Abstract
Background
Left atrial function (LAF) plays an interactive role between pulmonary and systemic circulation. Cardiac biomarkers, such as amino-terminal pro-brain natriuretic peptide (NT-proBNP) and troponins, might reflect cardiac function. This study aims to evaluate the association between high sensitivity troponins (hsTn) and left atrial function in patients undergoing cardiac magnetic resonance imaging (cMRI).
Methods
Patients undergoing cardiac magnetic resonance imaging (cMRI) were enrolled prospectively. Patients with right ventricular dysfunction (<50%) were excluded. Blood samples for measurements of hsTn and NT-proBNP were collected at the time of cMRI.
Results
Eighty-four patients were included. Median LVEF was 59% (IQR 51–64%). HsTn correlated inversely with LAF within multivariable linear regression models (hsTnI: Beta −0.46; T −4.44; P = 0.0001; hsTnT: Beta −0.29; T −3.06; P = 0.003). High sensitivity troponins increased significantly according to decreasing stages of impaired LAF (P = 0.0001). High sensitivity troponins discriminated patients with impaired LAF < 55% (hsTnT: AUC = 0.80; P = 0.0001; hsTnI: AUC = 0.74; P = 0.0001) and <45% (hsTnT: AUC = 0.75; P = 0.0001; hsTnI: AUC = 0.73; P = 0.001) and were still significantly associated in multivariable logistic regression models (LAF < 55%: hsTnT: OR = 21.78; P = 0.0001; hsTnI: OR = 5.96; P = 0.009; LAF < 45%: hsTnT: OR = 10.27; P = 0.0001; hsTnI: OR = 12.56; P = 0.001).
Conclusions
This study demonstrates that hsTn are able to reflect LAF being assessed by cardiac magnetic resonance imaging.
Introduction
Left atrial function (LAF) plays an interactive role between pulmonary and systemic circulation.1,2 The left atrium (LA) modulates left ventricular (LV) filling both within an active and passive phase during LV diastole and serves as a reservoir for pulmonary venous return during LV systole. 3 LAF might be impaired due to progressive atrial fibrosis and enlargement over years leading to an adverse structural remodeling of the LA. Accordingly, this adverse structural remodeling predisposes to the development of atrial fibrillation (AF).4–6 Recent studies demonstrated that an impaired LAF might be associated with a higher risk of re-hospitalization in patients suffering from heart failure with preserved ejection fraction (HFpEF).7–9 Accordingly, LA fibrosis was shown to be associated with a poor prognosis in both patients with reduced and preserved EF (HFrEF and HFpEF).4,5 Therefore, an accurate assessment of LAF might lead to an earlier detection of chronic heart failure (CHF) development.3,9 The assessment of LAF was shown recently to be more accurate by cardiac magnetic resonance imaging (cMRI) compared to echocardiography.10,11
Cardiac biomarkers, such as amino-terminal pro-brain natriuretic peptide (NT-proBNP) and cardiac troponins (cTn), were shown to be associated with short- and long-term prognosis in CHF.12–14 Particularly, NT-proBNP has emerged to the reference cardiac biomarker improving the diagnostic assessment of acute (AHF) and CHF. 15 With regard to LAF natriuretic peptides, such as atrial natriuretic peptide (ANP) and NT-proBNP were shown to correlate with LAF. 16 Numerous studies revealed that NT-proBNP correlated significantly with LAF in patients with HFpEF and HFrEF.17–19 CTn represent the central cardiac enzymes sustaining myocardial contractility and represent the reference biomarker defining the diagnosis of non-ST segment elevation myocardial infarction (NSTEMI).14,20,21 Recently developed high sensitivity troponin (hsTn) assays reveal an approximately hundredfold lower detection limit compared to contemporary troponin assays.22,23 Consequently, these hsTn allow an even faster “rule-in” and “rule-out” of NSTEMI within 1 h compared to 3 h with contemporary troponin assays.20,24
Furthermore, hsTn concentrations were shown to correlate significantly with LV ejection fraction (LVEF) and LV end-diastolic pressure (LVEDP) being assessed by transthoracic echocardiography. 25 Within a community registry of patients with atherosclerosis elevated hsTn were shown to be associated with an increased incidence rate of AF. 26 However, hsTnI, hsTnT as well as NT-proBNP have never been evaluated comparatively regarding their diagnostic value for LAF being assessed by cMRI. 27
Therefore, this study aims to investigate whether concentrations of hsTn are able to reflect LAF being assessed by cMRI.
Methods
Study population
The ‘Cardiovascular Imaging and Biomarker Analyses' (CIBER) study (clinicaltrials.gov identifier: NCT 03074253) represents a clinically prospective, controlled and monocentric study at the University Medical Centre Mannheim (UMM), Germany. The study was carried out according to the principles of the Declaration of Helsinki and was approved by the local ethics committee. Written informed consent was obtained from all participating patients or their legal representatives.
For the present study, patients undergoing cMRI during routine clinical care were included consecutively from February 2015 until June 2015 within an all-comers design. In order to perform valuable cMRI examination, all patients had to be in stable clinical condition without acute clinical symptoms, such as acute dyspnea or extensive peripheral edema. The indications for cMRI were not restricted to any specific disease entity. Exclusion criteria for cMRI accorded to commonly known exclusion criteria, such as claustrophobia and metal implants. 28 Specifically for the present study, patients with a reduced right ventricular function (RVF) below 50% were excluded. All patients included were scheduled for 6 months and 12 months of follow-up by telephone visits.
All available clinical information of the study patients was documented, such as detailed findings of patients’ medical history, laboratory values and medical therapies. Blood samples for biomarker measurements were collected once within 24 h following cMRI examination.
Measurements of biomarkers
All expressed biomarkers were measured in the serum of patients' blood. All samples were obtained by venipuncture into serum monovettes® and centrifuged at 2500 g for 10 min at 20℃. The aliquoted samples were cooled down with liquid nitrogen before being stored at −80℃ until analysis. The complete processing was conducted within 2 h after blood extraction. After thawing, the samples were mixed gently by inverting and centrifuged with 2500 g for 10 min at 20℃, respectively, 3000 g for 30 min for hsTnI at 4℃.
