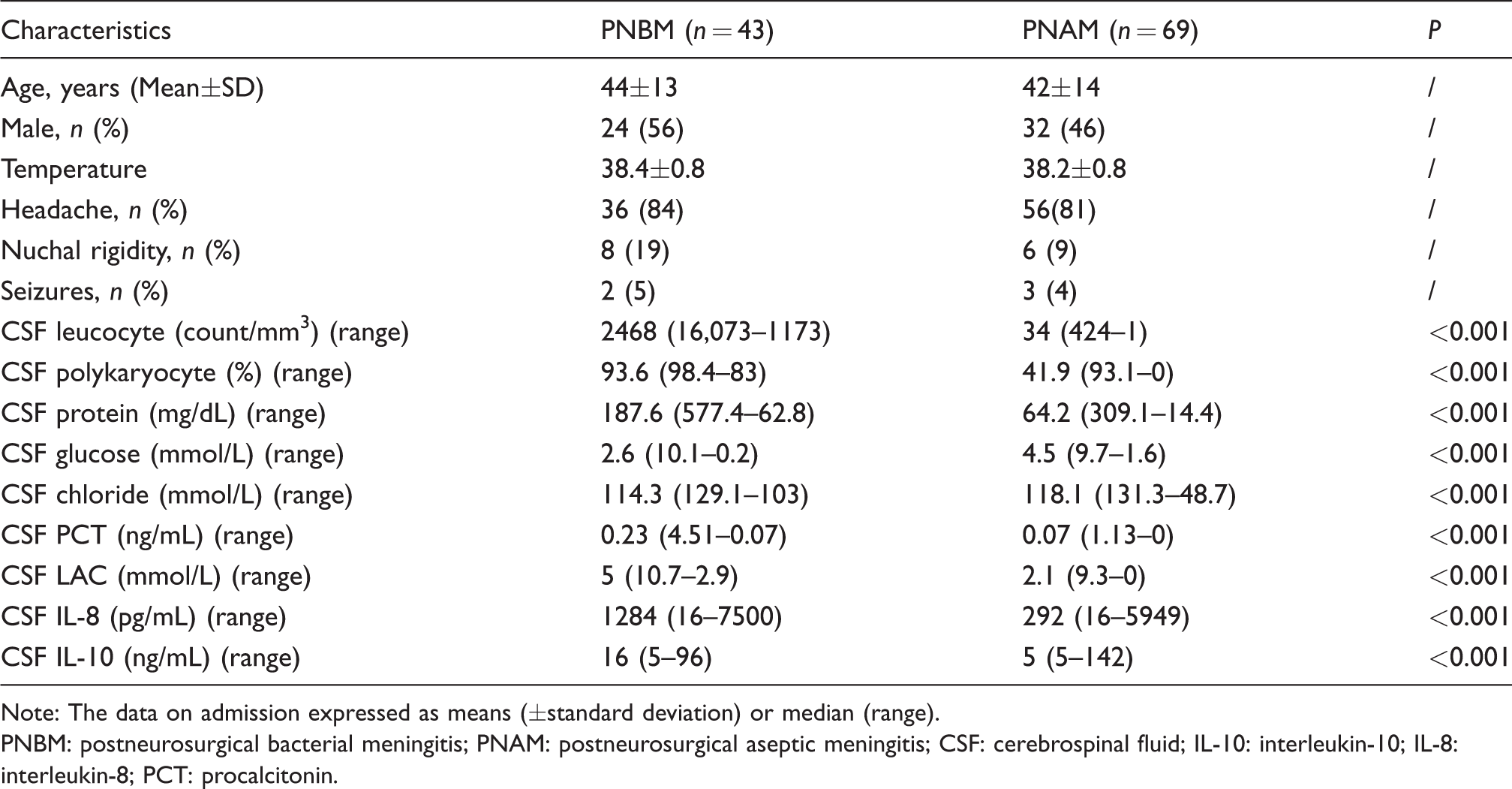
Note: The data on admission expressed as means (±standard deviation) or median (range).
PNBM: postneurosurgical bacterial meningitis; PNAM: postneurosurgical aseptic meningitis; CSF: cerebrospinal fluid; IL-10: interleukin-10; IL-8: interleukin-8; PCT: procalcitonin.
