Abstract
Background
This study evaluates the associations between high-sensitivity troponin I and T (hs-TnI/hs-TnT) and the stages of heart failure with preserved ejection fraction (HFpEF)/diastolic dysfunction.
Methods
Blood samples for biomarker measurements (hs-TnI/hs-TnT/NT-proBNP) were collected within 24 h of routine echocardiographic examination. Patients with left ventricular ejection fraction <50%, right ventricular dysfunction and moderate-to-severe valvular heart disease were excluded. Graduation of diastolic dysfunction was determined according to current guidelines.
Results
A total of 70 patients were included. Hs-TnT concentrations increased significantly according to the progression of diastolic dysfunction (P = 0.024). Hs-TnT was able to discriminate patients with diastolic dysfunction grade III (AUC = 0.737; P = 0.013), while NT-proBNP revealed a greater AUC (AUC 0.798; P = 0.002). Concentrations of hs-TnI increased only numerically according to the increasing stages of diastolic dysfunction (P = 0.353). In multivariable logistic regression models, hs-TnT concentrations > 28 ng/L were associated with diastolic dysfunction grade III (OR = 4.7, P = 0.024), even after adjusting for NT-proBNP.
Conclusion
Increasing concentrations of hs-TnT may reflect the stages of diastolic dysfunction being assessed by echocardiography, whereas hs-TnI does not show any association with diastolic dysfunction.
Keywords
Introduction
Heart failure with preserved ejection fraction (HFpEF) accounts for 50% of all heart failure syndromes, and its rising prevalence, often credited to demographic variations and a widening cardiovascular risk factor profile, represents a growing epidemiological problem.1–4
The pathophysiology of HFpEF and the cardiac phenotypes in these patients are typically heterogeneous.1,5 Myocardial relaxation, myocardial compliance and left ventricular (LV) pressure are the triad of factors serving the basis of our understanding of diastolic function. Causal connections like age, ischaemia and hypertrophy have been associated with impaired myocardial relaxation, while scarring and fibrosis reduce myocardial compliance necessitating higher filling pressures. 6 A recent study has suggested that chronic inflammation and coronary microcirculatory disturbances may also influence the pathophysiology of HFpEF. 1 The integration of these influences and contrasting views facilitates the opening of various target points for novel diagnostic biomarkers and pharmacological therapies.
Echocardiography is the most common imaging technique in clinical cardiology practice. As a non-invasive method, it allows a real-time overview of the cardiac function. 7 The use of echocardiographic parameters to assess impaired myocardial diastolic function is influenced by contextual factors such as emphysema and adiposity. These factors serve as an impediment against a fully objective and accurate assessment of the cardiac status. The use of biomarkers to quantify cardiac dysfunction in HFpEF is naturally free of such disadvantages; however, early studies researching these have yielded disappointing results. This has been partly attributed to their poor reliability. 8
B-type natriuretic peptide (BNP) and its amino-terminal precursor (NT-proBNP) shows significant association with echocardiographic parameters used to diagnose diastolic dysfunction. Although there has been no conclusive evidence demonstrating a specific association of serum BNP and NT-proBNP concentrations to HFpEF patients, it has been implied that a non-linear correlation between varying serum concentrations of NT-proBNP and increased risks in cardiovascular mortality as well as re-hospitalization exists. 9
Increased concentrations of cardiac troponin (cTn) in patients with HFpEF vary between 22% to 44% (conventional assay) and 55% to 72% (hs-assay). 10 Additionally, serum concentrations of cTn correlate significantly with LV mass and size, left atrial (LA) diameter and the severity of diastolic dysfunction as determined by echocardiography. A comparison of cTn concentration in patients with HFrEF and HFpEF showed lower values for patients diagnosed with the latter variant. 10 Nevertheless, it appears that high-sensitivity troponin T (hsTnT) has significant prognostic value for overall survival and re-hospitalization among HFpEF patients. 11 A recent study from Japan reported that an increased high-sensitivity troponin I (hsTnI) concentration in combination with elevated serum concentrations of NT-proBNP correlates better to the risk of developing heart failure in patients diagnosed with hypertension and preserved ejection fraction. 12
These data serve as the basis of our study, which sets out to investigate the hypothesis that quantitative concentrations of hsTnI and/or hsTnT could reflect the severity of the diastolic dysfunction and correspond to echocardiographic parameters associated with HFpEF.
