Abstract
Background
Analytical comparisons between different high-sensitivity cardiac troponin (hs-cTn) assays are important for reassurance of results performed with different methodologies and to identify potential interferences or confounders to result interpretation. Our objective in the present study was to compare Beckman Coulter’s latest hs-cTnI assay to Abbott’s hs-cTnI assay and to assess agreement between results.
Methods
Two hundred ethylenediaminetetraacetic acid plasma samples that had clinically reported hs-cTnI results from the Abbott ARCHITECTi2000 that spanned the analytical range were stored (median = 4 h), re-centrifuged and retested for hs-cTnI on the Abbott ARCHITECTi1000 and Beckman Coulter Access2 analysers. Passing–Bablok regression and fold-differences were evaluated, with differences approximately three-fold between results further subjected to Roche hs-cTnT testing and polyethylene glycol precipitation.
Results
The Beckman and Abbott hs-cTnI concentrations were correlated (r = 0.95) with Beckman yielding proportionally lower concentrations (slope = 0.78; 95%CI: 0.74–0.85). There were 12 samples that yielded Abbott hs-TnI concentrations ≥3-fold higher than the Beckman hs-cTnI concentrations; of which nine samples from seven different patients had sufficient quantity for additional testing. All seven patients had macrocomplexes as determined with polyethylene glycol precipitation that affected the Abbott hs-cTnI assay. One patient with Abbott hs-cTnI results >1300 ng/L had polyethylene glycol, heterophile antibodies and creatine kinase-MB testing performed which confirmed that a macrocomplex most likely affected the Abbott and Roche (hs-cTnT = 65 ng/L) assays but not the Beckman (hs-cTnI = 12 ng/L) assay.
Conclusion
The hs-cTnI concentrations obtained from ethylenediaminetetraacetic acid plasma between the Beckman and Abbott assays are highly correlated, with large differences in concentrations (≥3-fold) between Abbott and Beckman assays possible due to macrocomplexes.
Introduction
Much of the focus with high-sensitivity cardiac troponin (hs-cTn) assays has been on ruling-out myocardial infarction (MI) at emergency department (ED) presentation by using low concentration cut-offs only measurable by these assays. However, there is emerging evidence that a single abnormal high concentration as measured by hs-cTn assays may be equally efficacious for predicting MI at ED presentation. 1 The improved precision and analytical design of the hs-cTn assays have eliminated some of the analytical noise of the assay; yet, there are still interferences that may affect result interpretation. Specifically, autoantibodies to cardiac troponin may result in lower concentrations of this analyte following acute MI. 2 There are also sample/matrix effects that result in abnormal cardiac troponin concentrations produced with some assays but not others. 3 Further to this, there are specific preanalytical factors that may result in a falsely decreased or increased cardiac troponin concentration. 4 An example of one known interference affecting many different cardiac troponin assays is macrocomplexes; however, the extent of this phenomenon has not been thoroughly accessed with hs-cTn assays.4,5
Accordingly, before clinically employing new hs-cTn assays, analytical comparison studies between hs-cTn assays are important for reassurance on analytical performance characteristics related to hs-cTn assays. Furthermore, comparison studies may also identify discordant results which may prove useful in discovering not only interferences but also in the development of procedures to mitigate these interferences. In this report, we undertook such an analytical comparison study between the Abbott hs-cTnI and Beckman Coutler hs-cTnI assays and further tested discordant results with the Roche hs-cTnT assay and with polyethylene glycol (PEG) precipitation.
Methods
Plasma (ethylenediaminetetraacetic acid [EDTA] samples) with clinically reported hs-cTnI concentrations (Abbott ARCHITECT i2000 over 12 days) were collected and separated (median 4 h after collection with an average of 17 samples per day). Selection of samples was based on sufficient volume for repeat testing and to obtain a range of concentrations that spanned the analytical range for the Abbott hs-cTnI assay. Prior to repeat testing, the samples underwent re-centrifugation (5 min at 3000 × g) and then were tested for hs-cTnI in the Clinical Research and Biobank Laboratory (CRLB, Hamilton Health Sciences) on the Abbott ARCHITECT i1000 and Beckman Coulter Access2 analysers. Re-centrifugation was performed to mitigate any outliers due to sample storage as previously described for the Abbott hs-cTnI assay. 6 Two hundred EDTA plasma samples were tested on all three platforms with Passing–Bablok regression comparison between the CRLB Access2 and ARCHITECTi1000 results; with fold-differences assessed and compared with the clinically reported hs-cTnI results (Analyse-it v4.80.9). Discordant concentrations (i.e. approximately 0.33 or three-fold difference) from the clinically reported hs-cTnI results were noted and samples with sufficient volume were stored and tested for the Roche hs-cTnT assay on the E-modular platform (CRLB) and were also subject to PEG precipitation (25% PEG6000) as previously described, depending on the remaining sample volume. 5 Chart review was also performed on the patients who yielded the discordant concentrations with all Abbott hs-cTnI results, and the hospital discharge diagnosis was extracted. Ethics approval for the study was obtained by the Hamilton Integrated Research Ethics Board (REB Project # 07-387).