The differences of PCT, lactate, interleukin-8 and interleukin-10 concentrations between the PNBM and the PNAM groups were statistically significant (P < 0.001). The individual baseline values and median levels of the four biomarkers are shown in Figure 1. To determine the diagnostic value of the CSF indicators for PNBM, ROC curve analyses of the CSF PCT, lactate, interleukin-8 and interleukin-10 were performed (Figure 2). The computational AUCs, specificities, sensitivities, Youden indices, positive predictive values and negative predictive values of the single markers and the composite markers with regard to diagnoses of PNBM are shown in Table 2. The AUCs for the detection of PNBM were 0.803 (95% confidence interval [CI], 0.724–0.883) for PCT, 0.936 (0.895–0.977) for lactate, 0.771 (0.683–0.860) for interleukin-8, and 0.860 (0.797–0.929) for interleukin-10 (Table 2). Based on the aforementioned criteria, the lactate test was classified as an excellent test, PCT and interleukin-10 were classified as good tests and interleukin-8 was classified as a fair test.
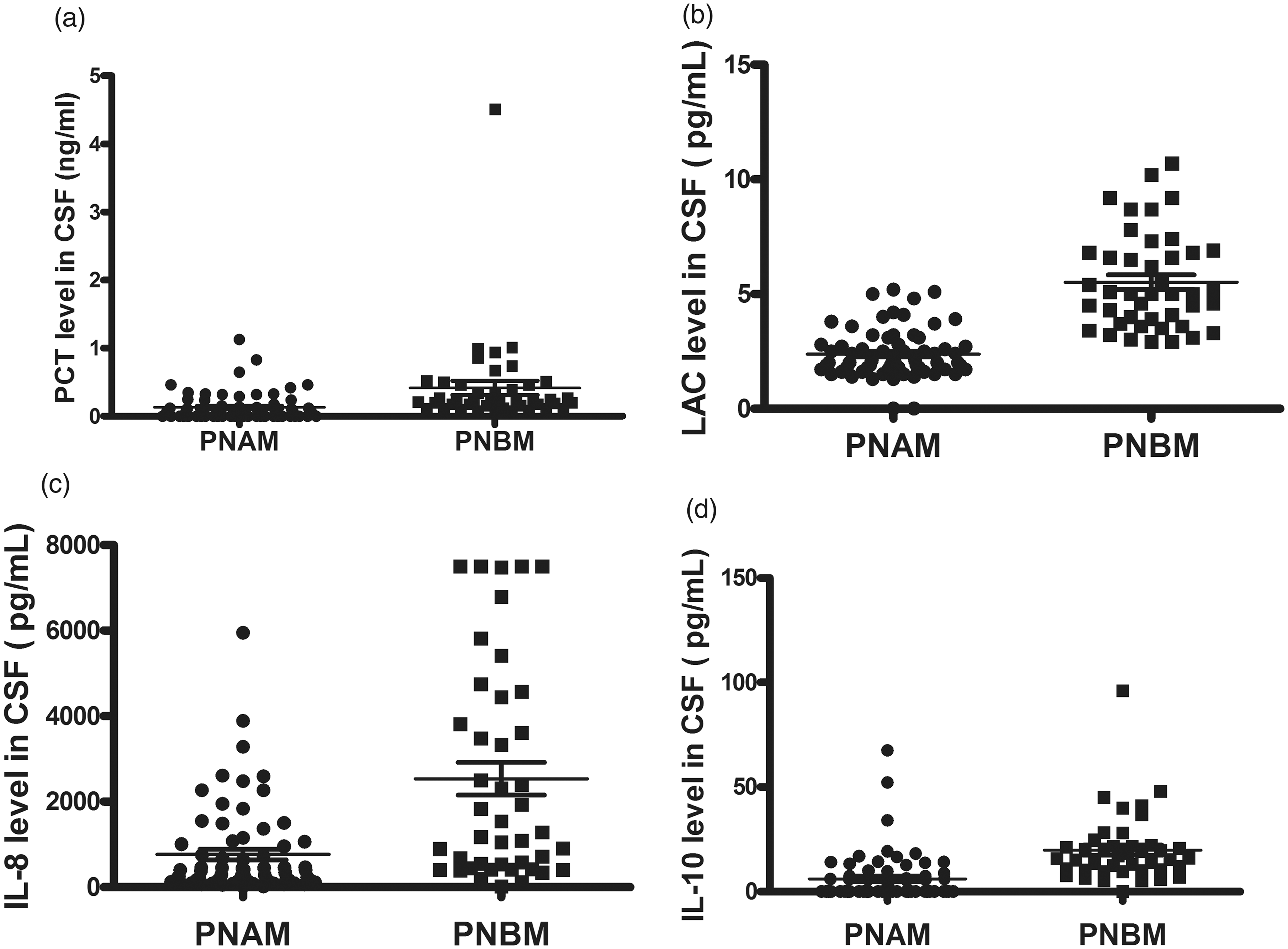
The difference of biomarkers’ concentration in CSF between the PNBM and the PNAM. (a) PCT, (b) lactate, (c) interleukin-8, (d) interleukin-10, both showed statistical significance (Mann-Whitney, P<0.001).