HsTnT was measured with the Troponin T hs STAT assay on a cobas e 602 analyzer (Roche Diagnostics, Mannheim, Germany). The limit of blank (LoB) for this assay was 3 ng/L and the limit of detection (LoD) was 5 ng/L as described in the instructions for use. 29 HsTnI was measured with the STAT High sensitivity Troponin-I assay on an Architect i1000 analyzer (Abbott, Wiesbaden, Germany). The limit of blank (LoB) was 0.7–1.3 ng/L for this assay as described in the instructions for use. 30 NT-proBNP was measured with the proBNP II STAT assay on a cobas e 602 analyzer (Roche Diagnostics, Mannheim, Germany). The LoD for this assay was 5 ng/L. 31 Creatinine was measured with the Creatinine Jaffe Gen.2 assay on a cobas c 702 analyzer (Roche Diagnostics, Mannheim, Germany).
cMRI acquisition
All studies were performed using a 1.5-Tesla whole body imaging system (Magnetom Avanto and Sonata, Siemens Medical Systems, Healthcare Sector, Erlangen, Germany) using a four-element (Sonata) or six-element (Avanto) phased-array body coil. Cine images were acquired using a retrospective electrocardiographic-gated, balanced segmented steady state free precession (trueFISP) sequence in three long-axis views (two-, three-, and four-chamber views) and in multiple short-axis views, covering the entire left ventricle from base to apex.
cMRI analysis
The cMRI image analysis was performed using the commercially available computer software program cvi 42 ® (Circle Cardiovascular Imaging Inc., Calgary, Canada). LA end-diastolic and end-systolic volumes (LAEDV, LAESV) were measured by biplane ellipsoid formula on four and two chamber long axis views using the area-length method (LA Volume = 0.85 × four-chamber area × two-chamber area/average of two-and four-chamber length). LAEDV was defined as LA volume (LAV) at end of LV systole before mitral valve opening. LAESV was defined as LAV at end of LV diastole right after mitral valve closure. LA stroke volume (LASV) and LA ejection fraction (LAF) were calculated using the formulae: LASV = LAEDV – LAESV and LAF = LASV/LAEDV × 100%. Three sub-groups were set according to LAF (LAF I: ≥55%; LAF II: ≥45%– < 55%; LAF III: <45%), referring to Aune et al., 32 where LAF > 45% was identified as normal in patients free from clinical and sub-clinical cardiovascular diseases in echocardiographic examination.
Statistical analysis
For data with normal distribution, the student t test was applied. Otherwise, Kruskal–Wallis test was used as a non-parametric test. Deviations from a Gaussian distribution were tested by the Kolmogorov–Smirnov test. Firstly, the clinical confounding factors influencing LAF in the total cohort were evaluated within a multivariable linear regression model adjusted for baseline (age, sex) and clinical parameters or comorbidities (coronary artery disease (CAD), valvular heart disease and AF), irrespectively of hsTn values. Secondly, the distribution of cMRI indices according to LAF subgroups was analysed. Thirdly, univariable correlations between hsTn and cMRI parameters according to LAF were analysed using Spearman’s rank correlation for non-parametric data. In a fourth step, multivariable linear regression models adjusted for baseline (age, sex, creatinine) and clinical parameters or cardiac comorbidities (LAF, LVEF, CAD, valvular heart disease and AF) were performed for evaluating influencing factors on hsTn in the present cohort. Thereafter, receiver operating characteristic curve analyses (ROC) with area under the curves (AUC) were determined in order to evaluate whether biomarkers are able to discriminate the presence of reduced LAF. Combined AUCs of biomarkers were calculated using plotted likelihoods of each biomarker. ROC curves were compared by the method of Hanley et al. 33 In a last step, multivariable logistic regression models were developed to confirm the diagnostic value of hsTn for LA dysfunction implicating pre-defined cutoffs, and these models were adjusted for baseline (age, sex, creatinine), clinical characteristics (LVEF) and NT-proBNP. Multivariable linear or logistic regression analyses were performed with backward elimination. Parameters in multivariable models were included to these models as independent variables, when they revealed a known clinical impact on or significant univariate correlations with the dependent variable. Data are presented as means with confidence intervals or medians with interquartile ranges (25th to 75th percentiles), depending on the distribution of the data. P-values <0.05 were considered as statistically significant. Cutoffs of biomarkers were set at the group specific medians of each biomarker for the groups of reduced LAF. The calculations were performed with GraphPad Prism software (GraphPad Software Inc., San Diego, CA, USA) and SPSS software (IBM SPSS Statistics, IBM Corp., Armonk, NY, USA).
Results
Study population
A total of 84 patients were enrolled in the present study (Figure 1). Median age was 55 years (range 18–85 years). Most patients were of male gender (n = 58). Twenty-six patients suffered from compensated CHF (according to LVEF < 55%) with only mild to moderate symptoms according to NYHA class I and II (n = 24). Thirteen patients suffered from AF, mostly paroxysmal AF (n = 8) (Table 1). Thirty-one patients suffered from valvular heart diseases, mostly from mitral valve regurgitation (MR) (n = 15), followed by tricuspid (n = 10) and aortic valve regurgitation (n = 5). Thirteen patients suffered from mild mitral valve regurgitation. Only one patient suffered from MR grade III and one patient from aortic valve stenosis grade III (data not shown). Twenty-six patients presented with CAD, and six of these already underwent aorto-coronary bypass (ACVB) surgery. Seven patients suffered from chronic kidney disease, while no patient suffered from end-stage renal failure or was in need for hemodialysis. The median glomerular filtration rate (GFR) was 89 ml/min (IQR 75–101 ml/min) (Table 1).
