Abstract
Background
Smoking cessation was associated with improved prognosis of coronary artery disease. This study was designed to investigate the effect of smoking cessation on high-density lipoprotein functionality in coronary artery disease patients.
Methods
In this prospective, randomized and parallel controlled study, coronary artery disease smokers (n = 28) and healthy smokers (n = 30) were divided into smoking cessation group and continuous smoking group, respectively. Blood samples were collected before and after three-month smoking cessation. Plasma high-density lipoprotein was isolated by density gradient centrifugation. The ability of high-density lipoprotein against copper-induced oxidation of lipoprotein was determined to evaluate the antioxidative property of high-density lipoprotein, and the macrophage migration inhibited by high-density lipoprotein was tested to identify the antichemotactic property of high-density lipoprotein. High-density lipoprotein-induced macrophage cholesterol efflux was measured by fluorescence spectrometry using NBD cholesterol analogue. Healthy non-smoking volunteers were enrolled as the baseline control.
Results
The baseline antioxidative, antichemotactic ability of high-density lipoprotein and high-density lipoprotein-induced cellular cholesterol efflux in coronary artery disease smokers and healthy smokers were significantly attenuated when compared with those in healthy non-smokers. After three-month smoking cessation, both the antioxidative ability and antichemotactic ability of high-density lipoprotein were improved significantly in coronary artery disease smokers. However, high-density lipoprotein-induced cellular cholesterol efflux was not increased by smoking cessation. In in vitro experiments, carbon monoxide reduced the antioxidative ability and nicotine enhanced the antichemotactic ability of high-density lipoprotein.
Conclusions
Smoking cessation is an effective measure to improve high-density lipoprotein functions in coronary artery disease smokers. Our study re-emphasizes the importance of smoking cessation in the secondary prevention of coronary artery disease.
Keywords
Introduction
Epidemiological data have demonstrated that the concentration of high-density lipoprotein cholesterol (HDL-C) is negatively correlated with the risk of cardiovascular events.1–3 The attempts aimed at increasing plasma HDL-C concentration to reduce the risk of cardiovascular events failed eventually.4,5 Thus, the focus has recently shifted toward HDL function. HDL has multi-antiatherogenic effects on coronary artery disease (CAD), such as antioxidation and anti-inflammation, in addition to being a key mediator of reverse cholesterol transport. Cholesterol efflux capacity has been proved inversely associated with the incidence of cardiovascular events. 6 Inflammation reduces the ability of HDL to participate in reverse cholesterol transport and to protect low-density lipoprotein (LDL) from oxidation, which may facilitate the development of atherosclerosis. 7 The HDL function is considered much more important than plasma HDL-C concentrations.
Cigarette smoking is an important risk factor for the development of cardiovascular disease. Tobacco contains thousands of harmful substances, and many of them are detrimental to the cardiovascular system, including carbon monoxide, tar, nitrosamines, polycyclic aromatic hydrocarbons, cyanide of hydrogen, aldehydes and heavy metals. 8 Epidemiological and clinical studies have shown that cigarette smoking dose- and time-dependently increased the risk of cardiovascular events. Smoking accelerated the occurrence of acute myocardial infarction nearly a decade earlier. 9 Smoking cessation is an effective measure to reduce the risk of myocardial infarction and all-cause mortality in CAD patients.10,11
Our previous study showed that exposure to cigarette smoking was inversely correlated to HDL-C concentration. 12 Smoking cessation increases the HDL-C concentration and reduces the risk of CAD.13 Previous studies in healthy smokers demonstrated that smoking cessation improved HDL function, increased cholesterol efflux capacity and reduced inflammation, without changing HDL-C concentrations or HDL subfractions. And the authors speculated that these might be the mechanisms by which smoking cessation reduces the risk of cardiovascular disease. 14
Smoking cessation improves the compliance of small arteries and the endothelial function, reduces platelet aggregation and attenuates platelet redox imbalance in long-term smoking patients. To date, the potential mechanisms by which cigarette smoking accelerates atherosclerosis and smoking cessation benefits the patients with vascular diseases have not yet been fully clarified. There are no data, so far, showing the impact of smoking or smoking cessation on HDL functionality in CAD individuals.
The purpose of our study is to investigate the antioxidative and antichemotactic properties of HDL and HDL-induced macrophage cholesterol efflux in CAD smokers before and after three-month smoking cessation. Furthermore, in this study, we try to explore the effects of nicotine and carbon monoxide on HDL function.
Materials and methods
Subjects
This is a randomized, prospective and parallel controlled study. A total of 84 male subjects were screened at Peking Union Medical College Hospital (PUMCH) (Beijing, China). Smoking subjects aged 40 to 80 years old, who had smoked at least 10 cigarettes per day for at least 10 years, were eligible for this study. All the CAD smokers had documented CAD with at least one major coronary artery stenosis ⩾50% by coronary angiography or a history of myocardial infarction. The subjects who had acute coronary syndrome, diabetes mellitus, history of renal or hepatic diseases, ischaemic cerebrovascular or cardiac episodes within three months preceding the randomization, taking lipid-lowering drugs within two weeks before randomization or serum triglyceride ⩾ 400 mg/dL were excluded. With the inclusion and exclusion criteria above, 35 healthy smoking volunteers (non-CAD [NCAD] group) and 32 CAD smokers (CAD group) were enrolled; 17 healthy non-smoking volunteers were used as the baseline control. Smoking subjects were randomized into smoking abstinence subgroup or continuous smoking subgroup. In the NCAD group, 17 subjects were randomized into smoking abstinence subgroup (NCAD-abs) and 18, the continuous smoking subgroup (NCAD-smo). In the CAD group, 16 CAD smokers were randomized into smoking abstinence subgroup (CAD-abs) and the other 16, continuous smoking subgroup (CAD-smo) (Figure 1). Five non-CAD and four CAD smokers interrupted during follow-up because of consent withdrawing or study protocol violation. Smoking subjects in the abstinence subgroups were requested to stop smoking during the three-month follow-up period. Urine cotinine concentrations and carbon monoxide in expiration air were monitored in smoking abstinence subjects to ensure the compliance of smoking cessation.
