Abstract
Background
To accurately assess hypogammaglobulinemia at birth, it is essential to determine the reference intervals of serum immunoglobulin (IgG) levels in newborns. In the present study, we determined the gestational age (GA)-/birth weight (BW)-dependent percentile-based reference intervals of serum IgG levels and converted them into simple formulas for practical use.
Methods
Serum IgG levels were measured in cord blood from 2902 newborns delivered at 22 to 41 weeks of GA or 264 to 4642 g of BW after exclusion of those with congenital disorders. Linear regression analysis was used to correlate GA and UC-IgG levels and BW and UC-IgG levels. After calculation of the percentile values of UC-IgG levels for each GA or BW, the distributions were approximated by the least-squares method. Fitness was evaluated by the coefficient of determination (R 2 ).
Results
Significant positive correlations were found both between GA and UC-IgG levels (r s = 0.790, P < 0.001) and BW and UC-IgG levels (r s = 0.626, P < 0.001). The distribution of the 5%ile of UC-IgG levels (Y) by GA or BW (X) was approximated as a straight line (Y = 37.5 *X – 775.8; Y = 0.161 *X + 95.34, respectively). The fitness was stronger in the GA-derived formula than the BW-derived formula (R 2 = 0.973 vs 0.913).
Conclusions
We established GA-/BW-dependent reference percentile-based intervals for serum IgG levels using cord blood from 2902 newborns without congenital disorders. Using GA-dependent reference intervals may be useful for assessing hypogammaglobulinemia at birth.
Introduction
Immunoglobulin G (IgG) is the only class of Ig that is transferred from the mother to the fetus across the placenta and plays a significant role for newborns as an immunological barrier against microorganisms. 1 Since most of the IgG transfer occurs in the third trimester, preterm newborns with gestational age (GA) < 28 weeks have lower serum IgG levels at birth. 2 Endogenous IgG synthesis does not begin until several months of life 3 ; thus, preterm and/or low-birth weight (BW) newborns have lower serum levels of IgG for several months after birth, which may account for their increased susceptibility to infection.
Especially for preterm and/or low-BW newborns, prophylactic administration of intravenous Ig (IVIG) has been used for preventing nosocomial infections in many studies. 4 Its effectiveness is controversial, which may be due to a lack of standardized definition of hypogammaglobulinemia at birth for IVIG administration. IVIG is not always well tolerated in newborns as it can increase serum viscosity in a dose-dependent manner, 5 which may result in unknown adverse effects. Thus, to accurately diagnose significant hypogammaglobulinemia at birth and further evaluate the effectiveness and safety of IVIG, it is essential to determine the reference intervals of serum IgG levels in newborns. Although several studies aimed to investigate reference intervals for GA and BW,6–8 they were limited by the small number of subjects. Moreover, the determined reference ranges were restrictive and inconvenient for practical use.
Recently, we established GA-dependent percentile-based reference intervals of serum albumin levels as a nutritional indicator, using cord blood from 2917 newborns without congenital disorders. 9 Furthermore, our determined reference intervals could be converted into simple formulas of linear approximations for each percentile of serum albumin levels for each GA. Herein, we determined the GA-/BW-dependent percentile-based reference intervals of serum IgG levels in a similar way. We then converted the percentile-based reference intervals into simple formulas for practical use and further investigated their clinical application.
Methods
Study design and participants
In this retrospective study, singleton newborns born at a tertiary perinatal center, Hyogo Prefectural Kobe Children’s Hospital Perinatal Center, between January 2004 and March 2020 were investigated. Since our hospital provides a full range of surgical services including cardiovascular and neurosurgery, sick newborns and fetuses with surgical diseases and/or chromosomal disorders are referred from the entire area of Kobe City.
This study was approved by the Ethics Committee of Hyogo Prefectural Kobe Children’s Hospital (R4-106). Written informed consent was received prenatally from the parents regarding the use of clinical data and umbilical cord venous blood for research purposes.
Clinical parameters
Clinical information was extracted from our hospital database. Maternal factors, such as mode of delivery and complications/medications during pregnancy, were studied. Neonatal factors including GA, BW, BW Z-score, Apgar scores, and sex were investigated. Small for GA (SGA) and large for GA (LGA) were defined as BWs below the 10%ile or above 90%ile for given GAs, respectively. The presence of chromosomal anomalies, congenital malformations, and serious congenital conditions were also retrieved.
Measurement method of UC-IgG levels
Cord blood was obtained from the umbilical cord vein by obstetrician-gynaecologists at delivery. After birth, the cord blood was centrifuged immediately at 1820×g for 10 min. As a routine assessment of fetal immunological status, IgG levels in umbilical cord serum (UC-IgG levels) were determined by turbidimetric immunoassay using Auto Wako IgG (Wako Pure Chemical Industries, Osaka, Japan) with the Alinity i system (Abbott Laboratories, IL, USA).
