Abstract
Purpose
The primary goal was to estimate reference values of parathyroid hormone (PTH) in very low birth weight infants without severe neonatal morbidity. A secondary objective was to assess the relationship between PTH serum levels and selected laboratory markers of bone metabolism.
Methods
Ninety two infants with birth weight less than 1500 g met the inclusion criteria of the study. Serum levels of PTH, 25-hydroxyvitamin-D [25(OH)D], C3-epi-25(OH)D, total calcium, phosphorus, and alkaline phosphatase, and urinary levels of calcium, phosphorus, and creatinine were examined on day 14 and subsequently every 2 weeks until discharge.
Results
Of the total 167 serum samples examined for PTH levels in infants without 25(OH)D deficiency the estimated range was 0.9–11.9 pmol/l (8.5–112.3 pg/mL). During the first month, no statistically significant correlation was observed between PTH level and that of 25(OH)D, C3-epimers of 25(OH)D, S-Ca, S-P, or ALP, nor with urinary excretion of calcium and phosphorus. From the second month of life, there was a moderately significant correlation between PTH and 25(OH)D (Rho = −0.40, P =< .001), between PTH and calcium/creatinine ratio (Rho = −0.56, P = < .001), and between PTH and phosphorus/creatinine ratio (Rho = 0.51, P = < .001).
Conclusions
The physiological range for PTH levels for preterm neonates without 25(OH)D deficiency was estimated as 0.9–11.9 pmol/l (8.5–112.3 pg/mL). It seems that elevation of serum PTH above this range can be considered as hyperparathyroidism in very low birth weight infants.
Introduction
The mechanisms governing calcium (Ca) and phosphorus (P) homeostasis and regulation in the foetus and early neonatal period differ significantly from those in children and adults. During foetal bone mineralisation, the primary regulators are parathyroid hormone (PTH) and parathyroid hormone-related peptide (PTHrP).1,2 Calcitonin, calcitriol, and fibroblast growth factor-23 (FGF-23), pivotal in the regulation of bone mineralization for both adults and children, are dispensable for the in utero transfer of minerals from the placenta to the foetus.1,2
After birth, the parathyroid gland releases a polypeptide known as PTH in response to the detection of low calcium levels in the bloodstream. Parathyroid hormone promotes the synthesis of active vitamin D and calcitriol (1,25-dihydroxycholecalciferol) in the kidneys, facilitating the absorption of calcium (and, to a lesser extent, phosphorus) from the intestine. In preterm infants, this pathway becomes fully operational by the end of the first month. Working in tandem with calcitriol, PTH regulates calcium and phosphate levels, exerting its effects in the bones, kidneys, and small intestine. 3
Metabolic bone disease (MBD) emerges as a complication of prematurity, marked by insufficient osteoid tissue and bone mineral content, accompanied by biochemical changes in phospho-calcium metabolism. Since the majority of calcium and phosphorus accumulation in utero takes place in the third trimester, many of these infants have insufficient mineral reserves, putting them at risk for calcium and phosphorus deficiencies. 4 It has been reported that the incidence rate of MBD is as high as 32 % of very low birth weight infants and 54 % of extremely low birth weight infants. 5 Metabolic bone disease can arise from various nutritional and biomechanical factors. The primary factor remains a deficiency of essential nutrients such as vitamin D, calcium, and phosphorus. However, factors like prolonged immobilization, extended parenteral nutrition (particularly exceeding 3 weeks), and the use of medications that oppose or antagonize bone metabolism can also contribute significantly to MBD. 6 Screening for MBD in premature infants relies heavily on identifying risk factors. Despite the absence of normative data and clinical trials, there is no established evidence-based guideline for the diagnosis and timing of potential treatment for MBD in premature infants. 4 Common biochemical screening tools for MBD include assessing serum levels of ALP, phosphorus, and calcium, as well as urinary calcium and phosphorus excretion. Nevertheless, there is no singular precise biochemical marker exclusively used for diagnosing MBD.3,4,7 Some studies have suggested a connection between secondary hyperparathyroidism and MBD, particularly in newborns with the lowest weight category and multiple morbidities.8–12
Even so, only recently was serum PTH included in the investigation protocol of MBD in preterm infants.7,13 The reference range of plasma PTH in neonates is not well-established. It is speculated that preterm infants have PTH levels that are similar to those of adults. Based on our pilot study concerning 20 healthy preterm infants (birth weight of 1000–1500 g, gestational age of 27–31 weeks), it is possible to support this claim. 3 The aim of this study is to confirm in a larger cohort of preterm infants our previous conclusion, and to estimate reference values of PTH in very low birth weight infants with vitamin D serum level >50 nmol/l and without severe morbidity.
Methods
Design
This was a single-centre, prospective, observational study.
Study population
The study was approved by the local Ethics Committee of The University Hospital Hradec Kralove (reference number 2013010 S11P and 201510 S14P). Preterm newborns from two different periods were included to the study in the Perinatology Centre of The University Hospital Hradec Kralove. At first, we used data from our pilot study concerning all newborns with birth weight 1000–1500 g admitted to the neonatal intensive care unit in the period from November 2013 until September 2014 (and born inclusively between the 27th and 31st gestational week). 3 Secondly, to encompass a larger number of preterm infants we included all neonates with birth weight under 1500 g in the period from January 2015 until December 2016. We considered only preterm newborns without other severe neonatal morbidity; exclusion criteria were congenital developmental defect, intrauterine growth restriction (diagnosis of IUGR was made prenatally by obstetricians using a stage-based approach 14 ), broncho-pulmonary dysplasia (BPD, ≥ moderate), necrotizing enterocolitis (all stages), intraventricular haemorrhage (Grade III–IV), cystic periventricular leukomalacia, sepsis with multiple organ dysfunction syndrome, and newborns who died during the hospital stay.
Clinical and laboratory characteristics
The followed clinical characteristics included respiratory distress syndrome (RDS; diagnosis was based on the presence of respiratory distress in conjunction with radiological chest X-ray findings for RDS), surfactant administration, broncho-pulmonary dysplasia (definition of BPD according to Jobe and Bancalari 15 ), medically treated patent ductus arteriosus, pneumothorax, necrotizing enterocolitis (defined using the Bell staging criteria 16 ), intraventricular haemorrhage (defined by Volpe classification 17 ), cystic periventricular leukomalacia, early and late onset sepsis (culture-proven or clinically suspected sepsis treated with antibiotics for at least 5 days), retinopathy of prematurity (all ROP), length of hospital stay, discharge weight and height and days on respiratory support before discharge (nasal continuous positive airway pressure or mandatory ventilation), and bone mineral content and density before discharge from hospital stay.
