Abstract
Objectives
Cholesterol efflux capacity (CEC), an atheroprotective function of high-density lipoprotein, is expected to be a potential biomarker for cardiovascular disease. However, CEC has not been widely introduced for application in clinical laboratories because of the complexity of the conventional CEC assay using cells and radioactive materials. Previously, we developed a novel CEC assay using immobilized liposome-bound gel beads (ILG), which solves these issues. We aimed to confirm the validation and further improve the ILG method for application in the clinical setting.
Methods
Cholesterol efflux capacity values by the ILG method assayed for shorter incubation time (4 h) were compared to those assayed for 16 h (our previous ILG method). To investigate a reference material that can correct the variation between ILG manufacturing lots, bovine serum albumin, human gamma-globulins, and globulin complexes were evaluated. CEC values were also estimated in plasmas obtained with different anticoagulants, serum treated with freeze-thaw cycles, and serum mixed with several interference substances.
Results
The CEC of 4- and 16-h incubation times were well correlated. Globulin complexes may be used as a reference material. Plasma can be used as the specimen. The serum and stored temperature of the specimen did not largely affect CEC. Hemoglobin and chyle did not have an effect on CEC, whereas high-bilirubin serum showed elevated CEC. The effect of bilirubin was nearly canceled by subtracting basal fluorescence intensity.
Conclusions
Present ILG method further fulfills some requirements for application in clinical laboratory. Using this reliable simple method, evaluation for clinical significance of CEC is expected.
Keywords
Introduction
Early detection and prevention of cardiovascular disease (CVD) is a pressing issue because of its asymptomatic development and lethality. Although many novel drugs have been developed and used to prevent atherosclerosis, which causes CVD, prediction and estimation of atherosclerosis development remain unclear due to measuring serum lipid levels. 1
To estimate the risk of CVD, quantification of high-density lipoprotein (HDL) cholesterol (HDL-C) levels has been widely performed for a long time. 2 High-density lipoprotein is known to exert an atheroprotective function in vivo through antioxidative and anti-inflammatory properties to prevent foam cell formation in atherosclerotic plaques and cholesterol efflux capacity (CEC) to diminish atherosclerotic lesions.3–6 Despite its prevalence and well-established evidence, several studies have suggested that HDL-C does not always indicate CVD risk.7,8 Moreover, previous studies showed that too high HDL-C levels due to genetic polymorphisms, 9 pharmaceutical intervention, 10 other disease, 11 and life habit 12 do not necessarily contribute to CVD prevention. Therefore, additional biomarkers have been sought, and CEC is thought to be one of the potential biomarkers for CVD risk. Cholesterol efflux capacity is defined as the capacity of HDL to accept excess cholesterol from foam cells in atherosclerotic lesions. Because CEC has already been reported as a better biomarker than HDL-C, it is expected that CEC may be introduced in clinical laboratories for CVD risk assessment.13,14
One of the reasons why CEC has not been tested in the clinical settings lies in its assay. To date, CEC has been measured primarily using artificially prepared foam cells composed of cultured macrophages and radioactive substances (3H-cholesterol). 15 However, this conventional method is not suitable for application in clinical laboratories because of the lack of simplicity and safety. Recently, we reported a novel CEC assay, called the immobilized liposome-bound gel beads (ILG) method. 16 Immobilized liposome-bound gel beads, which is used as an alternative to foam cells, comprises gel beads and fluorescently labeled cholesterol (BODIPY-cholesterol) instead of macrophages and 3H-labeled cholesterol, respectively. In addition to ILG, using apolipoprotein B (apoB)-depleted serum (BDS) obtained by polyethylene glycol (PEG) treatment, which has already been used instead of HDL as a cholesterol acceptor, this novel method can be completed using a simple process to be conducted in the clinical laboratory. We have already reported its basic properties, good correlation with the conventional method, validity of using BDS in the ILG method, 17 and a detailed comparison with the conventional method using some HDL subclasses. 18
In the present study, we aimed to validate the ILG method in terms of application for the actual measurement in a clinical laboratory.
Subjects and methods
Chemicals
All chemicals were purchased from FUJIFILM Wako Pure Chemical Corporation (Osaka, Japan), unless stated otherwise.
Ethics approval
Normal serum and plasma were obtained from blood samples collected from healthy volunteers who provided written informed consent at Tokyo Medical and Dental University. This study was approved by our institutional research ethics committee (approval no. M2015-546). Human experiments complied with all relevant national regulations and institutional polices were performed in accordance with the tenets of the Declaration of Helsinki.
Polyethylene glycol precipitation
Polyethylene glycol precipitation was performed as described previously.
19
Briefly, 16 μL of 20% PEG (molecular weight 6000) in 200 m
Preparation of ILGs
Immobilized liposome-bound gel beads were prepared as described previously.
17
Briefly, egg lecithin (10.6 mg) and cholesterol (2.3 mg) were dissolved in 12 mL of chloroform, and 30 μL of 0.5 m
Cholesterol efflux assay using ILGs
The cholesterol efflux assay was performed as previously described. 19 Briefly, ILG was uniformly suspended, and an aliquot (100 μL) was dispensed into a 2-mL Eppendorf tube. Next, 150 μL of CA solution (diluted BDS) or Buffer A was added to ILG followed by incubation in the dark at RT for 16 h unless stated otherwise. The final concentration of the CA solution was 2% as a serum. The mixture was then resuspended by vortexing and centrifugation. The fluorescence intensity (FI) of the supernatant was measured (Ex: 485 nm, Em: 538 nm). The values were normalized using BDS from a reference serum evaluated in every measurement, and the ratio was represented as CEC with the exception that “CEC without correction” indicates measured values before such normalization. All samples were assayed in triplicates.
