Abstract
Background
Methods
Porcine unconjugated bilirubin solution, chemically synthesized ditaurobilirubin solution, and chemically synthesized delta bilirubin solution were used as surrogates of naturally occurring unconjugated bilirubin, conjugated bilirubin, and delta bilirubin, respectively. The total bilirubin and direct-reacting bilirubin concentrations were measured by three bilirubin oxidase methods and one vanadic acid method, and the observed concentrations were compared with those obtained by the diazo-based reference measurement procedure.
Results
The unconjugated bilirubin and delta bilirubin concentrations were similar when any of the four
Conclusions
We revealed the reactivity of IVD-TB and IVD-DB reagents to artificially prepared bilirubin materials, and their consistency with reference measurement procedure. The delta bilirubin data results vary depending on the reagents used.
Keywords
Introduction
In 2020, on behalf of the Committee on Enzymes and Analytical Reagents, Japan Society of Clinical Chemistry (JSCC), we aimed to define the nomenclature of serum bilirubin subfractions needed in clinical laboratory medicine.
1
Japanese manufacturers have developed various types of
In 1981, Murao and Tanaka
5
in Japan isolated an enzyme that catalysed bilirubin oxidation (BOX, EC: 1.3.3.5) from
Based on this background, a fundamental question of how each IVD bilirubin reagent developed in Japan reacted with DLB and accurately reacted with CB and UCB is raised. In the quality assurance survey
8
for TB and DB carried out by the Japan Medical Association in 2018, five kinds of IVD bilirubin reagents were compared. Of all laboratories (
Currently, ditaurobilirubin (DTB), a synthetic water-soluble bilirubin, is added in survey serum as a substitute for natural CB (College of American Pathologists survey). 10 Furthermore, DLB can be chemically synthesized using UCB and peptide synthesis reagent (Woodward's reagent K). 11 Additionally, crystalline powder of UCB isolated from porcine bile is commercially available. To our knowledge, no systematic study has been performed comparing the reactivity with UCB, DTB, and DLB simultaneously using the IVD bilirubin reagents developed in Japan. The aim of this study was to determine how the IVD bilirubin reagents developed in Japan reacted with DLB, DTB, and UCB. We also wanted to determine whether the assay values of these IVD bilirubin reagents are consistent with those determined by RMP. The main aim was to clarify the difference in DB results depending on these reagents.
Materials and methods
Specimens
UCB powder was purchased from Sigma-Aldrich Co., St. Louis, MO, USA (B4126,
To make UCB solution (defined as UCB level 2), 1.2 mg of the UCB powder was dissolved in 0.5 mL of dimethyl sulfoxide and 1.0 mL of 0.1 mol/L aqueous sodium carbonate and diluted to 25 mL with 40 g/L (606
To make DTB solution (defined as DTB level 2), 1.1 mg of the DTB powder was dissolved in 25 mL of 0.1 mol/L Tris buffer (pH 8.5) containing 40 g/L BSA.
12
The weighed-in DTB concentration was expected to be 42.7 mg/L (73.0
DLB solution (defined as DLB level 2) was synthesized from UCB and human serum albumin (HSA) according to our previously reported method using Woodward's reagent K.
13
In synthetic DLB, UCB that was not covalently bound to HSA was removed by washing with a caffeine-benzoate solution to yield > 99% purity using Centrifree® Ultrafiltration Devices (Merck Millipore Ltd, Cork, Ireland). DLB level 1 was prepared by a two-fold dilution of DLB level 2 with phosphate-buffered saline (pH 7.4) containing 2.1 g/L (32
Each bilirubin specimen was divided into six aliquots. They were wrapped in aluminium foil to protect from light, frozen and distributed on dry ice to the laboratories of the four manufacturers providing IVD bilirubin reagents in Japan (Table 1) and two other laboratories (the Chiba Institute of Science and Tenri Hospital) to measure TB and DB concentrations using the laboratory’s assay procedure. The latter two laboratories measured bilirubin using the diazo method.