Determination of reference intervals for serum IgG levels
To establish reference intervals for UC-IgG levels in a homogeneous population of newborns, we excluded those with congenital disorders, including chromosomal anomalies, congenital malformations, and serious congenital conditions, as in our previous report. 9
Scatter plots were used to visualize the relationship between GA and UC-IgG levels and BW and UC-IgG levels in study newborns. To determine percentile-based reference intervals, the 5%ile, 10%ile, 50%ile, 90%ile, and 95%ile of UC-IgG levels (Y) for GA and BW (X) were calculated. Moreover, distributions were approximated by least-squares regression lines. Finally, their fitness was assessed by the coefficient of determination (R 2 ).
Biological differences related to sex and intrauterine growth
To evaluate the biological differences related to sex and intrauterine growth, scatter plots for UC-IgG levels by GA and BW were compared between male and female newborns, and among SGA, appropriate for GA (AGA), and LGA newborns.
Clinical application of the determined reference intervals for serum IgG levels
For the verification of the determined linear approximation for the 5%ile UC-IgG levels, UC-IgG levels in newborns with hydrops fetalis, gastroschisis, and fetomaternal haemorrhage (FMH) were used as known diseases causing hypogammaglobulinemia at birth.10–12 Hydrops fetalis was defined as the presence of abnormal accumulation of two or more fluids including ascites, pleural effusion, pericardial effusion, and skin edema. FMH was diagnosed in maternal blood using Kleinhauer–Betke stain.
