Abstract
Background
There is interest in utilizing different cut-offs per sex for high-sensitivity cardiac troponin I (hs-cTnI) but less so for high-sensitivity cardiac troponin T (hs-cTnT) for patient management in the acute setting. Our objective was to assess if differences in hs-cTn concentrations exist between males and females for an acute cardiac outcome following the presentation measurement in the emergency department.
Methods
An observational emergency department population with hs-cTn measurements (Roche Diagnostics and Abbott Diagnostics) at presentation with seven-day outcomes for a composite acute cardiac outcome (i.e. myocardial infarction, unstable angina, ventricular arrhythmia, heart failure or cardiovascular death) (ClinicalTrials.gov: NCT01994577). Receiver operating characteristic curve analyses were performed for each sex with both hs-cTn assays.
Results
In those patients who had a composite acute cardiac outcome (n = 128 females; n = 145 males), there was no difference in hs-cTn concentrations between the sexes (median [IQR] female hs-cTnT = 35 ng/L [21–69] vs. male hs-cTnT = 38 ng/L [19–77], P = 0.95; and median [IQR] female hs-cTnI = 27 ng/L [12–75] vs. male hs-cTnI = 26 ng/L [12–85], P = 0.97]. There was also no difference in the area under the curve between the hs-cTn assays and between the sexes (P > 0.10). Comparing hs-cTn concentrations in those patients with the composite outcome between the sexes <60 years and ≥60 years of age also did not yield significant differences (P > 0.70).
Conclusions
The concentrations and area under the curves of hs-cTnT and hs-cTnI at patient presentation in the emergency department for an acute composite cardiac outcome were similar between the sexes in this exploratory study.
Keywords
Introduction
There is ample evidence to support the superior diagnostic and health service performance of high-sensitivity cardiac troponin (hs-cTn) testing over contemporary cardiac troponin testing. 1 Where evidence is conflicting, and there is also much debate, is whether sex-specific 99th-percentiles for hs-cTn should be used for clinical care.2,3 One of the confounding issues when assessing different cardiac troponin assays and cut-offs for myocardial infarction (MI) is the incorporation bias that is present (i.e. cardiac troponin is used for the diagnosis of non-ST-segment elevation MI). However, MI is only one of the many serious cardiac outcomes that emergency physicians are concerned about in patients presenting with symptoms of acute coronary syndrome (ACS). Our study’s objective was to assess in patients with a serious cardiac outcome if there were differences in hs-cTn concentrations at emergency department (ED) presentation between males and females.
Methods
Patients enrolled in the ROMI-3 study with presentation hs-cTn measurements (n = 1137 patients from May 2013 to August 2013; ClinicalTrials.gov Identifier: NCT01994577) who identified themselves as male or female were included in this study cohort (n = 1136; 1 transgender patient was excluded from this sex-specific analysis). Details on the study population have been previously described with adherence to the STARD reporting guideline.4,5 In brief, consecutive adult patients presenting with ACS symptoms and had cardiac troponin clinically ordered by the emergency physician met the inclusion criteria. We excluded patients with ST-segment elevation MI, ventricular tachycardia or fibrillation, or if their symptoms were determined not to be due to ACS (i.e. chest trauma), or if they experienced a previous MI, cardiac intervention, or pulmonary embolus within the past month, or if they were determined to have sepsis, an active malignancy or a non-cardiac fatal illness.4,5
Blood samples (EDTA plasma samples; non-frozen) were measured with the high-sensitivity cardiac troponin T (hs-cTnT) assay (Roche E-modular analyzer) and the high-sensitivity cardiac troponin I (hs-cTnI) assay (Abbott ARHCITECT i2000 analyzers) with the imprecision of the measurements meeting acceptable standards (as previously reported). 6 The adjudicated composite acute cardiac outcome (adjudicators blinded to hs-cTn results) has been previously reported.5,6 In short, the primary outcome was a composite that consisted of MI (based on the Third Universal Definition of MI and the Abbott ARCHITECT sensitive cTnI assay), unstable angina, serious ventricular cardiac arrhythmia, decompensated congestive heart failure, or cardiovascular death within seven days of ED presentation.5,6
Receiver operating characteristic (ROC) curves were generated for each sex and the hs-cTn assay with the area under the curve (AUC) used to compare the diagnostic accuracy (with the 95% confidence intervals [CI] calculated). Differences in hs-cTn concentrations between sexes were also assessed with the Mann-Whitney test. All analyses were performed via Analyse-it and MedCalc software with P < 0.05 considered significant.