Results
Over the study period, using the same patient pool materials (n = 12 normal; n = 12 high concentrations) the following performance characteristics were observed: Abbott ARCHITECT i1000 normal mean (SD) = 4.5 ng/L (0.4) and high mean (SD) = 32.3 ng/L (1.2) and the Beckman Coulter Access2 normal mean (SD) = 3.4 ng/L (0.7) and high mean (SD) = 23.5 ng/L (1.3). In the 200 patient comparison sample set, the Beckman Coulter Access2 hs-cTnI concentrations (range: 1.1–18,551 ng/L) were proportionally lower as compared with the Abbott ARCHITECT i1000 hs-cTnI concentrations (range: 1–31,539 ng/L) (Beckman Coulter hs-cTnI = 0.78 (95% CI: 0.74–0.85) Abbott hs-cTnI + 0.8 (95% CI: 0.5–1.1) (Figure 1(a)). Visual inspection of scatter plot indicated a few samples where the Abbott hs-cTnI assay yielded significantly higher results. Three-fold differences between the Beckman Coulter hs-cTnI and Abbott hs-cTnI results were only observed for concentrations below 1500 ng/L (n = 184 samples), with the preponderance of discrepant results being significantly lower for the Beckman Coulter hs-cTnI assay (Figure 1(b)). There were no discrepant results between the Abbott ARCHITECTi1000 and i2000 platforms. From the 184 samples, there were 12 (6.5%) with Abbott hs-cTnI concentrations ≥3-fold higher than Beckman Coulter hs-cTnI, with only nine samples with sufficient volume for subsequent testing for hs-cTnT and PEG precipitation (see Table 1).
(a) Method comparison between Beckman Coulter hs-cTnI and Abbott hs-cTnI (logarithmic scale) performed in the CRLB. (b) Fold-differences between Beckman Coulter Access2 hs-cTnI and Abbott ARCHITECTi1000 hs-cTnI (performed in the CRLB) versus clinically reported Abbott ARCHITECTi2000 hs-cTnI results (Hamilton General Hospital). Seven