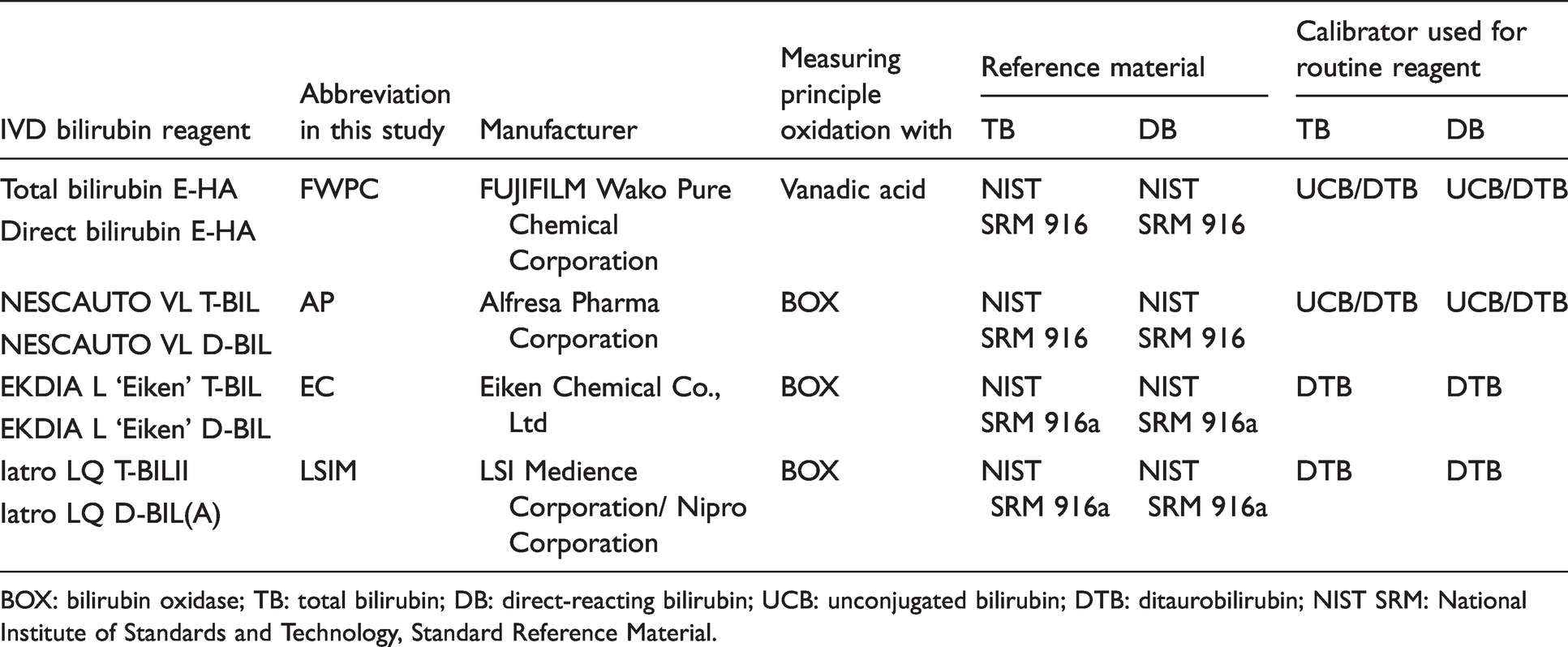
BOX: bilirubin oxidase; TB: total bilirubin; DB: direct-reacting bilirubin; UCB: unconjugated bilirubin; DTB: ditaurobilirubin; NIST SRM: National Institute of Standards and Technology, Standard Reference Material.
In this study, solution concentrations (mg/L) of UCB, DTB, and DLB were expressed as UCB equivalent. The molar concentration (
Assays
At the Chiba Institute of Science, TB concentration was measured by RMP according to the method reported by Doumas et al. 9 Briefly, bilirubin solution (0.25 mL) was mixed with diazo reagent (0.5 mL) in the presence of caffeine-benzoate solution (2.0 mL) and incubated at 25°C for 10 min (25 min for DLB measurement), followed by the addition of alkaline tartrate solution (1.5 mL), and then the absorbance at 598 nm was measured. The RMP was calibrated using reference standard certified by molar absorption coefficient of bilirubin azo-pigment at 598 nm (7664 m2/mol). 14 As an RMP for DB measurement has not been established, diazo-based RMP for TB was modified for DB measurement as reported by Doumas et al. 15 Briefly, bilirubin solution (0.25 mL) was preliminarily diluted with 0.05 mol/L hydrochloric acid (1.0 mL), to which diazo reagent (0.5 mL), ascorbic acid solution (0.1 mL), alkaline tartrate solution (1.5 mL), and caffeine-benzoate solution (2.0 mL) were added in this order. Ascorbic acid terminates the diazo reaction. As the addition of hydrochloric acid and ascorbic acid did not affect the absorption spectrum of azo pigment but merely increased the total volume of the reaction mixture, absorptivity at 598 nm in DB measurement was corrected for volume ratio by a factor of 5.35/4.25. To ensure observed concentrations of RMP, TB and DB concentrations were also measured by the acid diazo method without the addition of solutions of alkaline tartrate and hydrochloric acid, 16 which were in-house reagents adapted for automated analysis.