Protein solutions
Protein solutions for the ILG method were prepared at the following indicated final concentrations: bovine serum albumin (BSA, 0–4.5 g/dL); human gamma-globulins containing IgG (98.3%) and IgA (1.7%) (0–1.5 g/dL); and human globulins including alpha- (54%), beta-globulins (36%), and albumin (11%) (0–2.0 g/dL) (Sigma-Aldrich).
Interference of common blood components
Serum used for checking the interference of common blood components was prepared using interference Check A Plus (Sysmex) in accordance with the manufacturer’s protocol. Briefly, interference substances were added to serum samples from three healthy volunteers up to the following final concentrations: free-form bilirubin (0–19.1 mg/dL), conjugated-form bilirubin (0–21.2 mg/dL), hemolytic hemoglobin (0–490 mg/dL), and chyle (0–1610 FTU). B (apoB)-depleted serum was obtained from these samples and diluted to 2% serum, followed by measurement of their FI or their CEC, were measured as described above.
Statistical analysis
Correlation analyses were performed using Pearson’s test. The effects of storage temperature, freezing and thawing, and common blood components on CEC were expressed as mean ± SD and evaluated by one-way analysis of variance with Bonferroni correction. Differences were considered significant at P < 0.05.
Results
Shortening of the measuring period
The 16 h incubation time in the present ILG method is thought to be too long for the test in clinical settings. To complete the assay within the daytime, we tried to shorten the incubation time. First, the time-dependency of CEC was determined using four healthy sera. The result was expressed as the ratio to that of 16 h-incubation (the present method). Cholesterol efflux capacity increased after incubation in a time-dependent manner, but not linearly (Figure 1(a)). However, despite the fact that the measured value from the shorter incubation time was smaller, there was no difference in repeatability in triplicate between each assay at a given incubation time (Supplemental Table 1). Next, to investigate whether the CEC assay at 4 h incubation time can be an alternative to the current assay (16 h incubation time), the CEC of 10 BDS samples from healthy volunteers was measured by both assays. CEC measured in these two conditions was well correlated (Figure 1(b), r = 0.955, P < 0.005). Shortening of measuring time. Cholesterol efflux capacity (CEC) was determined using apolipoprotein B (apoB)-depleted serum (BDS) in four healthy sera with indicated incubation periods (a, 0.5, 1, 2, 4, 6, 8, 16, 24, and 48 h). The result was indicated as the ratio to that of 16 h incubation (the present incubation period). CEC of 10 BDS samples collected from healthy volunteers was measured at conditions of 4 h or 16 h incubation time and compared between both results (b). Correlation was evaluated using Pearson’s test. All samples were measured in triplicate.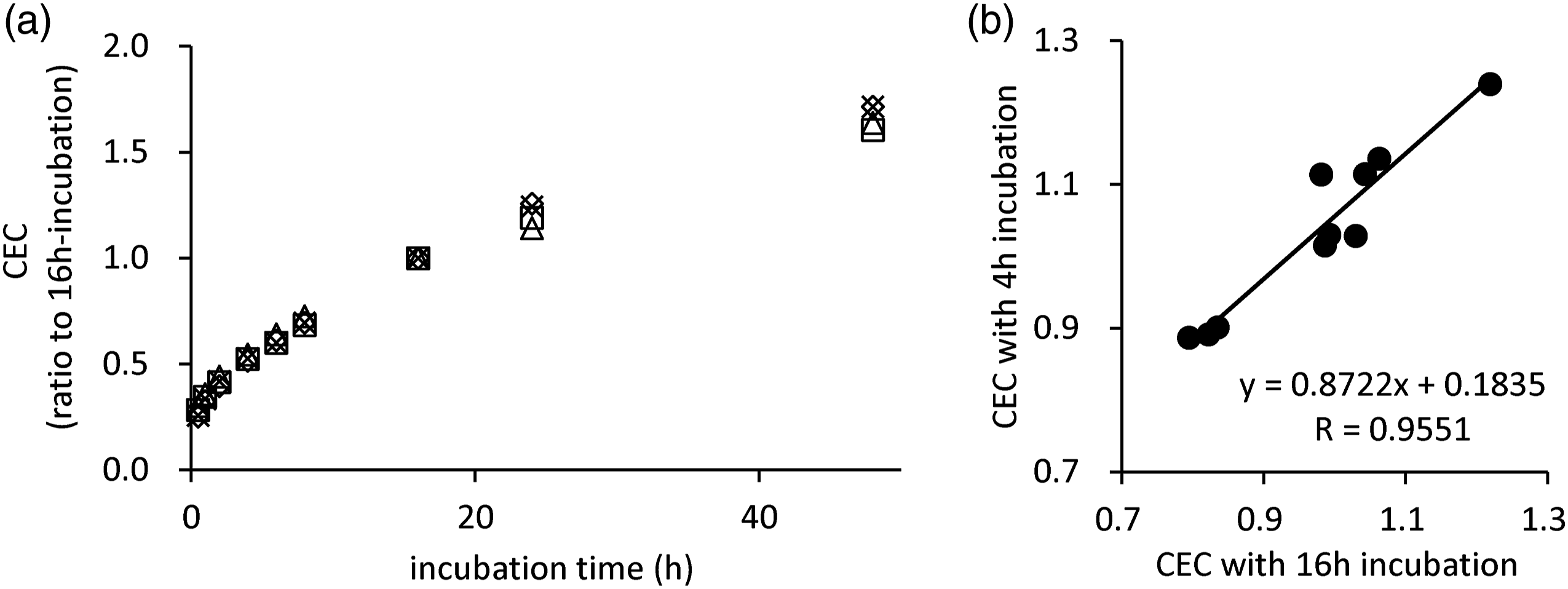
Search for a substitute for reference serum
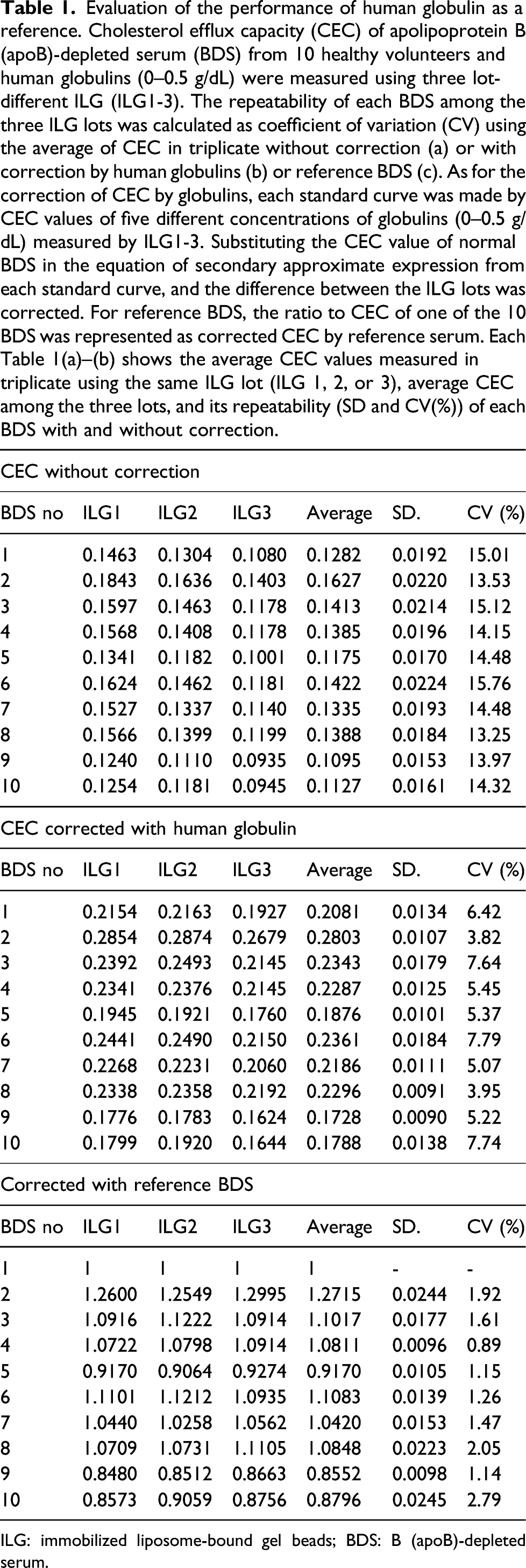
As stated in the Methods section, fluorescence-labeled liposomes are immobilized on the gel by freezing-thawing cycles in the preparation process of ILG, which produces a variation in the manufacturing ILG lot. To investigate the optimal material that can correct the variation among lots, we selected BSA, human gamma-globulin, and human globulins as the candidate. At first, CECs of various concentrations of BSA (0–4.5 g/dL), human gamma-globulin (0–3.0 g/dL), and human globulins (0–1.5 g/dL) were compared with those of BDS (2% as a serum) from three healthy volunteers. Within these ranges, the CECs of BSA and human gamma-globulin were obviously lower than those of normal serum (Figure 2(a) and (b)). In contrast, human globulins had higher CEC, suggesting that human globulins can be used for lot correction (Figure 2(c)). Next, lot variations of the three types of ILGs (ILG1-3) were evaluated by comparing the coefficient of variation (CV) (%) of CEC values in 10 healthy serum samples measured with or without lot correction. Without lot correction, the repeatability of CEC of 10 sera was approximately 15% as CV (Table 1(a)). Then, the performance of the human globulins as a reference was assessed. A standard curve was generated by CEC values of various levels of human globulins (0–0.5 g/dL). In total, three different standard curves were generated with CEC values of the same globulin specimens measured using three lot-different ILGs (ILG1-3). At the same time, CECs of BDS from 10 healthy sera were determined by three lot-different ILGs (Supplemental Figure 1). The equation of secondary approximate expression was calculated from each standard curve (e.g. y = −0.4154x2 + 0.7513x + 0.0037 for ILG1). The CEC corrected by human globulins was calculated by substituting the CEC value of each BDS for y in the above equation. The lot correction by human globulins