Blood samples were taken from the umbilical cord and for the infants every 2 weeks until discharge from hospital stay. Blood withdrawals were made between 7 and 8 a.m. Blood was collected into a standard tube and was centrifuged for at least 10 min after blood draw. The umbilical cord blood and serum samples of preterm infants were subsequently analysed for PTH, calcium, phosphorus, and alkaline phosphatase (ALP). Residual serum was immediately taken to −80°C and stored until analysis for 25(OH)D2 and 25(OH)D3 levels and their C3-epimers. Nocturnal urine collection was taken from the second week of life until discharge from hospital stay. A 6-h nocturnal urine collection has been proved as the best correlation with the 24-hour one. 18 Urinary calcium and phosphorus excretion was subsequently investigated and expressed as calcium/creatinine and phosphorus/creatinine ratio.
PTH analysis
Parathyroid hormone was analysed by chemiluminescence immunoassay on Liaison XL (DiaSorin Deutschland GmBH, Dietzenbach, Germany) intended for the quantitative determination of 1–84 PTH without cross-reaction to the 7–84 PTH fragment in human serum. This is one of the two third-generation immunoassays. The reference range for adults is 1–7 pmol/l (9.4–66 pg/mL). The coefficient of variation is 5.3 %. 19 Serum samples had never been thawed.
Vitamin D analysis
The umbilical cord blood, and for preterm infants serum samples, were analysed for levels of 25(OH)D2 and 25(OH)D3, and their C3-epimers. 25-hydroxyvitamin D and the C3-epimers were determined using a modified method of isotope-dilution liquid chromatography-tandem mass spectrometry ID-LC MS/MS. 20 In order to resolve the isomers by LC-MS/MS, we used a pentafluorophenyl stationary phase.21,22 The measurement was performed on an Agilent 6490 Triple Quadrupole connected to an Agilent Infinity 1290 liquid chromatography system. The assay quantitatively measured both major vitamin D metabolites 25(OH)D2 and 25(OH)D3 independently, as well as C3 epimers. For the purpose of this study, we define total 25(OH)D as the sum of 25(OH)D2 and 25(OH)D3 levels. Likewise, total C3-epi-25(OH)D is the sum of C3-epi-25(OH)D2 and C3-epi-25(OH)D3 levels.
Parenteral and enteral nutrition protocol
Parenteral and enteral nutrition of the included newborns was administered according to the nutritional protocol in respect of current nutrition recommendations for preterm newborns.23–25 Enteral and parenteral vitamin D supply to all newborns included in the study was consistent with the ESPGHAN recommendation. 24
Dual-energy X-ray absorptiometry
Bone mineralisation was assessed by densitometry using a commercially available Dual-energy X-ray absorptiometry (DXA) system (Hologic Discovery A, manufacturer, Hologic Inc., 250 Campus Drive, Marlborough, MA 01752 USA) in AP Spine mode with Infant spine analysis protocol (APEX for Windows 7 version 4.5.3.2; Hologic) with an examination performed at hospital discharge. Dual-energy X-ray absorptiometry evaluated the bone mass of the lumbar spine (L1–L4). A single technician performed all evaluations. Bone mineral density (g/cm2) was obtained by dividing bone mineral content (g) by the projected bone area (cm2).
Statistical analysis
Data were analysed using NCSS 2021 Statistical Software (2021) (NCSS, LLC. Kaysville, Utah, USA, ncss.com/software/ncss). Qualitative data were presented as frequencies and percentages, while quantitative data were presented as mean with standard deviation (SD) or as median with interquartile range (IQR). Nonparametric Spearman correlation coefficient (Rho) was used to evaluate the relationship between quantitative parameters. The highest level of statistical significance for rejecting zero hypotheses considered in the study was P < .05. The biochemical results of PTH serum levels showed non-normal distribution, and reference ranges were established using a non-parametric method as the 2.5 and 97.5 percentiles after exclusion of outliers. 26 In addition, the PTH concentrations were profoundly analysed by various types of statistical tests (e.g. classical ANOVA with F-statistics, non-parametric Kruskal–Wallis ANOVA with Scheffe post hoc test of the mean dependence, Wilcoxon rank-sum tests, two-sample two-tailed Student’s t-tests, normal probability plots, histograms) to evaluate differences between the means and standard deviations for the particular age data subgroups. Finally, power analyses assuming the same or different standard deviations within the data subgroups were performed employing Matlab R2024 and IBM SPSS Statistics software, respectively. Using these statistical tests, significance of the mean changes at different ages with respect to the mean for the whole dataset and the sample size influence could be judged. However, it is necessary to highlight that the parametric tests are very sensitive to detect differences in skewed data, and caution should be especially taken if the parametric tests indicate significant population differences. Where applicable, additional non-parametric tests were performed and the results were briefly given comment in this study.
Results
Between November 2013 and September 2014 there were 45 newborns with birth weight 1000 g to 1500 g. 25 were excluded, most often due to serious morbidity. Between January 2015 and December 2016 there were 183 newborns admitted to NICU with birth weight below 1500 g. Of these, 111 newborns failed to enter the study (most often due to exclusion criteria). From both periods, 92 infants met the entry criteria for this study (Figure 1 –Flow diagram of the study group). Flow diagram of the study group.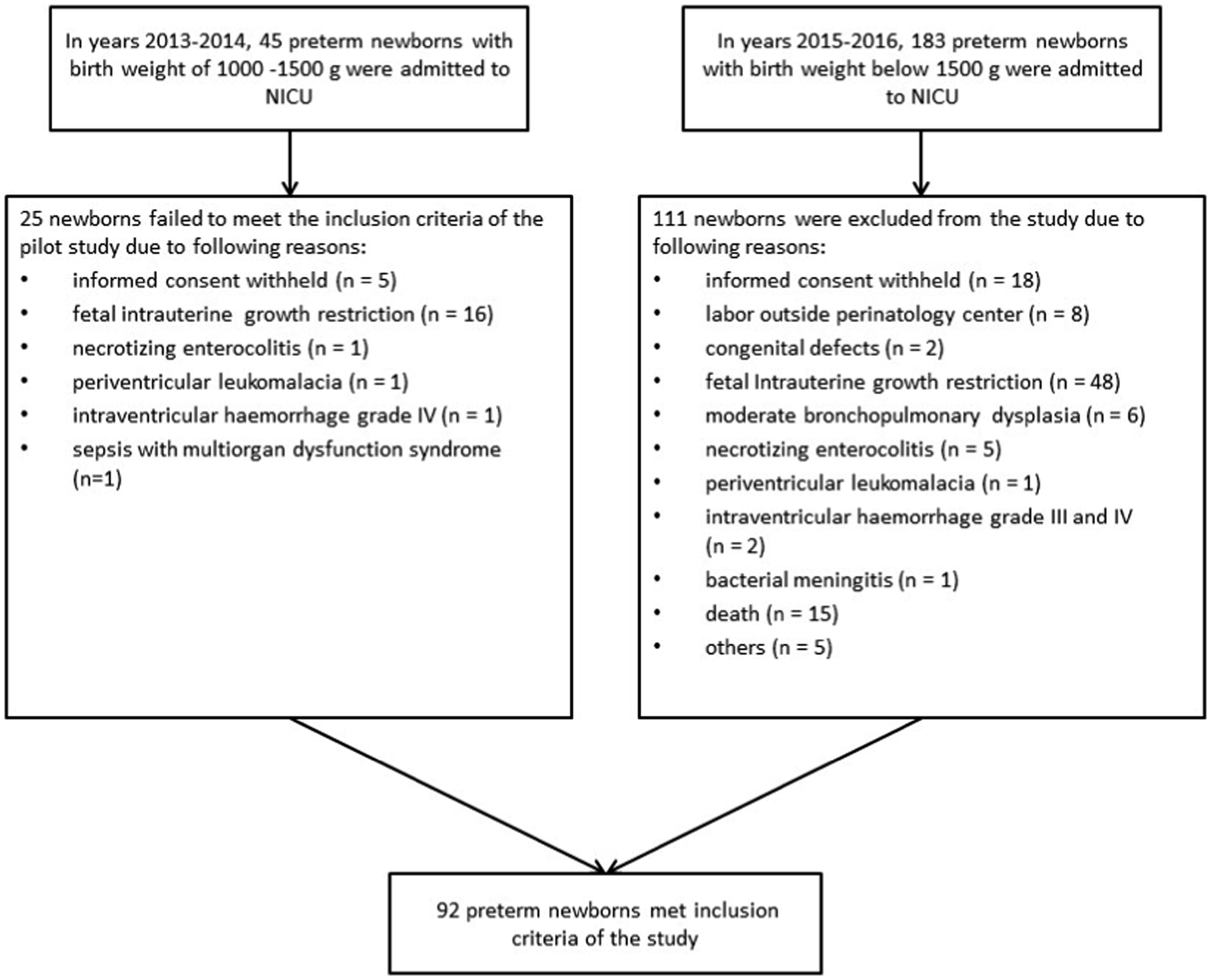
Demographic and clinical characteristics of the study group.