Furthermore, TB and DB concentrations were measured by IVD bilirubin reagent in the laboratories of the four manufacturers (Table 1). Each measurement was calibrated and performed according to the manufacturer’s instructions with the use of automated analysis (model 7170 or 7180 Hitachi automatic clinical analyser, Hitachi High-Tech Corporation, Tokyo, Japan). The IVD bilirubin reagent from FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan (FWPC), is based on the VA oxidation method. 4 Three other reagents from Alfresa Pharma Corporation, Osaka, Japan (AP), 3 Eiken Chemical Co., Ltd, Tokyo, Japan (EC), 17 , 18 and LSI Medience Corporation, Tokyo, Japan (LSIM), 3 are based on the BOX method. Henceforth, the name of each manufacturer is the shortened form. LSIM collaborated with Nipro Corporation, Osaka, Japan, to develop the IVD bilirubin reagent. All experiments including the preparation of bilirubin solutions were performed under dim light.
Statistical analysis
The data were analysed using the Wilcoxon signed-rank test, and differences of
Results
Certified concentration
As we weighed a small aliquot of bilirubin powder (1.2 mg for UCB and 1.1 mg for DTB) on the balance, solute concentrations (i.e. ‘weighed-in concentration’ in Table 2) were certified using RMP. The concentrations of UCB level 1 and level 2 were determined to be 38.0 ± 0.0
Observed concentration values of total bilirubin (TB) in artificially prepared bilirubin materials measured by RMP, in-house acid diazo method, and four IVD reagents.
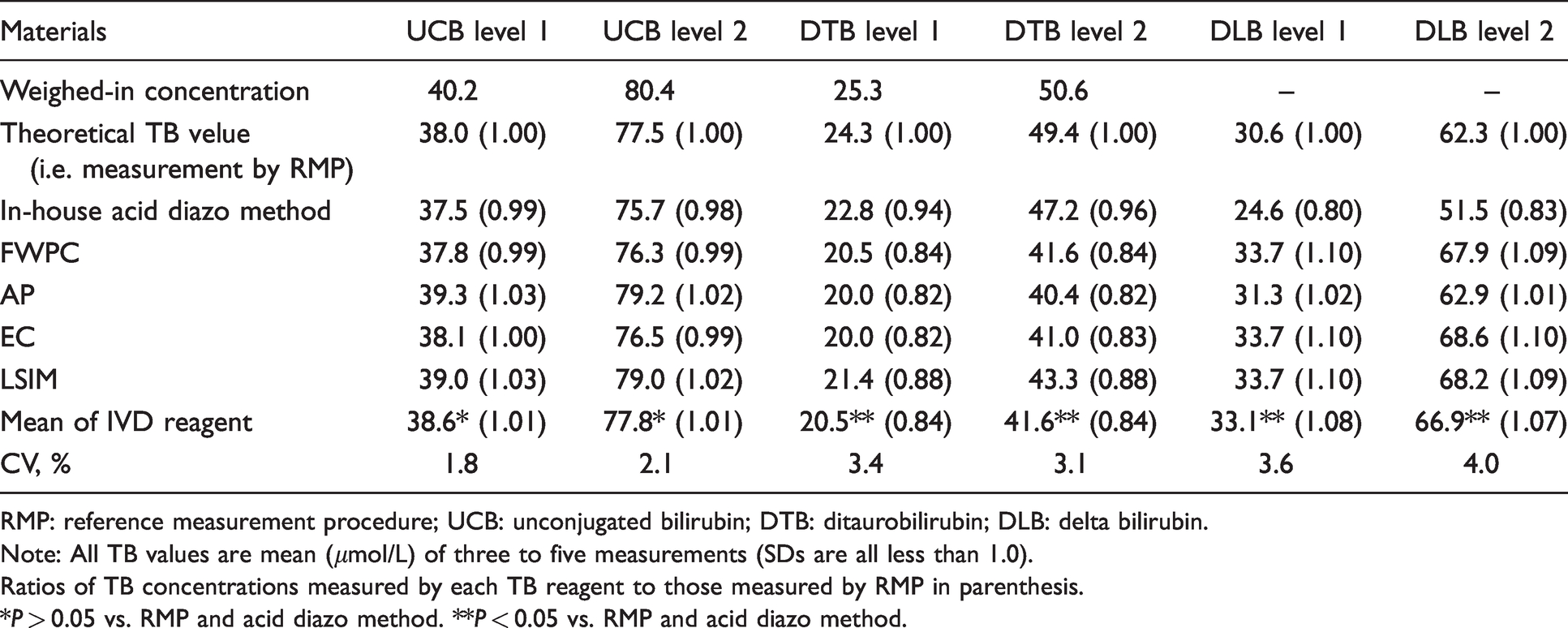
RMP: reference measurement procedure; UCB: unconjugated bilirubin; DTB: ditaurobilirubin; DLB: delta bilirubin.
Note: All TB values are mean (
Ratios of TB concentrations measured by each TB reagent to those measured by RMP in parenthesis.