For the exploratory application of the determined linear approximation for the 5%ile UC-IgG levels, UC-IgG levels in newborns with the following congenital disorders that are potentially associated with hypogammaglobulinemia at birth were used: chylothorax, congenital diaphragmatic hernia, congenital heart disease except patent ductus arteriosus, early-onset sepsis, gastrointestinal atresia/stenosis, meningomyelocele, omphalocele, trisomy 21, and trisomy 13/18.
Statistical analysis
Linear regression analysis was used to determine correlation between GA and UC-IgG levels and BW and UC-IgG levels. Correlation coefficients (r s ) were calculated using Spearman’s rank test. The distributions of UC-IgG levels by GA and BW were approximated by least-squares regression lines. Fitness was evaluated by the coefficient of determination (R 2 ). All analyses were conducted using GraphPad Prism V8.4 (GraphPad Software, San Diego, CA, US).
Results
Study population
Clinical characteristics of study newborns.
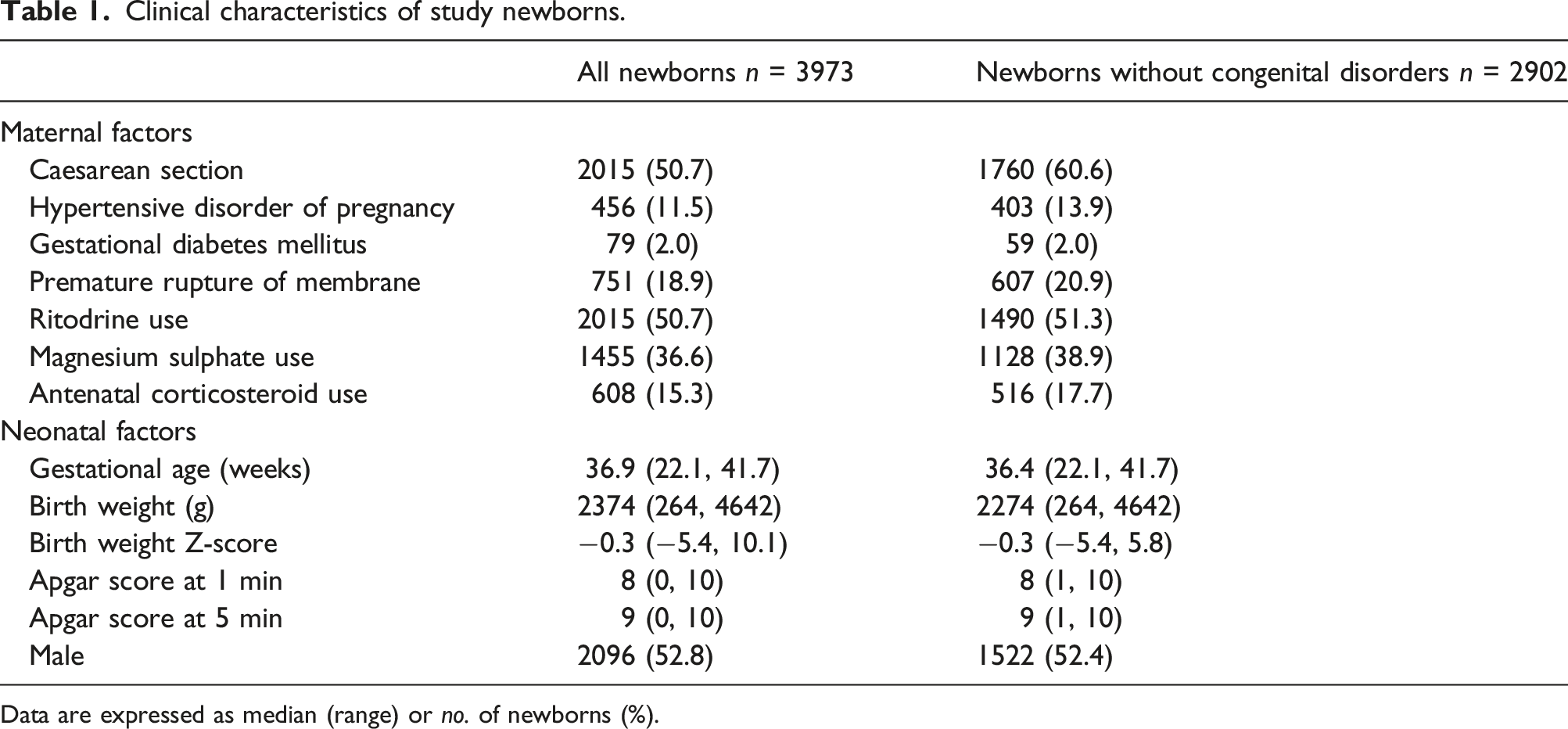
Data are expressed as median (range) or no. of newborns (%).
Determination of reference intervals of UC-IgG levels
