Abstract
Introduction
Despite some new treatment possibilities, the improvement in survival rate for hepatocellular carcinoma (HCC) patients is still poor due to late diagnosis. The aim of this study was to investigate the diagnostic sensitivity and specificity of protein induced by vitamin K absence or antagonist-II (PIVKAII), Glypican-3 (GP3), Cystatin B (CSTB), squamous cell carcinoma antigen 1 (SCCA1) and hepatocyte growth factor (HGF) as potential tumour markers for HCC in patients with alcoholic liver cirrhosis (ALC) using imaging techniques (MSCT and MRI) as reference standards.
Patients and methods
Eighty-three participants were included: 20 healthy volunteers, 31 patients with ALC and 32 patients with HCC. Peripheral blood sampling was performed for each participant, and serum concentrations of PIVKAII, GP3, CSTB, SCCA1 and HGF were determined using commercial ELISA kits.
Results
Only serum concentrations of PIVKAII were significantly higher in HCC patients as compared with ALC and healthy controls (cut-off: 2.06 µg/L; AUC: 0.903), whereas individual diagnostic performance of other individual compounds was inadequate. The ‘best’ combination of tumour markers in our study includes all tested markers with AUC of 0.967.
Conclusion
While novel diagnostic tumour markers are urgently needed, the examined potential tumour markers, with the exception of PIVKAII seem to be inadequate for diagnosing HCC in ALC. Furthermore, probably the future is in finding the best optimal combination of tumour markers for diagnosing HCC based on cost-effectiveness.
Introduction
Hepatocellular carcinoma (HCC) is the third most common cause of cancer-related deaths worldwide, and accounts for 90% of all primary liver malignancies. 1 Incidence of HCC in developed countries is not high when compared with other malignancies, but has high mortality rate, despite new treatment possibilities, indicating that early diagnosis is crucial in order to improve survival.1–4 Between 90 and 95% of HCC cases are diagnosed in patients with pre-existing cirrhotic livers. 1 Among risk factors for both cirrhosis and HCC, the most prevalent are hepatitis B (HBV) and hepatitis C (HCV) infections, alcoholism, fatty liver, haemochromatosis and primary biliary cirrhosis.1,2 Since the treatment is most effective when administered in the early stage of HCC, there is an obvious need for accurate diagnostic tool(s) for patients at risk. Although HCC meets the criteria for a surveillance programme, poor diagnostic performance of available tools has prevented its widespread use.3,5–7 Up to now, HCC screening has been based on alpha-fetoprotein (AFP) and liver ultrasound (US) both of which have a limited diagnostic performance.3,4,8 According to many international guidelines and reports, US is the most appropriate tool for HCC screening.6,7 Recent research on 2293 high-risk patients has shown that US could detect almost all HCC lesions with a sensitivity of 94% and specificity of 82% and more efficiently than using serum AFP. 6 The combined use of AFP and US seems to be useless because of increased false-positive results which lead to increased costs. 6 Therefore, implementation of more sensitive and specific tumour markers for HCC detection is the focus of many researches in order to improve sensitivity and objectify diagnosis.5–7,9 Besides AFP, some of the potential serum tumour markers for HCC detection include protein induced by vitamin K absence or antagonist-II (PIVKAII),7,10,11 Glypican-3 (GP3),7,10,12,13 squamous cell carcinoma antigen 1 (SCCA1),14–16 while data on the performance of Cystatin B and hepatocyte growth factor (HGF) are still unclear.17,18
PIVKAII is the second most commonly investigated marker for HCC besides AFP. It has been shown that increased concentrations of PIVKA II may be independent of AFP increase in HCC patients. 11 Furthermore, the Japanese Evidence-based Clinical Practice Guidelines and Consensus-based Clinical Practice Manual recommend measurement of PIVKA and AFP (or AFP-L3/AFP) for HCC screening in high-risk patients. 19
Recently, research on AFP, AFP-L3 and PIVKAII diagnostic and prognostic performance using two statistical models (GALAD and BALAD-2) for estimating the likelihood of the presence of HCC in individual patients with chronic liver disease and the survival of patients with HCC, were published. The authors concluded that these models might be used in HCC surveillance and determination of patient prognosis. 20
Glypican-3 (GP3) is an oncofetal protein and is one of the members of heparin sulphate proteoglycans anchored to the plasma membrane. Since GP3 has been exclusively detected in HCC cells and not in benign liver tissues, it has been investigated as another potential tumour marker for HCC detection.10,12,13 Results of meta-analysis conducted in 2014 indicate that serum GP3 is elevated in HCC patients when compared with healthy individuals, but it is still unclear whether it is useful in differentiation of patients with cirrhosis or HCC12.