Flow-chart showing patient selection criteria of the present study. Baseline characteristics of study patients. Confidence interval. Interquartile range.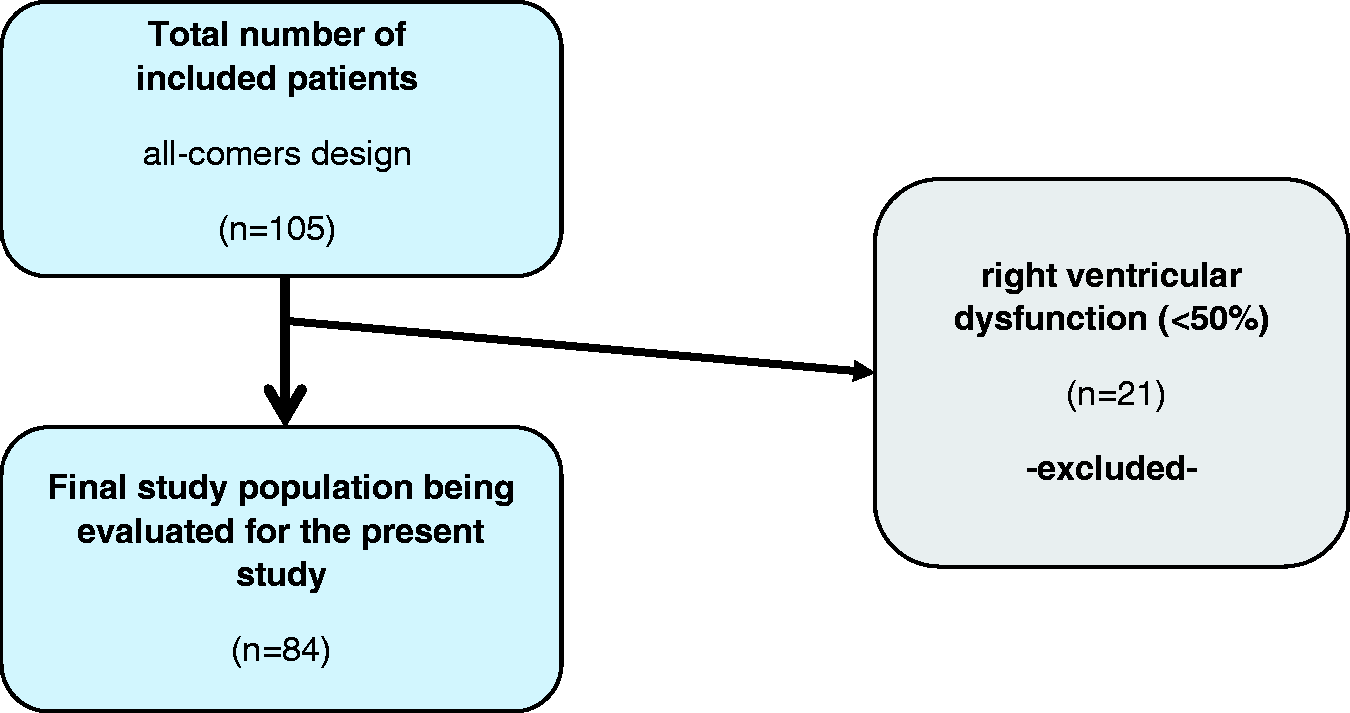
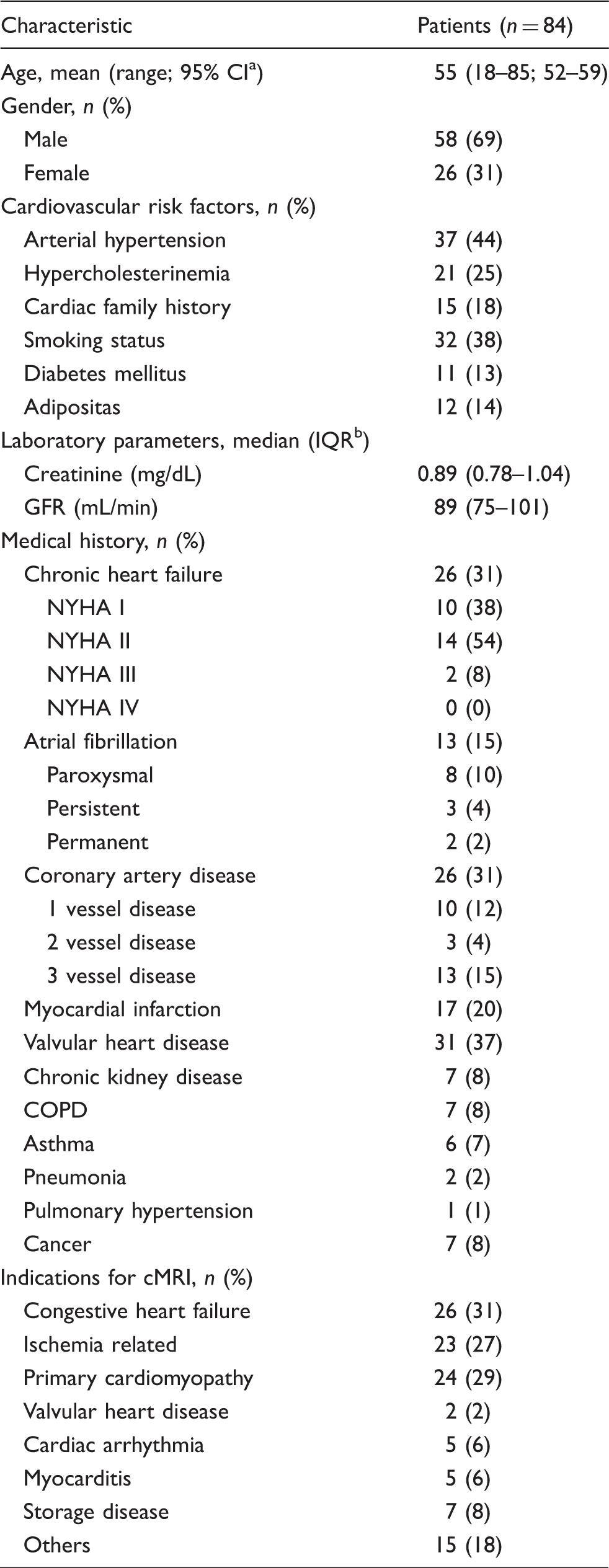
According to the extent of LAF, three subgroups were defined as follows: LAF I: LAF ≥ 55% (n = 31), LAF II: LAF ≥ 45–<55% (n = 22), LAF III: LAF < 45% (n = 31). AF (n = 13) was only present in patients with LAF < 55%, according to groups LAF II and III. Ten patients with LAF III suffered from AF.
Distribution of cMRI indices according to LAF subgroups
Distribution of cardiac MRI indices according to LAF subgroups.