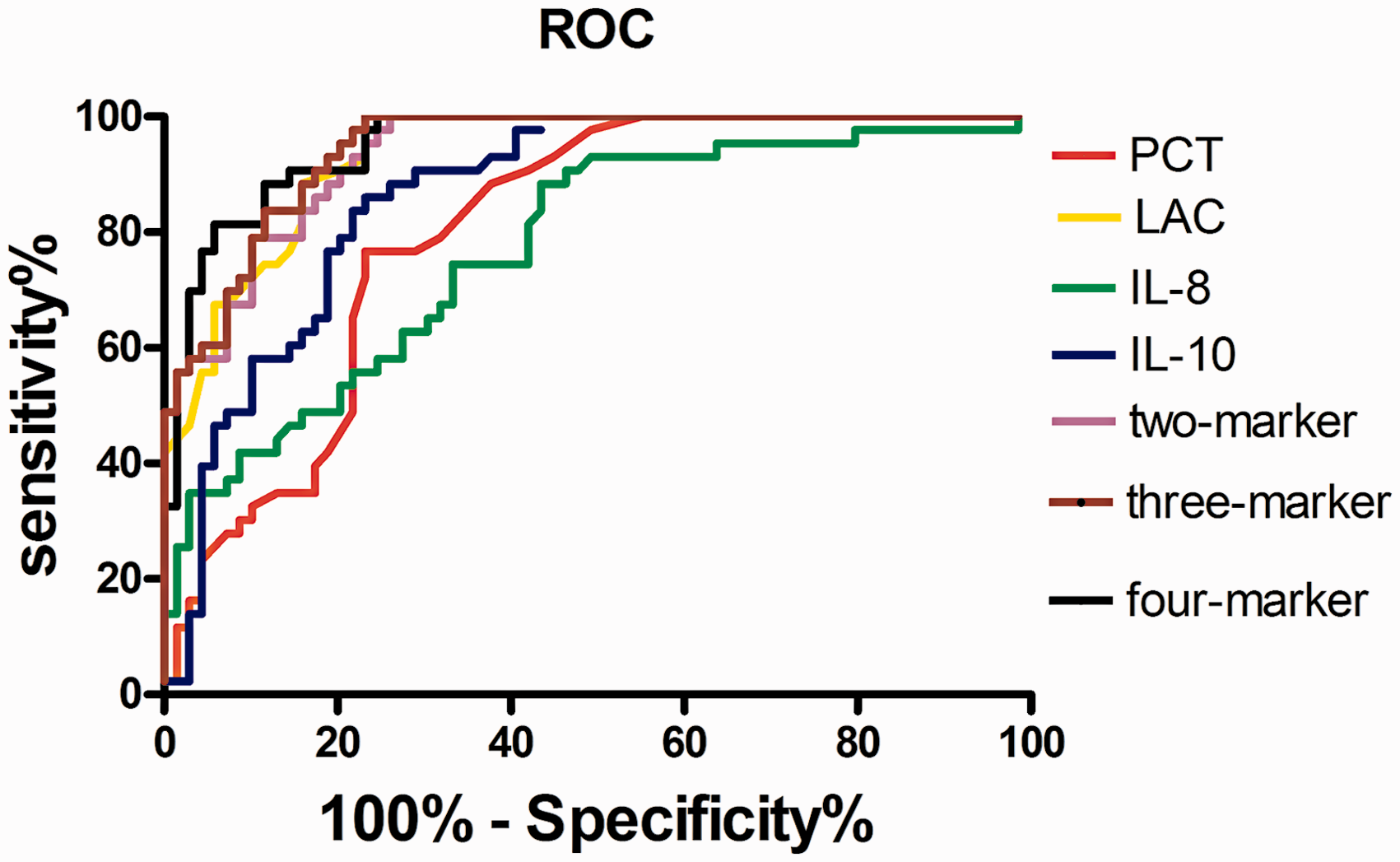
Receiver operating characteristic (ROC) curves comparing procalcitonin, lactate, interleukin-8, interleukin-10 and the combined two-marker, three-marker and four-marker tests for detection of postneurosurgical bacterial meningitis (PNBM) versus postneurosurgical aseptic meningitis (PNAM).