SCCA is a member of serine protease inhibitor family, and recent studies have shown that SCCA consists of two homologous proteins SCCA1 and SCCA2. 16 Although SCCA1 is found to be elevated in patients with HCC, its usefulness in diagnosing HCC is still doubtful.14–16
Cystatins are endogenous inhibitors of lysosomal cysteine proteinases (cathepsin L, H, and S). 17 Overexpression of Cystatin B (CSTB), a member of the cystatin superfamily, has been reported in patients with HCC. 17
HGF is produced by stromal cells and stimulates proliferation of epithelial cells, motility, morphogenesis and angiogenesis in various organs. 21 Increased HGF serum concentrations have been reported in HCC patients as compared with patients with cirrhosis or chronic hepatitis. 18
The diagnostic performance of the mentioned HCC tumour markers has been mostly investigated in patients with cirrhosis of viral aetiology. Considering the possible differences in pathogenic pathways leading to cirrhosis and HCC development,22–24 the choice of marker(s) for screening might differ in relation to the aetiology of liver disease. Since alcohol has become an important cause of liver cirrhosis in Europe, 23 an increasing number of HCC patients of this aetiological background should be expected. Keeping in mind these epidemiological trends and unmet need for an accurate diagnostic test, we aimed to investigate both an individual and combined diagnostic performance of PIVKAII, GP3, SCCA1, CSTB and HGF as potential tumour markers for HCC detection in patients with alcoholic liver cirrhosis (ALC) when compared with imaging techniques as a diagnostic standard for HCC screening in a high-risk population.
Subjects and methods
Eighty-three participants were included in the study: 20 healthy volunteers (HV), 31 patients with ALC and 32 patients with ALC and HCC. The study was approved by the Institutional Ethics Committee, and all participants signed informed consent.
Patients, hospitalized at the Departments of Gastroenterology and Hepatobiliary surgery of a tertiary care University hospital, were consecutively recruited in the study (during a three-year period (2009–2011)), when alcohol cirrhosis with HCC was suspected according to the physical examination, anamnesis (where history of alcohol exposure was recorded) and results of laboratory tests (alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), total bilirubin, albumin, creatinine, complete blood count (CBC) and prothrombin time (PT/INR)). The presence of cirrhosis was defined upon histology or combination of common clinical, ultrasonographic, laboratory and endoscopic criteria. All patients underwent contrast-enhanced multislice computed tomography (MSCT) and/or magnetic resonance imaging (MRI) in order to confirm or to reject presence of HCC in cirrhotic liver. In doubtful cases tissue diagnosis was obtained by needle biopsy of the tumor. After diagnosis, patients were divided into subgroups.
Healthy participants were included in order to confirm the reference range of HCC tumour markers. They were recruited from the cohort of persons coming for routine annual check-up at outpatient clinic. They had no history of liver disease, whereas physical examination, liver biochemistry and abdominal US were normal as well.