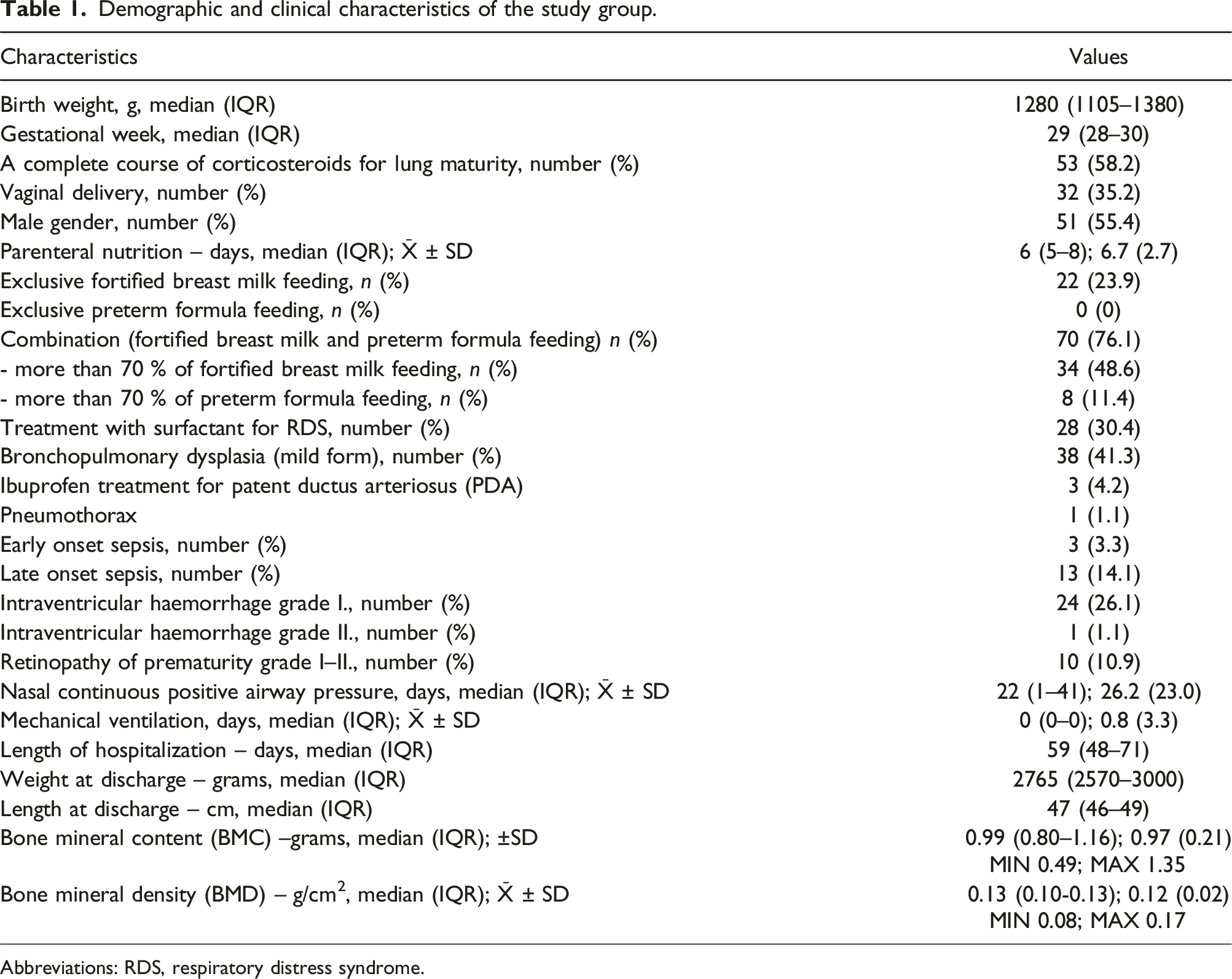
Abbreviations: RDS, respiratory distress syndrome.
Parathyroid hormone
Serum levels of parathyroid hormone (PTH), total calcium, phosphorus, alkaline phosphatase (ALP), vitamin D, and its C3-epimers over time.
