Abstract
Background
Our aim was to determine whether the postnatal age or postmenstrual age is a more appropriate criterion for evaluating foetal haemoglobin concentrations.
Methods
Blood samples (n = 1095) were obtained from 394 infants and were divided into two groups based on gestational age at birth: <37 weeks (n = 491) and ≥37 weeks (n = 604). (1) Foetal haemoglobin concentrations divided by one month at age after birth were compared between the groups. (2) Foetal haemoglobin concentrations divided into ≤9 months from last menstruation and one month thereafter were compared between the groups.
Results
In samples from infants ≥37 weeks’ gestational age at birth, the median foetal haemoglobin concentrations were 69.5%, 21.4% and 3.6% at 0–1 month, 2–3 months and ≥5 months after birth, respectively. The median foetal haemoglobin concentrations in infants <37 weeks’ gestational age at birth were 75.5%, 62.7% and 5.1% at 0–1 month, 2–3 months and ≥5 months after birth, respectively. The median foetal haemoglobin concentrations in infants <37 weeks’ gestational age at birth were significantly higher than that in infants ≥37 weeks’ gestational age at birth at all postnatal age points. (2) There was no significant difference between the groups at all age points after nine months of postmenstrual age: 72.5 and 75.3% at 9–10 months, 25.1 and 26.6% at 11–12 months and 5.5 and 4.6% at >13 months after last menstruation in infants ≥37 and <37 weeks’ gestational age at birth, respectively.
Conclusions
Evaluation of foetal haemoglobin concentrations at postmenstrual age is unaffected by gestational age at birth.
Keywords
Introduction
Foetal haemoglobin (HbF) is present in the blood of foetuses and neonates. The concentration of HbF in the blood gradually decreases after birth and is replaced with adult haemoglobin 1 ; at six months after birth, HbF accounts for less than 1% of the total haemoglobin. HbF is reportedly elevated in chronic lung diseases and sudden infant death syndrome.2,3 HbF concentrations could therefore be used as a predictive marker for these diseases. However, an established criterion for the HbF concentrations does not exist, because HbF concentrations at various postnatal ages show variations due to the gestational age (GA, a time elapsed between the first day of the last menstrual period and the day of delivery) at birth. 4 In the present study, we assessed whether the postnatal age or postmenstrual age (PMA, gestational age plus chronological age that is a time elapsed from birth) is more appropriate as a criterion for determining the HbF concentration.
Materials and methods
Blood samples (n = 1095) that were collected from 394 infants <12 months of age were analysed. No infants with sudden infant death syndrome and chronic lung disease that required home oxygen therapy were included. Infants with congenital diseases or after blood transfusion were excluded in this study, as these conditions could have influenced the HbF concentration. The blood samples were divided into the following two groups based on the GA at birth: <37 weeks (n = 491) and ≥37 weeks (n = 604). The HbF concentrations divided by one month at age after birth were compared between the groups. Additionally, the HbF concentrations divided into ≤9 months from last menstruation and one month thereafter were compared between the groups. Statistical analyses were performed using the Mann–Whitney U test. Differences were determined to be statistically significant when P < 0.05.
PMA and GA at birth were calculated from the last menstrual period and corrected by sonographic foetal measurement data, if needed.
HbF concentrations in all samples were measured by high-performance liquid chromatography using an ADAMS A1c HA-8180T (Arkray Inc., Japan) with the appropriate calibration based on the manufacture’s recommendation. The results of intra-day assay (n = 20) using a sample with a mean HbF value of 4.67% and inter-day assay (n = 20) using a sample with a mean HbF value of 4.93% gave coefficients of variation of 0.2% and 0.4%, respectively (Supplementary Table).
Results and discussion
Patient background
The median (range) of GA at birth and the birth weight of the infants were 36 (22–41) weeks and 2541 (478–4176) g, respectively. The preterm and term groups had a GA at birth of 32 (22–36) and 38 (37–41) weeks, respectively, and birth weight of 1834 (478–3460) and 2996 (2320–4176) g, respectively. The median postnatal HbF concentration at 0–1 month after birth was 72.0% regardless of GA at birth.