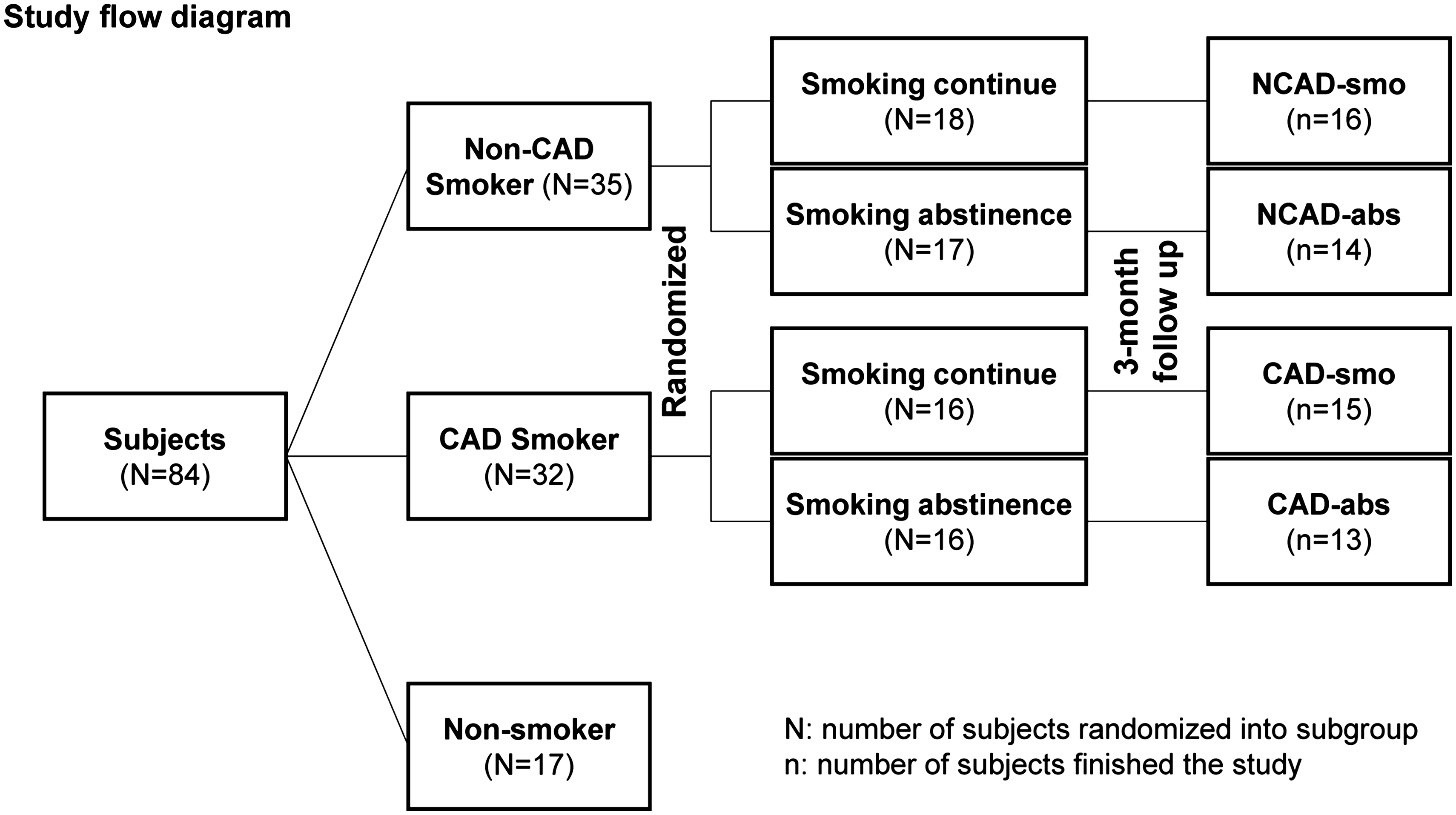
Study flow chart. Thirty-five healthy (non-CAD) smokers and 32 CAD smokers were enrolled and randomized into four subgroups; 17 non-smoking volunteers were enrolled as the baseline control. Five non-CAD smokers (three in NCAD-abs and two in NCAD-smo) and four CAD subjects (three in CAD-abs and one in CAD-smo) interrupted during follow-up because of consent withdrawing or study protocol violation.
Ethics statement
Registration number: ChiCTR-RCH-10000748 (Chinese clinical trial registry). Ethical approval was received from the Human Ethics Committee of Peking Union Medical College Hospital (PUMCH, approval No. S262). All participants had provided written informed consent. The study protocol conformed to the ethical guidelines of the Helsinki Declaration.
Cells and reagents
The human acute monocytic leukaemia cell line THP-1 was obtained from the cell bank of The Institute of Basic Medical Sciences of the Chinese Academy of Medical Sciences (Peking, China). Reagents and kits are shown in detail in the supplementary material.
Blood samples and biochemical analysis
Peripheral venous blood was sampled between 8:00 and 9:00 in the morning after an overnight fast at randomization and the end of follow-up. All subjects were requested to take low-fat diet for three days and not to smoke for 6 h before blood collection. Blood samples were collected into serum tubes and K3-tetraacetic acid (EDTA) tubes. Plasma was separated by centrifugation (2000 r/min, 20 min), and stored at 4°C for further analysis. Serum biochemical tests were performed in the Clinical Laboratory of PUMCH, including blood lipid profile, kidney function, liver function, blood electrolytes (Olympus AU5400, Japan). Complete blood count (Sysmex XE-5000–2, Japan) and urinalysis (SIEMENS Clinitek Atlas Analyzer, Germany) were also detected in Clinical Laboratory of PUMCH.
Isolation of lipoprotein
The isolation of lipoprotein process was conducted in Central Lab. of Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences (Beijing, China). The separation and purification of lipoprotein were described by Karlsson et al. 15 Briefly, 13 mL of plasma mixed with solid NaBr (0.4 g/mL) was layered at the bottom of a centrifuge tube. The EDTA plasma fraction was gently over-layered with NaBr solution (density 1.210 g/mL). After the first ultracentrifugation (Beckman L8–55 equipped with Ti 55.2 rotor, Beckman Coulter Inc., Brea, USA) at 42,000 r/min (109,000×g) for 12 h at 4°C, all lipoproteins were in the upper part of the tube. After gently adding 10 mL of NaBr solution (density 1.063 g/mL and 1.006 g/mL) to the top of lipoprotein fraction successively, the lipoproteins were further fractionated by a second centrifugation at 45,000 r/min (125,000×g) for 2 h at 4°C. Chylomicrons and very low-density lipoprotein were on the top, LDL in the middle and HDL at the bottom (Supplementary Figure 1). The isolated lipoproteins were dialysed against 20 mM Tris-HCl/0.9% normal saline (pH 7.4) for 48 h at 4°C to remove EDTA. Lipoproteins were collected and stored at 4°C. The concentration of isolated lipoprotein was measured by Lowry method, 16 HDL concentration was 1844 ± 912 μg/mL, and the purity was confirmed by agarose gel electrophoresis (Supplementary Figure 2).
Analysis of urinary cotinine and exhaled carbon monoxide
To ensure the compliance of study intervention, the urine of each smoker was collected to test urinary cotinine concentration using human cotinine kit (Immunalysis Co., Pomona, CA, USA), and the exhaled carbon monoxide was detected by carbon monoxide smoking analyzer (Smokerlyzer®-compactTM, Bedfont Scientific Ltd, UK) at randomization, every month of follow-up and the end of the study. The cut-off point of exhaled carbon monoxide between non-smokers and smokers was 6 ppm of carbon monoxide, and the cut-off point of urinary nicotine was 30 ng/mL. The smoking abstinence subjects whose parameters were more than these cut-off points or those of the continuous smokers were lower than these cut-off points would be excluded from the study. 17
Copper-induced oxidation of lipoproteins and antioxidation of HDL
The measurement of lipoproteins oxidation was described by Esterbauer et al. 18 Oxidation of LDL (diluent final concentration 50 μg/mL) was initiated by adding 50 μL of CuSO4 at a final concentration of 10 μM Cu2+ at 37°C. The kinetics of LDL oxidation was detected by the change of 234-diene absorption using spectrophotometer (Synergy4, BioTek, Winooski, VT, USA). The mixture of LDL samples from healthy non-smokers was regarded as the normal state of LDL (150 μg/mL), which were labelled as LDLpool. The LDL and HDL were obtained from the plasma of CAD and non-CAD subjects. The HDL+LDLpool was the HDL samples adding into LDLpool, for detecting the antioxidation ability of the HDL. The enzyme reaction rate was automatically calculated by preprogrammed kinetics software Gen 5 (BioTek, USA). Several characteristic oxidation parameters were obtained from the resulting OD value versus time curves, including basal OD value (reflecting basal oxidation state, detecting by the LDL sample), lag time (oxidation resistance, oxidative susceptibility of lipoprotein was negatively correlated with lag time), Vmax (maximum oxidation speed, the growth of OD value divided by the time in propagation phase) and maximum OD value (the maximum oxidation state, which is the maximum oxidation level of lipoprotein) (Supplementary Figure 3).