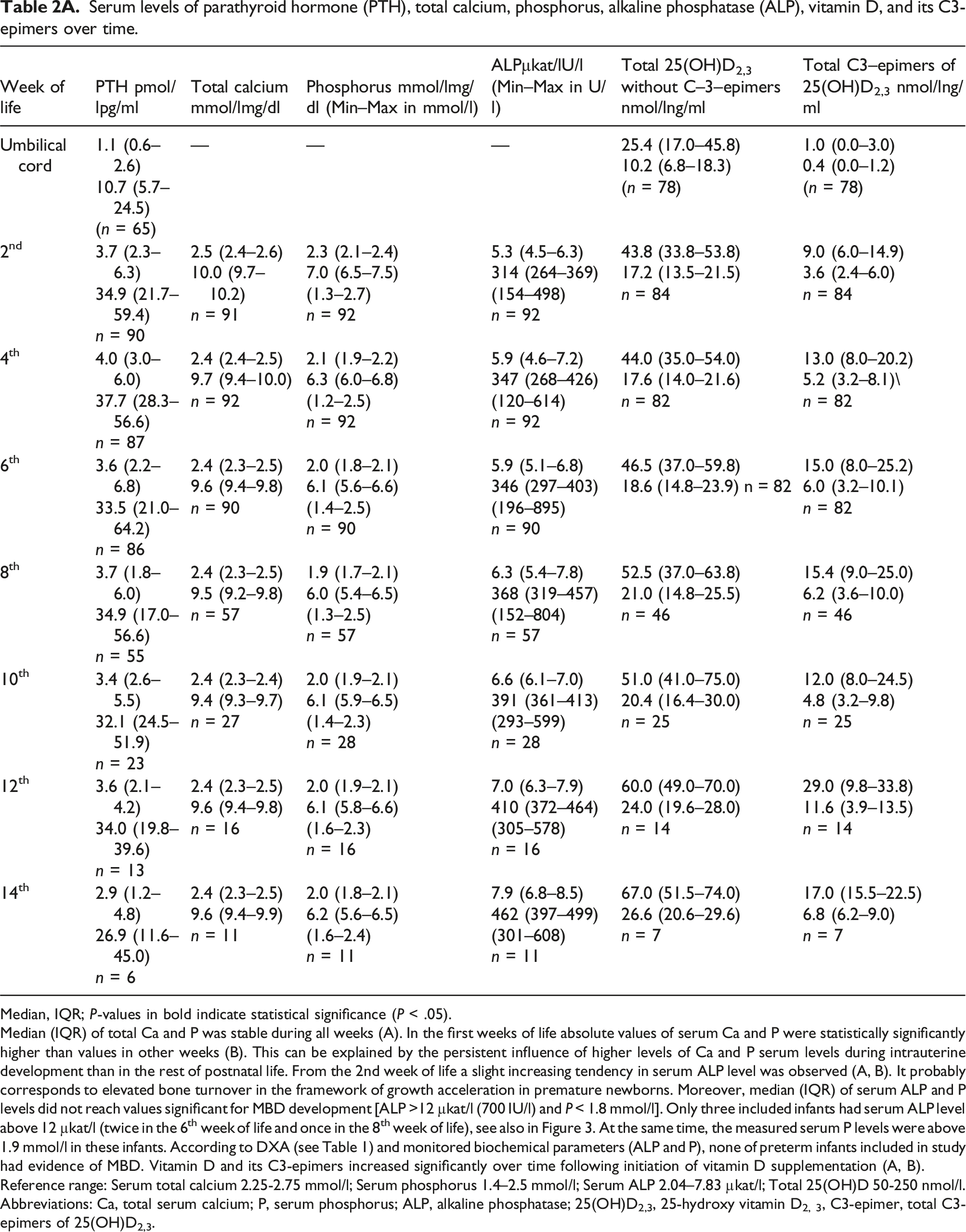
Median, IQR; P-values in bold indicate statistical significance (P < .05).
Median (IQR) of total Ca and P was stable during all weeks (A). In the first weeks of life absolute values of serum Ca and P were statistically significantly higher than values in other weeks (B). This can be explained by the persistent influence of higher levels of Ca and P serum levels during intrauterine development than in the rest of postnatal life. From the 2nd week of life a slight increasing tendency in serum ALP level was observed (A, B). It probably corresponds to elevated bone turnover in the framework of growth acceleration in premature newborns. Moreover, median (IQR) of serum ALP and P levels did not reach values significant for MBD development [ALP >12 µkat/l (700 IU/l) and P < 1.8 mmol/l]. Only three included infants had serum ALP level above 12 µkat/l (twice in the 6th week of life and once in the 8th week of life), see also in Figure 3. At the same time, the measured serum P levels were above 1.9 mmol/l in these infants. According to DXA (see Table 1) and monitored biochemical parameters (ALP and P), none of preterm infants included in study had evidence of MBD. Vitamin D and its C3-epimers increased significantly over time following initiation of vitamin D supplementation (A, B).
Reference range: Serum total calcium 2.25-2.75 mmol/l; Serum phosphorus 1.4–2.5 mmol/l; Serum ALP 2.04–7.83 µkat/l; Total 25(OH)D 50-250 nmol/l.
Abbreviations: Ca, total serum calcium; P, serum phosphorus; ALP, alkaline phosphatase; 25(OH)D2,3, 25-hydroxy vitamin D2, 3, C3-epimer, total C3-epimers of 25(OH)D2,3.
The level of significance (p-values) reported for the changes in bone markers between weeks.
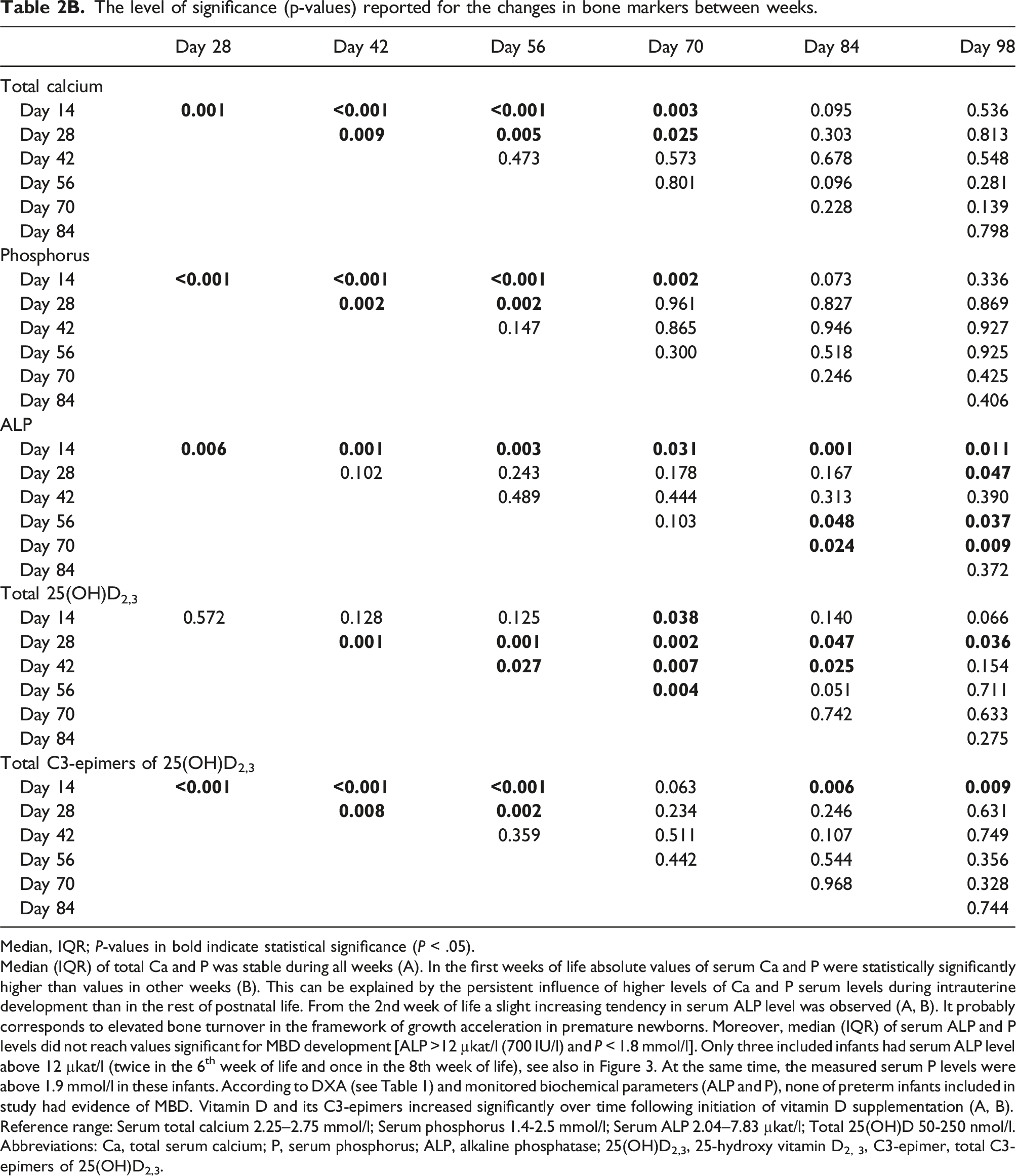
Median, IQR; P-values in bold indicate statistical significance (P < .05).