Accuracy of the four markers and the combined two-marker, three-marker and four-marker tests in diagnosing PNBM.
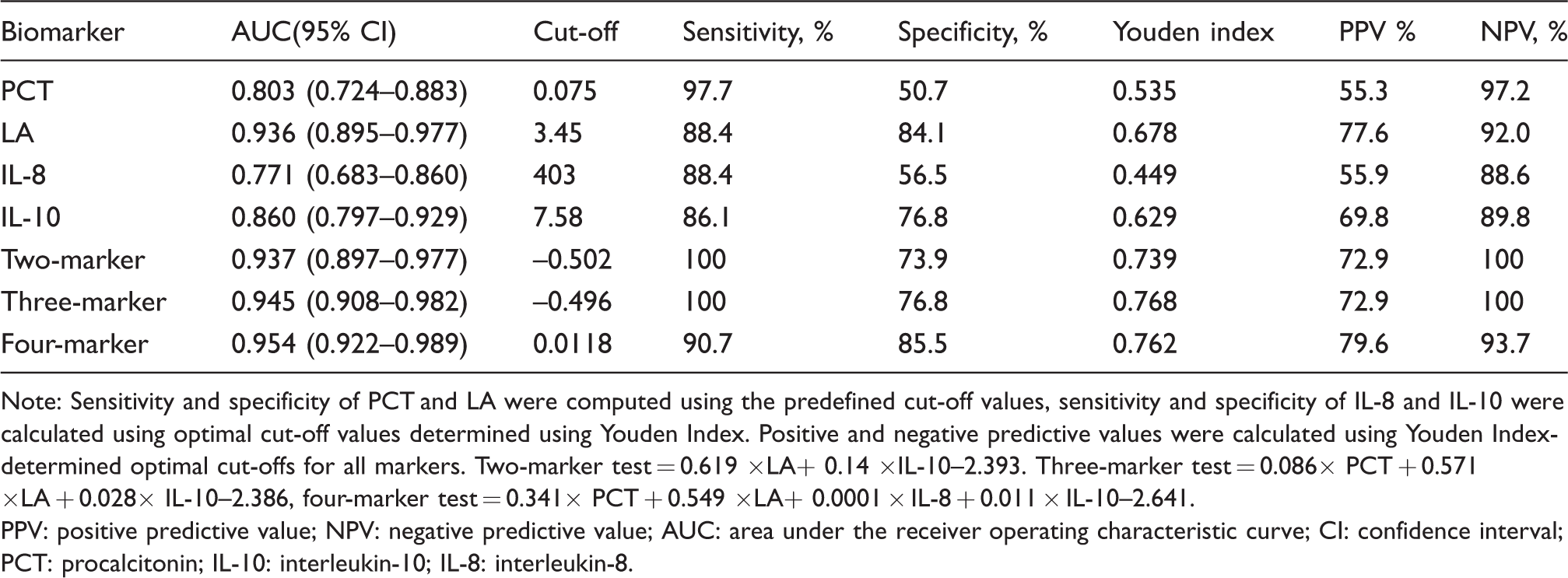
Note: Sensitivity and specificity of PCT and LA were computed using the predefined cut-off values, sensitivity and specificity of IL-8 and IL-10 were calculated using optimal cut-off values determined using Youden Index. Positive and negative predictive values were calculated using Youden Index-determined optimal cut-offs for all markers. Two-marker test = 0.619 ×LA+ 0.14 ×IL-10–2.393. Three-marker test = 0.086× PCT + 0.571 ×LA + 0.028× IL-10–2.386, four-marker test = 0.341× PCT + 0.549 ×LA+ 0.0001 × IL-8 + 0.011 × IL-10–2.641.