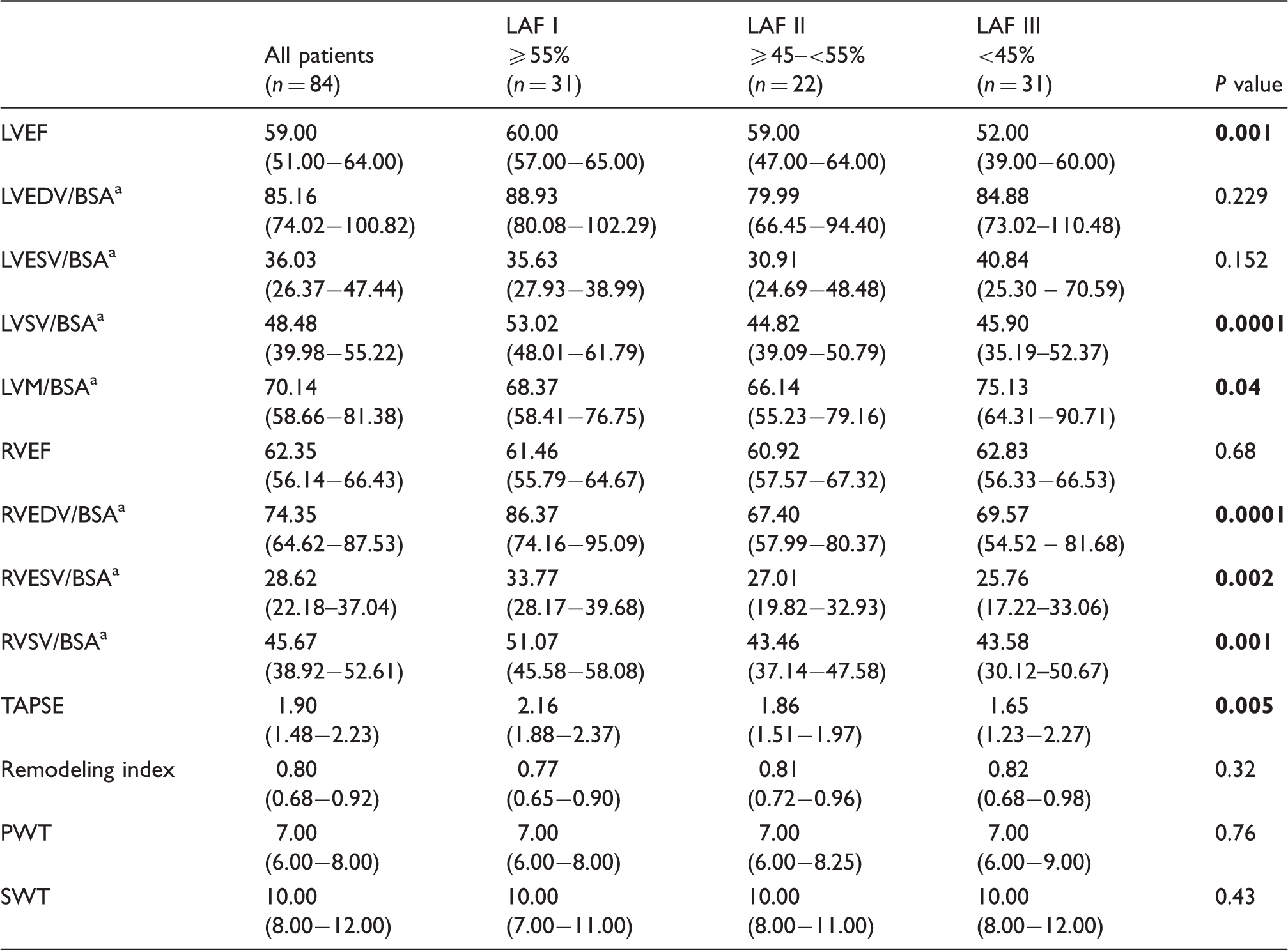
Note: Data presented as median with interquartile ranges (IQR). Bold values indicate statistically significant P values (P < 0.05).
Body surface area.
LVEDV: left ventricular enddiastolic volume; LVEF: LV ejection fraction; LVESV: LV endsystolic volume; LVM: LV mass; LVSV: LV stroke volume; PWT: posterior wall thickness; RVEDV: right ventricular enddiastolic volume; RVEF: RV ejection fraction; RVESV: RV endsystolic volume; RVSV: RV stroke volume; SWT: septal wall thickness; TAPSE: tricuspid annular plane systolic excursion.
Clinical confounding factors influencing LAF
Multivariable linear regression model for evaluating associations between LAF and clinical characteristics and cardiac comorbidities.
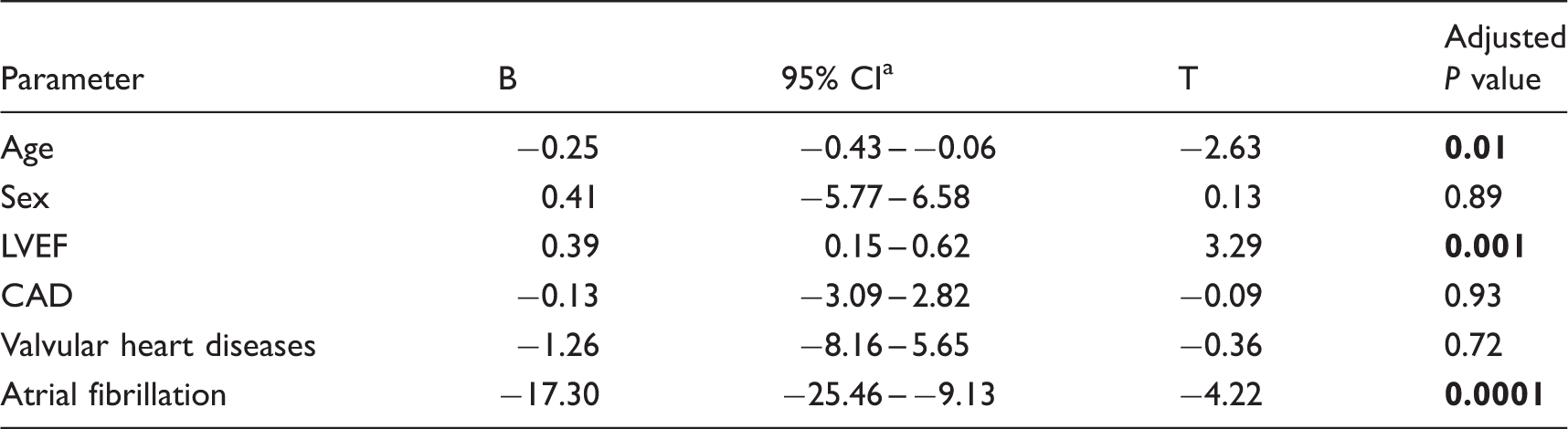
Note: Bold values indicate statistically significant P values (P < 0.05).
Confidence interval.
CAD: coronary artery disease; LVEF: left ventricular ejection fraction.