Median (IQR) of total Ca and P was stable during all weeks (A). In the first weeks of life absolute values of serum Ca and P were statistically significantly higher than values in other weeks (B). This can be explained by the persistent influence of higher levels of Ca and P serum levels during intrauterine development than in the rest of postnatal life. From the 2nd week of life a slight increasing tendency in serum ALP level was observed (A, B). It probably corresponds to elevated bone turnover in the framework of growth acceleration in premature newborns. Moreover, median (IQR) of serum ALP and P levels did not reach values significant for MBD development [ALP >12 µkat/l (700 IU/l) and P < 1.8 mmol/l]. Only three included infants had serum ALP level above 12 µkat/l (twice in the 6th week of life and once in the 8th week of life), see also in Figure 3. At the same time, the measured serum P levels were above 1.9 mmol/l in these infants. According to DXA (see Table 1) and monitored biochemical parameters (ALP and P), none of preterm infants included in study had evidence of MBD. Vitamin D and its C3-epimers increased significantly over time following initiation of vitamin D supplementation (A, B).
Reference range: Serum total calcium 2.25–2.75 mmol/l; Serum phosphorus 1.4-2.5 mmol/l; Serum ALP 2.04–7.83 µkat/l; Total 25(OH)D 50-250 nmol/l.
Abbreviations: Ca, total serum calcium; P, serum phosphorus; ALP, alkaline phosphatase; 25(OH)D2,3, 25-hydroxy vitamin D2, 3, C3-epimer, total C3-epimers of 25(OH)D2,3.
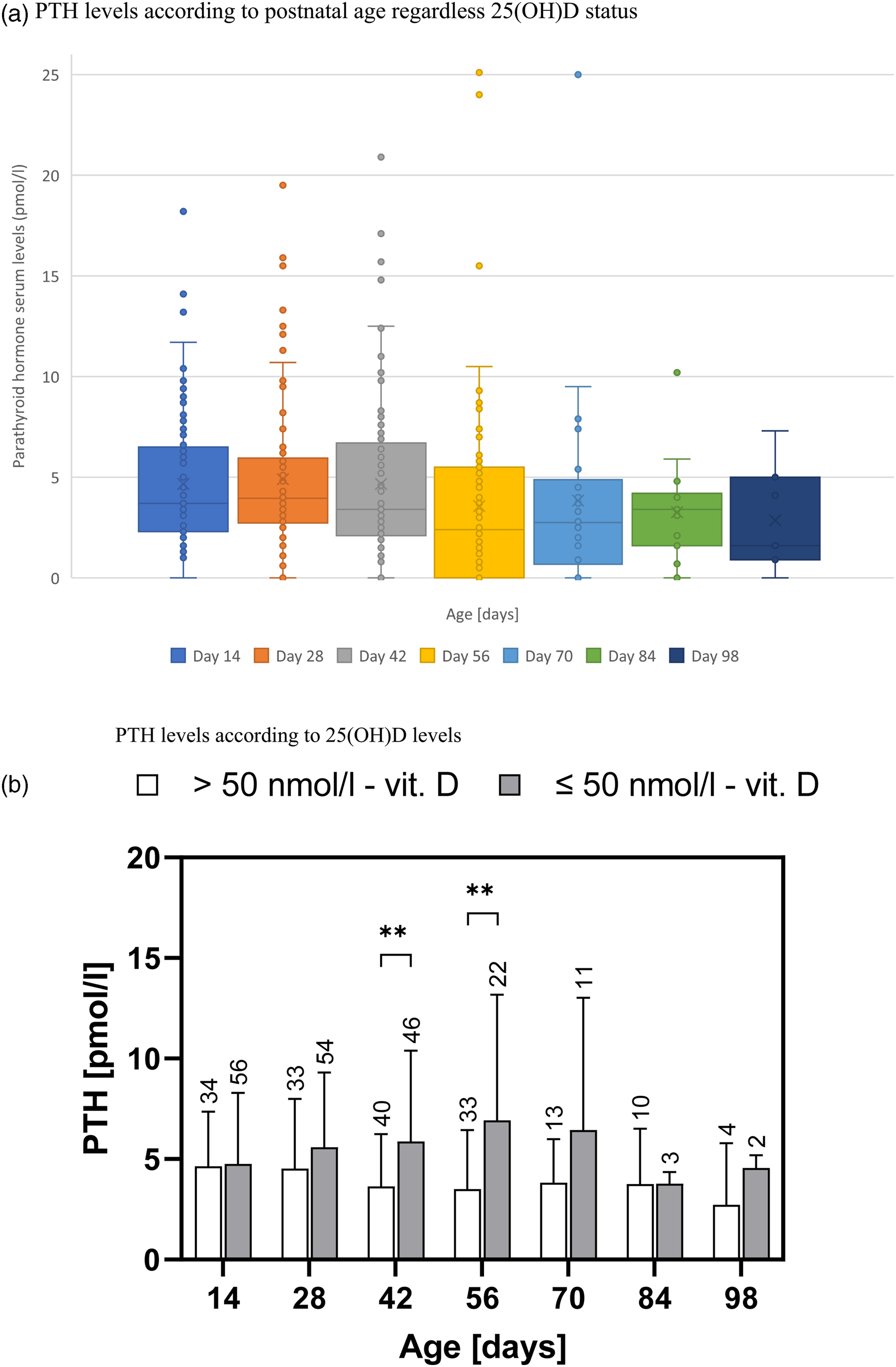
Parathyroid hormone serum levels over time in preterm infants. (a) PTH levels according to postnatal age regardless 25(OH)D status, (b)PTH levels according to 25(OH)D levels. (a) Each boxplot graph shows the whiskers from minimum [Q1–1.5xIQR] to maximum [Q3 + 1.5xIQR], first quartile [Q1], median, mean, third quartile [Q3], and outliers. (b) From the second month of life serum PTH level is significantly higher in preterm newborns with 25(OH)D deficiency. PTH levels for newborns with 25(OH)D levels <50 nmol/l (black) and for newborns with 25(OH)D levels >50 nmol/l (white) are expressed as the mean ± SD. P-values of the Student’s t-tests are annotated as * for P < .05, P < .01 as ** and P < .001 as ***. The numbers over the SD bars describe numbers of samples in each subgroup.