Methods
Study population
The ‘Cardiovascular Imaging and Biomarker Analyses’ (CIBER) study (clinicaltrials.gov identifier: NCT 03074253) represents a clinically prospective, controlled and monocentric study conducted at the University Medical Center Mannheim, Germany. The research adhered to the principals outlined in the Declaration of Helsinki and was approved by a regional ethics committee. Written informed consent was obtained from all patients.
For the present study, patients presenting to our echocardiography laboratory with compensated HFpEF as being assessed by routine echocardiographic imaging were included between 2014 and 2016. Patients under the age of 18 years, left ventricular ejection fraction <50%, reduced right ventricular function (RVF) corresponding to a tricuspid annular posterior systolic excursion (TAPSE) <17 mm and moderate to severe valvular heart disease were excluded from this study.
All relevant clinical data including the medical history, results of echocardiographic assessment and the laboratory blood values at baseline were collected from the hospital documentation system and summarized in a database for further reference. Follow-ups were scheduled for all patients after 6 and 12 months by telephone or personal visits in case of re-hospitalization.
Transthoracic echocardiography
Transthoracic echocardiography was carried out by independent echocardiographic specialists/physicians using standard techniques according to the guidelines established by ASE/EACVI to acquire reproducible images.13,14 The results were checked and revised by an independent study physician being well experienced in the assessment of HFpEF and being unaware of the patients clinical and biomarker data.
Assessment of the systolic function with the ejection fraction (EF using Simpsons biplan method), end-systolic and -diastolic volumes and diameters, right ventricular (RV) function with TAPSE and valvular heart diseases were included in the investigation. The classification of the diastolic dysfunction was determined by the mitral inflow velocities examination (E and A) and the mitral annular velocities examination (E'). In patients with atrial fibrillation, the LV end-diastolic pressure (LVEDP) was measured to evaluate the diastolic function. Patients were divided into the four groups: Patients with good diastolic function, diastolic dysfunction grade 1, grade 2 or grade 3. Details for HFpEF classification based on echocardiography are outlined in supplemental Table 1 for concise presentation purpose.
Laboratory analysis
All expressed biomarkers were measured in the serum of patients’ blood. Within 24 h before or after the echocardiographic imaging, peripheral venous blood samples were taken from each patient and collected in serum monovettes® tubes and centrifuged at 2500 g for 10 min at 20°C. The aliquoted samples were cooled down with liquid nitrogen before being stored at –80°C until analysis. The whole processing took part within 2 h after blood extraction. After thawing, the samples were mixed gently by inverting and centrifuged at 2500 g for 10 min at 20°C for Troponin T and N-terminal pro-brain natriuretic peptide (NT-proBNP) and creatinine analysis.
For TnI measurement, the samples were gently mixed by inverting after thawing and centrifuged for 30 min at 3000 g at 4°C. Troponin I was measured with the STAT High Sensitive Troponin-I assay on an Architect i1000 analyzer (Abbott, Wiesbaden, Germany) with a limit of blank (LoB) of 0.7–1.3 ng/L and a limit of detection (LoD) of 1.1–1.9 ng/L. The 10% imprecision was at 4.7 ng/L. The 99th percentile was at 26.2 ng/L. 15 The reportable range for our study was 0–50,000 ng/L. Troponin T was measured with the Troponin T hs STAT assay on a cobas e 602 analyzer (Roche Diagnostics, Mannheim, Germany). The LoB for this assay was 3 ng/L and the LoD 5 ng/L. The 10% imprecision was 13 ng/L. The 99th percentile was at 14 ng/L. 16 The reportable range for our study was 3–10,000 ng/L. NT-proBNP was measured with the proBNP II STAT assay on a cobas e 602 analyzer (Roche Diagnostics, Mannheim, Germany). The LoD was at 0.6 pmol/L. The reportable range for our study was 0.6–4130 pmol/L. 17 Serum creatinine was measured with the Creatinine Jaffe Generation 2 assay on a cobas c 702 system. The LoD was 15 μmol/L. The reportable range for our study was 41–2226 μmol/L. 18 C-reactive protein (CRP), gamma-glutamyl-transferase (GGT) and lactic dehydrogenase (LDH) were determined under ISO-15189 accreditation conditions. Samples in Li-Hep Monovettes® (Sarstedt, Nümbrecht, Germany) were centrifuged at 2500 g for 10 min at 20°C and subsequently analysed on Vista Dimension 1500 platform (Siemens, Eschborn, Germany). CRP was determined by microparticle-enhanced nephelometric immunoassay (Siemens, Eschborn, Germany) with analytical measuring range (AMR) from 0.29 to 190 mg/L. GGT and LDH assays are both based on bichromatic kinetics (Siemens, Eschborn, Germany) and show an AMR from 3 to 800 U/L for GGT, and 6 to 1000 U/L for LDH.