improved the repeatability to 3.82–7.78% (Table 1(b)). Finally, the correction by reference serum was performed as the present method: the ratio to CEC of one of the 10 BDS (No. 1) indicated corrected CEC. Unexpectedly, the best repeatability was observed when one of the 10 BDSs was used as the reference material with within 3% CV (Table 1(c)). Search for potential reference protein instead of serum. Cholesterol efflux capacity (CEC) of bovine serum albumin (BSA; 0–4.5 g/dL, a), human gamma–globulin (0–3.0 g/dL, b), and human globulin (0–1.5 g/dL, c) were evaluated and compared with those of BDS from three healthy volunteers. All samples were measured in triplicate. Evaluation of the performance of human globulin as a reference. Cholesterol efflux capacity (CEC) of apolipoprotein B (apoB)-depleted serum (BDS) from 10 healthy volunteers and human globulins (0–0.5 g/dL) were measured using three lot-different ILG (ILG1-3). The repeatability of each BDS among the three ILG lots was calculated as coefficient of variation (CV) using the average of CEC in triplicate without correction (a) or with correction by human globulins (b) or reference BDS (c). As for the correction of CEC by globulins, each standard curve was made by CEC values of five different concentrations of globulins (0–0.5 g/dL) measured by ILG1-3. Substituting the CEC value of normal BDS in the equation of secondary approximate expression from each standard curve, and the difference between the ILG lots was corrected. For reference BDS, the ratio to CEC of one of the 10 BDS was represented as corrected CEC by reference serum. Each Table 1(a)–(b) shows the average CEC values measured in triplicate using the same ILG lot (ILG 1, 2, or 3), average CEC among the three lots, and its repeatability (SD and CV(%)) of each BDS with and without correction. ILG: immobilized liposome-bound gel beads; BDS: B (apoB)-depleted serum.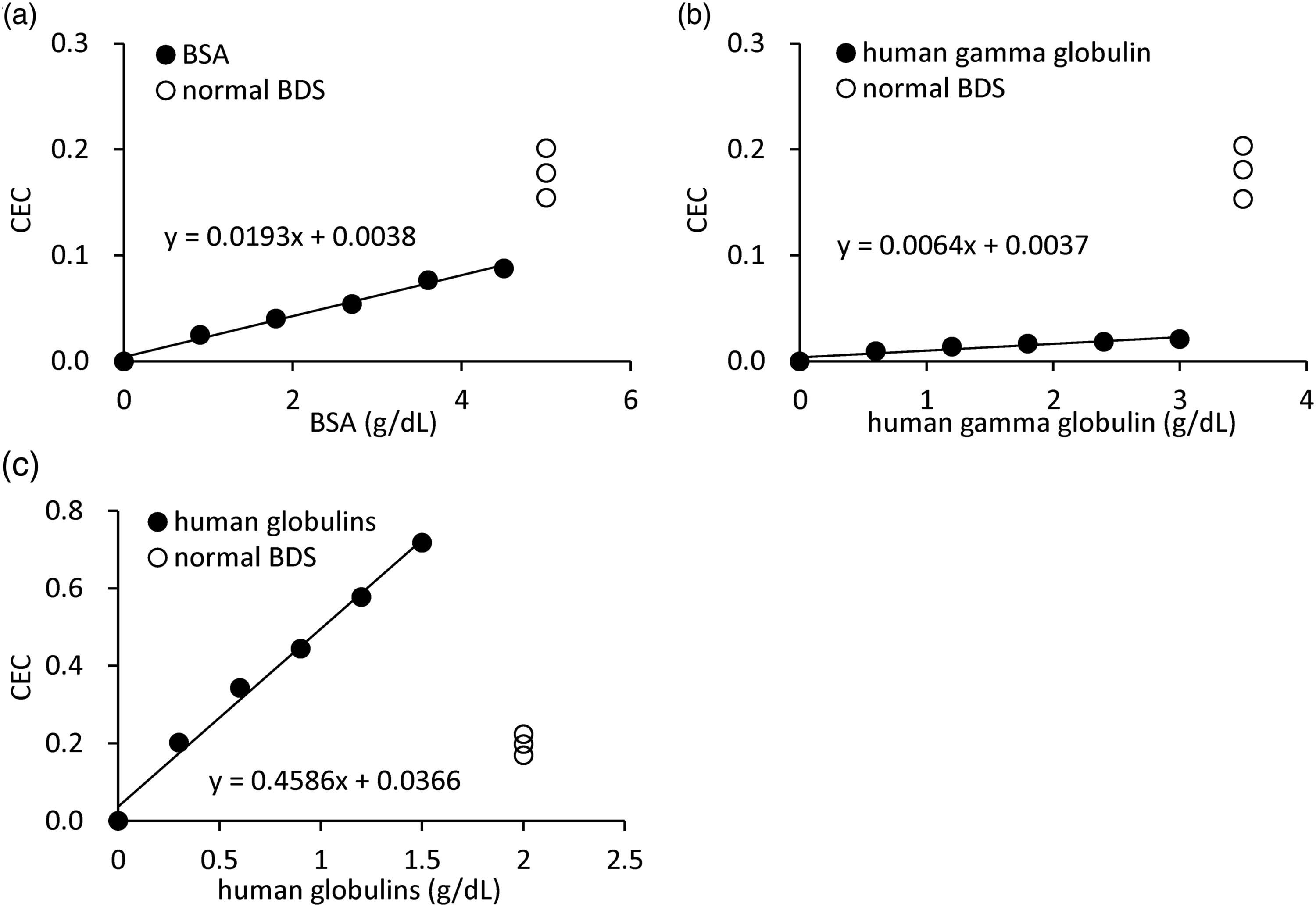
Effect of anticoagulants in blood-collecting tubes
To investigate the effect of various anticoagulants used in routine laboratory testing on CEC values, blood samples were collected from 10 healthy volunteers in three different types of blood collection tubes (EDTA-2K, EDTA-2Na, and lithium heparin), and after the PEG precipitation procedure, the CEC of each sample was compared to that of BDS from the same individual. CECs of all types of plasma were well correlated with those of serum (Figure 3(a)–(c), r = 