HsTn and NT-proBNP in LAF sub-groups
Figure 2(a) and (b) demonstrates significantly increasing hsTn values according to subgroups of decreased LAF (P = 0.0001). Biomarker concentrations were as follows:
Distribution of hsTnI (panel a) and hsTnT (panel b) serum concentrations according to subgroups of reduced LAF. Data are presented as medians with 25th and 75th percentiles (boxes) and 5th and 95th percentiles (whiskers).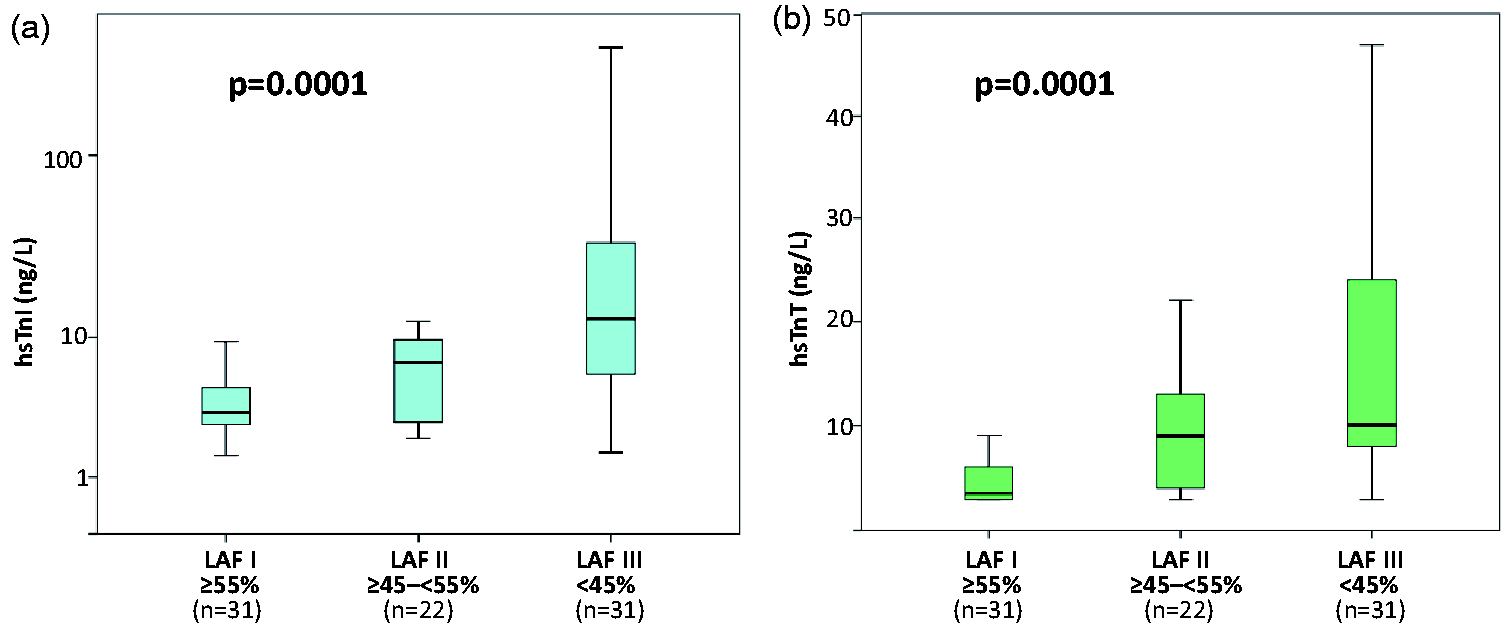
HsTnT: LAF I (median 4 ng/L, interquartile range (IQR) 3–6 ng/L), LAF II (median 9 ng/L, IQR 4–13 ng/L), LAF III (median 10 ng/L, IQR 8–29 ng/L). HsTnI: LAF I (median 3.40 ng/L, IQR 2.80–5.10 ng/L); LAF II (median 7.1 ng/L, IQR 2.88–10.15 ng/L); LAF III (median 12.70 ng/L, IQR 5.70–36.70 ng/L). NT-proBNP (data not shown): LAF I (median 57.07 ng/L, IQR 32.62–115.00 ng/L); LAF II (median 131.95 ng/L, 20.38–391.80 ng/L); LAF III (median 749.90 ng/L, IQR 121.30–1475.00 ng/L) (P = 0.0001).
Correlations of hsTn with cMRI parameters and clinical characteristics
HsTn correlated significantly with LAF (hsTnI: r = −0.47; P = 0.0001; hsTnT: r = −0.55; P = 0.0001), age (hsTnI: r = 0.45; P = 0.0001; hsTnT: r = 0.65; P = 0.0001) and NT-proBNP (hsTnI: r = 0.64; P = 0.0001; hsTnT: r = 0.69; P = 0.0001) in all patients. Only hsTnT correlated significantly with creatinine (r = 0.24; P = 0.03), whereas hsTnI did not (r = 0.09; P = 0.4).
Univariable correlations between hsTn and cardiac MRI parameters according to LAF.
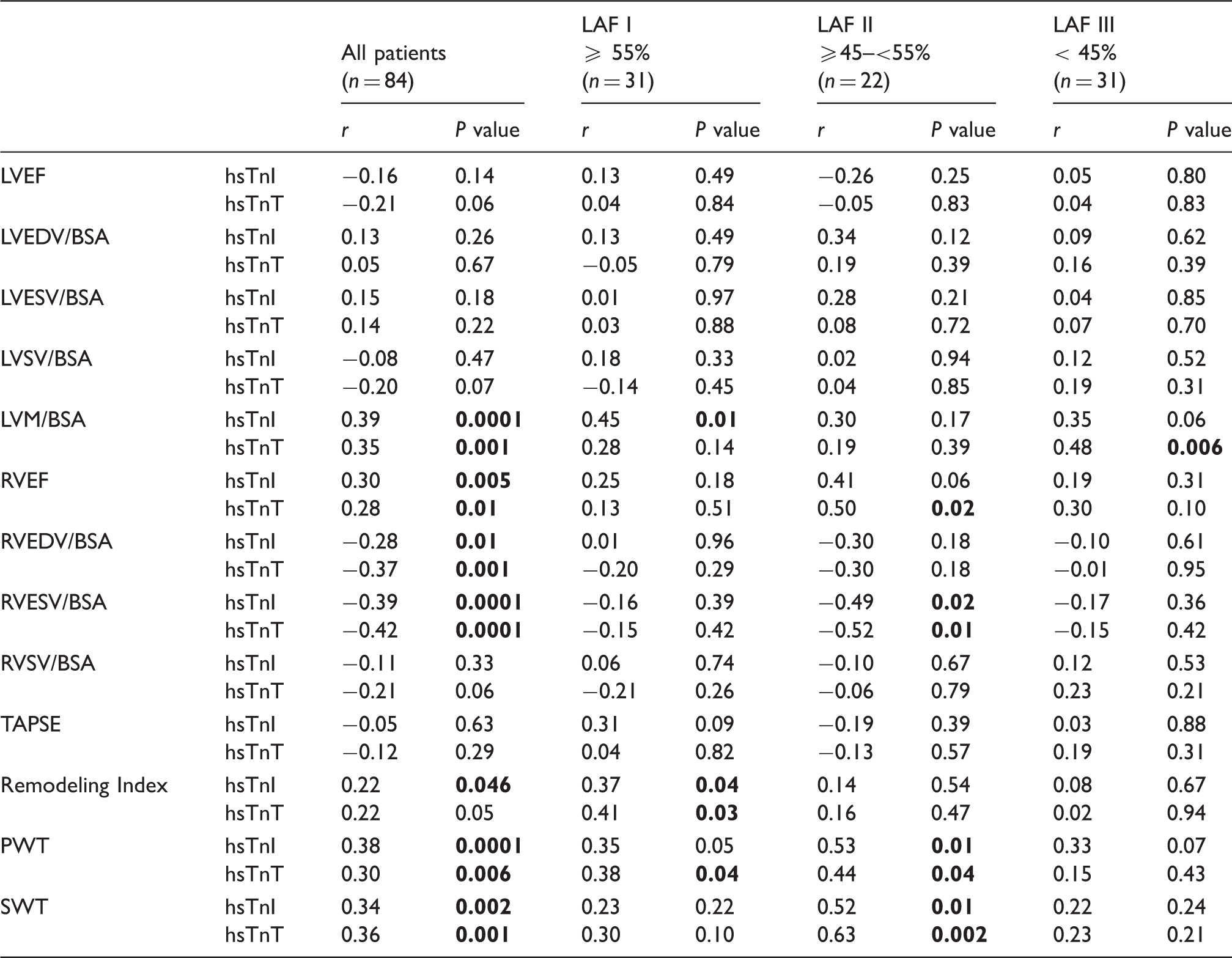
Note: Bold values indicate statistically significant P values (P < 0.05).
