Abstract
Background
United Kingdom national guidelines do not recommend routine testing for thyroid disease in people with type 2 diabetes mellitus (T2DM). However, some studies suggest an increased risk of thyroid dysfunction in T2DM. The aim of this study was to evaluate the current practice of screening for thyroid disease in patients with T2DM.
Methods
Patients with pre-existing T2DM (n = 339) were selected from records for routine glycated haemoglobin testing performed in December 2008. Using routinely collected primary/secondary care data from 2009 to 2017, we examined longitudinal thyroid-stimulating hormone and free thyroxine requests to determine the overall proportion of patients screened for thyroid dysfunction and the time interval between thyroid tests requested.
Results
Thirty-three patients (9.7%) had pre-existing thyroid disease. Of the remaining 306 patients, 96.4% had at least one thyroid test during the follow-up period. When the time interval between tests was evaluated in these patients, there was a discrete peak in thyroid function test requests at 12 months, consistent with routine annual testing. Most requests (77%) originated from a general practice setting.
Conclusions
Contrary to current guidelines, we have provided evidence suggestive of regular screening for thyroid dysfunction in patients with T2DM, particularly in general practice. The appropriateness of this practice remains unclear, but may warrant further examination to assess the clinical benefits of screening, balanced against cost.
Introduction
United Kingdom (UK) national guidelines recommend annual screening for thyroid disease in people with type 1 diabetes mellitus (T1DM), but not type 2 diabetes mellitus (T2DM) (https://www.nice.org.uk/guidance/NG17). Previous joint guidelines from British Thyroid Association and Association for Clinical Biochemistry and Laboratory Medicine state that people with T2DM should have their thyroid function checked at diagnosis, but not routinely thereafter (http://www.acb.org.uk/docs/default-source/guidelines/TFTguidelinefinal.pdf). However, several studies have illustrated a higher prevalence of thyroid disorders in patients with T2DM, 1 suggesting screening may be of value.
Anecdotal evidence suggests that thyroid screening was indeed taking place for people with T2DM as well as T1DM. We aimed, therefore, to determine the proportion of patients with T2DM, without thyroid disease, who had thyroid function tests (TFT), along with the frequency of this testing.
Methods
A cohort of people with pre-existing T2DM were selected from records of all routine glycated haemoglobin (HbA1c) tests, for diabetes monitoring, performed in the month of December 2008 at the University Hospitals of North Midlands NHS Trust. Electronic patient records were used to confirm diabetes type and thyroid disease status. Of 339 patients with T2DM, 33 (9.7%) had existing thyroid disease and were excluded from subsequent analysis, leaving 306 patients registered across 88 general practices (median age 64 years, 58.8% male). In these patients, we collected all routinely performed primary and secondary care TFT results between January 2009 and December 2017. Excluding tests that were requested following an abnormal result gave a data-set comprising 1827 TFTs.
Of the 306 patients, 11 had no TFTs during follow-up and 24 had only one test. Determination of monitoring frequency was based on the remaining 271 cases that had at least two TFTs (1532 test intervals), the first of which was required to be between January 2009 and December 2015, with subsequent test data collected up to December 2017. Repeat testing intervals (time between two consecutive tests) were grouped by month and the relative frequency of each group compared. Where repeat tests were not recorded within 24 months, intervals were categorized as >24.
During the study period, thyroid-stimulating hormone (TSH) and free thyroxine (fT4) were measured using the Siemens ADVIA Centaur XP. TSH was measured front line; if found to be abnormal, fT4 was also tested. Reference ranges used were TSH: 0.1–5.0 mIU/L and fT4: 8–19 pmol/L. Sustained thyroid dysfunction was defined as an abnormal TSH on ≥2 consecutive occasions and results further classified as subclinical hypothyroidism on the finding of a high TSH concentration with a fT4 within range. Thyroid peroxidase (TPO) antibody status, if performed, was recorded.
Results
Overall, 295 (96.4%) cases had at least one TFT during the follow-up period. When the distribution of repeat testing intervals between all pairs of tests was examined, there was a clear peak around 12 months (Figure 1); 29.9% of test intervals were at 12 ± 2 months. Between January 2009 and December 2015 (seven years), the median number of tests per patient was 7 (IQR = 4–9); median time between tests was 10 months (IQR = 8–13).