In 2902 newborns without congenital disorders, UC-IgG levels for each GA were scattered (Figure 1(a)). A significant positive correlation was found between GA and UC-IgG levels (Y = 55.7 *X – 1163.0, r
s
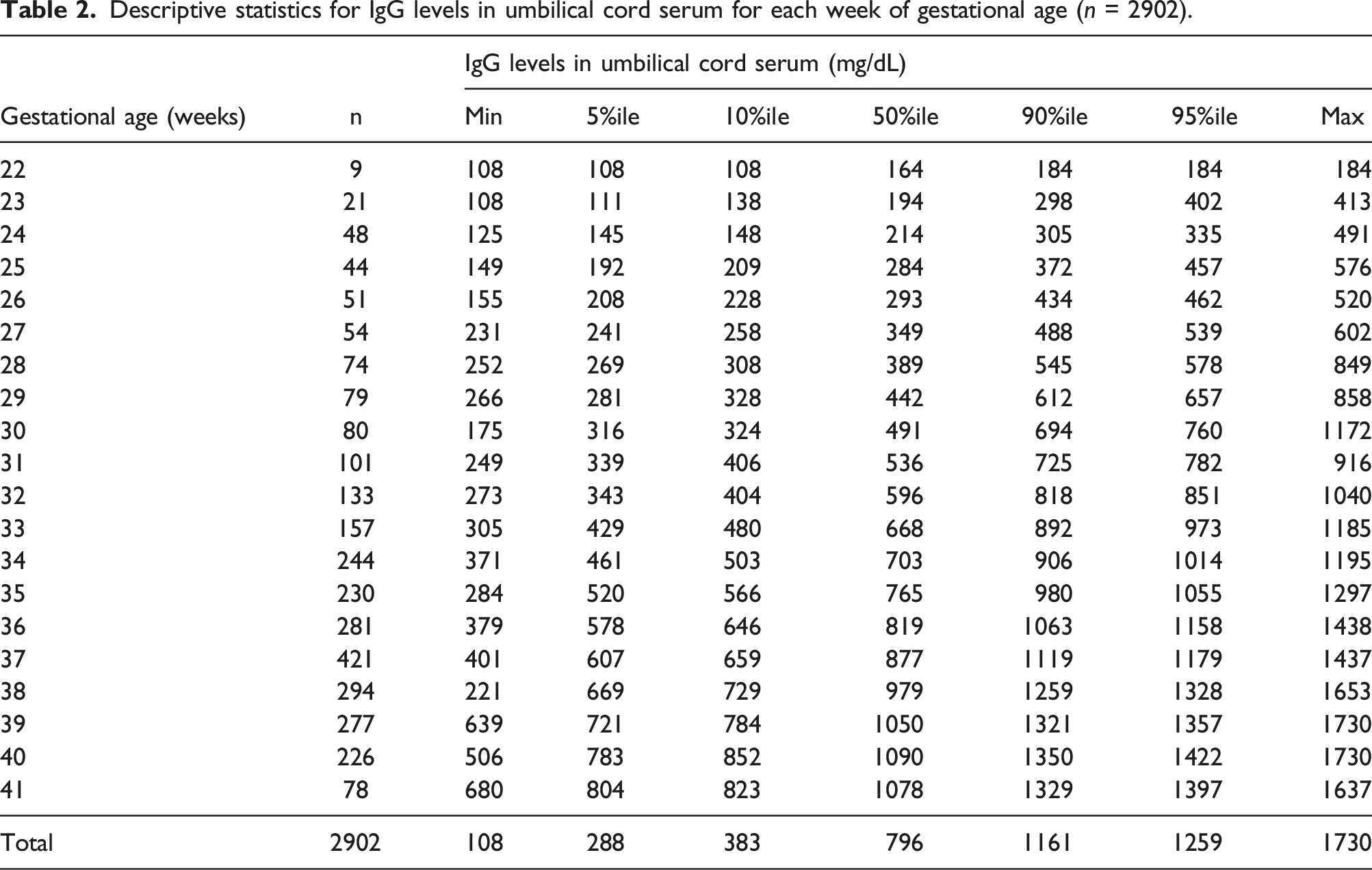
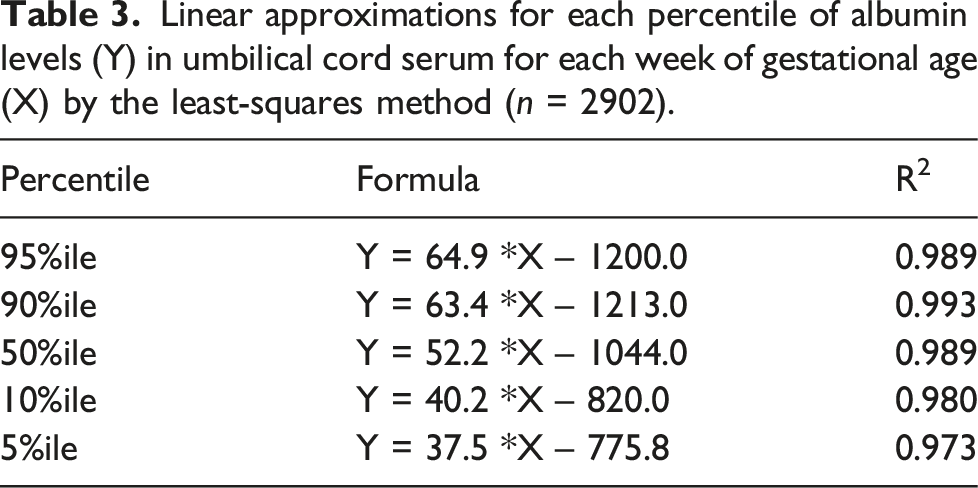
= 0.790, P < 0.001). The distributions of the 5%ile, 50%ile, and 95%ile of UC-IgG levels (Y) were calculated for each GA (X) (Table 2) and approximated as straight lines by the least-squares method (Y = 37.5 *X – 775.8, Y = 52.2 *X – 1044.0, and Y = 64.9 *X – 1200.0, respectively) (Figure 1(b)). These formulas strongly fit each percentile of the UC-IgG levels for each GA (R
2
= 0.973, 0.989, and 0.989, respectively) (Table 3). Gestational age (GA)-/birth weight (BW)-dependent distribution of serum immunoglobulin G levels in umbilical cord serum (UC-IgG) in newborns without congenital disorders (n = 2902). (a) Scatter plot for UC-IgG levels by GA in newborns without congenital disorders. A significant positive correlation was found between GA and UC-IgG levels (Y = 55.7 *X – 1163.0, r
s
= 0.790, P < 0.001). (b) After determination of the 5%ile (▲), 50%ile (◯), and 95%ile (▼) of UC-IgG levels for each week of GA, linear approximation for the three percentile values (Y) by GA (X) was performed by the least-squares method (Y = 37.5 *X – 775.8, Y = 52.2 *X – 1044.0, and Y = 64.9 *X – 1200.0, respectively). (c) Scatter plot for UC-IgG levels by BW in newborns without congenital disorders. A significant positive correlation was found between BW and UC-IgG levels (Y = 0.244 *X + 247.9, r