Statistical analysis
Statistical analysis was conducted for all patients’ data as well as the four predefined subgroups according to the severity of HFpEF. The Student’s t-test was used with variables following normal distribution, otherwise the Kruskal-Wallis test (non-parametric test) was appointed as the method of choice. Discrepancies from Gaussian distribution were furthermore screened by the Kolmogorov-Smirnov test to accommodate and adjust for the relatively small sample size. Depending on the distribution, all data are presented as mean with confidence interval (CI) or median with interquartile ranges (IQR; 25th to 75th percentiles).
Correlation of hsTnT and hsTnI serum concentrations with non-parametric data, such as echocardiographic indices and medical parameters, is determent by the Spearman's rank correlation coefficient. Cut-offs for the biomarkers was set at the group-specific medians of each biomarker. Multivariable linear or logistic regression analysis with backward elimination was performed to identify any potential source of irritation. Subsequently, depending on the significance of those irritations’ adjustments of several clinical parameters or biomarkers were carried out. The Hanley and McNeil method were implemented to identify if hsTnT or hs TnI was more accurate than NT-proBNP in the identification of HFpEF. 19
Tests were carried out using SPSS software (SPSS Software GmbH, IBM, Armonk, NY) and statistical significance was attained whenever the observed P-value of a test statistic was less than 0.05.
Results
Study population