THP-1 cell culture and treatment
The THP-1 human monocytic cell line was cultured in RPMI 1640 medium (Sigma-Aldrich, St. Louis, MO, USA) supplemented with 20% heat-inactivated FBS and 100 μg/mL penicillin-streptomycin at 37°C with 5% CO2. The THP-1 cells were seeded at a density of 2 × 106 cells/mL in a 12-well plate (Costar; Corning Inc., NY, USA) and transformed into macrophages by treatment with phorbol 12-myristate 13-acetate (PMA) (100 ng/mL) for 72 h and synchronized with RPMI-1640 medium containing 0.4% FBS for 12 h at 37°C and 5% CO2. To confirm the THP-1-derived macrophages, the cells were dyed with Giemsa and immunohistochemically stained with CD68, and observed using a microscope (Supplementary Figure 4(a)).
Antichemotactic effects of HDL
Migration of THP-1 monocytes towards monocyte chemoattractant protein-1 (MCP-1) was determined using 24-well plates (CoStar Corp, USA) carrying 12 transwell inserts of 8 μm pore diameter. The method was described by Kintscher et al. 19 with slight modifications. After THP-1 cells (2.0 × 105 cells/mL) were re-suspended in RPMI 1640 medium containing 0.4% FBS for 12 h at 37°C and 5% CO2 in the upper chamber, THP-1 incubated with LDLpool (50 μg/mL) alone or LDLpool plus HDL (50 μg/mL). One millilitre of 25 ng/mL MCP-1 and RPMI 1640 complete medium containing 10% FBS were added into the lower chamber to induce migration, while 1 mL of saline containing RPMI 1640 and 10% FBS was used as negative control. Cell migration was assessed after 24 h, and the number of migrated cells passing through and attaching to the polycarbonate membrane was counted by DAPI (6-diamidino-2-phenylindole dihydrochloride) dyeing under a fluorescence microscopy (EVOS-fl; Thermo Fisher Scientific, Waltham, USA), the images processed by Image J (NIH, USA) (Supplementary Figure 4(b)).
HDL-induced cholesterol efflux
HDL-induced cholesterol efflux from THP-1-derived macrophages was tested by fluorescence spectrometry using NBD cholesterol (Thermo Fisher Scientific Inc., Waltham, MA, USA) analogue (Invitrogen, N1148, USA), as described by Song et al. 12 The time and concentration of incubation were determined by the fluorescence–concentration curve and fluorescence–time curve, which were drawn according to the results of preliminary experiments. THP-1-derived macrophages (2 × 105 cells/mL) were incubated with RPMI 1640 containing 0.2% BSA for 12 h for equilibration of the intracellular-free cholesterols. Then the cells were labelled with NBD cholesterol (10 μg/mL) for loading of cholesterol for 6 h, and induced cholesterol efflux in serum-free RPMI 1640 medium with HDL (50 μg/mL) at 37°C for 4 h. The supernatant was removed, and the cells were washed with PBS and lysed by Triton X-100 (concentration 0.5%). Medium and lysate were centrifuged separately at 2000 r/min for 10 min. The fluorescence intensity and images were captured using fluorescence microscopy and a multilabel counter (PerkinElmer, Inc., Waltham, USA) at excitation 470 nm and emission 540 nm. The percentage of NBD cholesterol efflux was calculated by dividing the fluorescence intensity in the medium by the sum of the fluorescence intensity in the medium and cell lysate. Individual efflux values were averaged from three measurements in different wells.
Effects of nicotine and carbon monoxide on HDL function
Nicotine was dissolved in normal saline (0.9% NaCl), and the pH of the solution was adjusted to 7.4 using NaOH. Normal HDL (150 μg/mL) was incubated with different concentrations of nicotine (final concentration 25 μM, 50 μM, 75 μM and 100 μM) or CORM-2 (a release of carbon monoxide compounds, final concentration 0.1 μM, 1 μM, 5 μM and 10 μM), at different durations of incubation time (0 h, 12 h, 24 h, 3 days, 7 days). The LDL and HDL used in this part of the experiment were obtained from the healthy non-smokers. All the experimental conditions were determined by preliminary studies. After the co-incubation, lipoprotein dialysis, filtration and concentration measurement were conducted as described previously. HDL antioxidative, antichemotactic ability and HDL-induced cholesterol efflux were determined as mentioned above.
Statistical analysis
Data were analysed using the SPSS Statistical Analysis System V-17.0 (SPSS, Chicago, IL, USA). The data were expressed as the mean ± standard deviation (SD). After three-month follow-up, the changes of study parameters from baseline were defined as: the value at the end of study−the value at baseline. Paired t-test, independent-sample t-test, One-way ANOVA and Turkey’s test were used for comparisons, and Wilcoxon signed-rank tests were used for ordinal variables. A P value of less than 0.05 was considered to be statistically significant. Data were presented by GraphPad Prism 6.0 (GraphPad Software, Inc., La Jolla, CA, USA).
Results
Study subjects
After three months of follow-up, 58 subjects (15 in CAD-smo, 13 in CAD-abs, 16 in NCAD-smo, 14 in NCAD-abs subgroup) and 17 non-smokers completed the study. Subjects who failed to stop smoking in the abstinence subgroups (three subjects in NCAD-abs and three in CAD-abs) were excluded by assaying of urinary cotinine and expiration carbon monoxide, two subjects in NCAD-smo and one in CAD-smo subgroup withdrew from the study during the follow-up period. General characteristics and laboratory profiles of the study subjects at baseline are shown in Table 1, and the changes in blood lipid profile from baseline are shown in Supplementary Table 1. The concentrations and changes of plasma PON-1, IL-6, MMP-9 and hs-CRP are shown in Table 2.
General characteristics and laboratory profiles of study subjects at baseline.