*
Comparison of TB concentration
We investigated whether the four IVD-TB reagents yielded consistent TB concentration results. For UCB solutions, TB concentration determined by all the IVD bilirubin reagents were similar (Table 2) and averaged 38.6 ± 0.7
For the DTB solutions, although TB concentrations determined by IVD-TB reagents converged to 20.5 ± 0.7
For the DLB level 1 and level 2 solutions, TB concentrations determined by IVD-TB reagents from FWPC, EC, and LSIM were higher (
Comparison of DB concentration
Bilirubin concentrations were determined using the IVD-DB reagents and two diazo methods (RMP modified for DB measurement and in-house acid diazo method); bilirubin concentrations were determined (Table 3) for the same solutions of UCB, DTB and DLB used in the above-mentioned TB measurement. In principle, although UCB solutions contain 38.0
Observed concentration values of direct-reacting bilirubin (DB) in artificially prepared bilirubin materials measured by RMP, in-house acid diazo method, and four IVD reagents.
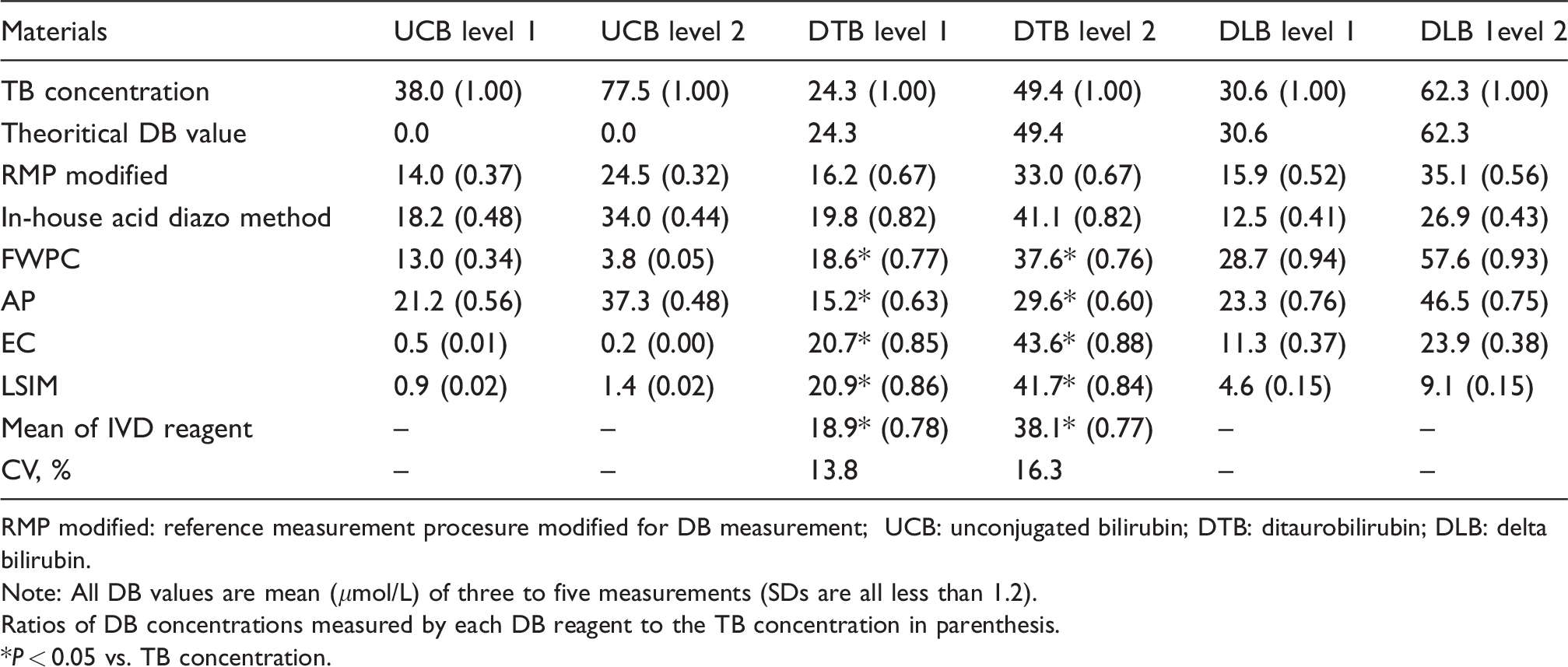
RMP modified: reference measurement procesure modified for DB measurement; UCB: unconjugated bilirubin; DTB: ditaurobilirubin; DLB: delta bilirubin.
Note: All DB values are mean (
Ratios of DB concentrations measured by each DB reagent to the TB concentration in parenthesis.
*
However, in fact, every bilirubin assay method doubtfully reacted with UCB which was behaving as a direct-reacting form of bilirubin (Table 3), and the magnitude of doubtful reactivity was highest in the reagent from AP (the ratios of DB to TB was 0.56 for UCB level 1 and 0.48 for UCB level 2), followed by the diazo methods and FWPC. Reagents from EC and LSIM reacted less with UCB (the ratios of DB to TB were 0.00–0.02 both for UCB level 1 and 2). It was notable that the observed DB concentrations in UCB level 1 were higher than those in UCB level 2 as measured by reagents from FWPC and EC.