HbF concentrations at postnatal age
HbF concentrations at postnatal age are shown in Figure 1(a) and (c). In samples from infants ≥ 37 weeks’ GA at birth, the median HbF concentration was 69.5% at 0–1 month after birth, and gradually declined thereafter, measuring 10.9% at 4–5 months and 3.6% at ≥5 months after birth. Colombo et al.
1
reported that the mean HbF concentration obtained using electrophoresis in 30 samples of umbilical cord blood in full-term infants was 64.8%, which was close to our result in spite of the different materials and different measurement methods used. The mean HbF concentration decreased to approximately 5% by five months of age.
1
Garby et al.
5
also measured HbF concentrations from 0 to 140 days after birth in 19 infants ≥37 weeks’ GA at birth and found that the HbF concentration was 50–60% immediately after birth and decreased to approximately 5% at 100–140 days after birth. These are similar to the results obtained in our study. In samples from infants <37 weeks’ GA at birth, the median HbF concentrations were 75.5% at 0–1 month after birth, 28.2% at 4–5 months and 5.1% at ≥5 months after birth. The decrease in the HbF concentrations was delayed compared with those of infants ≥37 weeks’ GA at birth. The HbF concentration of an infant with 22 weeks’ GA at birth was approximately 30% even at six months of age (the value was over the reference range, as seen in Figure 1(a) and within the reference range, as seen in Figure 1(b)). Cottom
4
has shown that the HbF concentrations in umbilical cord blood are higher in infants with a lower GA. Our results also show that the HbF concentrations in infants <37 weeks’ GA at birth were significantly higher than those in infants with a GA at birth ≥37 weeks. These findings suggest that the HbF concentrations at a certain postnatal age can have variations due to the GA at birth.
(a) Plots and reference ranges of HbF concentration at postnatal age. (b) Plots and reference ranges of HbF concentration at postmenstrual age. (c): HbF concentrations at postnatal age and postmenstrual age in infants <and ≥37 weeks’ GA at birth. Data are shown as median (range).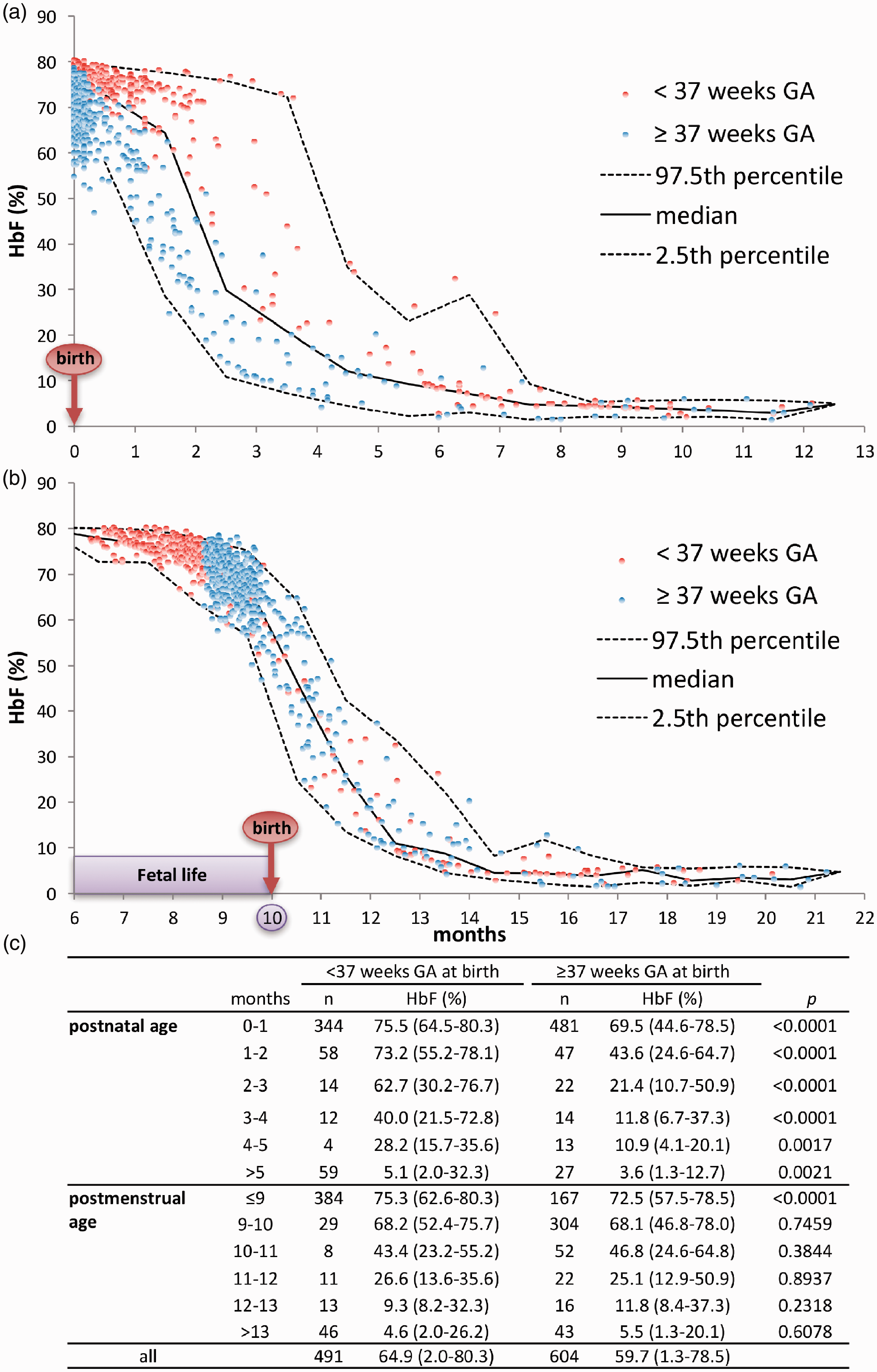