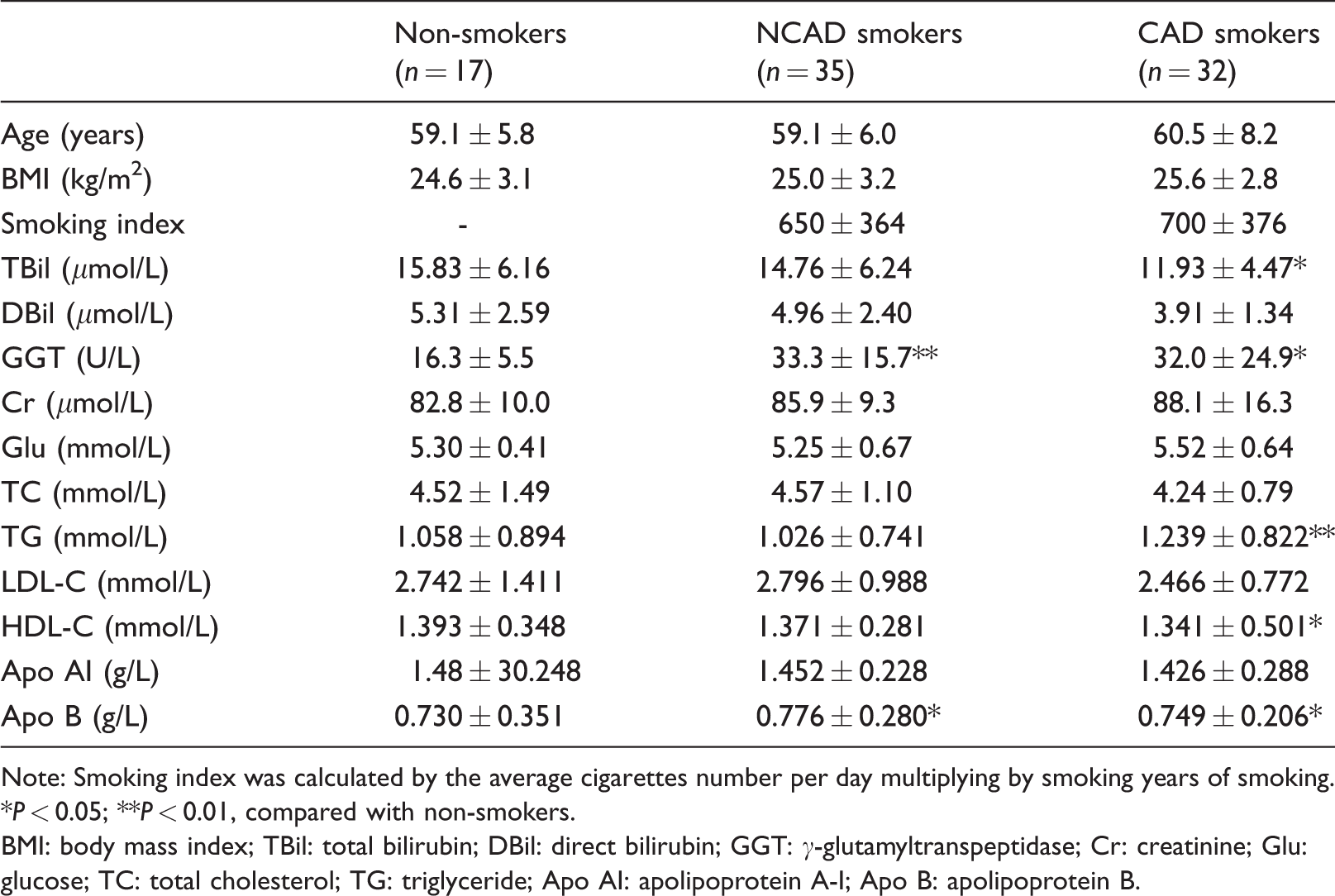
Note: Smoking index was calculated by the average cigarettes number per day multiplying by smoking years of smoking. *P < 0.05; **P < 0.01, compared with non-smokers.
BMI: body mass index; TBil: total bilirubin; DBil: direct bilirubin; GGT: γ-glutamyltranspeptidase; Cr: creatinine; Glu: glucose; TC: total cholesterol; TG: triglyceride; Apo AI: apolipoprotein A-I; Apo B: apolipoprotein B.
The inflammatory markers, antioxidation, antichemotactic effects and cellular cholesterol efflux of HDL at baseline and at the end of follow-up.
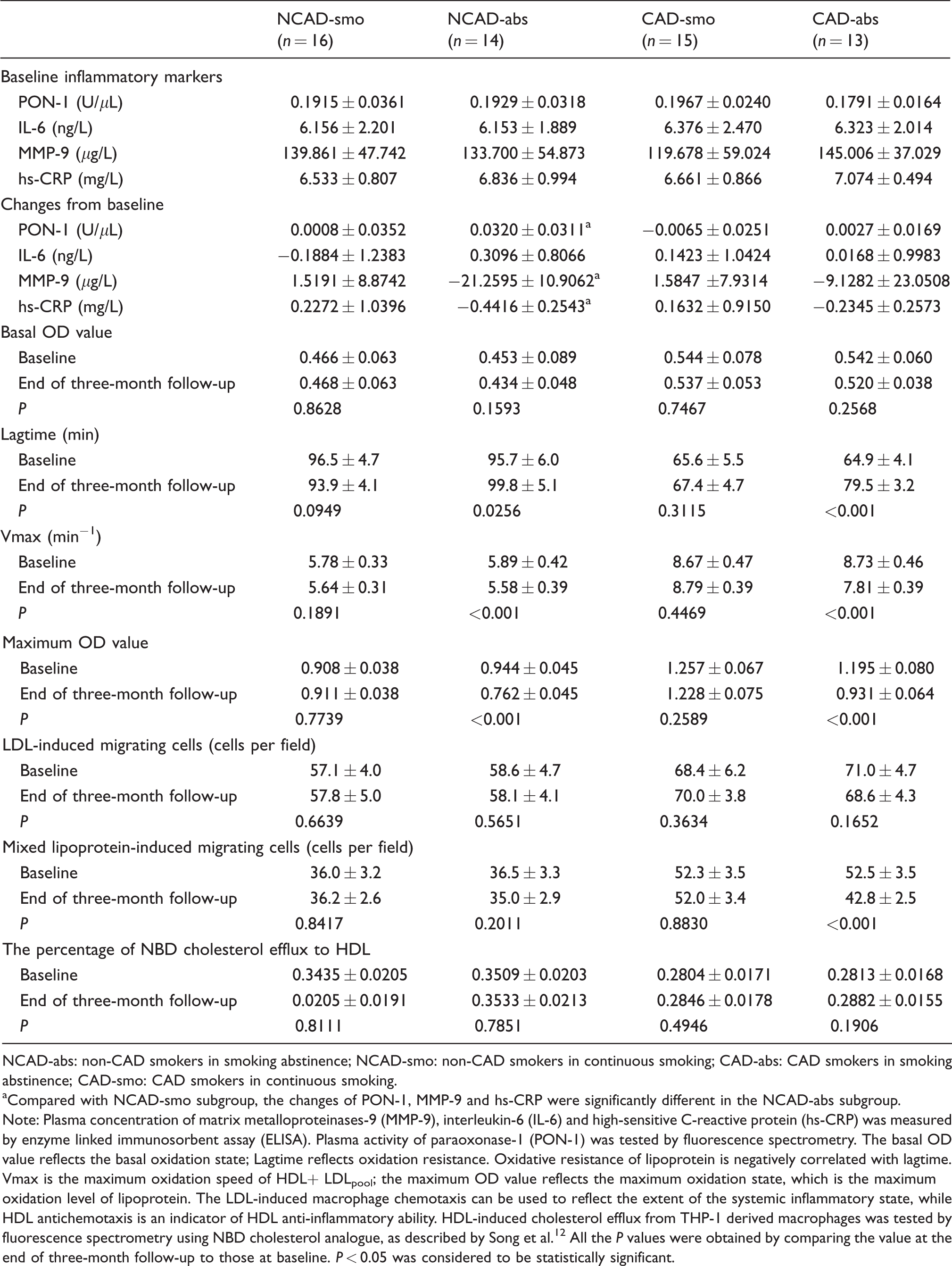
NCAD-abs: non-CAD smokers in smoking abstinence; NCAD-smo: non-CAD smokers in continuous smoking; CAD-abs: CAD smokers in smoking abstinence; CAD-smo: CAD smokers in continuous smoking.
aCompared with NCAD-smo subgroup, the changes of PON-1, MMP-9 and hs-CRP were significantly different in the NCAD-abs subgroup.
