Abstract
Iodine deficiency is a significant global health concern, and the single greatest cause of preventable cognitive impairment. It is also a growing public health concern in the UK particularly among pregnant women. Biomarkers such as urinary iodine concentration have clear utility in epidemiological studies to investigate population-level iodine status, but determination of iodine status in individuals is much more problematic with current assays. This article reviews the available biomarkers of iodine status and their relative utility at the level of both populations and individuals for the investigation of iodine deficiency and iodine excess.
Introduction
Iodine is an essential trace element. It is a constituent of the thyroid hormones thyroxine (T4) and tri-iodothyronine (T3) which are themselves essential for life. Environmental sources of iodine include soil and sea water. Iodide ions are present in sea water at a concentration of approximately 50 μg/L, 1 and they can become oxidized resulting in elemental iodine which evaporates due to its high volatility. Following precipitation, iodine is then returned to the soil in rain. In some regions, this cycle is inadequate leading to iodine deficiency in the soil, and crops grown in these regions may also be deficient in iodine. 2 Iodine deficiency can subsequently develop in the populations that live in these regions, and some countries have sought to address this by artificially adding iodine back into the food chain through salt iodization programmes. 3
Approximately 90% of ingested iodide ions are absorbed in the stomach and duodenum. Once in the circulation, a sodium/iodide symporter transports iodide into the thyroid gland, establishing a concentration gradient of between 20 and 50 times between thyroid tissue and plasma. 4 Iodine then re-enters the circulation in the form of T4 and T3 released by the thyroid, and iodine forms the majority of the weight of both. This circulating iodine can then be taken up by the thyroid again or excreted by the kidneys, and the fate of approximately 90% of absorbed iodine is ultimately renal excretion.
The World Health Organization (WHO) issue recommended daily intake values for iodine stratified by age with allowances made for pregnancy and lactation. These are summarized in Table 1.
Recommended daily intake for iodine.
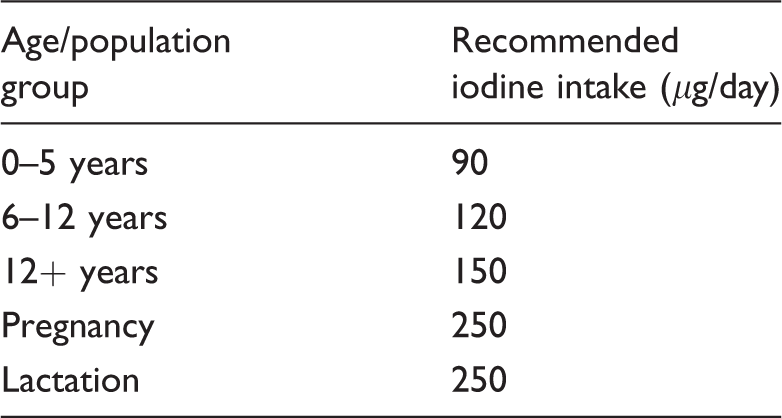
Note: Values taken from WHO 2001. 2
When dietary requirements for iodine are not met, synthesis of thyroid hormones may become impaired resulting in hypothyroidism. If this occurs during pregnancy, growth and development for the offspring may be impaired.5–7 Iodine deficiency is a clinical issue of great worldwide importance, such that the WHO considers it to be the most important global preventable cause of brain damage and estimates that approximately two billion individuals have an insufficient iodine intake. 2 The problem of iodine deficiency is not, however, unique to developing nations, and evidence is accumulating that it is a significant clinical concern in the United Kingdom. There has been particular concern that this may be an issue in pregnant women, 8 and more generally in those of child-bearing age.9,10 Additionally, a recent study of 810 school girls from nine different cities across the UK found evidence for marked iodine deficiency felt to be of significant public health importance. 11 Then in 2013, the landmark Avon Longitudinal Study of Parents and Children (ALSPAC) was published which investigated this issue further by examining mother and child pairs from the UK in a longitudinal study, demonstrating a link between inadequate iodine status in first trimester pregnancy and lower IQ and reading ability in the offspring at age 9. 12
Given the above, there is clear clinical need for diagnostic tests that can assess iodine status. At the same time, recent evidence of iodine deficiency in the UK has led to renewed interest in iodine status, and clinical laboratories may start to receive more enquiries from clinicians regarding how to assess this. Much of the evidence in the literature comes from studies using methods designed to look at iodine status at a population level, not necessarily addressing what a result may mean for an individual. It is important to consider the differences in what can be learned from the use of biomarkers of iodine status in populations as opposed to individuals. However, interpreting such results is fraught with difficulty, and there are definite limitations in what can and cannot be inferred from the use of such biomarkers.