HbF concentrations at PMA
The HbF concentrations at PMA are shown in Figure 1(b) and (c). When we compared HbF concentrations between in infants with GA at birth ≥ and <37 weeks, there was no statistically significant difference between them at the age points after the PMA of nine months. The HbF concentrations at PMA were not affected by the GA at birth, which confirms the report by Colombo et al. 1 Our results are compatible with those of a previous animal study, in which it was confirmed that the switching from foetal to adult haemoglobin production occurred at a time related to the postmenstrual age. 6
Conclusion
The HbF concentrations at the postnatal age are higher when the GA at birth is low. However, the HbF concentrations at the PMA did not differ according to the GA at birth. Therefore, it is better to evaluate the HbF concentration using the PMA rather than using the postnatal age.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is partially supported by the Practical Research Project for Intractable Diseases from Japan Agency for Medical Research and Development, AMED. This work was supported in part by grants from Arkray, Inc.
Ethical approval
This study was approved by the institutional review board of the Kobe University Graduate School of Medicine (reference number: 1652). All procedures performed in studies involving human materials were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Anonymous residual blood samples, obtained after performing routine laboratory tests, were used. We also opened this study project to the public using the notice board in our hospital and our homepage; therefore, formal consent was not required for this type of study.
Guarantor
IM.
Contributorship
All authors contributed to the intellectual content of this manuscript. YW, KO and IM wrote the first draft of this manuscript. YW, IS, SI, RK, IH, NH and JS collected the clinical samples and data. YW, KO, IS, SI and IM performed analysis of the clinical and laboratory findings. KO, IS, KI and IM designed this study. YW and KO contributed equally to this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