Results
The female population (n = 601; median age [interquartile] = 72 y [57–83]) was significantly older than the male population (n = 535; median age [interquartile] = 64 y [54–77]) (P < 0.01). ROC curve analyses using the presentation hs-cTn concentration demonstrated no difference in AUC between hs-cTnI and hs-cTnT for the composite acute cardiac outcome in females (hs-cTnI AUC = 0.87; 95% CI: 0.83–0.90 vs. hs-cTnT AUC = 0.86; 95% CI: 0.83–0.90, P = 0.60) and males (hs-cTnI AUC = 0.85; 95% CI: 0.81–0.88 vs. hs-cTnT AUC = 0.82; 95% CI: 0.78–0.86, P = 0.12) and between the sexes using the same hs-cTn assay (P > 0.10). In those patients who had a composite acute cardiac outcome (n = 128 females; n = 145 males), there was also no difference in hs-cTn concentrations between the sexes (median [IQR] female hs-cTnT = 35 ng/L [21–69] vs. male hs-cTnT = 38 ng/L [19–77], P = 0.95; and median [IQR] female hs-cTnI = 27 ng/L [12–75] vs. male hs-cTnI = 26 ng/L [12–85], P = 0.97) (Figure 1). For patients <60 years with the composite outcome, the median (IQR) concentrations in females (n = 16 with median age = 54 y; hs-cTnT = 28 ng/L [9–61]; hs-cTnI = 24 ng/L [5–50]) were not significantly different than males (n = 27 with median age = 55 y; hs-cTnT = 27 ng/L [13–44]; hs-cTnI = 20 ng/L [8–61] [P = 0.91;0.72]). For patients ≥60 years, there was also no difference in concentrations between the sexes in patients with the composite outcome (n = 112 females [median age = 82 y] hs-cTnT = 37 ng/L [22–70]; hs-cTnI = 28 ng/L [13–79]) vs. n = 118 males (median age = 75 y) hs-cTnT = 39 ng/L (21–82); hs-cTnI = 26 ng/L (13–86) (P = 0.76;0.95).
Concentrations of hs-cTnT (a) or hs-cTnI (b) in females and males with a composite cardiac outcome (note: the central box represents the 25th to 75th percentile (IQR) with the middle line representing the median).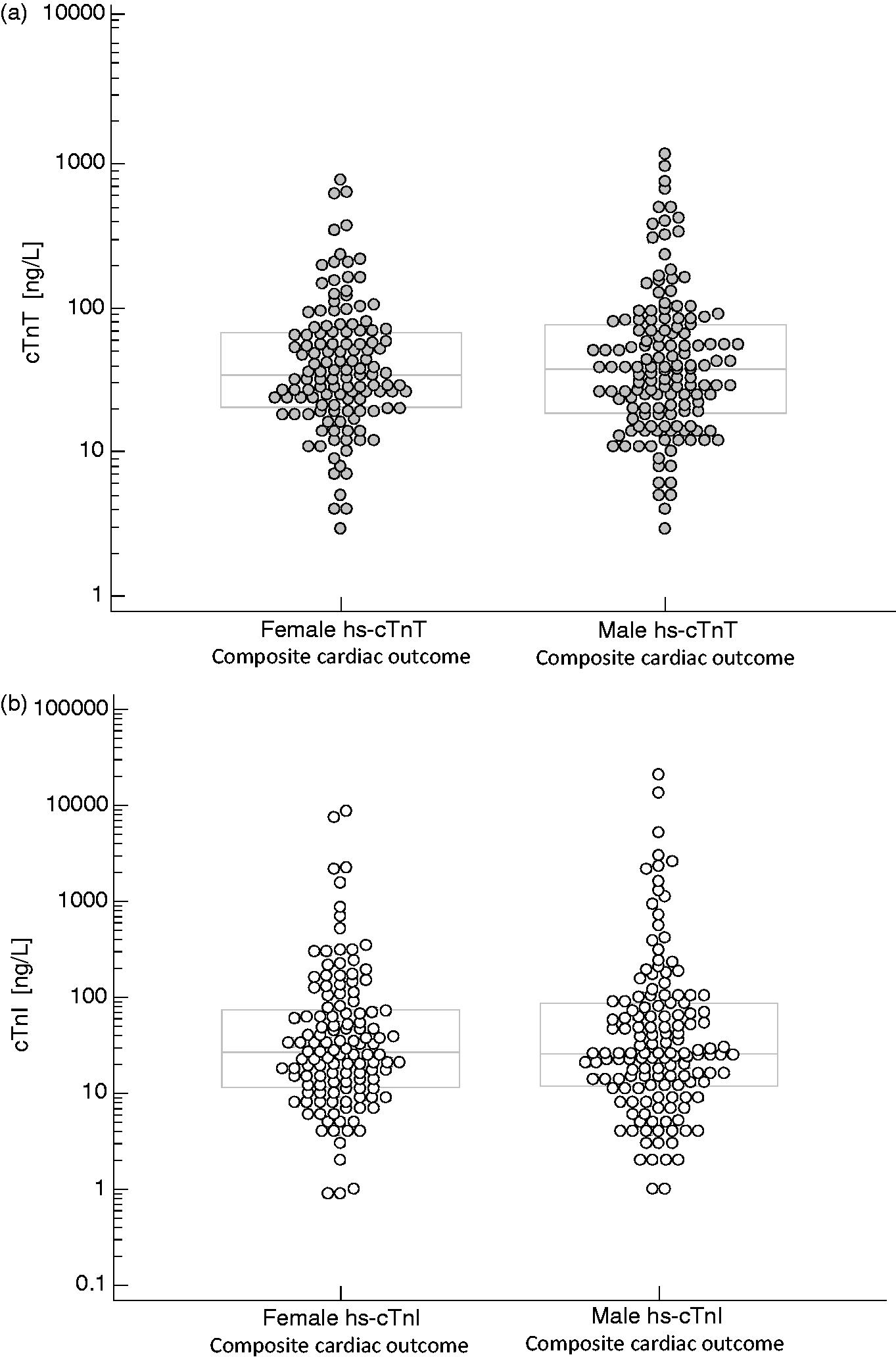
Discussion
Despite healthy reference populations yielding different hs-cTn 99th percentiles between males and females, many algorithms that have been developed for early decision-making have not used the patient’s sex as a variable. The present study findings indicated similar diagnostic performance between presentation hs-cTnI and hs-cTnT concentrations in both the female and male population in the ED for detecting an acute composite cardiac outcome despite the significant age difference between the two groups. This finding reaffirms that cardiac specific and sensitive tests, such as hs-cTnT and hs-cTnI, detect patients in the ED with myocardial injury and who are at an increased risk for an adverse cardiac event. The overall finding that there was no difference in hs-cTn concentrations in females vs. males with the composite acute cardiac outcome may support a risk cut-off approach as opposed to one overall cut-off.5,6