Biomarkers of iodine status
There are a number of potential biomarkers available for the assessment of iodine status including urine iodine (UI), thyroid-stimulating hormone (TSH), thyroglobulin and the thyroid hormones T3 and T4.
TSH
TSH is a direct marker of thyroid function, and thus could be considered to be in some way reflective of iodine status. It is used as part of newborn screening in many developed countries in order to detect cases of congenital hypothyroidism. 13 Neonates have higher iodine turnover in the thyroid than children and adults, and neonatal TSH should be a sensitive indicator of population iodine deficiency; however, when this has been scrutinized, it has been concluded that the rise in TSH seen is not sufficient for this to be a useful marker. 14 Additionally, in children and adults, TSH concentrations may rise slightly in iodine deficiency, but values commonly remain within the reference interval, rendering it a relatively insensitive marker.
Thyroid hormones
The main bioactive thyroid hormones comprise T4 and T3, and they represent a direct reflection of thyroid function. However, they are poor biomarkers of iodine status at both a population and an individual level. Changes in thyroid hormone concentration in relation to iodine status commonly occur within the reference interval, and they lack the sensitivity to be truly reflective of iodine status in all but the most iodine-deficient areas. 15
Thyroglobulin
Thyroglobulin is an iodoglycoprotein synthesized in the follicular cells of the thyroid in response to TSH and acts as the precursor to the thyroid hormones. Its main clinical utility is as a tumour marker in patients who have undergone total thyroidectomy for differentiated thyroid cancer. Thyroglobulin concentration broadly reflects overall thyroid cell mass, and thus it has been suggested that it may be useful as a biomarker of population iodine status in areas with endemic goitre and indeed has been shown to decrease at a population level after the implementation of a salt iodization initiative. 16 A more recent study used a dried blood spot method to examine thyroglobulin concentrations and UI concentration in over 2500 school age children from 12 different countries. 17 Using the WHO-recommended UI concentration cut-offs for possible iodine deficiency, sufficiency and excess and comparing this to thyroglobulin concentrations, the authors demonstrated a U-shaped curve with increased thyroglobulin in those with low UI concentration, decreasing in those with an ‘iodine-sufficient’ UI concentration and increasing again in those with increased UI concentrations suggestive of excess intake. They concluded from this that thyroglobulin was a sensitive marker of decreased and excess iodine intake. However, there was very wide scatter when plotting UI concentration against thyroglobulin concentration precluding thyroglobulin as a test that could reflect iodine status in an individual. Further, thyroglobulin concentrations were similar at the iodine-deficient and iodine-sufficient UI concentrations meaning that thyroglobulin lacks diagnostic specificity.
Urine iodine
UI is the most widely studied and used biomarker for the assessment of iodine status. Dietary iodine is cleared from the circulation by either the thyroid or the kidneys. While thyroid clearance of iodine varies depending on the level of iodine intake and can increase the fraction of absorbed iodine taken up into the thyroid from less than 10% in times of plenty to over 80% in times of iodine deficiency, 18 renal excretion of iodine remains fairly constant as a proportion of that which has been absorbed. 15 The literature contains studies outlining many different methodologies for measuring UIC including gas chromatography mass spectrometry, catalytic spectrophotometry, atomic absorption spectrometry, inductively coupled plasma atomic emission spectrometry, inductively coupled plasma mass spectrometry (ICP-MS), capillary electrophoresis and UV-Vis spectrophotometry. 19 Worldwide, two main methodologies have become dominant and they occupy opposite ends of the sophistication spectrum: spectrophotometric methods based on the Sandell-Kolthoff reaction and ICP-MS methods.