Note: Plasma concentration of matrix metalloproteinases-9 (MMP-9), interleukin-6 (IL-6) and high-sensitive C-reactive protein (hs-CRP) was measured by enzyme linked immunosorbent assay (ELISA). Plasma activity of paraoxonase-1 (PON-1) was tested by fluorescence spectrometry. The basal OD value reflects the basal oxidation state; Lagtime reflects oxidation resistance. Oxidative resistance of lipoprotein is negatively correlated with lagtime. Vmax is the maximum oxidation speed of HDL+ LDLpool; the maximum OD value reflects the maximum oxidation state, which is the maximum oxidation level of lipoprotein. The LDL-induced macrophage chemotaxis can be used to reflect the extent of the systemic inflammatory state, while HDL antichemotaxis is an indicator of HDL anti-inflammatory ability. HDL-induced cholesterol efflux from THP-1 derived macrophages was tested by fluorescence spectrometry using NBD cholesterol analogue, as described by Song et al.12 All the P values were obtained by comparing the value at the end of three-month follow-up to those at baseline. P < 0.05 was considered to be statistically significant.
The cotinine concentration in urine of non-smoker group was significantly lower than that in smoker group (21 ± 16 vs. 1960 ± 287 μg/L, P < 0.001). After three-month follow-up, urinary cotinine concentration did not change significantly compared with that at baseline in the continuous smoking group (2011 ± 268 vs. 1953 ± 242 μg/L, P = 0.08), but it was declined in the smoking abstinence group (1896 ± 286 vs. 22 ± 5 μg/L, P < 0.001) (Supplementary Figure 5).
Antioxidation of HDL
The ability of HDL against LDLpool oxidation was evaluated by lag time and Vmax of oxidation. At baseline, basal OD value of LDL, the lag time of HDL+ LDLpool, Vmax of HDL+ LDLpool, maximum OD value of HDL+ LDLpool were significantly higher in smokers than those in non-smokers group (all the P < 0.001). Basal LDL OD value, Vmax and maximum OD value were significantly higher (all P < 0.001) in CAD smokers than those in NCAD smokers, and the lag time of HDL+LDLpool was shorter in CAD smokers than that in NCAD smokers (P < 0.001) (Supplementary Figure 6). All these findings demonstrated that, at baseline, smokers were in a high oxidative state, especially for CAD smokers.
At the end of three-month follow-up, the changes of antioxidation parameters of HDL (HDL against LDLpool oxidation) from baseline were compared among the four study subgroups. The change of lag time from baseline in CAD-abs was significantly more than that in CAD-smo subgroup (14.56 ± 4.53 vs. 2.21 ± 4.99, P < 0.0001) and so was the change of lag time in NCAD-abs than that in NCAD-smo (4.08 ± 6.84 vs. −2.41 ± 7.17, P = 0.0061). After three-month follow-up, Vmax in HDL+LDLpool curve was significantly reduced in CAD-abs when compared with that in CAD-smo subgroup (−0.92 ± 0.27 vs. 0.12 ± 0.38, P < 0.0001). The change of maximum OD value from baseline was significantly more in CAD-abs than CAD-smo subgroup (−0.264 ± 0.095 vs. −0.021 ± 0.106, P < 0.001), and so was the change of maximum OD in NCAD-abs than that in NCAD-smo subgroup (0.182 ± 0.063 vs. 0.004 ± 0.031, P < 0.001). Comparing with those in NCAD-abs subgroup, the changes of lag time, Vmax and maximum OD of HDL+ LDLpool from baseline were more significant in CAD-abs subgroup (all P < 0.0001) (Figure 2). These results showed that smoking cessation improved the antioxidation ability of HDL in both CAD and non-CAD smokers, which was more predominant in the CAD smokers.
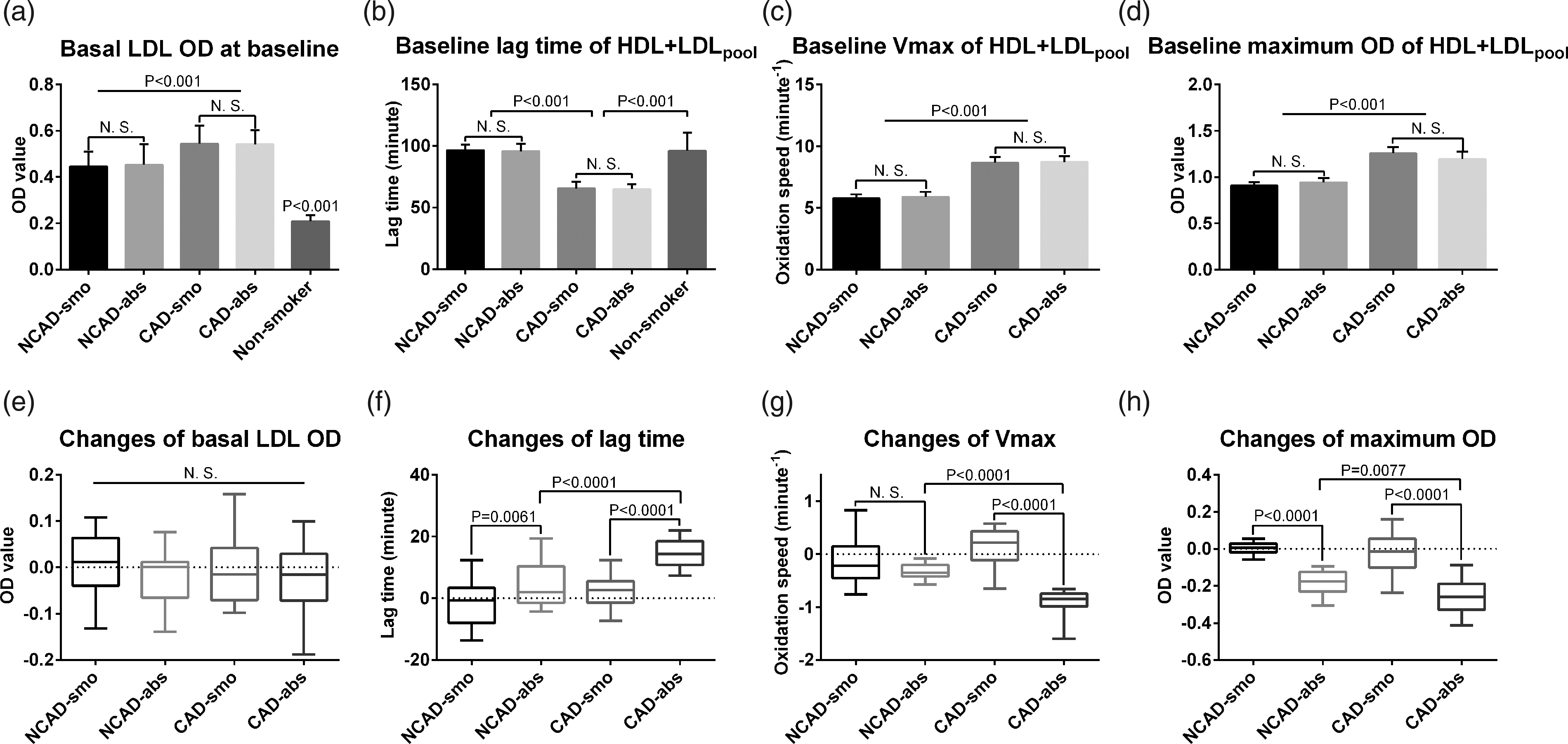
The baseline (a, b, c, d) values at randomization, and changes (e, f, g, h) of basal LDL OD value (basal oxidation state), lagtime (oxidative resistance of HDL, the longer the more resistance), Vmax (the maximum oxidation velocity) and maximum OD value (reflecting maximum oxidation state) from baseline at the end of three-month follow-up in the subgroups. The mixture of LDL samples from healthy non-smokers was labelled as LDLpool, the LDL and HDL were obtained from the plasma of CAD and non-CAD subjects. The changes from baseline were defined as and calculated as: the value at the end of study−the value at baseline (randomization). P < 0.05 was considered to be statistically significant.N. S: no significant difference; LDL: low-density lipoprotein; HDL: high-density lipoprotein; CAD: coronary artery disease.