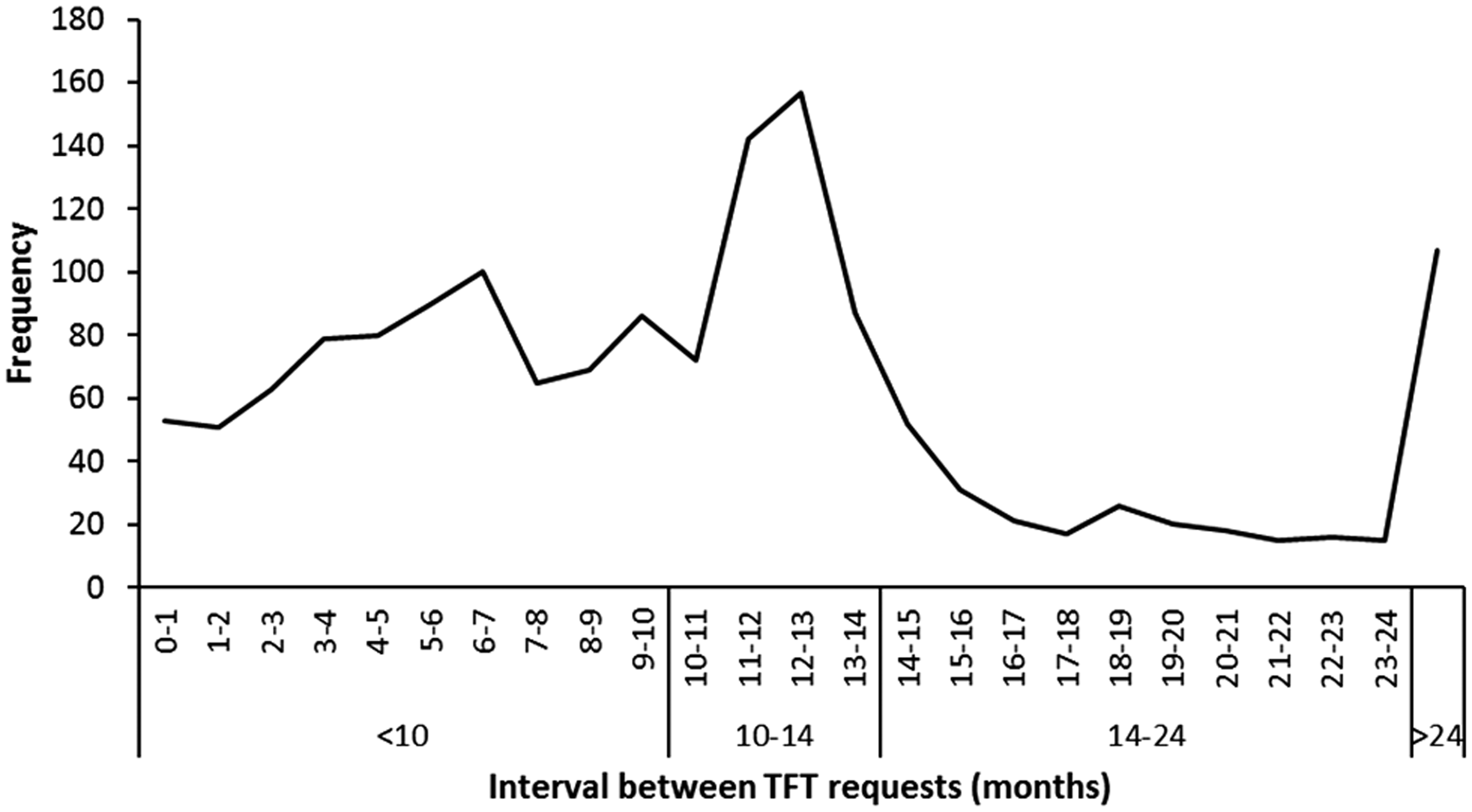
Observed frequencies of thyroid function testing interval.
The median interval for patients aged ≥65 years was significantly shorter (nine months, IQR = 5–12 months) compared with those aged <65 years (11 months, IQR = 6–14; P < 0.001, Mann-Whitney U test). There was no significant difference in pattern of testing between males and females.