The Sandell-Kolthoff reaction was first described in 1937,
20
and methods based around the reaction are likely to represent the commonest way to quantify urine iodine worldwide. The reaction itself is well described and involves the reduction of yellow Ce(IV) by As (III), resulting in the formation of colourless Ce(III). This reaction normally takes place very slowly but is catalysed by trace quantities of iodide as shown below
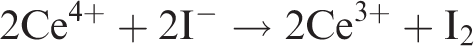
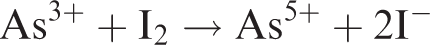
The rate of disappearance of the yellow colour can be measured and will be proportional to the urine iodine concentration of the sample, and absorbance is usually measured at 405 to 420 nm. Before the reaction can take place, however, it is necessary to release bound iodine from urine excretory compounds and remove potential interfering compounds by using a digestion step. Interfering compounds include thiocyanate, metal ions, such as mercury and silver and other compounds that can readily become oxidized such as nitrite, ascorbic acid and ferrous iron.19,21 In 1951, a method was published that utilized an alkaline digestion procedure combined with ashing, and this was widely used thereafter. 22 However, the ashing procedure is now known to result in significant loss of analyte leading to the possibility of falsely low results.23,24 Chloric acid has also been used and was shown to have good diagnostic performance and effective removal of interfering substances 25 ; however it is a hazardous substance requiring many safety precautions including an explosion proof fume hood. Ammonium persulphate digestion is now widely used for this purpose, having been initially demonstrated in 1998. 26 This is a safer, more convenient method and performs similarly well to chloric acid-based techniques. Such techniques can utilize a microplate format to minimize the production of toxic waste products and increase sample throughput. 24
ICP-MS methods are a more recent development, and now represent the methodology of choice for determination of iodine in all biological samples. An ICP-MS method for measuring UI concentration was first reported in 1990 using a prototype mass spectrometry instrument, and the authors reported a limit of detection of 1.6 μg/L (1.3 μmol/L). 27 This method was then modified and updated for use in the National Health and Nutrition Examination Survery (NHANES) in 2000. 28 Two separate groups have undertaken method comparison studies and have reported good agreement between this ICP-MS method and the more traditional Sandell-Kolthoff-based methods,29,30 although Macours et al. found that results generated by ICP-MS were slightly higher.
In general terms, ICP-MS is now in widespread routine clinical use for trace element analysis, and a typical analytical platform consists of a number of components; a sample introduction system, ICP torch, interface, vacuum system, ion optics, collision cell, mass spectrometer, detector and data handling system. Temperatures of up to 6000°C are generated by the ICP torch in order to convert the atoms of the element of interest into positive ions which are then separated in a quadrupole mass analyser on the basis of their mass to charge ratio, and then detected. Iodine atoms have a relatively high first ionization potential to reach I+, and consequently, the iodine present in the sample is only partially ionized, but despite this, ICP-MS techniques are able to achieve superior sensitivity than other methods, as well as a greatly increased linear dynamic range. 29 Mass spectrometry can also be used as a detector in conjunction with liquid or ion chromatography to provide information on iodine species.31,32 As with all trace element analysis, measurement of iodine is prone to contamination from a variety of sources including urine dipsticks as was recently reported. 33
Urine iodine in populations
UI concentration has long been used as the standard means with which to assess the iodine status within a given population. 34 Prior to this, studies had focused on examining the prevalence of goitre as a surrogate biomarker of population iodine status. 35
Urine iodine is an accurate reflection of recent dietary intake of iodine as approximately 92% of ingested iodine is absorbed from the gastrointestinal tract, and in adults who are healthy and replete in iodine over 90% of absorbed iodine is excreted in the urine.36,37 In the various population studies that have assessed use of UI concentration, spot urines have generally been used as opposed to 24-h collections or results expressed as a ratio of iodine/creatinine. This is generally due to difficulties in obtaining good quality 24-h urine samples in field studies and the higher variability of creatinine excretion seen in the often protein-malnourished populations that would be assessed in such studies. 38 It is important to consider that a relatively large sample size is required in studies of population iodine status in order to account for high inter- and intraindividual variation in UI concentration.39,40
A landmark study was published in 1970 which presented data from 186 areas and over 3000 people with goitres in Central America. 41 The authors measured urine iodine and creatinine, and estimated the 24-h excretion of iodine based on these values. They observed that goitre was endemic (defined as a prevalence of greater than 10%) in all areas where the mean estimated 24-h urine iodine excretion was less than 25 μg, in most of the areas where it was 25–49 μg, in a third of areas where it was 50–99 μg and in no areas where is was greater than 100 μg. It is upon these observations that the WHO bases their cut-off values for UI concentration.
The WHO advises that a median UI concentration of greater than 100 μg/L indicates population iodine sufficiency. 42 This is the most widely used indicator of population iodine status and is considered to be reliable in the context of school-aged children. The most appropriate cut-off to use for adults in this situation is not clear, although some authorities suggest 60–70 μg/L. 34
Urine iodine in individuals
More problematic and less well studied is the use of urine iodine measurement to assess iodine status in individuals. As mentioned before, it is known that UI concentration reflects recent dietary intake of iodine and issues arise when attempting to extrapolate from this value clinically useful information regarding overall iodine status given the high variability of UI concentration measurements.
There is currently no clear consensus on the most appropriate way to use urine iodine measurement to assess iodine status in an individual, or even whether this is in fact a useful thing to do. However, it is clear that one cannot simply use the WHO endorsed cut-off of less than 100 μg/L from a single-spot urine as this figure refers to a population median.