For DTB solutions, DB concentrations determined by the IVD-DB reagents averaged 18.9 ± 2.6
For DLB level 1 and level 2 solutions, DB concentrations observed by IVD bilirubin reagents differed from reagent to reagent. Reagents from FWPC and AP reacted strongly with DLB; however, those from EC and LSIM reacted less with DLB. Magnitude of reactivity with DLB (i.e. DB to TB ratio; sixth and seventh columns in Table 3 and shown in Figure 1) was the highest in FWPC (0.94 for DLB level 1 and 0.93 for DLB level 2), followed by AP (0.76–0.75), RMP (0.52–0.56), acid diazo method (0.41–0.43), EC (0.37–0.38), and LSIM (0.15–0.15).
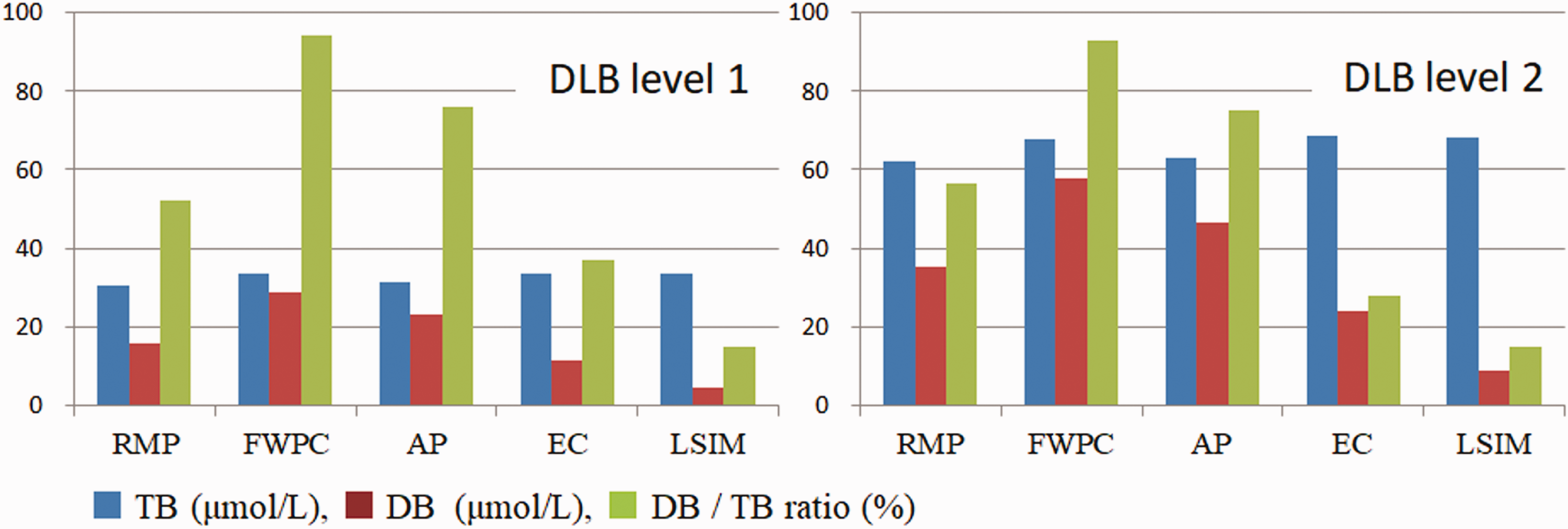
Comparison of reactivity of RMP and four IVD bilirubin (TB and DB) reagents to DLB level 1 and level 2. DLB was chemically synthesized from UCB and human serum albumin using Woodward's reagent K. In-house acid diazo method is excluded from this figure, since it could not completely react with DLB in TB measurement.
Discussion
Weiss et al.
19
reported that serum DLB decreased more slowly than CB during the recovery period in patients with jaundice. DLB is formed from CB and is covalently bound to albumin in serum samples of patients with cholestasis.