PTH reference range for preterm infants without 25(OH)D deficiency.
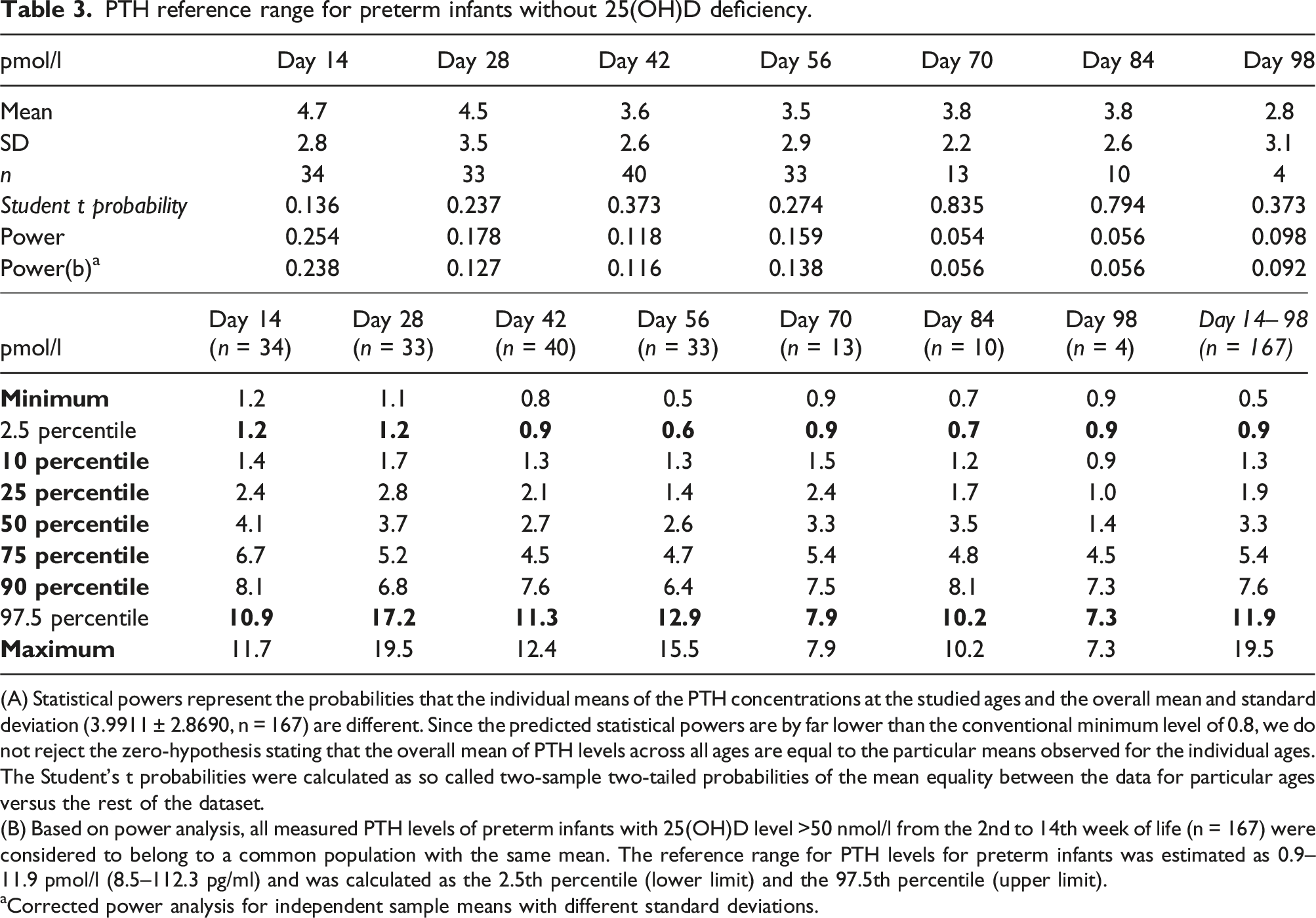
(A) Statistical powers represent the probabilities that the individual means of the PTH concentrations at the studied ages and the overall mean and standard deviation (3.9911 ± 2.8690, n = 167) are different. Since the predicted statistical powers are by far lower than the conventional minimum level of 0.8, we do not reject the zero-hypothesis stating that the overall mean of PTH levels across all ages are equal to the particular means observed for the individual ages. The Student’s t probabilities were calculated as so called two-sample two-tailed probabilities of the mean equality between the data for particular ages versus the rest of the dataset.
(B) Based on power analysis, all measured PTH levels of preterm infants with 25(OH)D level >50 nmol/l from the 2nd to 14th week of life (n = 167) were considered to belong to a common population with the same mean. The reference range for PTH levels for preterm infants was estimated as 0.9–11.9 pmol/l (8.5–112.3 pg/ml) and was calculated as the 2.5th percentile (lower limit) and the 97.5th percentile (upper limit).
aCorrected power analysis for independent sample means with different standard deviations.