It is well appreciated that UI concentration from a spot sample is subject to significant variation. A small study published in 1999 demonstrated this, looking at urine iodine parameters in 10 healthy volunteers. 43 The authors observed considerable between-day and within-day variation in 24-h urine excretion of iodine and also UI concentration from spot samples. One study performed in Switzerland in the mid 1990s looked at UI concentrations in spot samples in 1729 volunteers. 44 The authors observed significant seasonal variation in UI concentration in children with differences of between 5 and 30% in mean values between summer and winter, with values being higher in the summer months. They attributed this principally to known differences in cow’s milk iodine concentration, and differences in milk consumption. A further more recent study investigated this seasonal effect in 401 healthy adults living in Belgium. 45 They too observed that UI concentration varied significantly with the seasons, and this difference persisted after controlling for age, sex, ethnicity and BMI. There was a greater proportion of individuals with a UI concentration less than 100 μg/L in the winter than there was in the summer.
There is also evidence that variation in iodine excretion may follow a circadian rhythm. A study from Switzerland looked at over 3000 spot samples from 42 adults and children over the course of two years and uncovered circadian rhythmicity that was independent of season, age and gender. 46 They observed that UI concentration is lowest at 0800–1100 h, and increases progressively between 1200 and 0000 h and that concentrations generally peak 4–5 h after meals. They concluded that studies using spot urine samples from fixed times of the day such as the morning could not be reliably compared with studies where sampling occurred at different times of the day or where sampling times were not fixed. Additionally, this introduces interpretative issues when considering UI concentration data in individuals. There is also evidence to suggest that hydration status is a significant source of variation in UI concentration, and use of single-spot samples for UI concentration as opposed to 24-h collections in individuals may provide misleading results. 47
Alternative methods have been suggested for use in individuals such as iodine to creatinine ratios on spot urine samples as a way of estimating 24-h iodine excretion using age- and sex-adjusted reference data, and there is some evidence to suggest that this may be superior to UI concentration. One study from Denmark was published in 2009 and looked at urinary iodine excretion in 16 healthy male volunteers living in an area of mild to moderate iodine deficiency. 48 They collected monthly morning spot urine samples for 13 months and analysed them for iodine and creatinine, and once again observed significant variability in UI concentration and also estimated 24-h iodine excretion. They concluded that in order to achieve a precision range ± 20% for an individual, 12 spot urine samples are required, or 7 spot samples with an iodine to creatinine ratio to calculate estimated iodine excretion. They also suggest that based on their results, less than 10 urine samples in an individual may provide a misleading indication of their iodine status.
A more recent study from 2011 compared measured 24-h urine iodine excretion with estimated urine iodine excretion in 22 healthy female Swiss volunteers. 49 The authors showed that repeat spot urine samples used to estimate urine iodine excretion can provide results which are comparable to formal measured 24-h urine collections and could thus provide a reliable alternative. Similarly to the previously described study, they concluded that a minimum of 10 repeated spot urine samples are required to achieve a precision of 20%. In this context, estimated urine iodine excretion can be considered more reliable than simple UI concentration measurements as it accounts for hydration status. It may also be associated with lower intraindividual variation. 50 However, it is important to remember that urine creatinine measurement used to generate estimated iodine excretion is unreliable in the context of malnutrition.
It is crucial to consider whether or not urine iodine excretion in an individual, regardless of the precision of measurements, is actually truly reflective of individual iodine status in a way that is clinically meaningful. The thyroid gland is able to store large quantities of iodine; it is known that 70–80% of the body’s total iodine stores, comprising 15–20 mg, are found within the thyroid. 51 These stores are unaffected by short-term changes in iodine intake, and urine iodine excretion is predominantly a measure of recent iodine intake as the vast majority of iodine absorbed by the gastrointestinal tract is excreted within 24–48 h. 52 Therefore, any form of urine iodine measurement is unlikely to be useful in determining whether or not an individual is iodine deficient unless samples are collected over a prolonged period of time, which will severely limit the clinical utility. Additionally, sampling would ideally cover a sufficiently long period of time to account for seasonal variations in intake of iodine, and this is clearly beyond the realms of routine clinical practice.