0.9875, r = 0.9860, and r = 0.9831, respectively, P < 0.005). Furthermore, the average ratio of CEC of the plasma to that of serum was 0.99, 0.97, and 0.98, and no significant difference was confirmed. Effect of anticoagulants in blood collection tubes. Serum and three types of plasmas, whose anticoagulants were EDTA-2K, EDTA-2Na, or lithium heparin, were obtained from the blood samples of 10 healthy volunteers. After the polyethylene glycol precipitation procedure, the cholesterol efflux capacity (CEC) was measured. The correlations between the CEC of serum and that of each type of plasma were determined. ((a) EDTA-2K, r = 0.991, P < 0.005, (b) EDTA-2Na, r = 0.968, P < 0.005, (c) lithium heparin, r = 0.959, P < 0.005). Correlation was evaluated using Pearson’s test.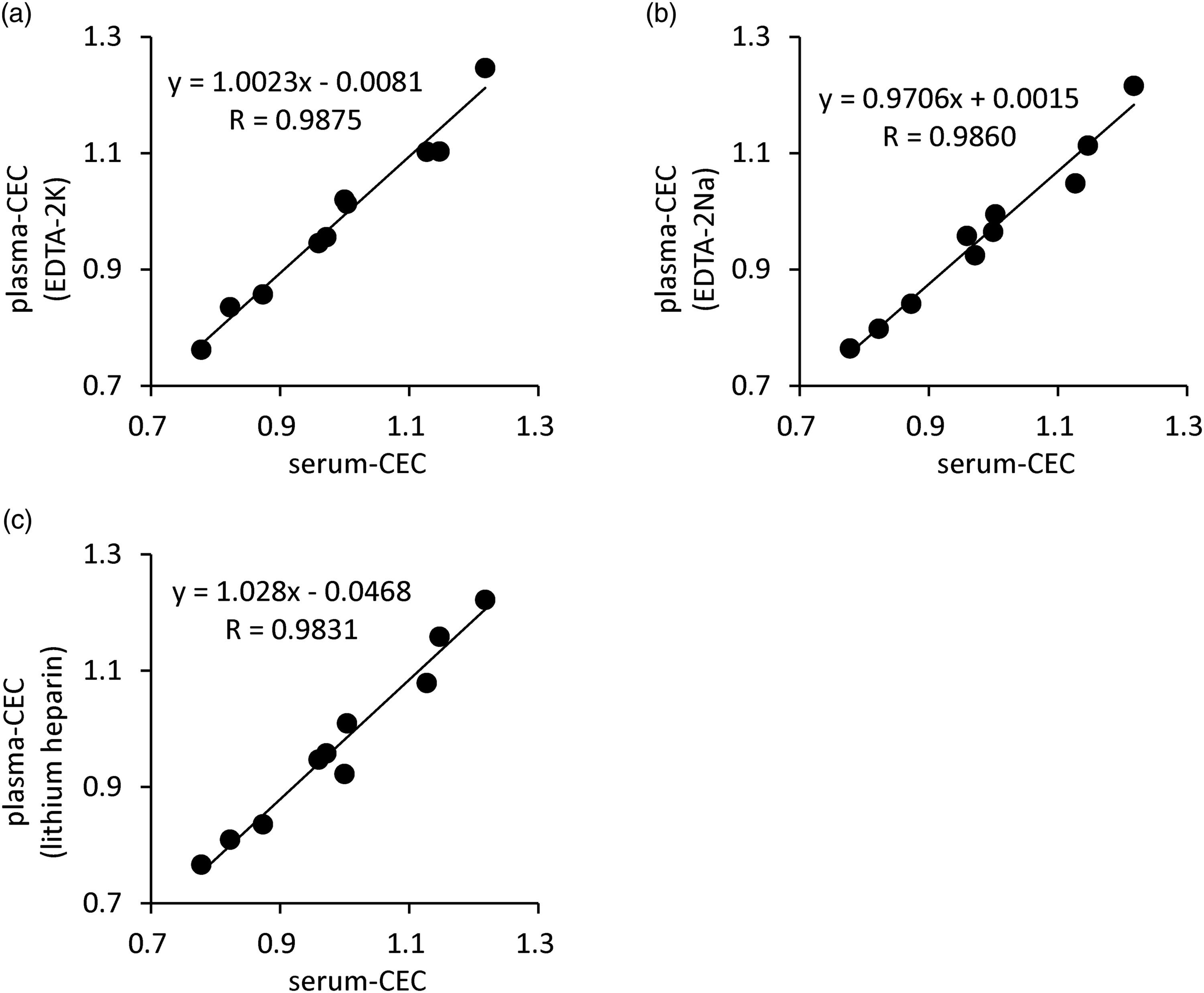
Effect of storage temperature of the serum specimen
Sera were obtained from four healthy volunteers and dispensed into 1.5 mL microcentrifuge tubes. The serum-containing tubes were stored for 8 h at the indicated temperature or at 4°C for the indicated time followed by storage at −80°C for the remaining time until the total storage time reached up to 5 days. Then, the CEC values of BDSs obtained from the stored sera were measured using the ILG method. No significant difference was caused by both the 8 h storage temperature (Figure 4(a)) and storage at 4°C and −80°C (Figure 4(b)). Effect of storage temperature and freezing and thawing of serum specimen. Serum was obtained from four healthy volunteers and dispensed. They were stored for 8 h in the indicated temperature (a), stored at 4°C for the indicated period, and stored at −80°C until the total storage period reached 5 days (b), or frozen at −80°C and thawed at 4°C for the indicated times (c). Apolipoprotein B (apoB)-depleted serum (BDSs) were obtained from these sera and their cholesterol efflux capacity (CEC) were measured. All samples were assayed in triplicates. Values are presented as mean ± SD (n = 4).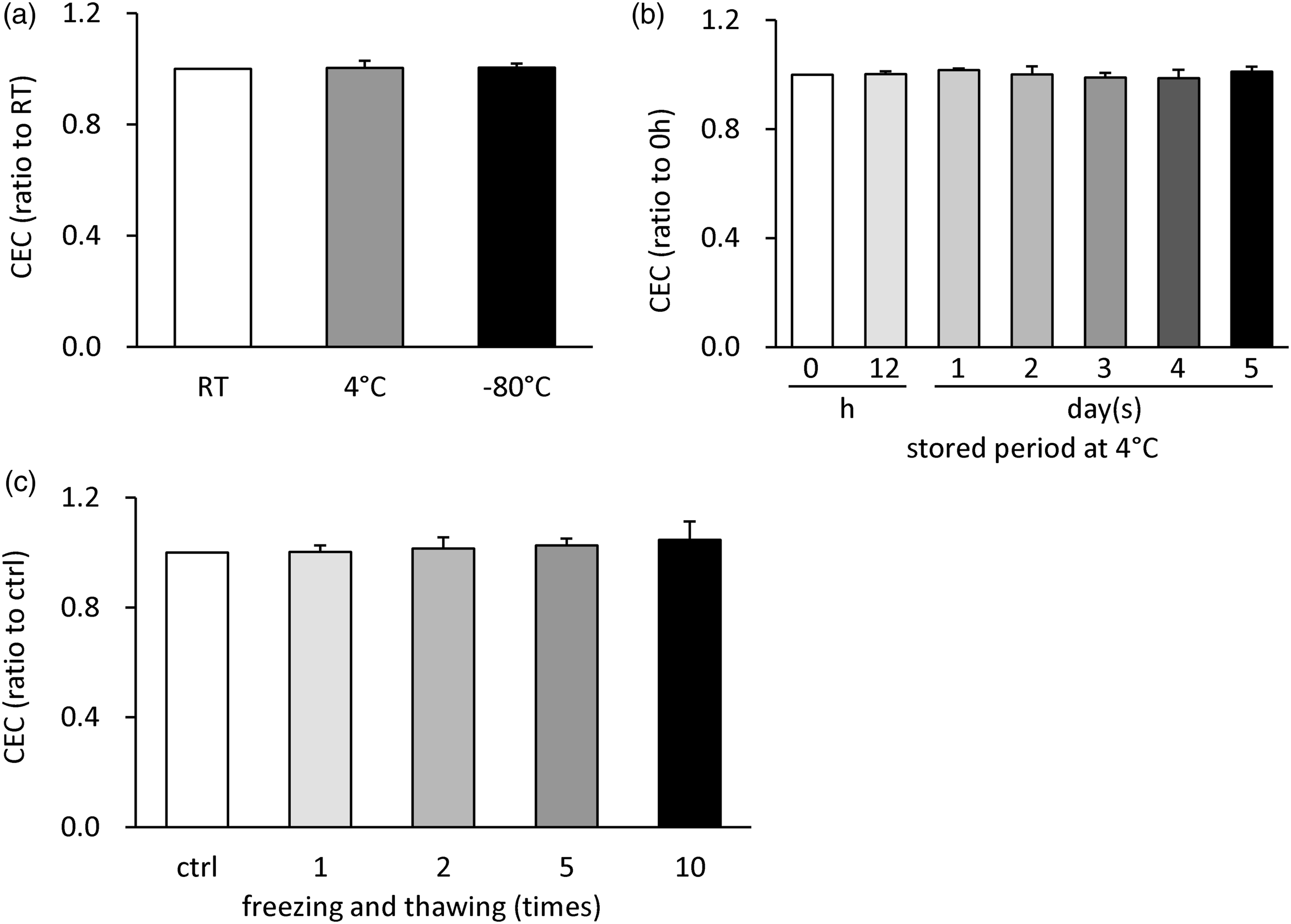
Effect of freezing and thawing of the serum specimen
Similarly, sera obtained from four healthy volunteers were dispensed into tubes. After the indicated number of freeze-thaw cycles (frozen at −80°C and thawed at 4°C), the CECs of BDSs from the treated sera were evaluated using the ILG method. No difference was observed by the time of freezing and thawing (Figure 4(c)).
Interference of common blood components on CEC