s
= 0.626, P < 0.001). (d) After determination of the 5%ile (▲), 50%ile (◯), and 95%ile (▼) of UC-IgG levels for each week of BW, linear approximation for the three percentile values (Y) by GA (X) was performed by the least-squares method (Y = 0.161 *X + 95.34, Y = 0.211 *X + 252.2, Y = 0.224 *X + 611.4, respectively). Descriptive statistics for IgG levels in umbilical cord serum for each week of gestational age (n = 2902). Linear approximations for each percentile of albumin levels (Y) in umbilical cord serum for each week of gestational age (X) by the least-squares method (n = 2902).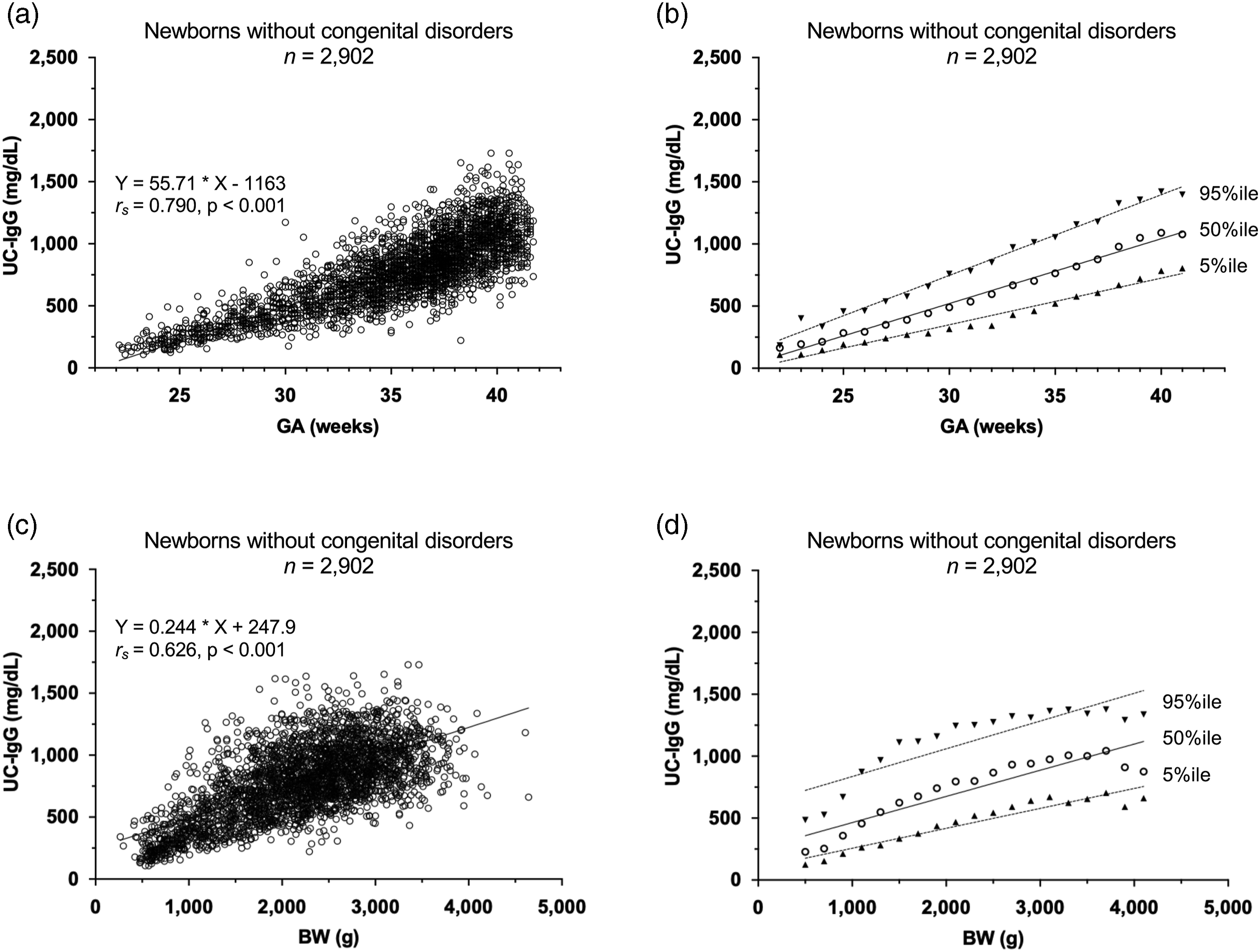
UC-IgG levels for each BW were scattered (Figure 1(c)). A significant positive correlation was found between BW and UC-IgG levels (Y = 0.244 *X + 247.9, r s = 0.626, P < 0.001). The distributions of the 5%ile, 50%ile, and 95%ile of UC-IgG levels (Y) were calculated for BW (X) (Table S2) and approximated as straight lines by the least-squares method (Y = 0.161 *X + 95.34, Y = 0.211 *X + 252.2, and Y = 0.224 *X + 611.4, respectively) (Figure 1(d)). These formulas fit each percentile of the UC-IgG levels for each BW (R 2 = 0.913, 0.840, and 0.766, respectively) (Table S3); however, the coefficients of determinations were relatively low. Based on the results of fitness tests, GA-dependent reference interval was used for subsequent studies.