A total of 70 HFpEF patients aged between 22 years and 97 years were included in this study. The baseline clinical characteristics are shown in Table 1. The mean age was 65 years and baseline demographics represented a balanced gender distribution, with 49% of the study population being women. The median creatinine value was estimated to be 82.21 μmol/L (range 67.76–101.2 μmol/L) in the total cohort. Interestingly, arterial hypertension was noticed in almost 80% of the study patients (n = 56), effectively proving arterial hypertension as a predominant risk factor for HFpEF patients. Cardiac co-morbidities such as atrial fibrillation was diagnosed in 38% of the patients, whereas 50% suffered from pre-existing coronary artery disease, although only 7% of these presented themselves with a prior history of myocardial infarction.
Baseline characteristics of study patients.
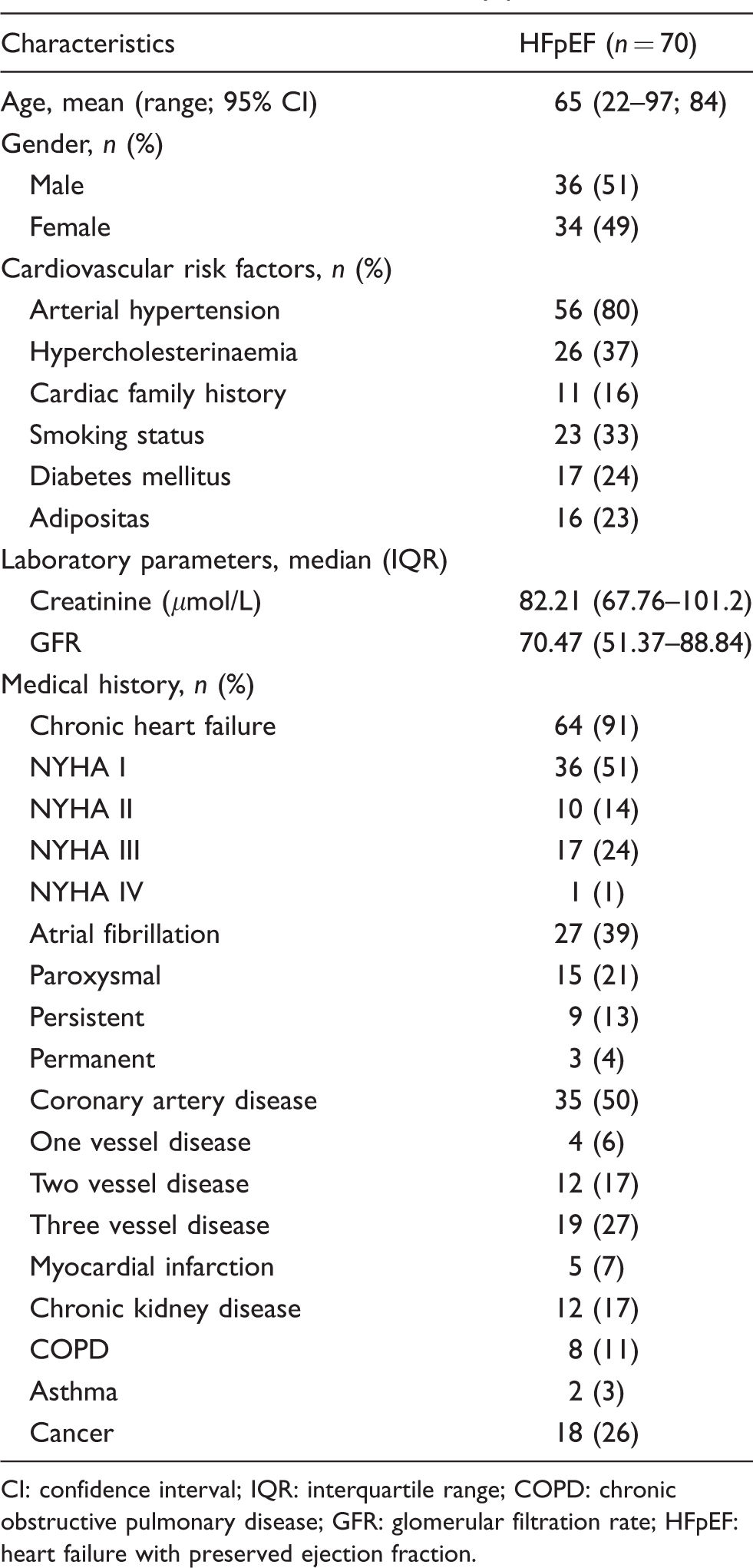
CI: confidence interval; IQR: interquartile range; COPD: chronic obstructive pulmonary disease; GFR: glomerular filtration rate; HFpEF: heart failure with preserved ejection fraction.
Echocardiographic assessment
Based on the significant correlation between HFpEF and E/A (P = 0.0001), E' lat (P = 0.009) and E/E' (P = 0.0001) parameters, the group population was segregated accordingly to allow differential analysis. Study patients were diagnosed with either a good diastolic function (20%; n = 14) or a diastolic dysfunction grade I (21%; n = 15), grade II (43%; n = 30) or grade III (16%; n = 11).
Detailed results of the distribution of echocardiographic indices among the HFpEF subgroups are described in Tables 2 to 4. Consistent with the trial inclusion criteria, neither the ejection fraction (P = 0.109) nor TAPSE (P = 0.41) differed significantly among the HFpEF subgroups. Echocardiographic parameters attributed to have statistical significance include the LV posterior wall thickness (LVPW; P = 0.045), LA dimensions (P = 0.044) and LA volume (P = 0.002).
Distribution of echocardiographic indices according to HFpEF subgroups.