Demographic structure and liver function data of the studied participants.
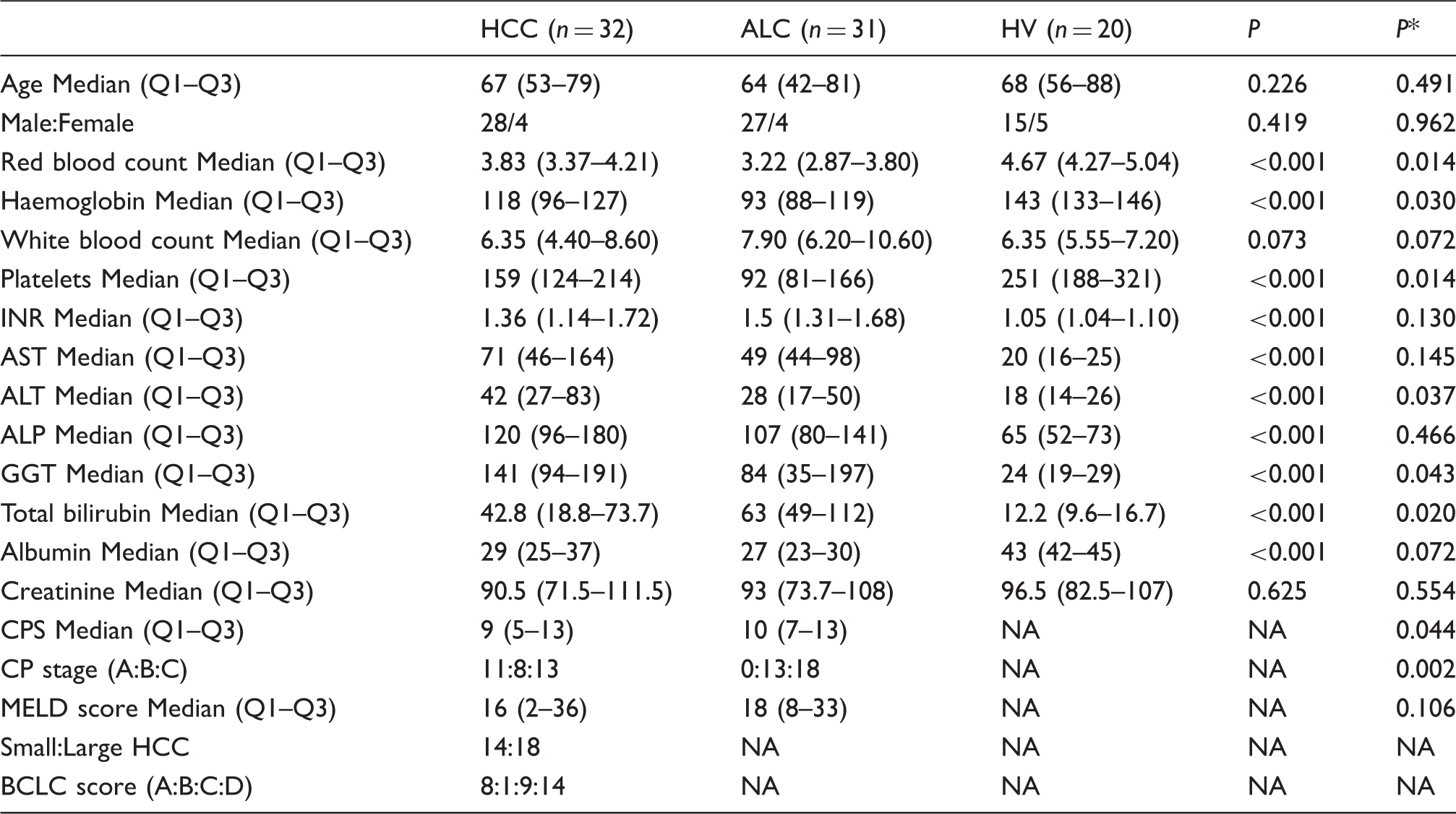
P: statistical difference between HV, HCC and ALC; P*: statistical difference between HCC group vs. ALC group.
CP: Child-Pugh; CPS: Child-Pugh score; HCC: hepatocellular carcinoma; ALC: alcoholic liver cirrhosis; HV: healthy volunteers; MELD: model for end-stage liver disease; small HCC: single nodule <5 cm or up to 3 nodules <3 cm; Large HCC: single nodule >5 cm or multinodular HCC; BCLC: Barcelona clinic liver cancer staging classification; NA: not applicable.
For the patients included in the study, additional laboratory tests were performed: hepatitis B surface antigen, anti-hepatitis B core IgG + IgM, hepatitis C antibody test and serum iron.
Exclusion criteria were: presence of HBV and/or HCV, suspected haemochromatosis, presence of metastases, prior treatment of HCC and history of other solid tumour (-s).
Sample size estimation using comparison of two means was performed in order to assure an adequate power to detect statistical significance in the study. We have estimated that in order to achieve a power of 80% (to detect a significant difference (P < 0.05)) between HCC and cirrhosis in PIVKA II four patients per HCC, and one for the cirrhosis group were needed with an assumed SD of 3.68 ng/mL and 0.80 ng/mL, respectively; 10 in GP3 minimally five patients per HCC, and five for cirrhosis group with an assumed SD of 3.25 ng/mL and 1.16 ng/mL, respectively; 13 in SCCA1 minimally eight patients per HCC, and five for cirrhosis group were needed with an assumed SD of 0.28 ng/mL and 0.10 ng/mL, respectively; 15 in CSTB four patients per HCC, and three for cirrhosis group were needed with an assumed SD of 35.23 ng/mL and 6.58 ng/mL, respectively; 17 in HGF 12 patients per HCC, and nine for cirrhosis group were needed with an assumed SD of 0.17 ng/mL and 0.05 ng/mL, respectively. 18
Child-Pugh score (CPS) and Model for end-stage liver disease (MELD) were used in order to estimate the severity of liver disease for each patient with ALC. CPS was calculated using serum values of total bilirubin, albumin, PT/INR and the presence and severity of ascites and encephalopathy.25,26 The MELD score was calculated by using serum values of bilirubin, creatinine and plasma INR. 27
The collection tubes used were provided by BD (Becton, Dickinson and Co, Italy). First peripheral blood sampling was performed for each participant upon hospitalization in fasting state. CBC was determined from 3 mL K2EDTA tubes on Advia2120 haematology analyser (Siemens, Frimley, Camberley, UK), PT/INR from 2.7 mL citrate-plasma tubes (buffered 0.109 mol/L sodium citrate) on BCS XP analyser (Siemens, Deerfield, USA) and ALT, AST, ALP, GGT, total bilirubin, albumin, creatinine were determined from 10 mL silicone coated, plastic tubes with clot activator on AU2700 plus (Beckman Coulter, Tokyo, Japan) after centrifugation at 1370 × g for 10 min in a Rotina 35 R Hettich centrifuge (Tuttlingen, Germany).