Plasma iodine concentration largely reflects the iodine found in circulating thyroid hormones, and thus is not a useful diagnostic test for iodine deficiency. 15
Iodine status and pregnancy
It is worth considering some specific issues with the assessment of iodine status in pregnancy, as pregnant women could be considered the population at highest risk of iodine deficiency; there are known consequences for iodine deficiency during pregnancy, and this population is commonly investigated in clinical studies and targeted by public health initiatives. Some of these important pregnancy-specific issues are discussed in a recent review. 53
There is evidence to suggest that renal clearance of iodine is increased in pregnancy, and therefore urine iodine measurement may overestimate dietary iodine intake in this population. 54 Reflective of this, the WHO advises higher iodine intake in pregnant women. 2 Additionally, the median population UI concentration for pregnant women to denote population iodine sufficiency as suggested by the WHO is higher at 150 μg/L. As with non-pregnant individuals, this population median cannot be used as a cut-off to assess the iodine status of the individual.
It has long been recognized that serum-free T4 concentrations decrease in pregnancy, particularly in the third trimester. 55 This has been investigated, and giving iodine supplements in pregnancy has no effect on this decrease. 56 However, there is some evidence to suggest that those who are iodine deficient may see a greater fall in free T4 than those who are iodine sufficient. 57
As described above, thyroglobulin can be used as a biomarker of iodine status and increased concentrations of serum thyroglobulin have been associated with iodine deficiency in population studies. Increased serum thyroglobulin concentrations have been reported in pregnant women in Denmark 58 ; however, such a finding is not specific for iodine deficiency. This finding has been investigated further and is not thought to be related to altered nutritional iodine status, but rather it is likely to be reflective of increased thyroid gland secretory activity in the pregnant state. 59
These issues are potentially of clinical significance as they could affect interpretation of biochemical testing used to assess iodine status in pregnant women.
Iodine excess
This review has focused on iodine deficiency; however, iodine excess is also a recognized clinical entity that could require biochemical investigation in a small number of situations.
It is possible to dramatically exceed recommended intake for iodine, for instance, some kelp and other edible seaweeds can contain up to 8 mg of iodine per gram. 60 There have also been cases reported of thyroid dysfunction associated with ingestion of large quantities of kelp-containing tea. 61 Other sources of potential iodine excess include over-the-counter iodine supplements, drugs such as amiodarone, iodinated contrast used in radiological investigations and antiseptic topical iodine preparations. The effects of excess iodine on the thyroid gland are complex and incompletely understood, and iodine excess can lead to both hyperthyroidism 62 and hypothyroidism. 63
A median concentration of greater than 300 μg/L is considered to represent excess intake in adults and children, and greater than 500 μg/L is considered so in pregnant women. 2 As discussed previously, there is evidence that dried blood spot thyroglobulin could have some utility in detecting iodine excess at a population level. 17
There are situations, however, where it might be useful to investigate for possible iodine excess in an individual. Factitious hyperthyroidism can occur when an individual takes too much thyroid hormone. It can also occur if too much iodine is ingested in the form of a nutritional supplement or iodine-containing food. Evidence is very hard to come by for this unusual group of patients; however, in this situation, UI concentration will provide a reliable indication of recent iodine intake and could be useful in making a diagnosis of iodine-induced hyperthyroidism. Additionally, iodine excess is known to be an issue in neonates with congenital heart disease who undergo surgery and have intravenous iodine injection as part of cardiac catheterization procedures. A case series has shown that such patients can develop transient hypothyroidism, and as one would imagine, they can have outlandishly high UIC of up to 1,200,000 μg/L. 64 However, biochemical tests of iodine status in this group of patients are of debatable clinical utility, as the cause of iodine excess will be clear and the important parameters to monitor will be simple thyroid function tests.
Conclusion
There is clear utility for the use of biomarkers of iodine status in epidemiological studies to assess for possible iodine deficiency, and this is a worldwide public health issue of immense importance. In this context, median UI concentration can provide a valuable insight. Determining the iodine status of an individual is a much more problematic proposition. UI is highly variable from day to day in a given patient and has low predictive value for iodine deficiency. Median urine iodine concentrations used in epidemiological studies should not be applied to individuals; if they are then iodine deficiency will be over diagnosed. We know from the evidence presented that even an accurate 24-h urine collection cannot be used to reliably assess iodine status. This is because all such tests can only provide information on recent iodine intake, whereas the thyroid gland can store large amounts of iodine. This issue can be partially addressed by performing multiple collections at different times in order to assess an individual’s iodine intake over a more prolonged period of time. If results are repeatedly low, then one could infer that they are likely to be iodine deficient. However, the need for multiple samples and a long sampling period severely limits the clinical utility of such tests, and it is important for laboratory and clinical staff to be aware of these limitations when considering the investigation of a patient with possible iodine deficiency.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PW.
Contributorship
Both authors contributed equally to the review.