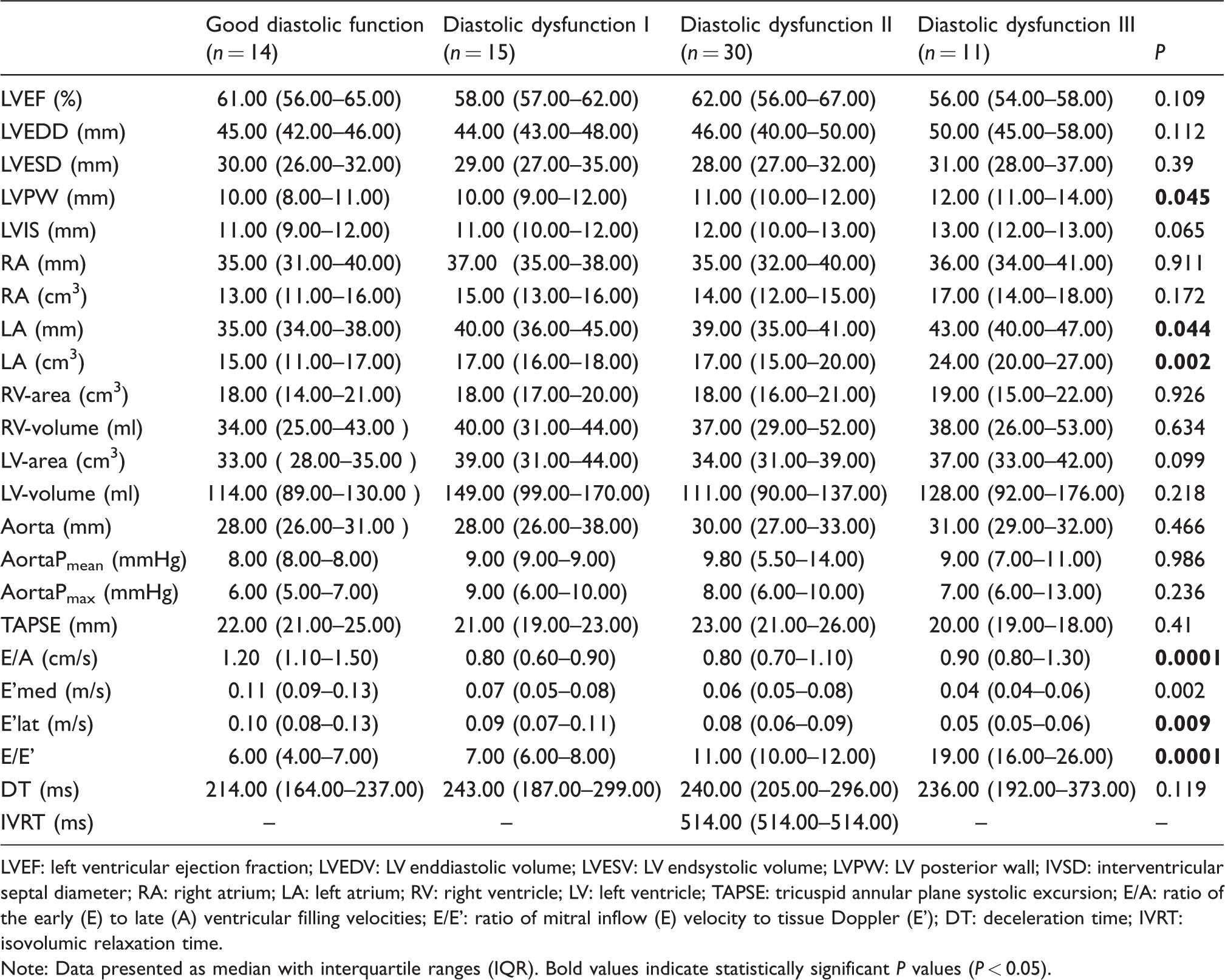
LVEF: left ventricular ejection fraction; LVEDV: LV enddiastolic volume; LVESV: LV endsystolic volume; LVPW: LV posterior wall; IVSD: interventricular septal diameter; RA: right atrium; LA: left atrium; RV: right ventricle; LV: left ventricle; TAPSE: tricuspid annular plane systolic excursion; E/A: ratio of the early (E) to late (A) ventricular filling velocities; E/E': ratio of mitral inflow (E) velocity to tissue Doppler (E'); DT: deceleration time; IVRT: isovolumic relaxation time.
Note: Data presented as median with interquartile ranges (IQR). Bold values indicate statistically significant P values (P < 0.05).
Univariable correlations between hsTNT and baseline characteristics, biomarkers and echocardiographic indices according to HFpEF subgroups.