PPV: positive predictive value; NPV: negative predictive value; AUC: area under the receiver operating characteristic curve; CI: confidence interval; PCT: procalcitonin; IL-10: interleukin-10; IL-8: interleukin-8.
Three algorithms were constructed with linear discriminant analyses (Figure 2), which is a classification method that integrated multiple dimensional data to maximize the capacity for differentiating between two groups. Algorithm 1 integrated two biomarkers that resulted in the greatest AUC. The AUC-ROC value of algorithm 1 was 0.937 (95% CI, 0.897–0.977), which was significantly higher than the values of interleukin-10 but was the same as lactate. Algorithm 2 integrated PCT, lactate and interleukin-10, with an AUC of 0.945 (0.908–0.982). Algorithm 3 integrated the four biomarkers with an AUC of 0.954 (0.922–0.989), which was greater than that of the single biomarkers. All composite results were classified as excellent tests.
Discussion
In the present study, we investigated the combined values of CSF PCT, lactate, interleukin-8 and interleukin-10 in detecting PNBM. The results showed that there was a significant gain in discriminative power of diagnostic biomarkers when a linear combination resulting in the highest AUC was used.
Previous studies have showed the diagnostic value of serum or CSF PCT assays for the differentiation of bacterial meningitis and viral meningitis.21–25 Serum PCT has been a proven newly identified biomarker for inflammation and sepsis. However, the usefulness of CSF PCT in the diagnoses of intracranial infection has not been well documented. Compared with a previous retrospective study, 11 CSF PCT showed a higher AUC (0.803 vs. 0.746) in this report. When 0.075 μg/L was used as a cut-off value for PCT, the computed sensitivity and specificity were 97.7% and 50.7%, respectively. However, when we determined the optimal cut-off values for PCT using the Youden index, it was 0.155 μg/L. The difference between the two cohorts may be partly due to the sampling. In this prospective study, patients showing clinical symptoms of bacterial meningitis within 48–72 h after surgery were asked to participate, and none of these patients were treated with antibiotics.
CSF lactate originates from anaerobic glycolysis inside bacteria and ischaemic brain tissue, which is caused by bacterial infection. It has been reported to be a good single indicator and a better marker compared with other conventional markers for discriminating PNBM from aseptic meningitis.9,26 Compared with our previous retrospective study, 11 the diagnostic efficacy of CSF lactate (AUC: 0.943 vs. 0.936) was comparable. The cut-off value of lactate obtained from a retrospective study 11 was applicable in the current study, and the computational specificities and sensitivities were 84.1% and 88.4%, respectively.
In the CNS, interleukin-8 and interleukin-10 are upregulated whenever neuroinflammation is involved. 27 Interleukin-8 acts as a chemoattractant of neutrophils to the site of inflammation. 12 Yao et al. reported that the corresponding summary performance estimates for interleukin-8 were as follows: sensitivity, 0.95; specificity, 0.89 and AUC, 0.95 (95% CI, 0.93–0.97). 12 However, in our study, the diagnostic accuracies of interleukin-8 (AUC, 0.779); sensitivity, 88.4 and specificity, 56.5 were fair in differentiating between PNBM and PNAM. There were differences between the two cohorts, both in terms of the spectrum and severity of the diseases. Interleukin-10 concentrations have also been found to increase in the serum or CSF of patients with meningitis.17,28,29 Oztuzcu et al. reported that a significant increase of interleukin-10 gene expression was present in patients with meningitis. 17 In the present study, the AUC of interleukin-10 (0.860) was second only to lactate and showed good diagnostic accuracy.