different patients with nine samples with sufficient volume for testing with Roche hs-cTnT and PEG treatment. Note: The following three samples were not available for subsequent testing: CRLB#50: clinically reported Abbott hs-cTnI = 40 ng/L; Beckman hs-cTnI = 6 ng/L; fold-difference = 0.15; CRLB#29: clinically reported Abbott hs-cTnI = 267 ng/L; Beckman hs-cTnI = 69 ng/L; fold-difference = 0.26; CRLB#189: clinically reported Abbott hs-cTnI = 223 ng/L; Beckman hs-cTnI = 71 ng/L; fold-difference = 0.32.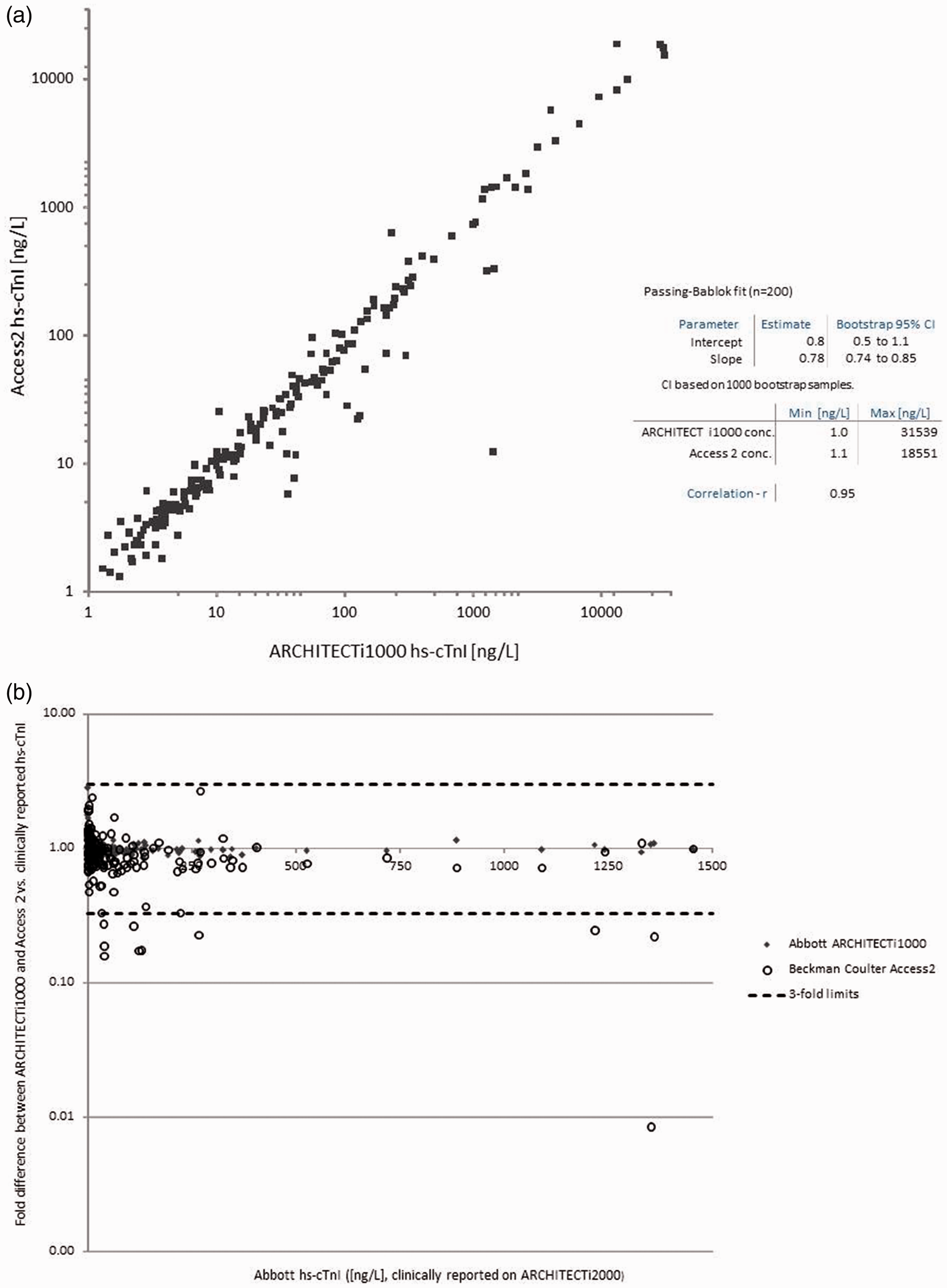
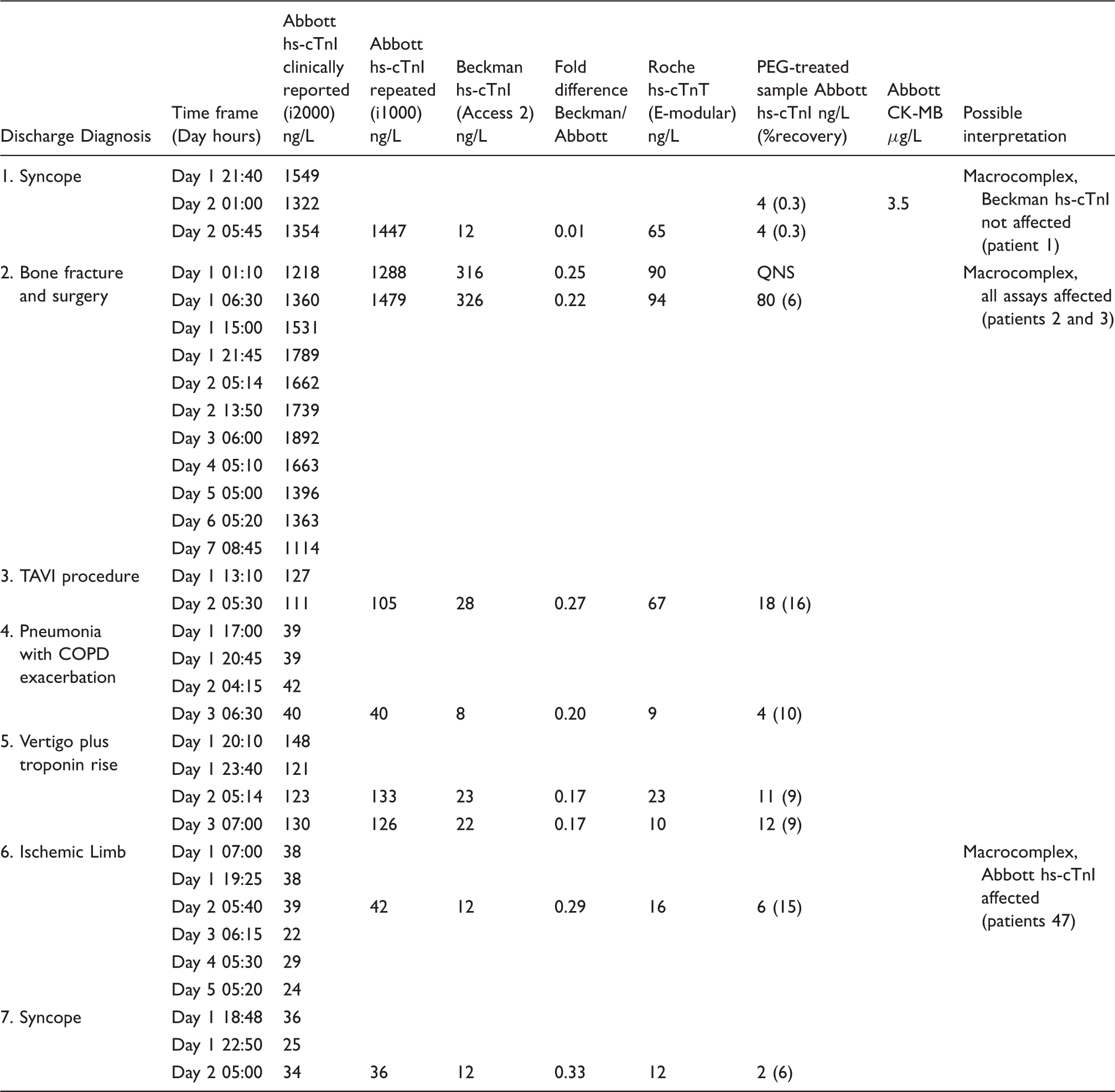
The nine samples were from seven different patients with a possible assignment in the following groups: (i) macrocomplexes most likely affecting all hs-cTn assays; (ii) macrocomplexes most likely resulting in the high Abbott hs-cTnI concentrations and (iii) macrocomplexes affecting Abbott hs-cTnI and Roche hs-cTnT (Table 1). Macrocomplexes were identified if the recovery after PEG precipitation was <20% for hs-cTnI concentrations 5 as recovery of normal/control samples has been reported between 33 and 61% (two control samples in our study recovered with 39% and 48%). Four patients with five samples appeared to have macrocomplexes resulting in the higher Abbott hs-cTnI concentrations (PEG recovery 6–15%). One patient with Abbott hs-cTnI results >100-fold higher than the Beckman Coulter hs-cTnI result had hs-cTnT, PEG, HAMA and CKMB testing performed. The hs-cTnT result was 65 ng/L, the CKMB result was 3.5 µg/L, the HAMA testing was negative for presence of antibody and PEG recovery was 0.3%. There were two patients with possible macrocomplexes affecting all three hs-cTn assays.
Discussion