BSA: body surface area; LAF: left atrial function; LVEDV: left ventricular enddiastolic volume; LVEF: LV ejection fraction; LVESV: LV endsystolic volume; LVM: LV mass; LVSV: LV stroke volume; RVEDV: right ventricular enddiastolic volume; RVEF: RV ejection faction; RVESV: RV endsystolic volume; RVSV: RV stroke volume; SWT: septal wall thickness; TAPSE: tricuspid annular plane systolic excursion.
Multivariable linear regression model for evaluating associations between hsTn and clinical characteristics and cardiac comorbidities.
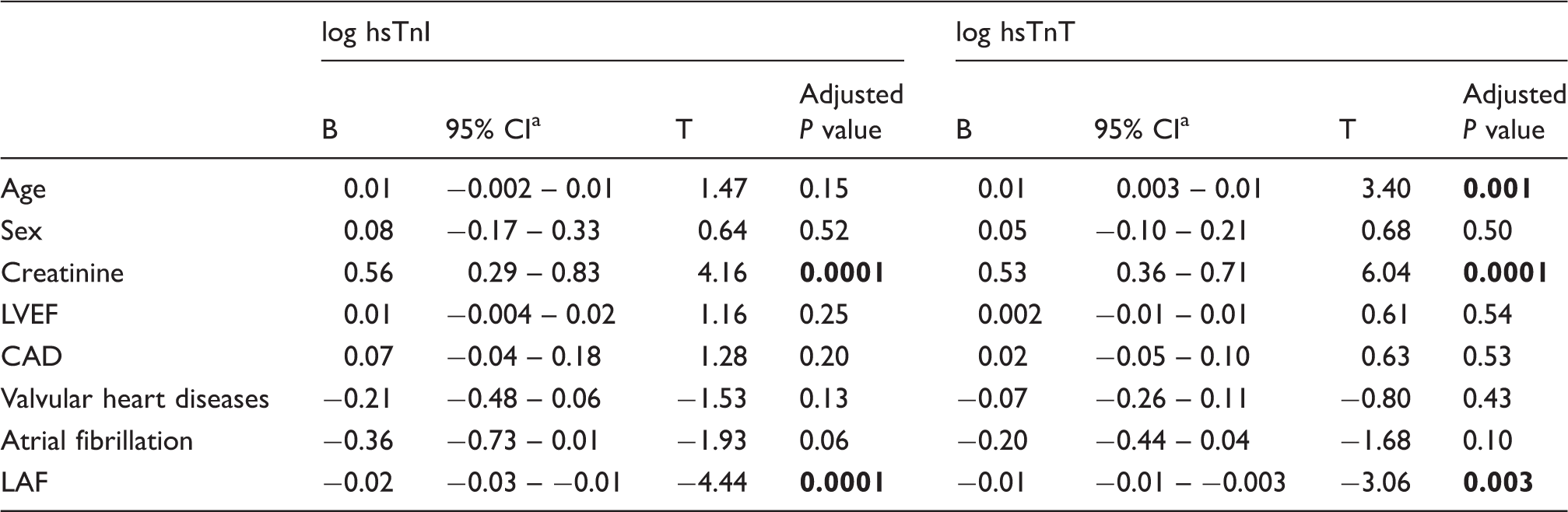
Note: Bold values indicate statistically significant P values (P < 0.05).
Confidence interval.
CAD: coronary artery disease; LAF: left atrial function; LVEF: left ventricular ejection fraction.
hsTn discriminate reduced LAF
As analysed by receiver-operating characteristic (ROC) curves, both hsTnT and hsTnI were able to discriminate significantly patients with impaired LAF < 55% from all others (hsTnT: AUC = 0.80; 95% CI 0.71–0.90; P = 0.0001; hsTnI: AUC = 0.74; 95% CI 0.63–0.85; P = 0.0001). Noteworthy, the AUCs of hsTn were numerically greater than the AUC of NT-proBNP (AUC = 0.72; 95% CI 0.61–0.83; P = 0.001) (P values for AUC comparisons: hsTnT versus NT-proBNP, P = 0.1; hsTnI versus NT-proBNP, P = 0.735) (Figure 3(a)).
Receiver-operating characteristic curves (ROC) revealing valuable discrimination of patients with reduced LAF of < 55% (panel a) and <45% (panel b).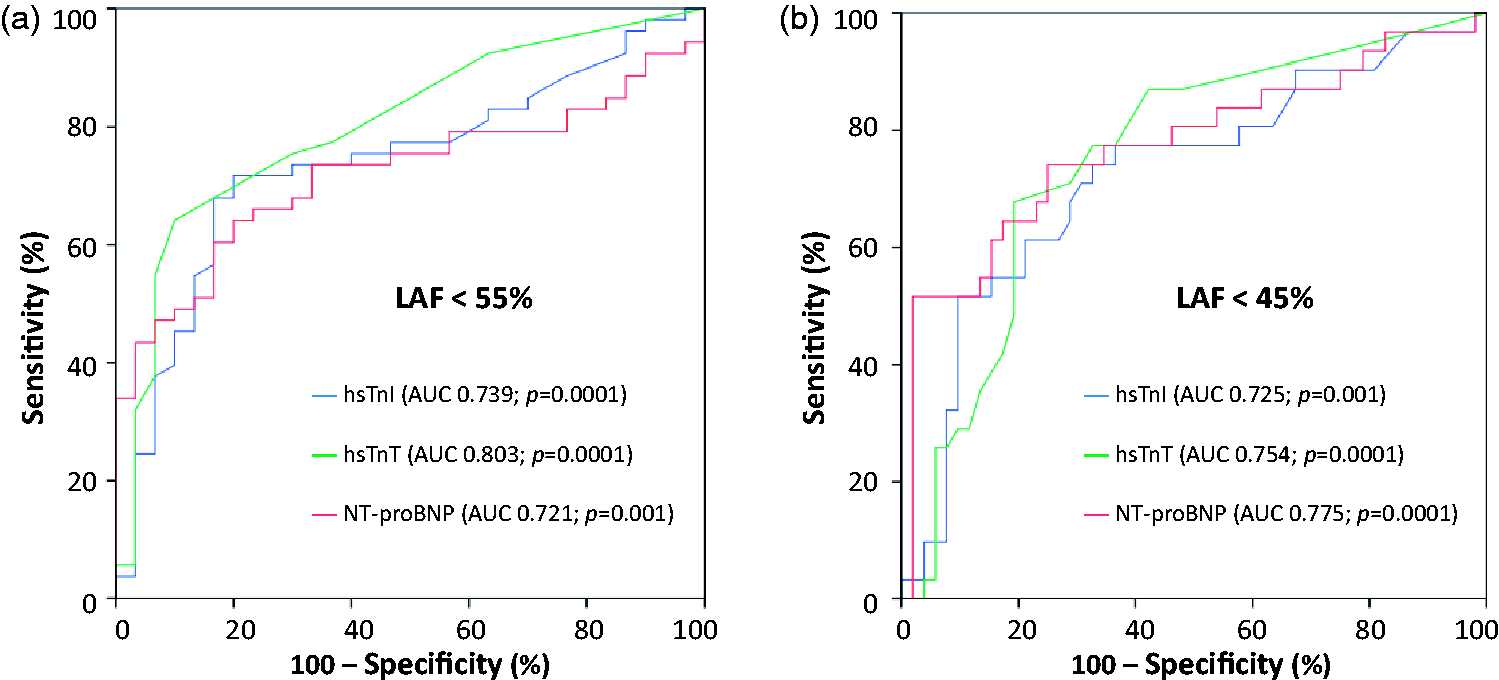
Furthermore, both hsTnT and hsTnI discriminated significantly patients with reduced LAF < 45%. AUCs of hsTn were comparable to the one of NT-proBNP (hsTnT: AUC = 0.75; 95% CI 0.65–0.86; P = 0.0001; hsTnI: AUC = 0.73; 95% CI 0.61–0.84; P = 0.001; NT-proBNP: AUC = 0.78; 95% CI 0.66–0.89; P = 0.0001) (P values for AUC comparisons: P > 0.05) (Figure 3(b)).