Strengths of our study include employing a composite cardiovascular outcome as the endpoint as opposed to MI alone. MI as an outcome is heavily influenced by the type of cardiac troponin assay used for adjudication and employing the composite cardiovascular outcome mitigates these inherent biases. Another strength is our consecutive patient enrolment and measuring the population with both an hs-cTnI and hs-cTnT assay. Limitations to the present analyses are: the sample sizes for each sex are not equivalent, the length of follow-up of only one-week and the older age of the study cohort. However, as physicians are concerned about identifying patients who are at risk for an event in the short-term, we feel that this time frame fulfils an important clinical gap in a representative population being assessed for ACS. It is worth noting that the objective of the present analysis (i.e. to assess the difference in hs-cTn concentration between males and females with the composite outcome) was not the primary objective in the original study grant (awarded by the Canadian Institutes of Health Research in 2013), and therefore the study was not designed to assess the difference between sexes. A post hoc power calculation indicates that this secondary analysis would require a sample size in excess of 1600 participants. Notwithstanding these limitations, these preliminary data suggest similar diagnostic information for an acute composite cardiac outcome between the sexes with the presentation hs-cTn concentration.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kavsak has received grants/reagents/consultant/advisor/honoria from Abbott Laboratories, Abbott Point of Care, Abbott Diagnostics Division Canada, Beckman Coulter, Ortho Clinical Diagnostics, Randox Laboratories, Roche Diagnostics and Siemens Healthcare Diagnostics. McMaster University has filed patents with Kavsak listed as an inventor in the acute cardiovascular biomarker field. High-sensitivity cardiac troponin reagents were provided by Roche Diagnostics and Abbott Diagnostics for this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Canadian Institutes of Health Research with high-sensitivity cardiac troponin tests provided by Roche Diagnostics and Abbott Diagnostics.
Ethical approval
Hamilton Integrated Research Ethics board; REB Project # 13-277.
Guarantor
PAK.
Contributorship
PAK and AW contributed equally to this manuscript. PAK, AW, CS, JM, NC, JS, SAH, MM, LG, SRM, PJD designed the study and experiments. PAK oversaw the analyses of samples. AW and PAK analysed the results and drafted the manuscript.