These data are noteworthy for both clinicians and laboratory professionals. First, the presence of macrocomplexes may explain why some patient results are so discrepant between hs-cTn assays. Second, macrocomplexes will result in concentrations often exceeding the 99th percentile (and multiples of it), so decision making based on serial hs-cTn results is essential. Third, not all macrocomplexes will affect all hs-cTn assays, with the Beckman Coulter hs-cTnI assay appearing to be less affected than the Abbott hs-cTnI assay in the present study. A previous study with the Beckman Coulter AccuTnI + 3 assay identified 1 macrocomplex from 1764 healthy subjects (0.06%). 4 Larger studies assessing more samples and hs-cTn assays are needed to provide a more robust estimate on the prevalence of macrocomplexes and their effects on hs-cTn results; however, these data are important for users to consider when investigating clinically discordant hs-cTn results.
Footnotes
Acknowledgements
The authors thank Beckman Coulter for the supplies to perform this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kavsak has received grants/reagents/consultant/advisor/honoria from Abbott Laboratories, Abbott Point of Care, Abbott Diagnostics Division Canada, Beckman Coulter, Ortho Clinical Diagnostics, Randox Laboratories, Roche Diagnostics and Siemens Healthcare Diagnostics. McMaster University has filed patents with Kavsak listed as an inventor in the acute cardiovascular biomarker field.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This analytical study was funded by Beckman Coulter.
Ethical approval
Hamilton Integrated Research Ethics board; REB Project # 07-387.
Guarantor
PK.
Contributorship
PK, CR, PM, SL, LC, designed the experiments. CR, PM, CM, TS, LC oversaw the analyses of samples. CA and PK analysed the results and drafted the manuscript.