hsTn reveal independent associations with impaired LAF
Finally, clinical characteristics, cardiac comorbidities, NT-proBNP and hsTn were adjusted within multivariable logistic regression models in order to evaluate their association with impaired LAF.
Multivariable logistic regression models for evaluating the diagnostic ability of hsTn to identify patients with reduced LAF of <55%.
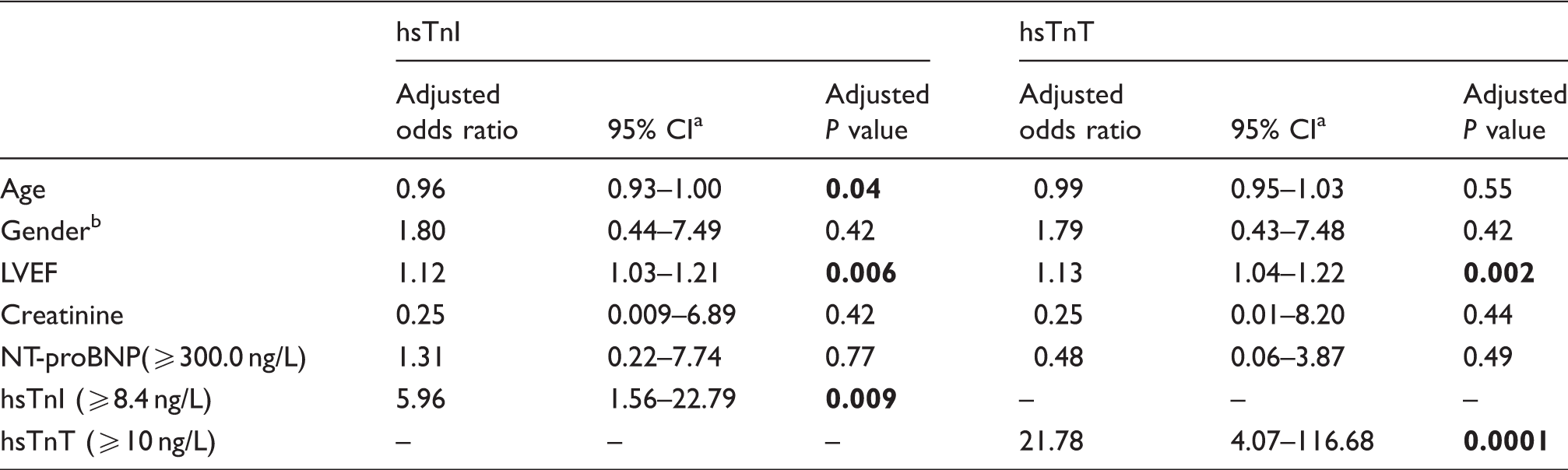
Note: Bold values indicate statistically significant P values (P < 0.05).
Confidence interval.
An adjusted odds ratio of <1 indicates an association of female gender with reduced LAF.
LVEF: left ventricular ejection fraction.
Multivariable logistic regression models for evaluating the diagnostic ability of hsTn to identify patients with reduced LAF of <45%.
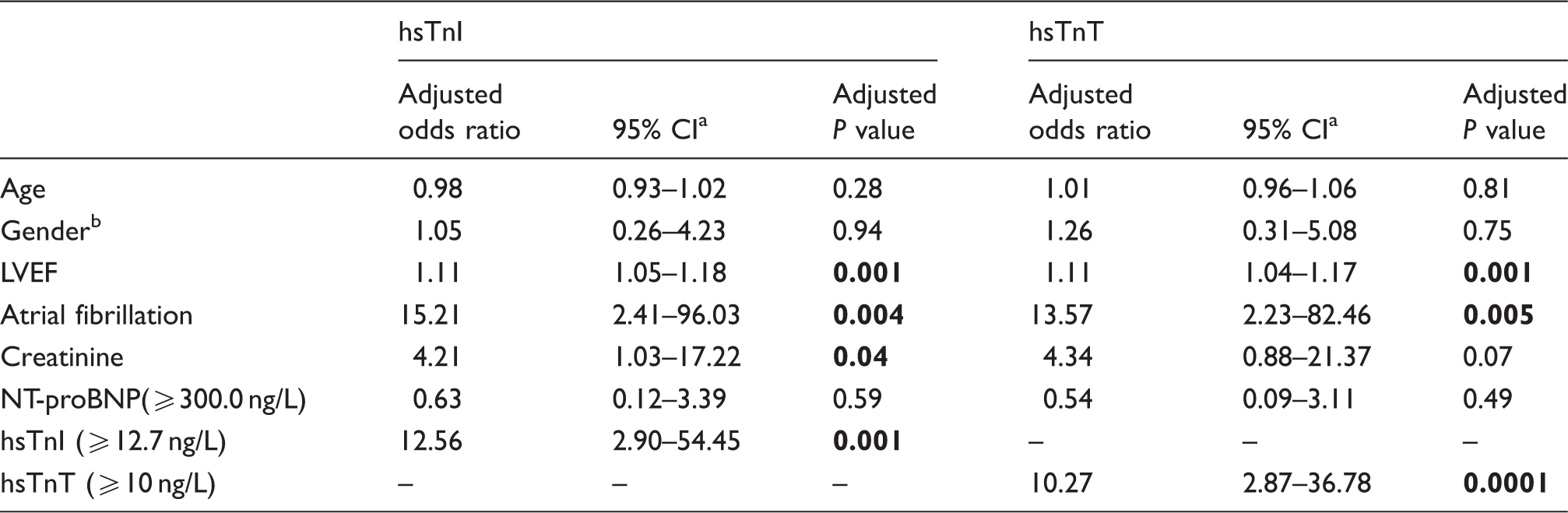
Note: Bold values indicate statistically significant P values (P < 0.05).