The interference of common blood components with CEC measurement was assessed by adding interfering substances to the serum samples. Four interference substances, free-form bilirubin, conjugated-form bilirubin, hemoglobin, and chyle were used. Free- and conjugated-type bilirubin had a great effect on CEC measurement (CEC was approximately doubled by the addition of 20 mg/dL bilirubin, Figure 5(a) and (b)), whereas hemoglobin and chyle did not have an effect on it (Supplementary Figure 2(a) and (b)). Interference of common blood components on cholesterol efflux capacity (CEC). A series of serum samples containing different concentrations of interfering substances were prepared by mixing the interfering substances obtained from Interference Check A (Sysmex) and healthy serum according to the attached manual. Then, apolipoprotein B (apoB)-depleted serum (BDS) was obtained from these samples followed by CEC measurement (Applied interference substances were free-form bilirubin (a) and conjugated-form bilirubin (b)). The results are indicated as the ratio to that of the sample that did not contain additive interfering substances. Corrected CEC was calculated by subtracting the basal fluorescence intensity measured using BODIPY-free ILG from CEC (c, d). Cholesterol efflux capacity (CEC) of 10 healthy volunteers with or without basal FI was compared between the groups. Each basal FI was measured using BODIPY-free ILG (E, r = 0.8871, P < 0.005). All samples were assayed in triplicates. Values are presented as mean ± SD (n = 4, A-D) and correlation was evaluated using Pearson’s test (e). *P < 0.05, **P < 0.01.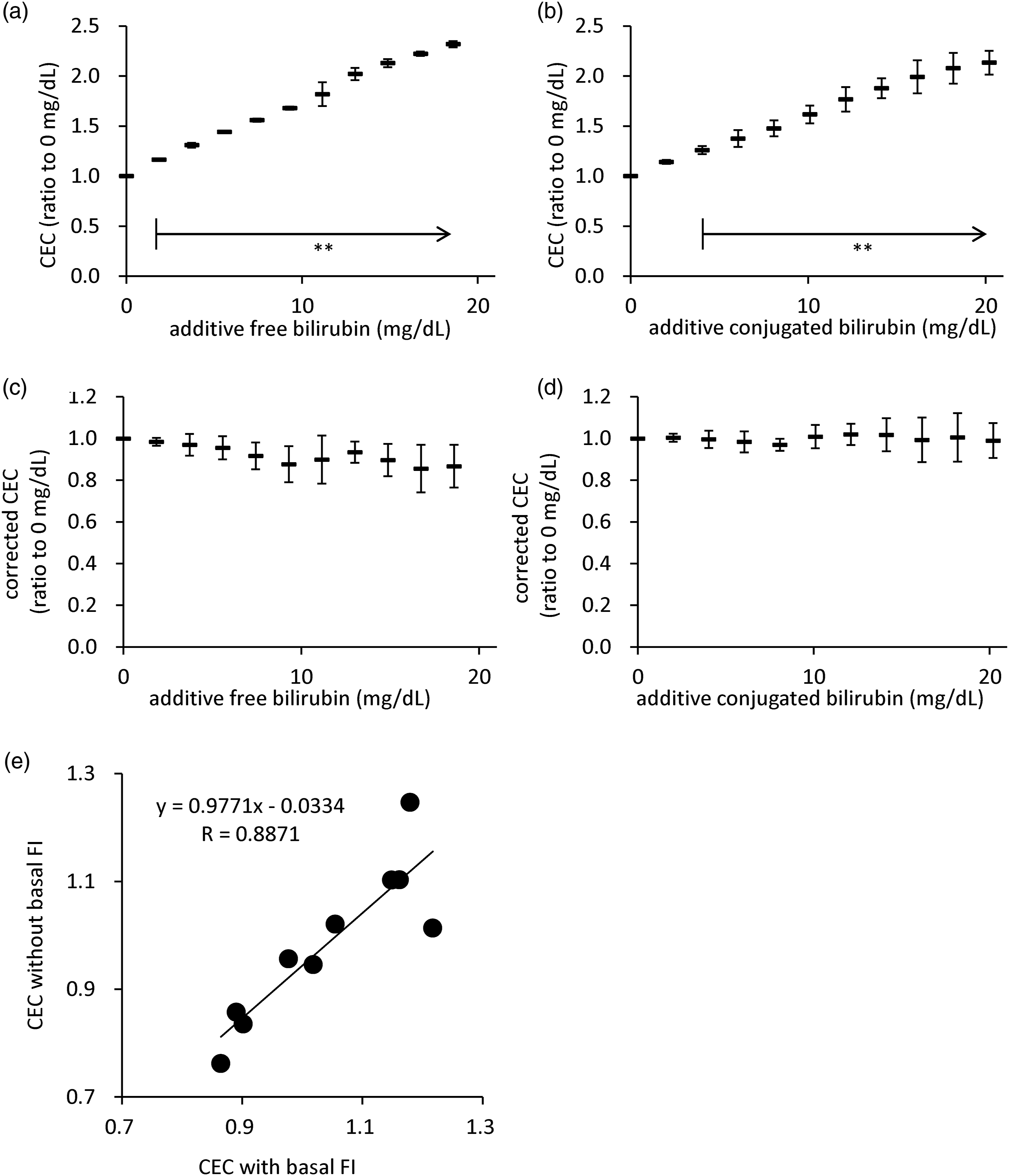
To investigate the cause of high CEC by bilirubin, the interference of bilirubin with FI was also assessed. The serum samples were prepared in the same way as those for the evaluation of CEC, and their FI was measured. Free- and conjugated-type bilirubin had a great effect on CEC measurement (20 mg/dL of both types of bilirubin made FI approximately 20 higher, Supplementary Figure 2(c) and (d)). To diminish the effect of basal FI from bilirubin in the serum, the CEC was corrected by subtracting the basal FI measured using BODIPY-free ILG. The correction by basal FI decreased the CEC of both types of high-bilirubin serum to almost the same level as that without additive bilirubin serum (Figure 5(c) and (d), no significance).
Subtraction of basal FI in healthy subjects