20
Possessing one molecule of glucuronic acid, DLB is hydrophilic and reacts with the diazo reagent as DB. Unfortunately, Weiss et al. were not aware of variation in reactivity with DLB for each IVD-DB reagent. In fact, literature values (case 1, 2, and 3 in Table 4)
21
,
22
for DB in the same serum samples showed differences caused by reagents potentially due to the presence of DLB in patients with cholestasis. In addition, recently, we were consulted regarding discrepancies in DB values of a patient with obstructive jaundice between two hospitals; 157
Literature values for comparison of total (TB) and direct-reacting (DB) bilirubin concentration values by different
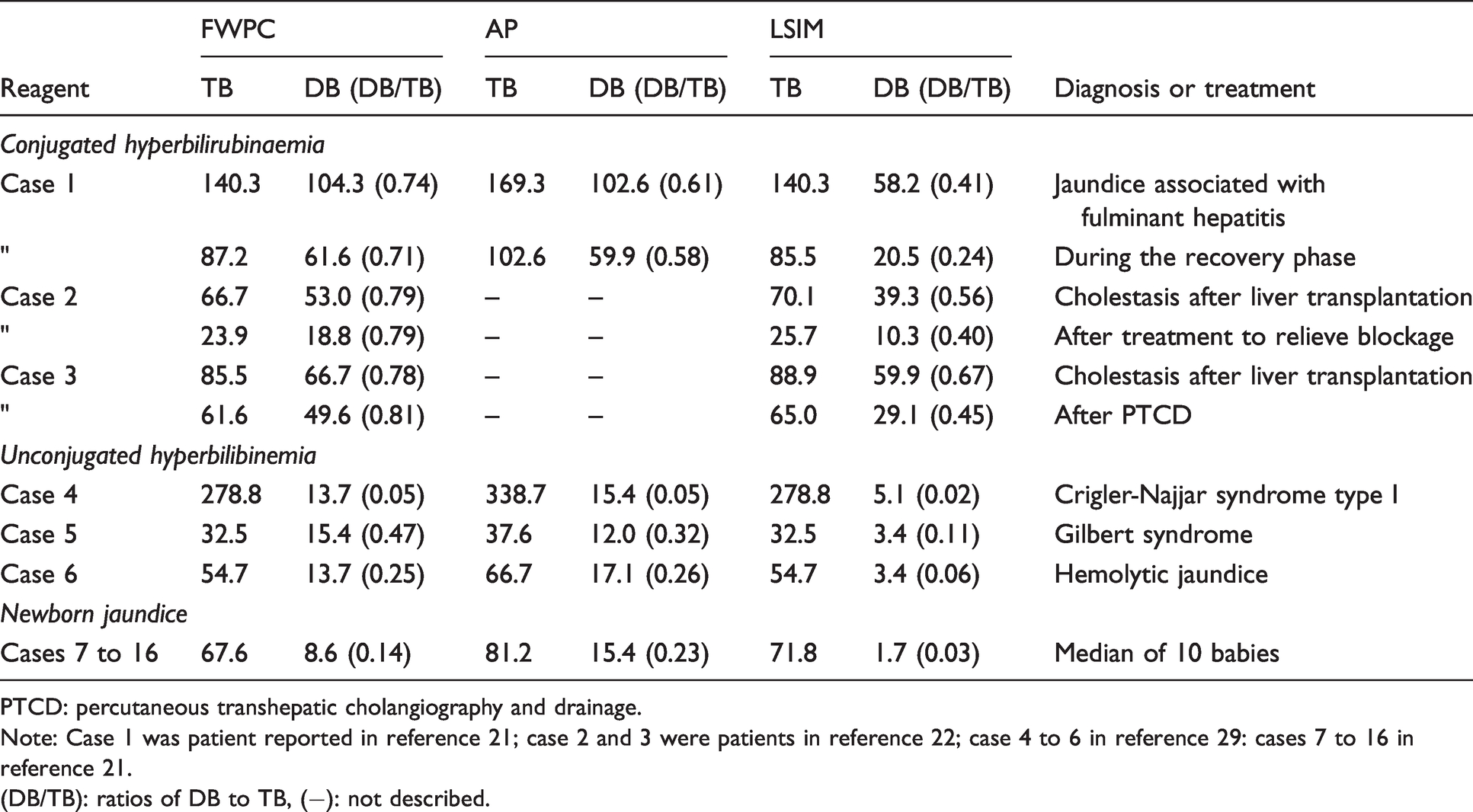
PTCD: percutaneous transhepatic cholangiography and drainage.
Note: Case 1 was patient reported in reference 21; case 2 and 3 were patients in reference 22; case 4 to 6 in reference 29: cases 7 to 16 in reference 21.
(DB/TB): ratios of DB to TB, (−): not described.
In TB measurement, inter-method variations for UCB, DTB, and DLB solutions were revealed to be converged as low as 1.8–4.0% (Table 2), satisfying the criteria for ‘TB measurement’ proposed by Dr James Westgard
23
(<10.9%) and the Committee on Quality Management of the JSCC
24
(<12.1%). In 2009, the Dutch External Quality Assessment Organization in Medical Laboratories conducted surveillance of pooled human serum spiked with UCB (Sigma) and assayed with diazo-based IVD-TB reagents (Roche, Beckman Coulter, Abbot, Ortho Clinical Diagnostics, Dade Behring, and Bayer).