Second peripheral blood sampling was performed after US, CT and MRI were performed and prior any therapeutic interventions. One K2EDTA, one citrate-plasma tube and two silicone-coated clot activator tubes were collected. CBC, PT/INR and ALT, AST, ALP, GGT, total bilirubin, albumin, creatinine and iron were determined as described above. Hepatitis B surface antigen, anti-hepatitis B core IgG + IgM and hepatitis C antibody test were performed on immunochemistry analyser Advia Centaur (Siemens Diagnostics, Tarrytown, NY).
Residual serum samples were aliquoted for PIVKAII, GP3, SCCA1, CSTB and HGF determination and aliquots were stored at −80℃ in DAIREI UPUL 580 freezer (DAIREI EUROPE, Esbejrg N, Denmark) until analysis. All markers were analysed using ELISA commercial kits (PIVKAII (Cusabio Biotech CO., LTD), GP3 (BlueGene Biotech CO. LTD, Shanghai, China), CSTB (Cloud-Clone Corp., Houston, USA), SCCA1 (USCN Life Science Inc., Houston, USA), HGF (Abcam plc, Cambridge, UK)) according to manufacturer’s instructions using commercial control samples which were part of each ELISA kit.
The Kolmogorov–Smirnov test was used to test for a normal distribution of the data. Chi-squared test was used to test the statistical difference between categorical data, Kruskal–Wallis and Mann-Whitney tests were used to test the statistical difference between numerical data. Performance of individual candidate tumour marker in differentiation of cirrhosis and HCC in the studied patients was tested using ROC curve analysis. Performance of combined potential tumour markers in differentiation of cirrhosis and HCC in the studied patients was tested using logistic regression. Correlation of candidate tumour markers and MELD score was tested using Spearman correlation test. A P < 0.05 was considered statistically significant.
Statistical analyses including sampling calculation were performed by using MedCalc Statistical Software version 14.8.1 (MedCalc Software bvba, Ostend, Belgium).
Results
There was no significant difference in the age or gender between the studied groups (Table 1). Although marginally different in absolute numerical values, liver function as reflected by CP appeared to be significantly better in patients with HCC as compared with patients with mere ALC (Table 1). ALT, GGT, RBC, HBG and PLT were significantly higher, while total bilirubin was significantly lower in patients with HCC (Table 1). All HCC patients were classified as early stage HCC (stage A; n = 8) and later stage disease (Stage B; n = 1, Stage C; n = 9 and stage D; n = 14) (Table 1), according to the Barcelona clinic liver cancer staging classification (BCLC).
Serum concentrations of candidate tumour markers in the studied groups.
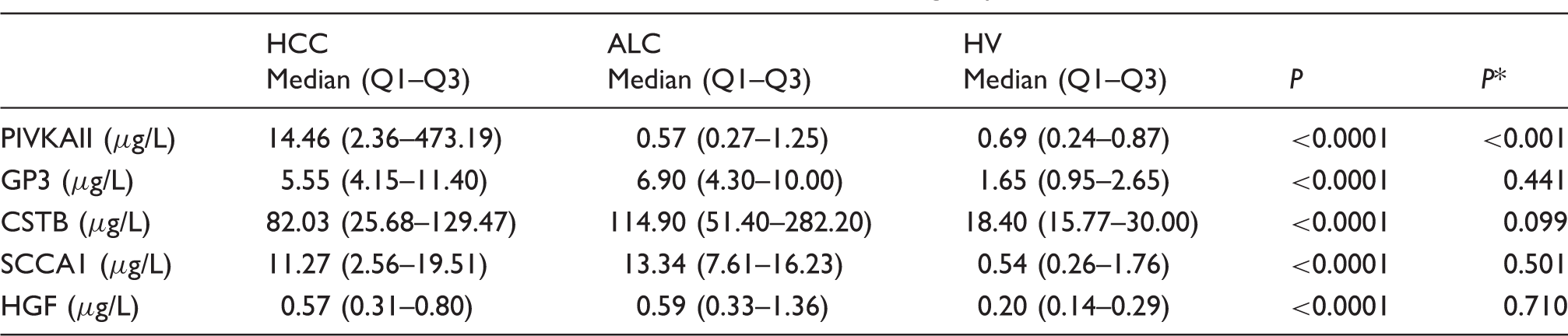
P: statistical difference between HV, HCC and ALC; P*: statistical difference between HCC group vs. ALC group.