These large influences of bilirubin indicate that bilirubin, despite being in the normal range, may also affect CEC values. To confirm this hypothesis, the CEC values of BDS assessed from 10 healthy volunteers with and without basal FI were compared. Basal FI was measured using BODIPY-free ILG. The values showed statistical correlations, but some showed apparent deviation from the correlation. (Figure 5(e), r = 0.8871, P < 0.005).
Discussion
The high mortality rate of CVD is a critical issue; therefore, more early and accurate evaluation of CVD risk is required. Although CEC is thought to be one of the potential biomarker for CVD, its measurement in clinical settings has not been put into practice yet by some reasons. One of the reasons is an unsuitable assay, in particular complicated, time-consuming, and unsafe aspects. Our novel ILG method was developed as a simpler, shorter time, and safer assay for CEC than the conventional method. 16 However, this method remained partially improved to adapt to clinical settings. In previous studies, we have examined the validity and usefulness of ILG method by various aspects: stability of the ILG, correction of measured values by the lot-different ILG using reference serum, strong correlation with the conventional method and measurement range of CEC for human serum. 16 In addition, PEG precipitation method was suitable as pretreatment procedure of patient serum samples in ILG method for evaluating HDL function. 17 Moreover, ILG method was greatly superior to the conventional method in aspects of both intra- and inter-assay repeatability, one of the demerits in the conventional method, and its CEC values of major HDL subclasses, HDL2 and HDL3, also corresponded to those from the conventional method. 18 This study is positioned as one of the series of such studies to further make practical use of ILG method in clinical laboratory.
In the ILG method, ILG and BDS were incubated to move BODIPY-cholesterol from the ILG to HDL in BDS for 16 h, which is too long as turnaround time (TAT) to be measured in clinical settings. To complete the assay within daytime, we tried to shorten the incubation time. Consequently, the CEC values determined by the shorter incubation time (up to half an hour) were still evaluable with sufficient repeatability (<5% CV). Moreover, CECs measured with 4 h and 16 h of incubation time were compared. A good correlation between these CECs indicated that the ILG assay is feasible with a shorter TAT.