25
Using their data, we calculated the inter-method variations as 7.6% and 6.1% at UCB concentrations defined by RMP as 26.7
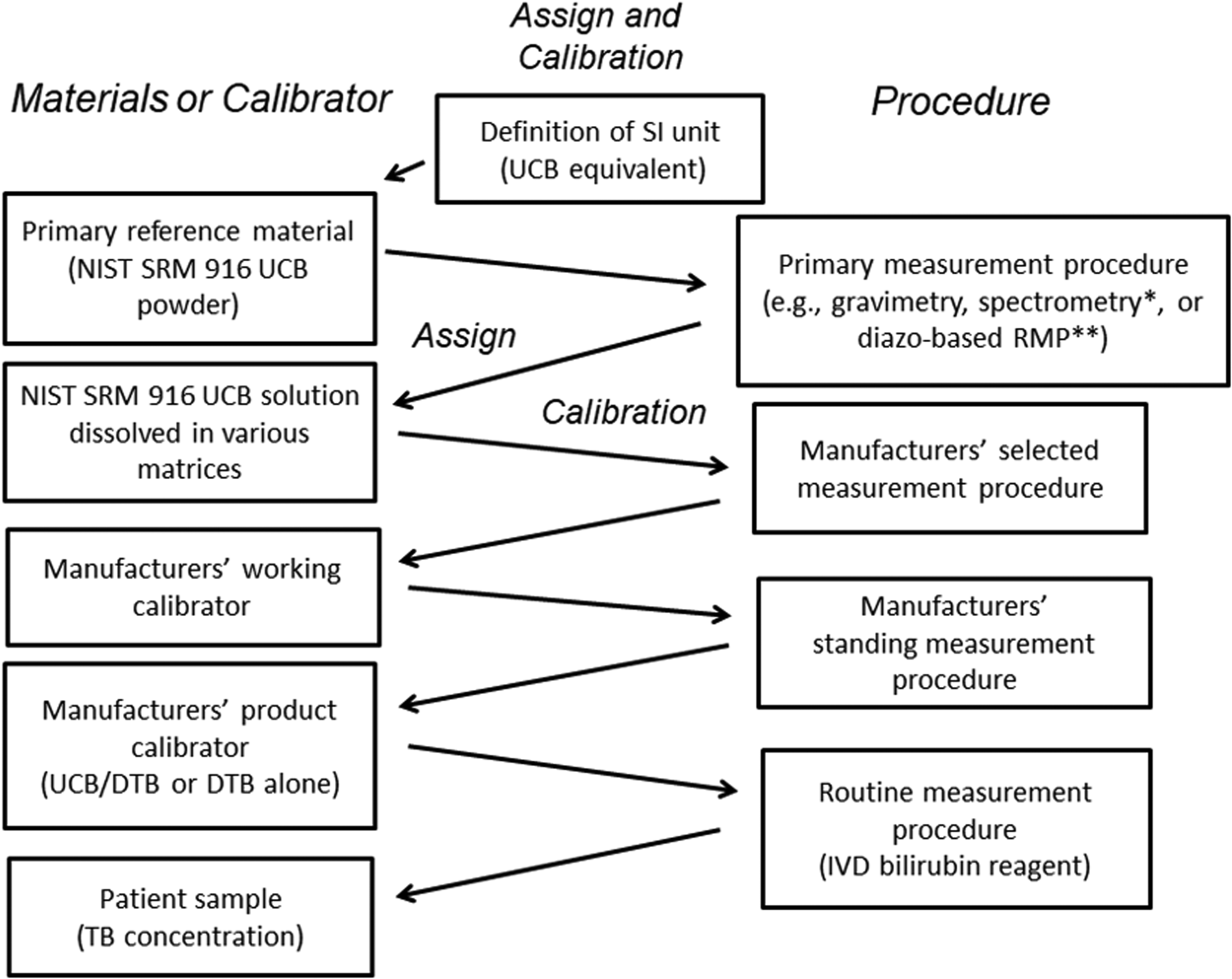
Traceability chain for TB measurement, in which NIST SRM 916 is used as a reference material. Some manufacturer used NIST SRM 916a (a different lot of SRM 916). *Bilirubin concentration was certified by molar absorptivity at 460 nm. **Bilirubin concentration was certified by molar absorptivity of azo-pigment at 598 nm.
In DB measurement, the inter-method variations for DTB solution (13.8% for level 1 and 16.3% for level 2) were a little higher than the criteria for ‘DB measurement’ proposed by Dr James Westgard
23
(<18.4%) and the Committee on Quality Management of the JSCC24 (<13.1%). The high CVs were caused by the unavailability of NIST standard reference combined with RMP to measure DB. While, in terms of accuracy, DTB concentrations measured by IVD-DB reagents showed lower values against target TB concentration (Table 3), and they needed further improvement by manufacturers. Doumas et al.
12
and, more recently, Kiuchi et al.