HCC: hepatocellular carcinoma; ALC: alcoholic liver cirrhosis; HV: healthy volunteers; MELD: model for end-stage liver disease; PIVKAII: protein-induced vitamin K absence or antagonist II; GP3: glypican-3; CSTB: cystatin B; SCCA1: squamous cell carcinoma antigen 1; HGF: hepatocyte growth factor.
Performance of individual candidate tumour markers in differentiation of cirrhosis and HCC in the studied patients.
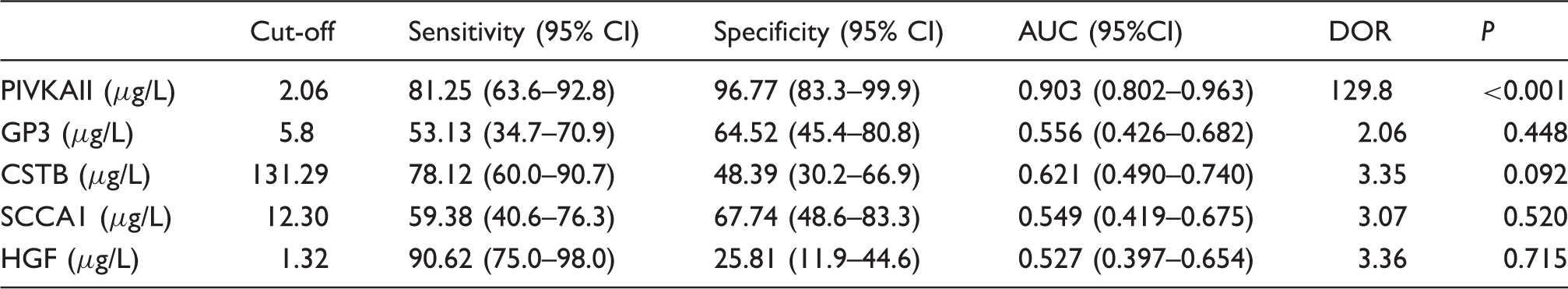
AUC: area under curve; DOR: diagnostic odds ratio; PIVKAII: protein-induced vitamin K absence or antagonist-II; GP3: glypican-3; CSTB: cystatin B; SCCA1: squamous cell carcinoma antigen 1; HGF: hepatocyte growth factor.
Performance of combined potential tumour markers in differentiation of cirrhosis and HCC in the studied patients.