Relationship between PTH and other bone metabolism markers
The measured levels of S-Ca, S-P, ALP, 25(OH)D, and its C3-epimers are summarised in Table 2A. The level of significance (P-values) reported for the changes in bone markers between postnatal weeks are presented in Table 2B. The relationship between PTH serum levels and selected serum and urinary bone metabolism markers is shown in Figure 3. In the first month of life, no significant correlation of PTH and other bone metabolism markers was found. From the second month of life, we can observe a slight tendency towards a moderately significant correlation only of 25(OH)D (Rho = −0.40, P = < .001; n = 164), urinary calcium/creatinine ratio (Rho = −0.56, P = < .001; n = 180) and urinary phosphorus/creatinine ratio (Rho = 0.51, P = < .001; n = 180). The correlation between PTH serum levels and selected serum and urinary bone metabolism markers. Legend: In the first month of life (column ‘A’) no significant correlation of PTH and other bone metabolism markers was found. From the second month of life (column ‘B’), we can observe a slight tendency towards a moderately significant correlation only with 25(OH)D (Rho = −0.40, P = < .001; n = 164), urinary calcium/creatinine ratio (Rho = −0.56, P = < .001; n = 180) and urinary phosphorus/creatinine ratio (Rho = 0.51, P = < .001; n = 180). Abbreviations: ALP, alkaline phosphatase.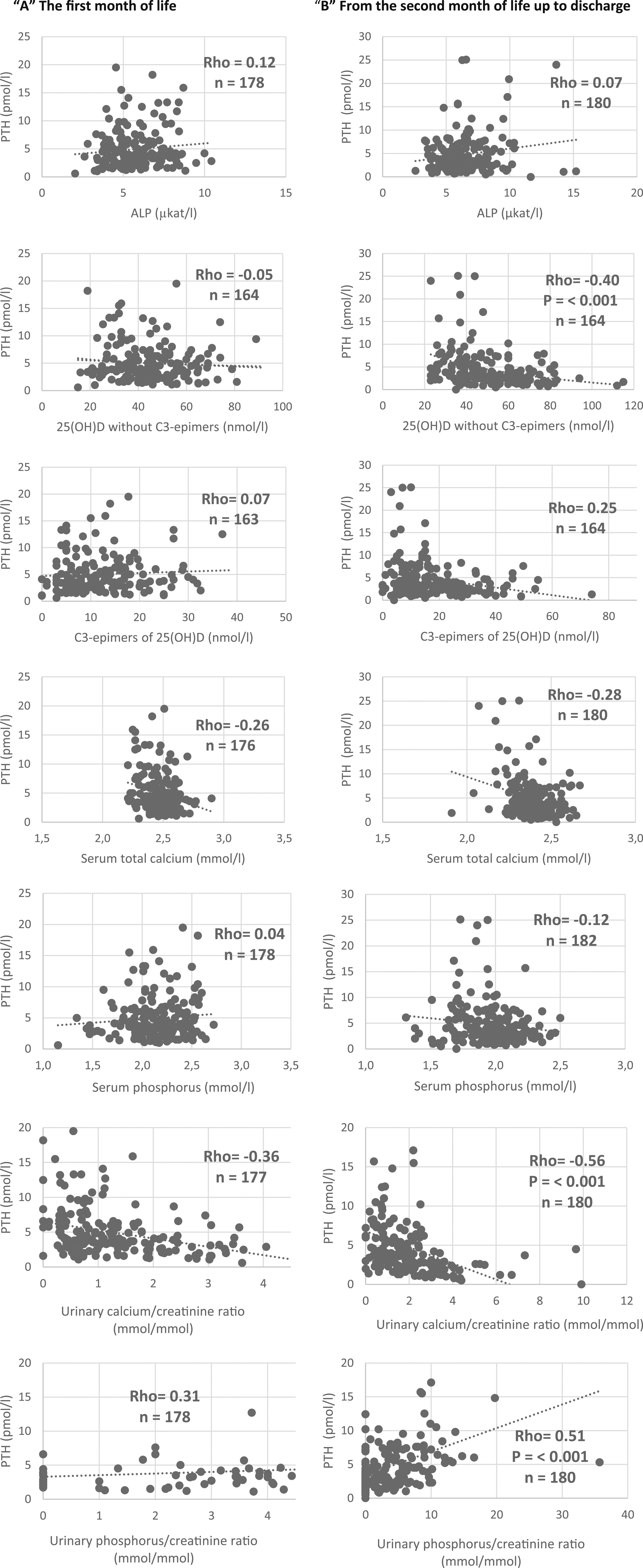
Discussion
Previously, we reported that the physiological range of serum PTH, indicated by the measurements in 20 preterm infants (birth weight 1000–1500 g only), was close to the reference limits for adults. 3 The primary goal of this prospective observational study was to estimate the physiological range of serum PTH levels using-third generation chemiluminescence immunoassay in a robust study group composed of the aforementioned 20 preterm infants and another 72 preterm infants with birth weight below 1500 g. To assess only ‘physiologically’ preterm infants, the study exclusively enrolled newborns devoid of severe morbidity and vitamin D deficiency, as these factors could substantially impact the measured values. In adults and children, the reference ranges for PTH typically show lower values when the serum level of 25(OH)D is taken into account.28,29 Our data are consistent with these observations and the difference reached statistical significance from the second month of life. Hence, based on prior suggestions, we opted to incorporate the 25(OH)D status for the calculation of PTH reference ranges.28,29 The PTH reference range was 0.9–11.9 pmol/l (8.5–112.3 pg/mL) and was calculated according to the latest recommendations.26,30
As far as we know, this is still the first study which has tried to assess physiological PTH values in very low birth weight infants. For this reason, we can only compare our results with several published reference ranges for the paediatric population. Ladang et al. 29 presented the PTH reference range in Belgian children according to age without 25(OH)D deficiency using the same third generation chemiluminescence immunoassay (LIAISON XL). In our cohort of preterm infants without 25(OH)D deficiency, the mean ± SD of PTH values were significantly higher compared to the children from 0 to 5 years old (n = 179) in the Ladang study (4.0 ± 2.9 vs 1.2 ± 0.4 pmol/l; P < .001 in two-sample two-tailed Student’s t-test). Despite the fact that PTH levels with second- and third-generation assays are not completely interchangeable, 30 we tried to compare our reference range with the CALIPER Cohort study 31 where immunoassay on the Abbott ARCHITECT i2000 was used to establish a reference range of intact PTH in Canadian infants up to 1 year (n = 193). Based on the statistical evaluation, it is possible to assert that both reference ranges are very similar (our study group 0.9–11.9 pmol/l vs CALIPER study group 0.7–9.3 pmol/l) and have the same mean (our study group 4.0 ± 2.9 pmol/l vs CALIPER study group 4.1 ± 4.7 pmol/l; P = .81 in two-sample two-tailed Student's t-test). It seems that the upper limit of the estimated range for PTH achieved higher values which are not observed in the paediatric or adult reference range (1–7 pmol/l; 9.4–66 pg/mL). This could be due to the low calcium stores in the foetal skeleton and the high prevalence of hypovitaminosis D in the preterm newborn population (to which the parathyroid glands respond by releasing PTH).1,32 After birth, this cannot be fully compensated by optimal parenteral or enteral nutrition in the population of preterm infants.