Confidence interval.
An adjusted odds ratio of <1 indicates an association of female gender with reduced LAF.
LVEF: left ventricular ejection fraction.
Discussion
The present study demonstrates that concentrations of hsTn are able to reflect LAF being assessed by cMRI. Both hsTnI and hsTnT were correlated inversely with LAF within multivariable linear regression models and increased significantly according to the different stages of impaired LAF. HsTn discriminated both patients with impaired LAF < 55% and <45%. In multivariable logistic regression models, hsTn were still associated significantly with impaired LAF after adjustment for age, sex, creatinine, LVEF, AF and NT-proBNP.
The cTn complex is composed of proteins taking part in cardiac contraction such as TnI, TnC and TnT. TnT is fixed to the cardiac myofibrillar troponin-tropomyosin complex. TnI inhibits troponin-tropomyosin interactions by reducing TnC affinity for calcium. 34 Both TnI and TnT are specific for cardiac injury.23,34,35 cTn are of major importance in the diagnosis of AMI. 20 Elevated hsTn were shown to be associated with an increased incidence rate of AF in patients with atherosclerosis. 26 In addition, hsTn correlated significantly with LVEF and LVEDP. 25 Furthermore, hsTn were shown to be associated with short- and long-term prognosis in CHF patients.12–14 Temporal increases of hsTnT were associated independently with incident coronary heart disease, death and HF events. 36 Several studies showed that cTn concentrations are associated with an increased risk of morbidity and mortality in both AHF and CHF. 34
HF represents a preventable and treatable disease. 37 In early stages of HF, patients can present with asymptomatic structural or functional cardiac alterations (systolic or diastolic LV dysfunction, or LA dysfunction), which are known as precursors of HF. It is of major importance to identify these precursors because they are related to a poor outcome. Starting clinical follow-up as well as targeted medical therapy at the precursor stages of HF may reduce mortality in patients with asymptomatic compensated HF.38,39 The present study shows that LAF is decreased despite a constant LVSV and only slightly reduced LVEF, which supports the hypothesis that LAF is a precursor of HF. Furthermore, the LA is the ultimate link between pulmonary and systemic circulation. It serves as a reservoir for pulmonary venous return during LV contraction and isovolumetric relaxation. As a conduit, LA modulates LV filling through passive blood transfer into the LV during LV diastole. During the final phase of diastole, LA contracts actively and contributes up to 30% of LVSV. 2 Furthermore, LA communicates with the neuro-hormonal systems via secretion of natriuretic peptides by stimulating atrial volume receptors through increased LA filling and diastolic stretch. It interacts with the sympathetic nervous system as well as the renin-angiotensin-aldosterone system.1,3
Dysfunctions of LA may lead to different problems. It has been shown earlier that HFrEF is characterized by greater eccentric LA remodeling, whereas HFpEF is characterized by increased LA stiffness and greater LA pressure pulsatility, which might contribute to greater AF burden. 8 AF correlated with an increased risk of morbidity and mortality in patients with HFrEF and HFpEF. The increasing risk being attributable to AF was even higher in patients with HFpEF. 5 In addition, LA dysfunction in general was associated more strongly with mortality as well as a higher risk of re-hospitalization in patients with HFpEF.7–9 LA fibrosis was associated with a poor prognosis in patients with HFpEF and HFrEF and adverse structural remodeling contributed to the development and attenuation of AF.4–6 LAF was shown to be a powerful predictor of incident AF, hospitalization for HF and of mortality in patients with HF and was related to major adverse cardiac events most. 40
To the best of our knowledge, only few prior studies suggested an association between LAF and hsTnT,16,27,41,42 whereas the present study is the first investigating the correlation between hsTnI and LAF being assessed by cMRI. Gupta et al. 41 found an association between cMRI-assessed LAF and hsTnT in participants from a general population in the Dallas Heart Study. Beatty 27 et al. demonstrated that elevated hsTnT concentrations were associated with greater severity of cardiac structural and functional abnormalities as measured inter alia by LAF index, assessed by echocardiographic examination. Furthermore, Ravassa et al. 16 showed that LA volume index (VI) being assessed by echocardiography increased with hsTnT in the general population. Circulating hsTnT was associated with LA (and LV) remodeling and LA enlargement. 16 An association between increased hsTnT and larger LA diameter (LAD) being assessed by echocardiography was shown in patients with HFpEF. 42
Kuhl et al. 11 showed that although transthoracic echocardiography (TTE) remains the simplest routinely available method of assessing the LA, it was less accurate than cMRI. TTE underestimated LAV systematically, whereas cMRI (and computed tomography) could assess LAV and LAF properties more precisely. This suggests that the chosen method of cardiac imaging via cMRI in the present study is advantageous in comparison to related prior studies, which used echocardiography for LA assessment mostly.16,27,42 It was shown that LAF, but not LAV was associated with incident HF hospitalization, cardiovascular mortality, all-cause mortality and AF. The assessment of LAF provided important information beyond LAV alone.40,41,43 This suggests that assessing LAF – as done in the present study – is superior and of more clinical relevance than assessing LA enlargement only – as done in prior studies.16,42
Taken together, the present study combines modern cardiac imaging by cMRI plus novel cardiac biomarkers (such as hsTnT, hsTnI and NT-proBNP) to assess LAF. The proven diagnostic value of hsTn might allow the detection of LA dysfunction already at very early and compensated stages of CHF with only mild to moderate symptoms revealing only mild to moderate reduction of LVEF and a still preserved LVSV, which turns LAF into a precursor of HF. Whether routine measurements of hsTn either with or without cMRI imaging within standard of care in such patients might lead to a more close-meshed follow-up of these patients or might initiate or improve HF treatment at earlier stages, needs to be addressed in upcoming larger prospective and randomized controlled studies. The clinical purpose for this approach is to avoid irreversible LA structural remodelling, development or attenuation of AF as well as poor cardiac outcome in terms of re-hospitalization.4,5,7–9
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TB performs reagent evaluation studies for Roche diagnostics. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the DZHK (Deutsches Zentrum fur Herz-Kreislauf-Forschung – German Centre for Cardiovascular Research).