For 77.0% of intervals, both tests in the pair originated from primary care. In these cases, the pattern of testing indicated the same clear peak at 12 months (34.3% of intervals at 12 ± 2 months). However, in cases where at least one of the pair of tests was requested in secondary care, the pattern indicated a shorter interval between tests, and the proportion of intervals at 12 ± 2 months was smaller (14.0%; P < 0.001, χ 2 test) than in cases where both tests were requested in primary care.
During follow-up, 16 patients (5.2%) developed sustained thyroid dysfunction, the most common form being subclinical hypothyroidism: 12 patients (3.9%). Just three patients with abnormal TFTs had TPO antibody status tested; all were negative.
Discussion
This assessment of current practice in a sample of patients over a nine-year period showed that the majority of people with T2DM were tested for thyroid disease. Furthermore, when the interval between pairs of tests were examined, the distinct peak of testing at 12 months suggested that a substantial proportion of testing was scheduled in nature, possibly as part of a diabetes annual review. This pattern was not reflected in tests involving secondary care where the overall interval was shorter and more diffuse, implying a more random approach to testing. Although the pattern of testing did not differ between males and females, older age was associated with a shorter interval between tests.
This study is the first to evaluate UK practice for the screening of thyroid disease in patients with T2DM. We recognize that it is a relatively small, single region service evaluation (although covering 88 different general practices) and therefore may not be representative of wider practice. However, in previous work using similar methodologies, we observed good reproducibility across multiple centres.2,3 During data analysis, we assumed that TFTs were requested for thyroid disease screening, but acknowledge that TFTs may have been ordered in other scenarios (e.g. to investigate thyroid disease symptoms). We did not utilize a comparison (non-T2DM) group. However, a recent UK study showed that in one GP surgery, 12% of all registered patients had a TSH test in one year from August 2012. 4 In our T2DM cohort, a substantially higher proportion of patients (53%) had at least one TSH test in this period. Despite having limitations, taking together the high overall proportion of patients tested with the high frequency of 12-month re-test intervals, our data do suggest scheduled screening practice in a significant proportion of cases.
Although not the main focus of this study, we did record cases of thyroid dysfunction. Prevalence of existing overt thyroid disease was 9.7%; after excluding these cases, an additional 5.2% developed thyroid dysfunction during follow-up. A number of cross-sectional studies have estimated an increased prevalence of thyroid dysfunction in patients with T2DM. 1 However, there is a dearth of high-quality studies in this area, and while our data are consistent with this notion, the limitations of our methodology mean firm conclusions cannot be made. Related to this, there are reports that thyroid dysfunction may correlate with deterioration of diabetes control,1,5 possibly supporting a case for targeted screening in the subset of T2DM patients with poor glycaemic control.
Despite guidance indicating that patients with T2DM do not require thyroid monitoring, our data suggest that this is taking place in practice, as is recommended in T1DM. This may reflect individual general practitioners’ views on the apparent high risk of thyroid dysfunction in T2DM, as suggested in some studies, 1 or possibly the use of a more pragmatic approach to screening, regardless of diabetes type. The identification of a prominent peak of testing at 12 months may be linked to routine testing at annual diabetes review. This finding of distinct peaks of activity has also been detected elsewhere in both diabetes and thyroid testing contexts.3,6 These studies also illustrate a lack of conformity to guidance on recommended testing frequency.
The identification of age as a significant factor in monitoring frequency has been identified in other contexts 7 and may reflect older patients being more engaged with health-care systems. 8 In the present study, testing patterns were also influenced by origin of request, with tests from secondary care generally requested more frequently and with a more sporadic pattern, consistent with observations made previously for HbA1c testing. 6
As described, many patients with T2DM appear to be tested for thyroid dysfunction on a regular basis. Hence, while there is no specific guidance to support regular TFT monitoring in patients with T2DM, screening may be considered, at least by some health-care professionals, to be necessary. Whether this testing behaviour represents inappropriate overuse of laboratory services, or prudent practice in this potentially at risk group, is still to be determined.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Routine data were retrospectively collected and analysed as part of a service evaluation; therefore, ethical approval was not required.
Guarantor
CJD.
Contributorship
All authors made substantial contributions to the study conception and design. TP and CJD collated the data. TP, CJD and AAF analysed the data. AHH provided interpretation and critique from a clinical endocrinology perspective. AAF and CJD provided interpretation and critique from a laboratory medicine perspective. All authors contributed to writing the article and approved the final version.