Biological differences related to sex and intrauterine growth
To evaluate the biological difference related to sex, scatter plots for UC-IgG levels by GA were compared between male (n = 1522) and female newborns (n = 1380). Similar significant positive correlations were observed between GA and UC-IgG levels in male and female newborns (Y = 56.3 *X – 1204.0, r
s
= 0.646, P < 0.001 and Y = 54.0 *X – 1120.0, r
s
= 0.651, P < 0.001, respectively) (Figure 2(a) and (b)). Scatter plot for immunoglobulin G levels in umbilical cord serum (UC-IgG) by gestational age (GA) between male (n = 1522) and female newborns (n = 1380) and among newborns with small for GA (SGA) (n = 702), appropriate for GA (AGA) (n = 1969), and large for GA (LGA) (n = 231). (a) A significant positive correlation was found between GA and UC-IgG levels in male newborns (Y = 56.3 *X – 1204.0, r
s
= 0.646, P < 0.001). (b) A significant positive correlation was found between GA and UC-IgG levels in female newborns (Y = 54.0 *X – 1120.0, r
s
= 0.651, P < 0.001). (c) A significant positive correlation was found between GA and UC-IgG levels in newborns with SGA (Y = 57.6 *X – 1218.0, r
s
= 0.551, P < 0.001). (d) A significant positive correlation was found between GA and UC-IgG levels in newborns with AGA (Y = 54.8 *X – 1154.0, r
s
= 0.679, P < 0.001). (e) A significant positive correlation was found between GA and UC-IgG levels in newborns with LGA (Y = 54.5 *X – 1042.0, r
s
= 0.672, P < 0.001).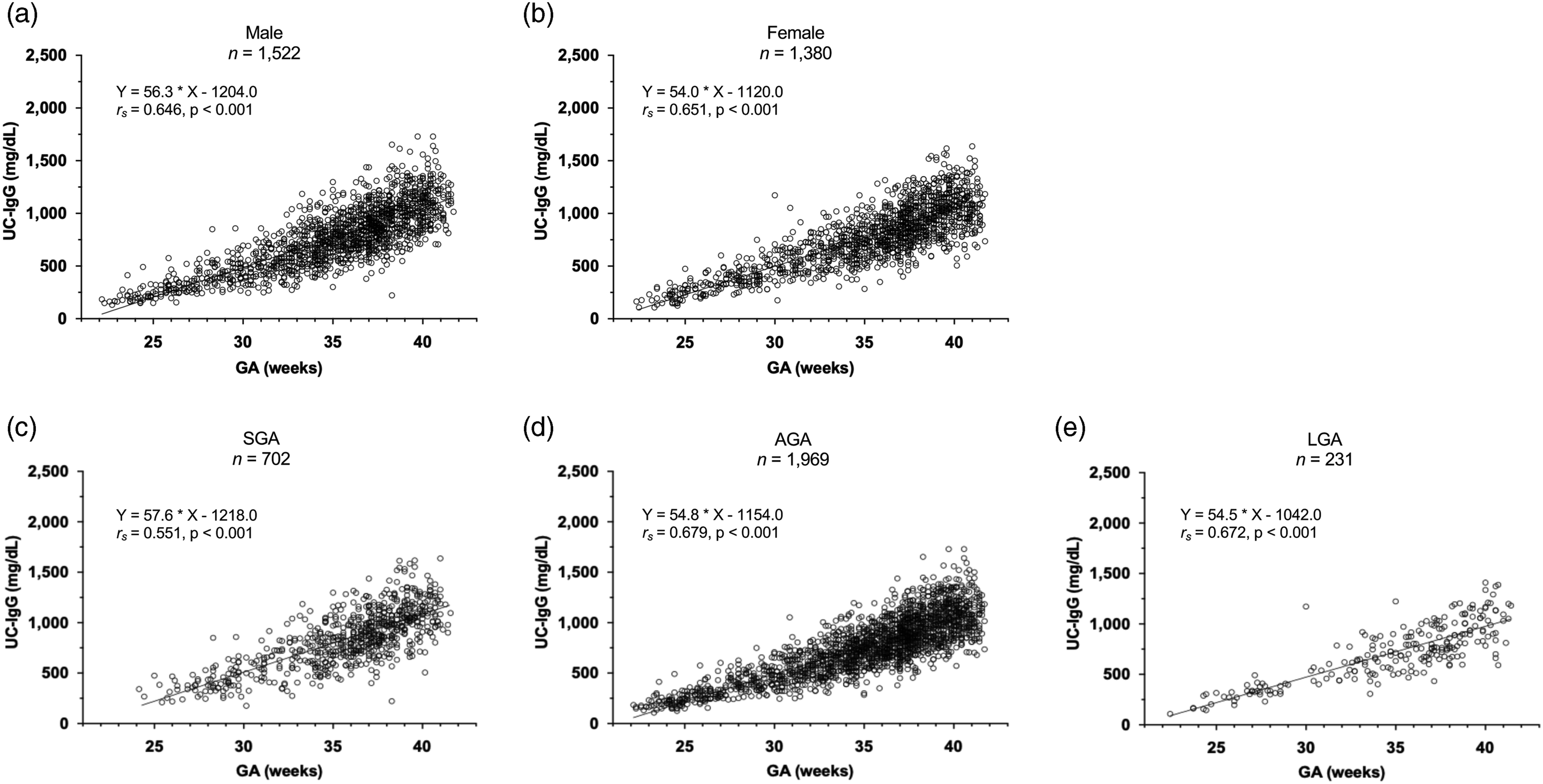
To assess the biological difference related to intrauterine growth, scatter plots for UC-IgG levels by GA were prepared among SGA (n = 702), AGA (n = 1969), and LGA newborns (n = 231). Furthermore, significant positive correlations were observed between GA and UC-IgG levels among SGA, AGA, and LGA newborns (Y = 57.6 *X – 1218.0, r s = 0.551, P < 0.001, Y = 54.8 *X – 1154.0, r s = 0.679, P < 0.001, and Y = 54.5 *X – 1042.0, r s = 0.672, P < 0.001, respectively) (Figure 2(c)–(e)).