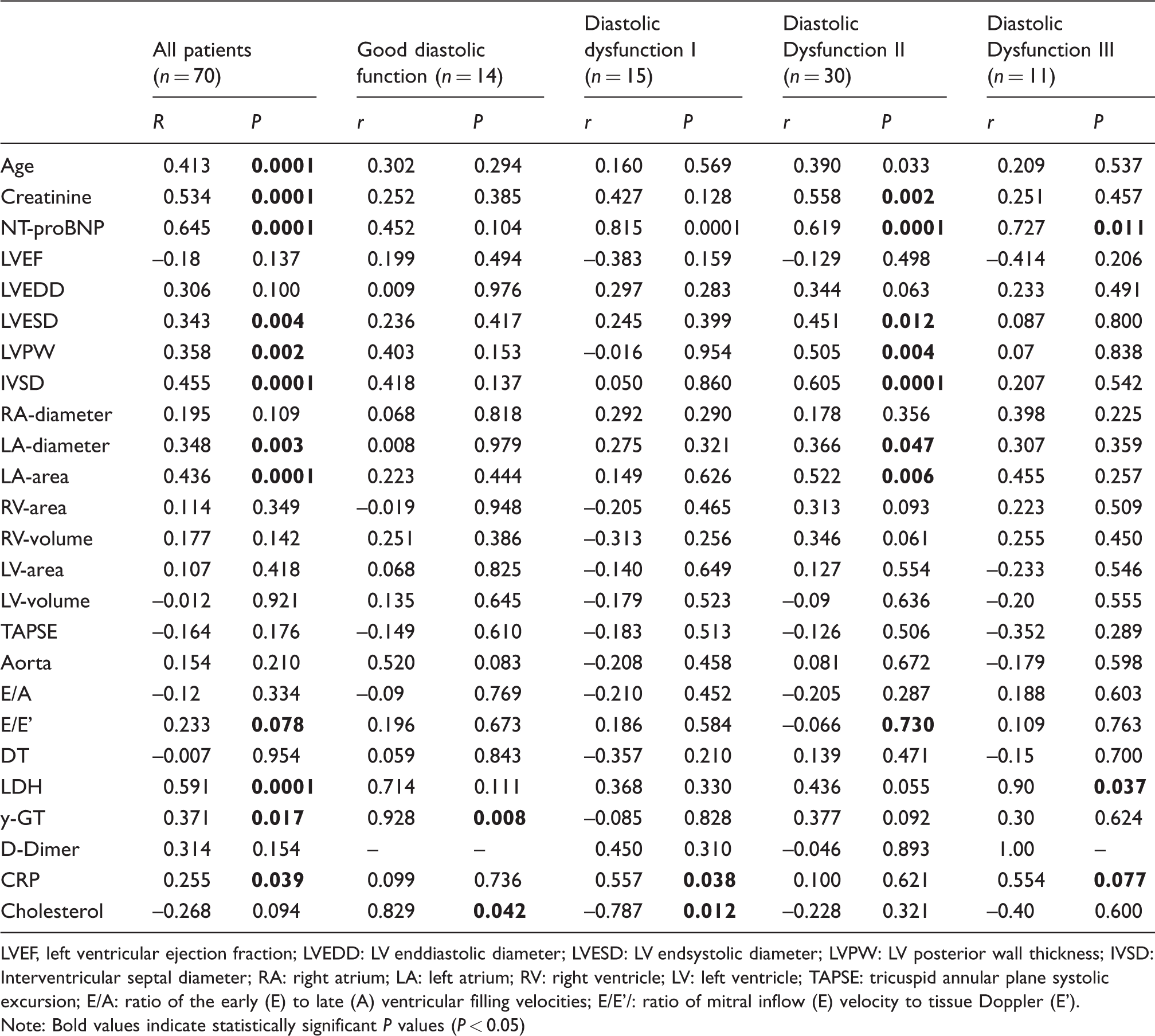
LVEF, left ventricular ejection fraction; LVEDD: LV enddiastolic diameter; LVESD: LV endsystolic diameter; LVPW: LV posterior wall thickness; IVSD: Interventricular septal diameter; RA: right atrium; LA: left atrium; RV: right ventricle; LV: left ventricle; TAPSE: tricuspid annular plane systolic excursion; E/A: ratio of the early (E) to late (A) ventricular filling velocities; E/E'/: ratio of mitral inflow (E) velocity to tissue Doppler (E').
Note: Bold values indicate statistically significant P values (P < 0.05)
Univariable correlations between hsTNI and baseline characteristics, biomarkers and echocardiographic indices according to HFpEF subgroups.