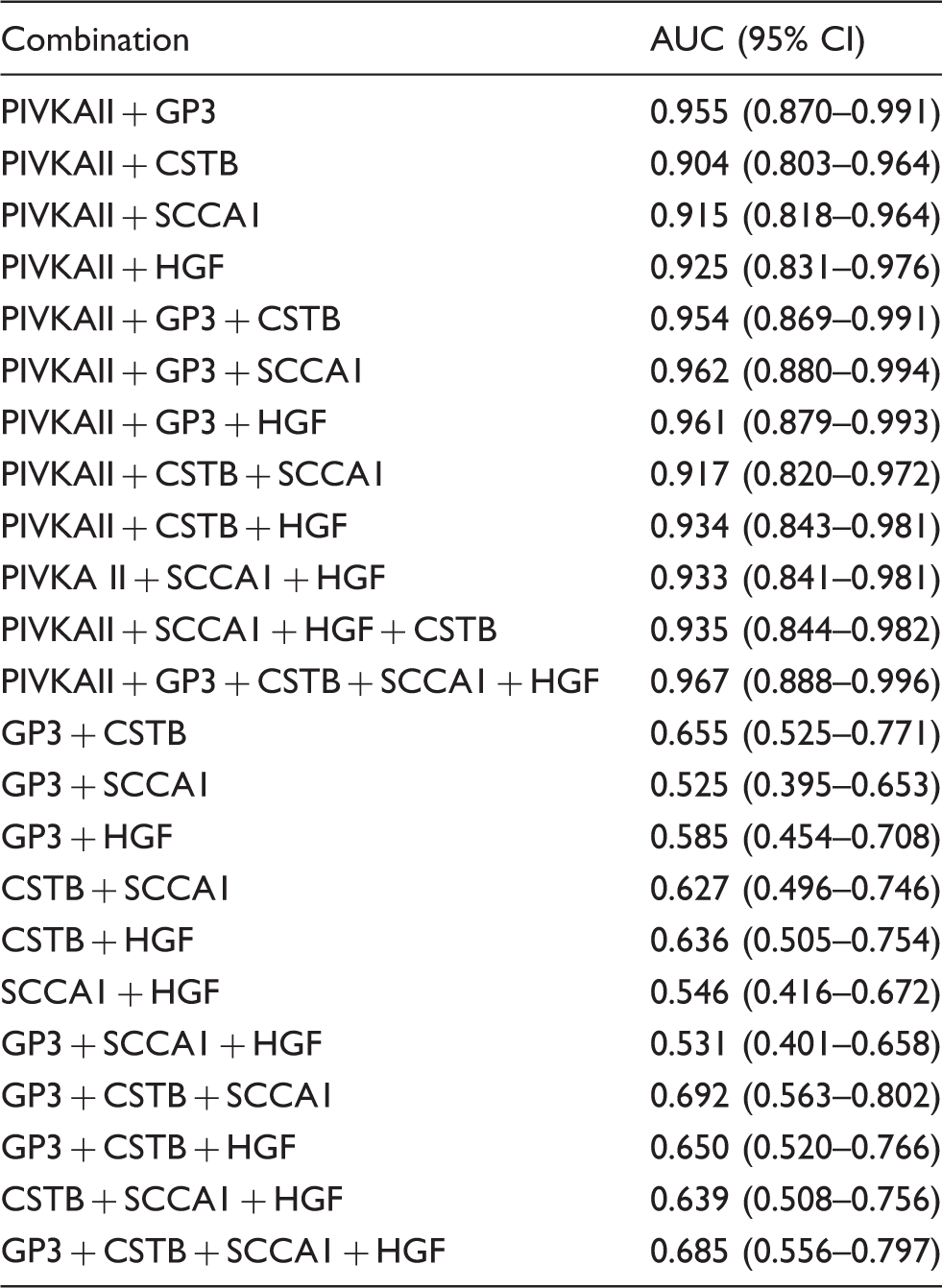
AUC: area under curve; PIVKAII: protein-induced vitamin K absence or antagonist-II; GP3: glypican-3; CSTB: cystatin B; SCCA1: squamous cell carcinoma antigen 1; HGF: hepatocyte growth factor.
All combinations that included PIVKAII showed better diagnostic performance than PIVKAII alone, while the best diagnostic performance was observed for combination of all tested markers with AUC of 0.967.
Serum concentrations of candidate markers in patients with cirrhosis according to the Child-Pugh categories.
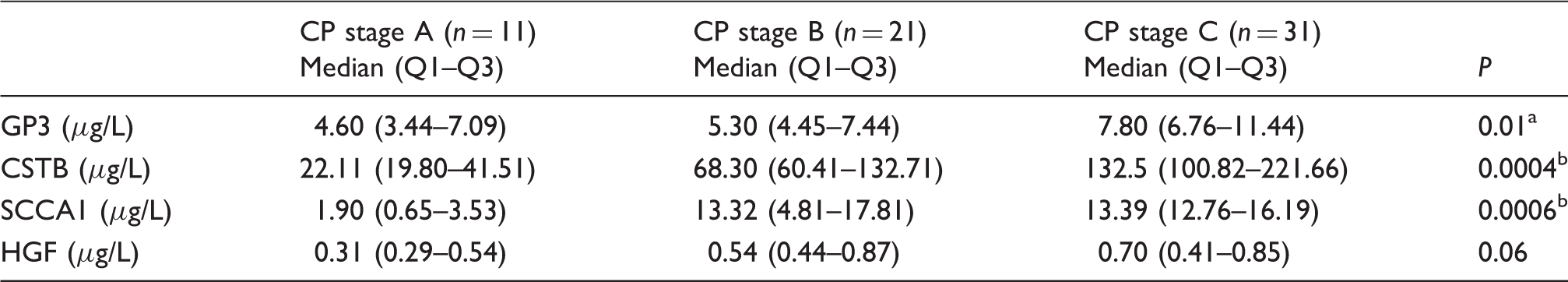
CPS: Child-Pugh stage; GP3: Glypican-3; CSTB: Cystatin B; SCCA1: Squamous cell carcinoma antigen 1; HGF: hepatocyte growth factor.