After three-month smoking cessation, if compared directly with baseline, the lag time of HDL+LDLpool was prolonged in CAD-abs and NCAD-abs subgroups (P < 0.001 and P = 0.0256, respectively). For both CAD and healthy smokers, Vmax and Maximum OD value in HDL+LDLpool curve was significantly lower in smoking abstinence arm than those in continuous smoking arm (both P < 0.001) (Table 2 and Supplementary Figure 6). These results, once again, showed that smoking cessation enhanced the antioxidative capacity of HDL in both CAD and non-CAD smokers.
Antichemotactic effects of HDL
The effect of HDL on THP-1-derived macrophage migration was evaluated at baseline and the end of three-month follow-up to investigate the antichemotactic effects of HDL. At baseline, chemotaxis wandering ability of THP-1-derived macrophages in smokers, either incubated with LDL alone (P < 0.001) or with HDL+LDLpool (P < 0.001) was significantly higher than that in healthy non-smokers (Supplementary Figure 7). LDL (69.6 ± 5.5 vs. 57.9 ± 4.4 cells per field, P < 0.001) or HDL+LDLpool (52.3 ± 3.4 vs. 36.2 ± 3.2 cells per field, P < 0.001) induced chemotaxis was also higher in the CAD smokers than that in the non-CAD smokers (Figure 3(a) and (b)). These findings revealed that cigarette smoking was associated with an activated state of macrophages, which was more predominant in CAD smokers.
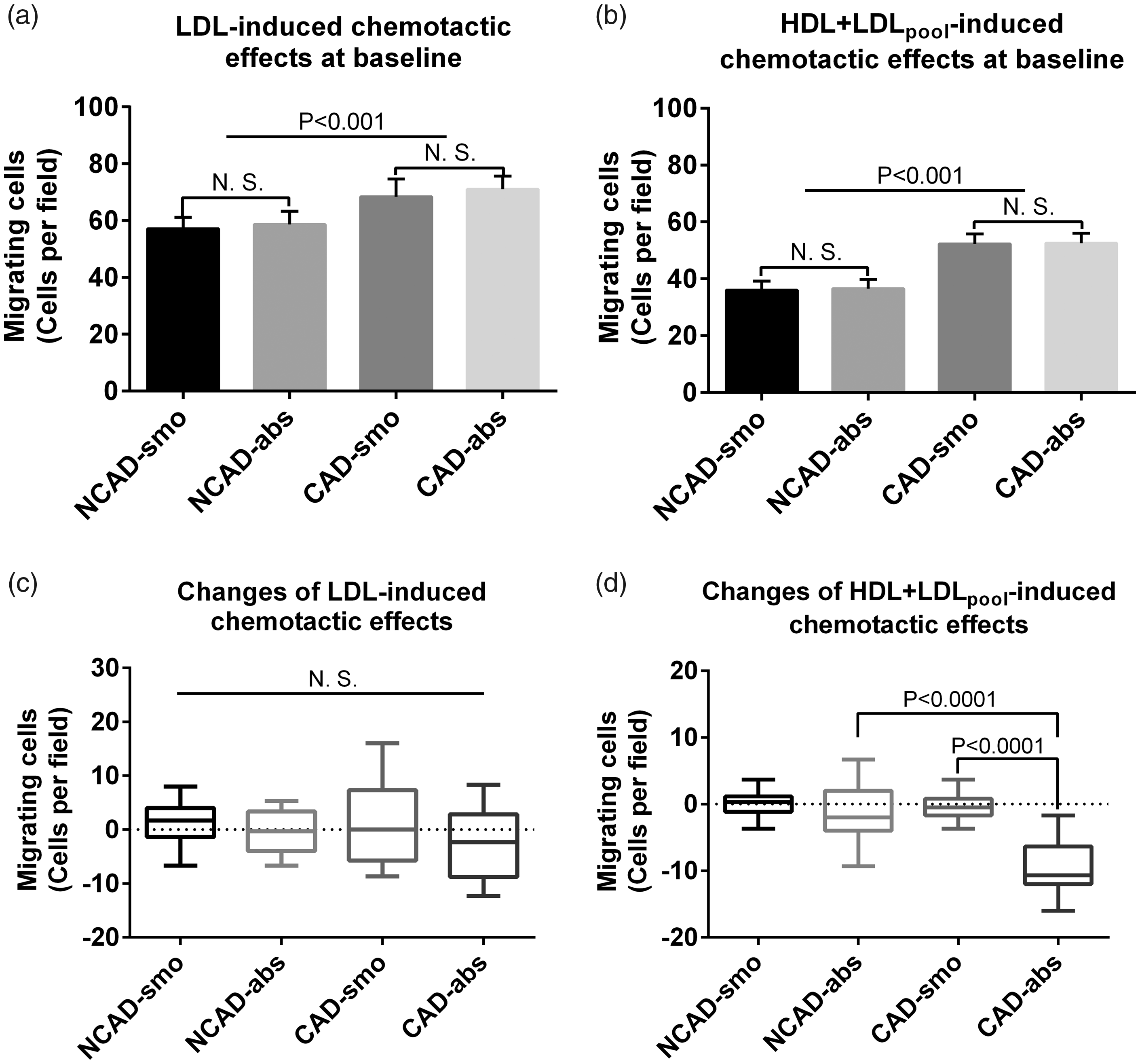
Chemotaxis assays by Transwell chamber induced by LDL or HDL+LDLpool at baseline (a, b) and the end of three-month smoking cessation (c, d). After incubation with LDL or HDL+LDLpool, the chemotactic ability of macrophages was evaluated by observing the number of migrated cells passing through the polycarbonate membrane of Transwell chamber. The LDLpool was the mixture of low-density lipoprotein from healthy non-smokers. The LDL and HDL were obtained from the plasma of CAD and non-CAD subjects. The change of chemotactic ability from baseline at the end of three-month smoking cessation was calculated as: the value at the end of study−the value at baseline (randomization). P < 0.05 was considered to be statistically significant.N. S: no significant difference; LDL: low-density lipoprotein; HDL: high-density lipoprotein; CAD: coronary artery disease.