Several studies have appeared in recent years describing the presence of secondary hyperparathyroidism in preterm infants as a sign of MBD development.8–12 It is now recommended to use measurement of PTH in preterm infants, because it helps in establishing underlying calcium or vitamin D deficiency as a cause for MBD.4,7 We agree with Chinoy et al. 7 that monitoring the trend of its values will certainly precede changes in other markers of bone metabolism. It seems that elevation of serum PTH above our suggested range (>11.9 pmol/l/112.3 pg/mL) can be considered as hyperparathyroidism in preterm infants. Several studies have supported our claim. Moreira et al. 13 found that at 3 weeks chronological age, a PTH level >19.0 pmol/l (180 pg/mL) or a P level <1.45 mmol/l (4.6 mg/dl) yielded a sensitivity of 100 % and specificity of 94 % for MBD. Moreover, Motte-Signoret et al. 33 found a sharp increase in serum PTH levels [median (IQR) approximately 20 (16–31) pmol/l] at the age of 1 month in a very small group of preterm infants with MBD of prematurity (n = 9; defined for their study as ALP levels greater than 600 IU/L). Another work by Czech-Kowalska et al. 34 showed that serum PTH might be a simple predictor of reduced bone mineral content (BMC) in preterm infants examined at term age using whole body DXA. ROC analysis showed that serum PTH >4.6. pmol/l (43.6 pg/mL) provided 40% sensitivity and 88% specificity. However, the PTH value didn’t reach significance for hyperparathyroidism, and sensitivity was also quite low.
A secondary objective of the study was to elucidate the correlation between PTH and additional markers of bone metabolism. In our previous research, we reported and discussed severe vitamin D deficiency in umbilical cord samples.3,32 As observed in the pilot study, the correlation between PTH and vitamin D became statistically significant starting from the second month of life. 3 It is crucial to recognize that the absorption of calcium from the intestine in very preterm neonates primarily occurs through passive diffusion during the initial month of life. Simultaneously, within the first three to four weeks of life, the maturation of the intestinal mucous membrane leads to an increase in the absorption of vitamin D and the expression of calcitriol receptors and calcium-associated transport proteins (calbindin D9K, D28K). Over time, the dominant mechanism for calcium absorption from the intestine shifts to the calcitriol-controlled transport, which is regulated by PTH.1,3 Immature and unstimulated parathyroid glands during the first 3 weeks of life combined with maturation of calcitriol-controlled transport of Ca could be the reason for the observed statistically significant correlation between PTH and vitamin D only from the second month of life. As per our recent findings, the synthesis of C3-epi-25(OH)D remains operative even in the most preterm newborns, originates during the foetal stage, and is predominantly reliant on the presence of circulating 25(OH)D. At the end of the first month of life, C3-epimers constitute over 20% of 25(OH)D. 35 However, we did not observe any association between PTH and levels of C3-epimers in this study.
We did not find any statistically significant relationship between PTH and Ca, P, or ALP. We believe that normal serum Ca, P, and ALP values were achieved by a very good composition of parenteral and enteral nutrition in the cohort of preterm infants without severe morbidity. It is possible to speculate that this is the reason for not finding a significant relationship between PTH and Ca, P, or ALP. In contrast, new information has been discovered about the relationship between PTH and excretion of urinary Ca and P. From the second month of life the values of calcium/creatinine ratio and phosphorus/creatinine ratio correspond to standard knowledge regarding PTH control of Ca and P homeostasis. As PTH values increase, urinary Ca excretion decrease and urinary P excretion increase. We see a possible explanation in the fact that urinary excretions are more sensitive to fluctuations in serum PTH levels and 1,25(OH)2D. While 1,25(OH)2D is the biologically active form of vitamin D, the exclusive biomarker for assessing vitamin D status should be 25(OH)D. Serum levels of 1,25(OH)2D do not closely correlate with the overall outcomes of vitamin D exposure and may remain normal until significant vitamin D depletion occurs. 4
The strength of the study is the addition of an objective skeletal imaging method for diagnosis of MBD to the validity of the estimated reference interval for PTH. Dual X-ray absorptiometry (DXA) is employed when X-rays can identify changes at the point of 20%–40% demineralization. 3 Dual-energy X-ray absorptiometry is regarded as the optimal technique for assessing bone mass in infants due to its speed, safety, accuracy, reproducibility, and minimal exposure of the patient to ionizing radiation. 36 For the purpose of this study, we used DXA of the lumbar spine to determine BMC and BMD before discharge from hospital stay. According to the study by Figueras-Aloy et al., 14 a BMD value for the lumbar spine >0.068 g/cm2 at discharge indicated a 90.3% probability of not developing MBD of prematurity. The mean (±SD) of BMD was 0.12 (0.02) g/cm2 in our cohort, which should indicate very good skeletal health. Our results are in accord with those from other studies that have measured DXA of the lumbar spine in preterm infants at discharge from hospital stay. The mean (±SD) of BMD was 0.139 (0.06) g/cm2 in the work by Yeste et al. involving 80 preterm infants. 36 In another study on 49 preterm infants, the mean (±SD) of BMD of the lumbar spine was 0.10–0.11 (0.02) g/cm2 at discharge from hospital stay. 37 Furthermore, we infer the favourable skeletal condition of our cohort by considering the normal values of commonly measured parameters in calcium-phosphate metabolism (S-Ca, S-P, and ALP). Among the potential limitations of this study, it is possible to include the use of third generation assays (1–84 PTH determination), since almost all cited studies used in the discussion determined intact PTH (iPTH).
Conclusion
Metabolic bone disease remains a significant comorbid condition in preterm, low birth weight infants. It is now recommended to use measurement of PTH in preterm infants, because it helps in establishing underlying calcium or vitamin D deficiency as a cause for MBD. This study has shown that the estimated reference range of PTH for very low birth weight infants is different to the reference range for children and adults. In preterm infants, serum levels of PTH above 11.9 pmol/l can be considered as hyperparathyroidism. Future studies are required to establish consensus guidelines for screening tests and monitoring parameters for infants at risk of MBD. In our opinion, with a knowledge of the reference range, PTH should already be included.
Footnotes
Acknowledgements
The authors are grateful to Ian McColl MD, PhD for assistance with the manuscript (proofreading) and to Rafael Dolezal, PhD for statistical analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University Hospital Hradec Kralove, Czech Republic project MH CZ - DRO (UHHK, 00179906). Access to statistical software was supported by the Ministry of Education, Youth and Sports of the Czech Republic through the e-INFRA CZ (ID:90254).
Ethical approval
The study was approved by the local Ethics Committee of The University Hospital Hradec Kralove (reference number 2013010 S11P and 201510 S14P).
Guarantor
TM.
Contributorship
Dr Tomas Matejek conceptualised and designed the study and the instrumental methods for data collection, coordinated and supervised data collection, drafted the initial manuscript, and critically reviewed and revised the final version of the manuscript. Dr Bara Zapletalova and Dr Jaroslav Stranik collected data, carried out the initial analyses, and critically reviewed and revised the manuscript. Dr. Lenka Zaloudkova coordinated and supervised biochemical analysis of the samples, and critically reviewed and revised the manuscript. Prof Vladimir Palicka conceptualized and designed the study, coordinated and supervised DXA investigation, and critically reviewed and revised the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Consent to participate statement
Informed written consent was obtained from parents before inclusion of the newborn into the study.
Data availability statement
The data that support the findings of this study are not publicly available due to their containing information that could compromise the privacy of research participants, but are available from the corresponding author (TM).