In post hoc analysis, GP3 was significantly lower in patients with CP-A compared with CP-C stage of cirrhosis.
CSTB and SCCA1 were significantly lower in patients with CP-A score of cirrhosis compared with both CP-B, and CP-C stages of cirrhosis.
Correlation of candidate markers and MELD score.
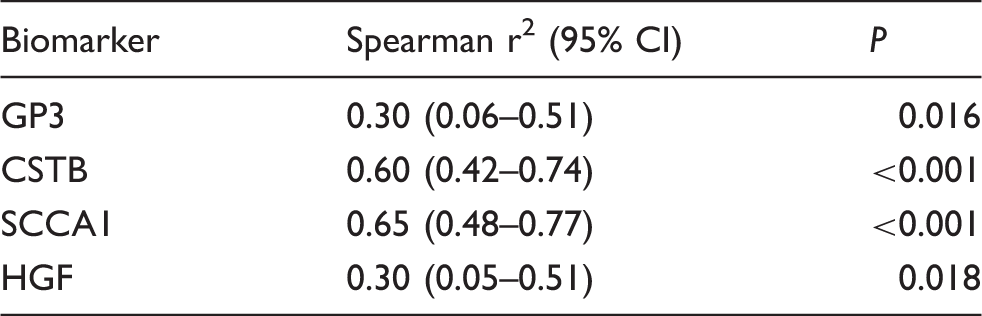
GP3: Glypican-3; CSTB: Cystatin B; SCCA1: Squamous cell carcinoma antigen 1; HGF: hepatocyte growth factor.
Discussion
In this study, we present data on the diagnostic performance of PIVKAII, GP3, CSTB, SCCA1 and HGF, for the detection of HCC in patients with ALC.
Over the past years, scientific efforts have resulted in a number of new tumour markers for HCC detection.2,3,5,7,9 Additionally, new cellular networks and signalling pathways have been discovered in liver cancer patients. 28 A candidate tumour marker for early diagnosis is expected to meet the following criteria: high accessibility (easy and non-invasive way of specimen collection), high accuracy and cost-effectiveness. Markers from body fluids (serum, plasma, urine) meet the first criteria. This makes them good candidates for HCC detection.
We performed our study in order to test the diagnostic performance of potential HCC tumour markers, as well as their combinations.
ROC curve analyses of the individual candidate markers for HCC in our study demonstrated the best performance for PIVKAII (cut-off: 2.06 µg/L (108 mAU/mL); AUC: 0.903). Different cut-off concentrations for PIVKAII have been proposed in different studies and in different HCC aetiologies. Abd El Gawad et al. reported a diagnostic sensitivity and specificity for PIVKAII of 95% and 90%, respectively, at the cut-off of 1.2 µg/L (63.1 mAU/mL) with AUC of 95% in patients with mainly HBV aetiology of HCC. 10 Meta-analysis by Zhu et al. has included 12 different studies on PIVKAII diagnostic performance with cut-offs of 40 mAU/mL, 84 mAU/mL, 125 mAU/mL and 150 mAU/mL with diagnostic accuracy from 63.7% to 93.7%. 11 Moreover, as authors also concluded, such different results might be related to the aetiologic difference of HCC of the studied population. 11
While serum concentrations of other candidate tumour markers were significantly higher in patients with ALC compared with healthy subjects, no significant difference could be observed between the concentrations detected in patients with and without HCC (all of them having liver cirrhosis as well). Although previous studies have shown that tissue expression and/or serum concentrations of these markers were higher in patients with cirrhosis and HCC compared with patients with cirrhosis and no HCC,10,13–18 our study failed to confirm the same relation in serum of tested patients.
There is a small number of studies that investigated potential tumour markers exclusively in alcohol aetiology of cirrhosis and HCC.29,30
Nault et al. 29 investigated diagnostic and prognostic value of three proteoglycans (endocan, syndecan-1 and GP3) and VEGF in 295 patients with alcoholic cirrhosis and HCC. The results of the study showed a significant increase in endocan, syndecan-1 and GP3 in advanced but not in early HCC. 29 Therefore, as our results also confirmed, GP3 alone is not a good tumour marker for early HCC detection in patients with alcoholic aetiology of disease.