At the end of follow-up, the change of HDL+LDLpool-induced macrophage migration from baseline in CAD-abs was more significant than that in CAD-smo (−9.688 ± 3.877 vs. −0.250 ± 2.279 cells/field, P < 0.0001). It was also more significant than that in NCAD-abs subgroup (−9.688 ± 3.877 vs. −1.412 ± 4.366 cells/field, P < 0.0001) (Figure 3(c) and (d)).
If compared directly with baseline, after three months of smoking cessation, HDL from CAD-abs significantly attenuated LDLpool-induced chemotactic ability (42.8 ± 2.5 vs. 52.5 ± 3.5 cells/field, P < 0.001) of THP-1-derived macrophages (Table 2 and Supplementary Figure 7). Our findings suggested that CAD smokers would benefit more from smoking abstinence concerning the antichemotactic ability of HDL.
HDL-induced cellular NBD cholesterol efflux
At the baseline, the percentage of macrophage NBD cholesterol efflux mediated by HDL in smokers was lower than that in non-smokers (31.49 ± 3.82% vs. 38.64 ± 1.00%, P < 0.001). Also, it was lower in CAD smokers than that in non-CAD smokers (28.09 ± 1.67% vs. 34.71 ± 2.04%, P < 0.001) (Supplementary Figure 8(a)). At the end of follow-up, the changes of HDL-induced cholesterol efflux from baseline were no different among the four study subgroups (Table 2 and Supplementary Figure 8(b)).
Effects of nicotine and carbon monoxide on HDL function
Lag time of HDL+LDLpool, HDL+LDLpool-induced macrophage migration, and HDL-induced cholesterol efflux was used to evaluate the effects of nicotine and carbon monoxide on HDL function. CORM-2 (a release of carbon monoxide compounds) dose- (P = 0.002) and time- (P = 0.0013) dependently reduced the antioxidative ability of HDL (the shortening of HDL+LDLpool lag time). Nicotine dose- (P = 0.007) and time- (P = 0.013) dependently enhanced the antichemotactic ability of HDL (fewer macrophages migration under HDL+LDLpool). No matter dose increased or time changed, both nicotine and CORM-2 had little effects on macrophage cholesterol efflux induced by HDL (Figure 4 and Supplementary Table 2).
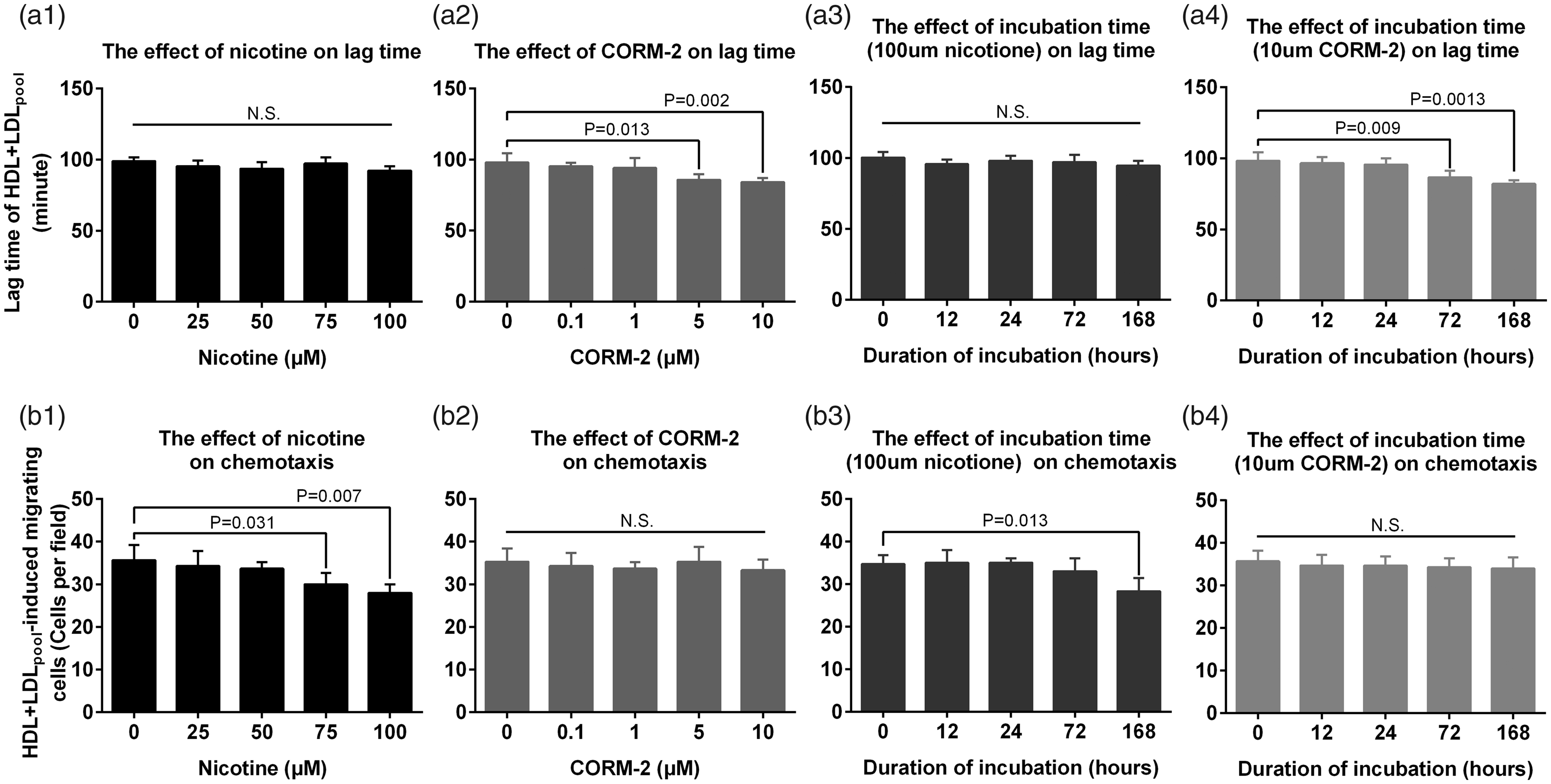
Effects of nicotine and carbon monoxide on HDL function. The ability of HDL against LDL oxidation was evaluated by lagtime of CuSO4-induced oxidation. The impacts of nicotine and CORM-2 on the lagtime of LDLpool+HDL in different concentrations (a1, a2) or at different durations (a3, a4) of incubation. The effects of nicotine and CORM-2 on macrophage chemotaxis in different concentrations (b1, b2) or at different durations (b3, b4) of incubation. CORM-2 (a release of carbon monoxide compounds) dose- and time-dependently reduced the antioxidant ability of HDL (the shortening of lagtime). Nicotine dose- and time-dependently enhanced the antichemotactic ability of HDL (less migrated macrophages). The LDLpool was the mixture of low-density lipoprotein from healthy non-smokers. The LDL and HDL were obtained from the plasma of CAD and non-CAD subjects. P < 0.05 was considered to be statistically significant.N. S: no significant difference; LDL: low-density lipoprotein; HDL: high-density lipoprotein; CAD: coronary artery disease.