Distribution of UC-IgG levels in newborns with congenital disorders
For the verification of the determined linear approximation for the 5%ile UC-IgG levels for each GA, UC-IgG levels for GA in newborns with hydrops fetalis, gastroschisis, and FMH were used. Among 64, 20, and 8 newborns presenting with hydrops fetalis, gastroschisis, and FMH, 51 (80%), 17 (85%), and 8 (100%), respectively, were lower than the line of linear approximation for the 5%ile of UC-IgG levels for each GA (Figure 3(a)–(c)). Scatter plot for immunoglobulin G levels in umbilical cord serum (UC-IgG) by gestational age (GA) in newborns with congenital disorders associated with hypogammaglobulinemia. The UC-IgG levels for GA in the newborns with hydrops fetalis, gastroschisis, and fetomaternal haemorrhage were scattered using three linear approximations for the 5%ile, 50%ile, and 95%ile of UC-IgG levels. (a) Among the 64 newborns with hydrops fetalis, 51 (80%) were below the line of linear approximation for 5%ile values. (b) Among the 20 newborns with gastroschisis, 17 (85%) were below the line of linear approximation for 5%ile values. (c) Among the eight newborns with fetomaternal haemorrhage, eight (100%) were below the line of linear approximation for 5%ile values.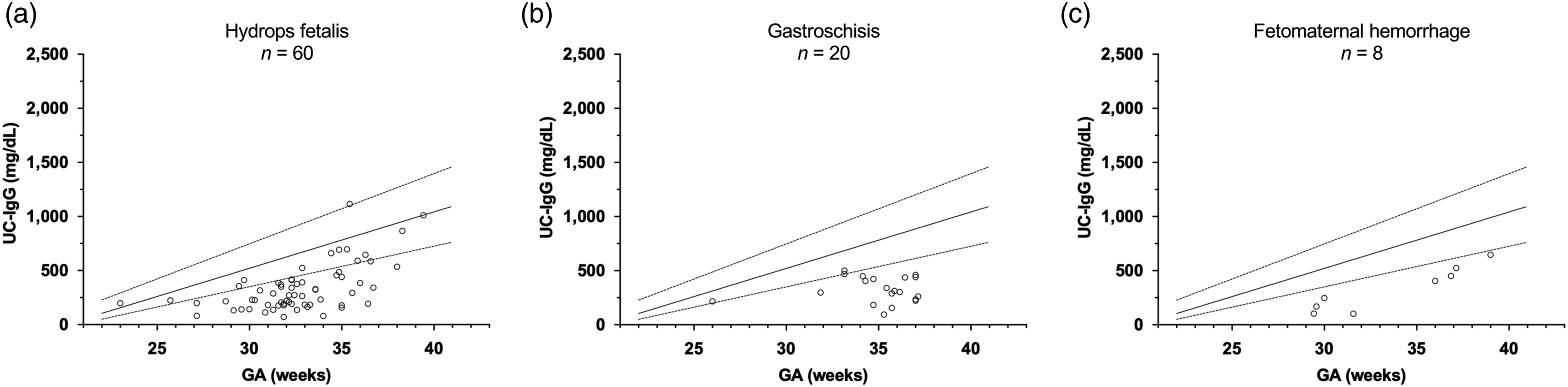
For the exploratory application of the determined linear approximation for the 5%ile UC-IgG levels for each GA, the UC-IgG levels in newborns with other congenital disorders were applied as potential diseases associated with hypogammaglobulinemia at birth (Figure S1(a)-(i)). The rates of newborns below the line of linear approximation were 78, 19, 17, 10, 25, 14, 33, 38, and 64% for those presenting with congenital chylothorax, congenital diaphragmatic hernia, congenital heart disease except patent ductus arteriosus, early-onset sepsis, gastrointestinal atresia/stenosis, meningomyelocele, omphalocele, trisomy 21, and trisomy 13/18, respectively.