To correct the variation among ILG lots, reference serum was measured in every measurement and CEC values of samples were represented as ratio to the measured value of the reference serum. However, reference serum is not ideal for clinical use because of its operational complexity. We considered that some types of protein can be used as alternative references and tried three types of proteins, namely, BSA, human gamma-globulin, and human globulins, as the first step to search for potential reference materials. The former two proteins did not have sufficient CEC values to be used as a reference for normal serum, while human globulins had more CEC than that of BDS from healthy subjects. Then, we tried to use human globulins instead of the reference serum for variation correction. Human globulins seemed to correct the difference among ILG lots; however, not all repeatability was less than 5% as CV. However, correction by the reference serum, which is currently in use, adjusted all repeatability within 3% as CV. Based on these results, the best reference was thought to be serum, but the usability of human globulins as a reference was not completely denied. Because the human globulins used in the present study were a mixture of multiple proteins, searching for a specific protein that is appropriate for reference may be necessary to obtain stable results. It is important to consider the suitability of the method to be used in clinical settings and the influence on CVD risk evaluation to decide what agent should be used for reference.
We examined the validity of the ILG method from various aspects in anticipation that serum is used as a specimen for CEC measurement. However, to apply the ILG method to clinical settings, the use of other specimens such as plasma also needs to be considered. There was no difference among CECs whose specimens were serum or plasma. The result indicates that plasma can also be used as a specimen for CEC.
In clinical laboratories at hospitals, unlike in research laboratories, there are some cases in which serum specimens are not frozen immediately after collection. We assessed whether the storage temperature influences the CEC value assuming the following two situations. One aspect for confirmation was the influence of storage temperature, RT, 4°C, and −80°C for 8 h, assuming that the specimens were left until after daytime operation. Another aspect for confirmation was the influence of storage temperature, 4°C and −80°C, for 5 days, assuming that the CEC assay was conducted once on the weekend. There was no difference among different storage temperatures, indicating that serum samples can be left at RT until the end of daytime operation and stored at 4°C until the weekend. To store serum specimens by freezing, the influence of freezing and thawing needs to be assessed. Although no significant difference was observed for many times of freezing-and-thawing cycles, a slight increase in CEC was observed along with the time of freezing and thawing in two of four serums (the ratios of ten-times freezing and thawing results to control in unchanged samples were 0.99 and 0.99, and those in the changed samples were 1.11 and 1.10, respectively). Smaller freezing and thawing times are thought to be the best conditions.
In clinical settings, measurement results sometimes do not reflect the actual conditions in vivo because it is largely affected by other components in the serum. Indeed, FI measurement in the CEC assay by the ILG method was largely affected by both free- and conjugated-form bilirubin, whereas hemoglobin and chyle did not interfere with it. Specifically, 20 mg/dL bilirubin addition increased FI to approximately 20 times. In fact, CEC was also affected by both free- and conjugated-form bilirubin, while no interference by hemoglobin and chyle was detected. The large influence of bilirubin in the serum indicates that bilirubin, even within the normal range, may also affect CEC. Using BODIPY-free ILG, subtraction of basal FI (FI of sample itself) from CEC reduced the effect of bilirubin considerably and produced good correlation of CEC with or without basal FI in healthy subjects. However, some values showed apparent deviation from the regression line. This suggests that it may be difficult to ignore the influence of basal FI even when the serum samples are collected from subjects with normal bilirubin levels. Further improvement of the present ILG method might be required.
The limitation of this study is that these examinations were conducted using specimens from healthy volunteers. The results examined in the present study may not always apply in clinical settings because specimens from patients often contain a particular substance at an extremely high level.
Conclusion
In summary, present ILG method seemed to fulfill some requirements to be conducted in clinical laboratory, such as short measurement time, availability of other blood specimens in addition to serum, the stability in the way of storage at clinical laboratories, and so on. To adapt novel methods to clinical settings, various confirmations, which do not require research use, must be considered. For example, short measurement time, adequately low cost, stability in the way of storage in clinical laboratories, interference from other components in serum, and so on are required. This study is performed in order to improve ILG method to application in clinical settings toward our final goal of CEC in use as a CVD risk marker. Although some issues remain to be solved, by using this reliable simple method, evaluation of the clinical significance of CEC as a biomarker for CVD is expected in the future.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632211054406 – Supplemental Material for Novel cholesterol efflux assay using immobilized liposome-bound gel beads: Confirmation and improvement for application in clinical laboratory
Supplemental Material, sj-pdf-1-acb-10.1177_00045632211054406 for Novel cholesterol efflux assay using immobilized liposome-bound gel beads: Confirmation and improvement for application in clinical laboratory by Yuna Horiuchi, Shao-Jui Lai, Takahiro Kameda, Minoru Tozuka and Ryunosuke Ohkawa in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for the Japan Society for the Promotion of Science Research Fellow [grant number: 20J13446 to Y.H.] and Grant-in-Aid for Scientific Research (C) [grant number: 19K07932 to R.O.] from the Japan Society for the Promotion of Science, and Medical Research Encouragement Prize from the Kondo Kinen Medical Foundation (R.O.).
Ethical approval
Approved by Graduate School of Medical and Dental Sciences research ethics committee (approval no. M2015-546).
Guarantor
RO.
Contributorship
YH, RO, and MT designed the study. RO and MT supervised the study. YH, and SJL carried out the research. YH and RO analyzed the data. YH and RO wrote the manuscript. TK and TM reviewed drafts of the manuscript. All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