27
suggested that observed DB results in DTB solution should be consistent with its TB concentration (i.e. 24.3
In this study, we have raised concerns that IVD-DB data results may have three problems; one is the underestimation of DB concentrations toward theoretical ‘true’ DTB concentration as mentioned above, and the other two were cross reactivity with UCB and different reactivity with DLB, all of which may mislead clinical interpretation. Specifically, IVD-DB reagent reacted with UCB and thus, was falsely measured out as DB. Magnitudes of increase in DB results appeared to be independent of UCB concentration, and the increase was occasionally higher at UCB level 1 than UCB level 2 (Table 3). These inversions of DB results have been reported by Doumas et al.
7
and Suzuki et al.
28
in BOX in-house reagent and IVD-DB reagent from AP, respectively, by addition of UCB into pooled human serum or into BSA solution. A similar tendency was observed in patient between case 4 (complete absence of hepatic uridine 5ʹ-diphospho-glucuronosyltransferase which produces CB)
29
and case 5 (FWPC) or case 6 (AP) (Table 4),
30
although reaction of UCB as DB in natural serum seemed to be lower than artificial UCB. As IVD-DB reagent was developed in order to quantify natural serum contained both DB and UCB, these reagents could not be adapt to measure artificial UCB solution, which does contain DB. However, reason for the inversion of DB results remains unresolved. In clinical paediatrics, Hodgson et al.
31
reported criteria of DB concentration as >25
On the other hand, IVD-DB reagents developed in Japan have varied reactivity with DLB; one reagent (FWPC) reacted strongly, another (AP) moderately, and the others (EC and LSIM) reacted weakly with DLB (Table 3). As mentioned above, DLB is bilirubin that is covalently bound to albumin and clearance of DLB reflects the half-life of albumin (approximately three weeks). Therefore, delayed clearance of DLB from circulation may have interfered with the interpretation of CB clearance when DB was measured by IVD-DB reagents that strongly reacted with DLB. Since elevated serum CB levels is a principal sign of cholestasis, physicians at the Japanese Society of Gastroenterology and the Japan Society of Hepatology asked our research group to measure CB that does not interfere with the presence of DLB. 1 Our results show that the IVD-DB reagents from EC and LSIM comply with this requirement. Since most laboratories worldwide fractionated serum bilirubin into TB and DB without measuring DLB, knowledge of how IVD-DB reagent we are using reacts with DLB is necessary both for laboratory personnel and physicians.
By contrast, the Intractable Hepato-Biliary Disease Study Group supported by the Ministry of Health, Labor and Welfare of Japan has established a new guideline 32 to predict the outcome of patients with fulminant hepatic failure after liver transplantation; the serum concentration of TB and the DB to TB ratio (critical values indicating the remaining activity of bilirubin conjugation in a patient's diseased liver; usually 0.7 and 0.5, respectively), along with other hepatobiliary test parameters, were measured. However, it is not clear whether the DB to TB concentration ratio considered reactivity with DLB. To improve the predictive ability of the outcome in these patients, the ratio needs to be reverified using CB without interference with DLB.
However, the present study has limitations. First, we used artificially prepared UCB, DTB, and DLB solutions to determine the reactivity of the IVD bilirubin (TB and DB) reagents. In the future, we would like to reconfirm the present results using naturally occurring authentic UCB, CB, and DLB of human origin. Second, an RMP for DB (or CB) measurements has not been established in the present study. Our research group is planning to establish an isocratic high-performance liquid-chromatography (HPLC) procedure reported by Osawa et al. 33 as the RMP for measuring serum bilirubin subfractions (i.e. UCB, CB, and DLB). Third, the effects of the bilirubin photoisomers generated under environmental lighting on the four IVD bilirubin reagents have not been thoroughly examined. 34 , 35 This requires future experimentation.
In conclusion, despite these limitations, we were able to unravel the individual reactivity of UCB, DTB, and DLB with IVD bilirubin reagents developed in Japan. TB concentrations measured by IVD-TB reagent were almost consistent with those by RMP even when UCB and/or DLB gave rise to hyperbilirubinaemia. However, DTB concentration as a surrogate for CB was underestimated, reported as 82–88% and 60–88% of RMP in TB and DB measurement, respectively. All IVD-DB reagents reacted with UCB resulting in overestimation of DB. Each IVD-DB reagents exhibited different reactivity with DLB; therefore, reagents from FWPC and AP measured DLB together with CB, and reagents from EC and LSIM exclusively measured CB.
Footnotes
Acknowledgements
We sincerely thank all the subjects for participating in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Foundations of Chiba Institute of Science and the Japan Society of Clinical Chemistry.
Ethical approval
Not applicable.
Guarantor
HI
Contributorship
HI, SK and SO designed the research; HI, SK, YH, KW, KT, HA, DT, AS and TI performed the research and interpretation of dada; HI, SK, SO and SI wrote the first draft of the manuscript; and all authors reviewed and edited the manuscript and approved the final version of the manuscript.