Discussion
To date, several reference intervals of serum IgG in newborns have been reported.6–8 However, the reference intervals determined based on GA and BW were limited due to the small sample size and restrictive application. Herein, we established GA-/BW-dependent percentile-based reference intervals of serum IgG levels (Table 2 and Table S2) and converted them into simple formulas for easier practical use (Table 3 and Table S3). Notably, the GA-dependent reference interval showed stronger fitness between the formulas and each percentile of the UC-IgG levels than the BW-dependent reference interval. These findings suggest that the placental transfer of IgG is primarily based on GA rather than fetal body growth. Thus, GA-dependent reference interval of serum IgG would be more appropriate for practical use in clinical settings.
In our assessment of biological differences related to sex and intrauterine growth, 12 the distributions of UC-IgG levels by GA were very similar between 1522 males and 1380 females, and among 702 SGA, 1969 AGA, and 231 LGA newborns. To the best of our knowledge, sex differences of serum IgG levels in newborns have not been reported. A previous observational study reported lower serum IgG levels in SGA newborns than AGA newborns; however, the sample size was small (47 SGA and 197 AGA newborns). 13 Our dataset with a larger sample size revealed that no clinically significant differences were observed in UC-IgG levels between male and female newborns, and among SGA, AGA, and LGA newborns. These findings support the reliability of our determined GA-dependent percentile-based reference interval for serum IgG levels at birth.
In verification analyses of the determined linear approximation for the 5%ile of UC-IgG levels for each week of GA, the rates of newborns below the line were 78, 85, and 100% among those presenting with hydrops fetalis, gastroschisis, and FMH, respectively (Figure 3), indicating adequate validation of GA-dependent reference ranges. Additionally, UC-IgG levels in newborns with other congenital disorders were applied as potential diseases associated with hypogammaglobulinemia at birth (Figure S1). Among the nine congenital disorders investigated, congenital chylothorax, trisomy 21, and trisomy 13/18 showed high rates (78, 38, and 64%, respectively) of newborns below the line. The pathological condition of congenital chylothorax may overlap with hydrops fetalis. Chromosomal anomalies frequently co-occur with congenital chylothorax and hydrops fetalis. Therefore, our GA-dependent percentile-based reference interval and the converted formulas should be validated for clinical assessment of fetal pathology related to the development of hypogammaglobulinemia at birth in such congenital disorders.
The definition of hypogammaglobulinemia at birth remains to be elucidated. Some studies reported that serum IgG level <200 mg/dL should be considered as physiologic; however, other studies reported that <600 mg/dL was pathologic. 14 When our determined formula for the 50%ile of UC-IgG levels for each week of GA was applied, preterm newborns with 24 weeks’ GA could have serum IgG levels of 200 mg/dL. Since such newborns cannot synthesize IgG for several months, they would have a higher risk for severe infection after birth. Although a cutoff value of serum IgG levels for adequately preventing infection in newborns remains to be elucidated, our derived formulas may be valuable for assessing the severity of hypogammaglobulinemia and determining the appropriate criteria for IVIG administration in preterm newborns.
Our study has several limitations. First, this study was performed at a single tertiary center; thus, unknown center-specific factors may have affected the distribution of UC-IgG levels. To validate our determined reference intervals, further multicenter studies are needed. Second, we could not analyze maternal serum IgG levels, which have been reported to affect serum IgG levels in newborns. 2 Third, multiple births were not investigated in this study. However, our determined reference intervals from singleton newborns could be useful to assess the pathological condition in multiple birth-related complications such as twin-to-twin transfusion syndrome.
Conclusion
We determined GA-/BW-dependent percentile-based reference intervals for serum IgG levels using cord venous blood from 2902 newborns without congenital disorders. Our findings suggest that GA-dependent reference intervals are more practical for assessing hypogammaglobulinemia at birth.
Supplemental Material
Supplemental Material - Determination and verification of reference intervals of serum immunoglobulin G at birth
Supplemental Material for Determination and verification of reference intervals of serum immunoglobulin G at birth by Toshihiko Ikuta, Sota Iwatani, and Seiji Yoshimoto in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We thank Drs. Emiko Takeoka, Sachiko Matsui, Shoko Tamaki, Hitomi Mimura, and Hideto Nakao, Department of Neonatology, for collecting clinical data. We also thank laboratory technicians at Hyogo Prefectural Kobe Children’s Hospital for their assistance with the serum immunoglobulin G measurements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Institutional Review Board of the Hyogo Prefectural Kobe Children’s Hospital (reference number: R4-106). All procedures performed in this study involving human materials were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained prenatally from the newborns’ parents regarding the use of clinical data and umbilical cord venous blood for research purposes.
Guarantor
S.I.
Contributorship
T.I. and S.I. designed and performed the study. T.I., S.I., and S.Y. are neonatologists who collected the clinical samples and data. T.I. and S.I. wrote the manuscript. S.Y. supervised the manuscript. All authors approved the final manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