Among this group of potential HCC tumour markers, SCCA1 was claimed to have higher tissue expression and serum concentrations in a cohort of HCV patients with HCC compared with cirrhotic patients of HCV aetiology and controls.14,15 This, however, was not confirmed in patients with HCC of alcoholic and non-alcoholic fatty liver aetiology, suggesting SCCA1 aetiology-related specificity with possibility that viral mechanisms might influence its pathogenic role and expression in HCC. 30 Our results, in accordance with Beale et al., 30 showed no significant difference in serum concentrations of SCCA1 between the patients with ALC and HCC.
CSTB was also recognized as potential tumour marker of HCC in the study of Lee et al. with mixed HCC aetiology patients. 17 On contrary, our study did not confirm above mentioned results indicating that CSTB is not a good tumour marker for diagnosing HCC of alcoholic aetiology. It is also interesting that proposed cut-off concentration in mentioned study of Lee et al. 17 was 5.34 µg/L, while our results showed that even patients with ALC and without HCC have higher median CSTB concentration (48.39 µg/L). These differences could be the result of different test sensitivities and different aetiologies of cirrhosis and HCC.
Additionally, among tested markers, HGF showed the highest diagnostic sensitivity of 90.62% at cut-off of 1.32 µg/L, but its specificity for HCC was very low (25.81%). High diagnostic sensitivity indicates that HGF could be a useful tumour marker when combined with another tumour marker of higher specificity for HCC.
Interestingly, as already mentioned, GP3, CSTB, SCCA1 and HGF concentrations in HV significantly differed from patient groups (all having ALC), pointing to possibility that their up-regulation might be an indicator of liver cirrhosis development. Whether this statement is true should be tested in properly designed prospective longitudinal studies and related to already-established reference tests for assessment of liver fibrosis.
Furthermore, GP3, CSTB and SCCA1 showed significant difference between the functional stages of liver cirrhosis as expressed by CPS and significant positive correlation with MELD score as well. Some previous studies have demonstrated that increased concentrations of these markers were in the relation to the progression of liver disease.17,29
Increased expression of GP3 was found in liver progenitor population (oval cells) when hepatocyte proliferation was blocked. 31 The other study revealed that decreased hepatocyte proliferation correlated with the severity of cirrhosis. 32 It has also been reported that liver regeneration was suppressed in alcoholic cirrhosis or alcoholic plus HCV cirrhosis compared with HCV cirrhosis alone, which was accompanied by decreased STAT3 activation. 33 The abovementioned findings could explain the increase of GP3 in relation to the liver disease progression shown in our study. Whereas SCCA1 overexpression was demonstrated to inhibit liver apoptosis and induce proliferation in the animal model of partial hepatectomy, 34 it has been related to higher risk of fibrosis progression and development of liver tumours in humans 35 as indicated by the results of our study.
When applied to our results and regardless pathophysiological mechanism, progressively increased serum concentrations of GP3, SCCA1 and CSTB might be considered as potential indicators of liver function that might be complementary used in follow-up studies of patients with chronic liver disease of alcohol aetiology. Again, this observation should be tested in prospective studies of patients with ALC longitudinally followed over a longer period of time.
Genotypes of heterogeneous patients and variety of risk factors make HCC a complex disease, 28 pointing to the difficulty of diagnosing HCC using a single tumour marker. Thus, a combination of markers might be better suited for the diagnosis, staging and/or prognosis of HCC.3,5 Indeed, some earlier studies suggested that combination of different tumour markers could improve overall performance in detecting HCC.10,14,17,30 The results of our study showed that every combination of tested tumour markers which include PIVKA II improves its diagnostic performance (Table 4). Although the ‘best’ combination of tumour markers in our study includes all tested markers, reduced combination of three markers PIVKAII, GP3 and SCCA1 reaches the comparably highest AUC of 0.962 which makes it optimal combination as regards cost-effectiveness.
In summary, while novel diagnostic tumour markers are urgently needed, the examined potential tumour markers, with the exception of PIVKAII seem to be inadequate for diagnosing HCC in ALC. Furthermore, the future is probably in finding the best optimal combination of tumour markers for diagnosing HCC based on cost-effectiveness.
However, the obtained results indicate CSTB and SCCA1 as potential non-invasive markers of liver function and cirrhosis development that should be further investigated.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from University Hospital Dubrava issued on 23 December 2008.
Guarantor
IG.
Contributorship
AU and IG were involved with study conception, design and drafting of manuscript. All authors were involved in acquisition, analysis and interpretation of data. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