Discussion
In this randomized controlled study, we demonstrated, for the first time, that in smoking CAD patients, smoking cessation could significantly improve the antioxidation and anti-inflammatory properties of HDL. Further experiments showed that carbon monoxide in cigarette smoke played roles in interfering the antioxidation property of HDL.
The capacities of HDL inhibiting oxidation, inflammation and promoting reverse cholesterol transport are interrelated. 20 Previous studies revealed that cigarette smoking had the impact on HDL metabolism, function and subfractions distribution,21,22 and the quality of HDL was more important than its quantity. Previous studies mainly focused on healthy smokers. This study, however, demonstrated more severe HDL dysfunction in CAD smokers and the favourable effects of smoking cessation on the recovery of HDL function. Our findings re-emphasized the rationality of smoking cessation as one of the most important measures in the secondary prevention of CAD.
Although Takata et al.’s study 14 showed that smoking cessation improved HDL function in chronic smokers, our results failed to reveal the beneficial effect of smoking abstinence on HDL-induced cholesterol efflux. ATP-binding cassette transporters (ABC) G1 is responsible for cellular cholesterol efflux to mature HDL.23,24 The precise role of ABCG1 in atherosclerosis remains controversy.25–27 Our previous studies27,28 found that downregulation of ABCG1 expression in human as well as in vitro was associated with attenuated macrophage cholesterol efflux, accumulated cellular oxysterols, accelerated cell apoptosis and attenuated development of CAD events. In the study concerning the effects of smoking cessation on macrophage cholesterol efflux, 12 we demonstrated that there was no significant difference in macrophage ABCG1 expression among CAD smokers, non-CAD smokers and non-smokers at baseline. After three months of smoking cessation, ABCG1 protein expression did not increase and ABCG1-mediated cholesterol efflux (to normal HDL) was not improved in both CAD and non-CAD smokers. Combining the finding in the present study, we believed that, in smokers, both ABCG1-mediated cholesterol efflux (to normal HDL) and the ability of smokers' HDL in promoting cholesterol efflux through ABCG1 are not affected by three-month smoking cessation. Since our results in this paper showed that long-term smoking was associated with the attenuated HDL-induced cholesterol efflux, especially in CAD smokers, we speculated that three-month smoking cessation might be too short to reveal the positive effect on HDL function in promoting cholesterol efflux in CAD and non-CAD smokers.
Inflammation plays an important role in the development of atherosclerosis. Cigarette smoking increases the concentrations of inflammatory cytokines and aggravates the chemotaxis of macrophages. 29 The LDL-induced macrophage chemotaxis can be used to reflect the extent of the systemic inflammatory state, while HDL antichemotaxis is an indicator of HDL anti-inflammatory ability. Our results confirmed that smoking promoted LDL chemotaxis and attenuated HDL anti-inflammatory ability, especially in CAD patients. Smoking cessation was an effective measure to improve the anti-inflammatory capacity of HDL in CAD smokers.
Nicotine is the most frequently studied cigarette ingredient, and carbon monoxide is an important toxic product of incomplete combustion of cigarettes. Studies have shown that the nicotine impaired anti-inflammatory effect and carbon monoxide exacerbated oxidative stress.30,31 Nevertheless, using HDL acquired from healthy non-smoking volunteers, we showed that nicotine treatment was associated with the enhanced antichemotactic property of HDL. Our previous experiment 12 also found that nicotine upregulated the ABCA-1 mRNA and protein expression in THP-1-derived macrophages. Also, quite a few papers reported that nicotine had the minor contribution to the initiation or propagation of atherosclerosis.32,33 Some studies even revealed that nicotine had anti-inflammatory property, which was mediated by cholinergic pathway. 34 Therefore, the exact role of nicotine on HDL function and atherosclerosis needs to be studied further.
Previous studies showed that carbon monoxide was associated with atherosclerotic cardiovascular disease.35,36 The impairment of the antioxidative function of HDL caused by carbon monoxide in our experiment was in line with the findings in our in vivo study and enriched, from the perspective of HDL dysfunction, the atherogenic mechanism of cigarette smoking. There are thousands of toxic components in cigarettes smoke, and which of them would play roles in lipid metabolism, and cholesterol deposition in vascular wall need to be investigated in future.
This study had several limitations. Firstly, the three-month duration of the follow-up might be too short to observe the full effects of smoking abstinence on HDL function. For instance, recently, Mandraffin et al. 37 showed an improvement of pro-atherogenic profile and biglycan expression after one year of smoke abstinence. Even so, we did acquire some meaningful findings that suggested smoking abstinence improved HDL function. A longer observation time could allow a better evaluation of overtime changes, including vascular and endothelial functions, which really deserve to be done in future. Secondly, a certain number of confounding factors might have impacts on the results of this study, such as diet, duration of smoking, number of cigarettes smoked per day and time-interval between last cigarette smoking and blood sampling. The strict inclusion and exclusion criteria, blood sampling stipulation, parallel controlled design, urine cotinine and carbon monoxide in expired air monitoring were carried out to minimize the possible bias as best as we can. Thirdly, drugs used in the treatment of CAD, such as aspirin, statins, renin-angiotensin system inhibitors, beta-blockers, and so on might affect the study parameters. We did our best to enrol stable CAD patients, set parallel controlled group and maintain treatment drugs and dosage unchanged throughout the study. Lastly, only two tobacco components (nicotine and carbon monoxide) were measured in the in vitro study. Had we performed more experiments concerning other components of cigarette smoke, we would have more important new findings.
In conclusion, cigarette smoking impairs the antioxidative and antichemotactic effects of HDL and attenuates the HDL-induced cellular cholesterol efflux, especially in CAD patients. Smoking cessation is an efficient way to improve the function of HDL in CAD smokers. Our findings re-emphasize the importance of smoking cessation in the secondary prevention of CAD.
Supplemental Material
Supplemental material for The effects of cigarette smoking and smoking cessation on high-density lipoprotein functions: implications for coronary artery disease
Supplemental material for The effects of cigarette smoking and smoking cessation on high-density lipoprotein functions: implications for coronary artery disease by Hong-Ying Chen, Shi-Cheng Li, Lian-Feng Chen, Wei Wang, Yu Wang and Xiao-Wei Yan in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
Part of this work was performed in Institute of Basic Medical Sciences (IBMS) of Chinese Academy of Medical Sciences.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This present study was supported by grants from the Pfizer Inc., NY, USA (grant number: WS554487).
Ethical approval
Human Ethics Committee of Peking Union Medical College Hospital (PUMCH, approval no. S262).
Guarantor
X-WY.
Contributorship
H-YC and S-CL contributed equally to this article. The study designed and prepared by XWY and H-YC. H-YC, L-FC, WW and YW performed the collection of samples and experiments. H-YC and S-CL performed the analyses. H-YC, S-CL and X-WY interpreted the results and wrote the manuscript. All the authors were involved in discussions and commented on the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