The ability of single biomarkers to discriminate between PNBM and PNAM was analysed, but these previous analyses did not evaluate the combination of multiple biomarkers. Measurements of each putative biomarker concentration with individual assays involve considerable time, cost and sample volume, thus limiting the ability to systematically examine the potential use of multiple biomarkers in combination. However, various methods can now measure biomarkers in parallel, 30 which had the advantages of less sample volume and reduced assay times, as well as an increase in the number of analytes that could be simultaneously measured.
Combining multiple biomarkers requires an algorithm to classify subjects into two groups: those suspected of having a disease and those not suspected of having a disease. Numerous types of analyses are available for classification, including normal linear discriminant analysis, 31 regression trees 32 and artificial neural networks. 33 Normal linear discriminant analysis derives a score but not a specific decision rule, which can be obtained using decision trees, Bayesian decisions and neural networks. 34 In many studies, linear discriminant analysis was adopted.35,36 In addition, the use of ROC curves and a comparison of AUCs for single biomarkers have become widespread.
The combination of diagnostic biomarkers to yield the greatest AUC has been a useful approach for improving the accuracy of diagnoses for many diseases, such an Alzheimer’s disease, sepsis and cancer. In the present study, three algorithms were constructed with linear discriminant analyses to integrate multiple biomarkers to maximize the capacity for differentiating between PNBM and PNAM. In algorithm 1, the combination of interleukin-10 and lactate was no better than lactate alone. In algorithm 3, the combination of four biomarkers had the greatest AUC (0.954) compared with single biomarker and other combinations. The AUC of CSF lactate is excellent (AUC 0.936) and the addition of the other three biomarkers improves a little. However, there are many factors that affect the detection for a single biomarker. In combination, the results between different makers can be mutually verified. The combination of four biomarkers not only improved AUC, but also sensitivity, specificity, positive predictive value and negative predictive value. Accurate and rapid diagnosis of bacterial meningitis is important in neurosurgery, since the infection outcome will depend on early and specific antibiotic therapy. Besides, we independently developed a parallel detection platform and this sensitive and cost-efficient platform is expected to be a powerful tool to measure combinations of various biomarkers. 37
Thus, we recommend 0.0118 in algorithm 3 as a score to distinguish between PNBM and PNAM. When algorithm 3 was >0.0118, we classified it as PNBM and started antibiotic treatment because the possibility of PNAM was <15% (the specificity was 85.5%). When the value was <0.0118, it was not necessary to start treatment immediately, because the possibility of PNBM was <10%. According to linear discriminant analyses, the diagnosis for PNBM involves the processing and analyses of data, which is based on the principle of automatic diagnosis found in modern hospitals.
Despite the strengths of our study, there were some limitations. Given that CSF bacterial cultures represented an unreliable test with a high number of false-negatives, the majority of PNBM diagnoses use the results of routine CSF examinations as inclusion criteria, such as cell counts, protein concentrations and glucose concentrations. We did not include these traditional markers in our linear combinations, which might have led to incorporation bias and thus an overestimation of the diagnostic power of these markers. In the following study, we would recruit PNBM patients based on CSF bacterial cultures and integrated all traditional and novel biomarkers into an ROC curve.
Conclusions
This study showed the value of combining CSF PCT, lactate and interleukin-10 values for detecting PNBM. The combination of diagnostic biomarkers improved the accuracy for diagnosing PNBM and may be also applicable to other complex diseases. However, it is important to consider that obtaining a larger AUC is not always clinically relevant.
Footnotes
Acknowledgements
We thank all clinicians who contributed to this study. The authors are grateful to the Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University and Beijing Neurosurgical Institute, Capital Medical University Beijing, China.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Capital Characteristic Clinical Application Research (Z151100004015144).
Ethical approval
Not applicable.
Guarantor
XK.
Contributorship
All three authors contributed to the design of the study. GZ researched the literature, wrote the first draft of the paper and edited the final draft of the manuscript. CY was responsible for the initial data acquisition and also contributed to the first draft of the paper. XK and ZG were responsible for the overall design of experiments and performed all statistical analyses. All authors agreed with the final draft of the manuscript. HW and YL contributed to gather clinical data.