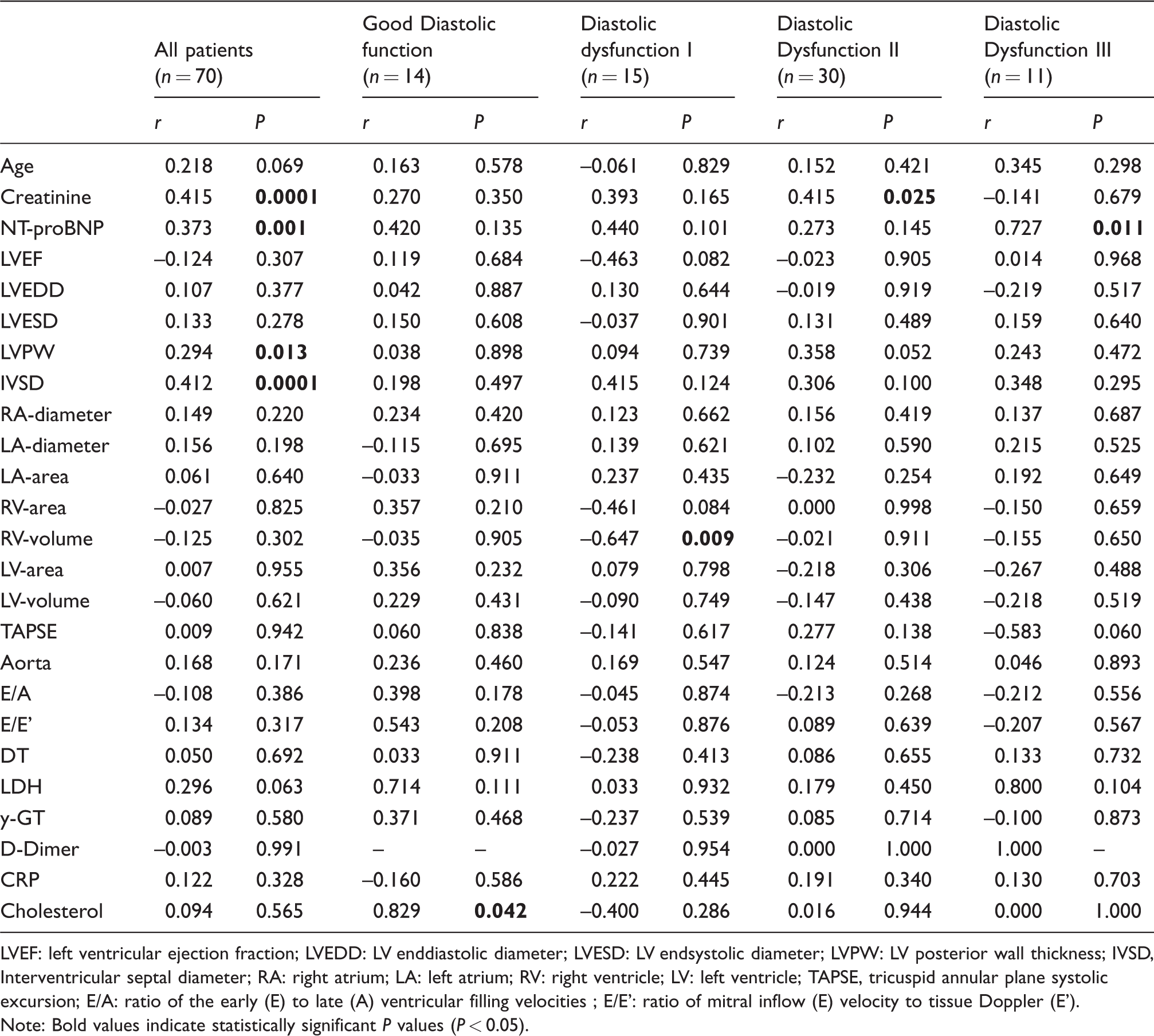
LVEF: left ventricular ejection fraction; LVEDD: LV enddiastolic diameter; LVESD: LV endsystolic diameter; LVPW: LV posterior wall thickness; IVSD, Interventricular septal diameter; RA: right atrium; LA: left atrium; RV: right ventricle; LV: left ventricle; TAPSE, tricuspid annular plane systolic excursion; E/A: ratio of the early (E) to late (A) ventricular filling velocities ; E/E': ratio of mitral inflow (E) velocity to tissue Doppler (E').
Note: Bold values indicate statistically significant P values (P < 0.05).
Characteristics of hsTnT and hsTnI in the present HFpEF cohort
HsTnT reflected the progression of HFpEF and increased concentrations corresponded to the development of diastolic dysfunction (P = 0.024). Concentrations of hsTnI increased only numerically according to increasing stages of HFpEF (P = 0.353). The distribution of hsTnT, hsTnI and NT-proBNP concentrations is graphically reproduced in Figure 1.
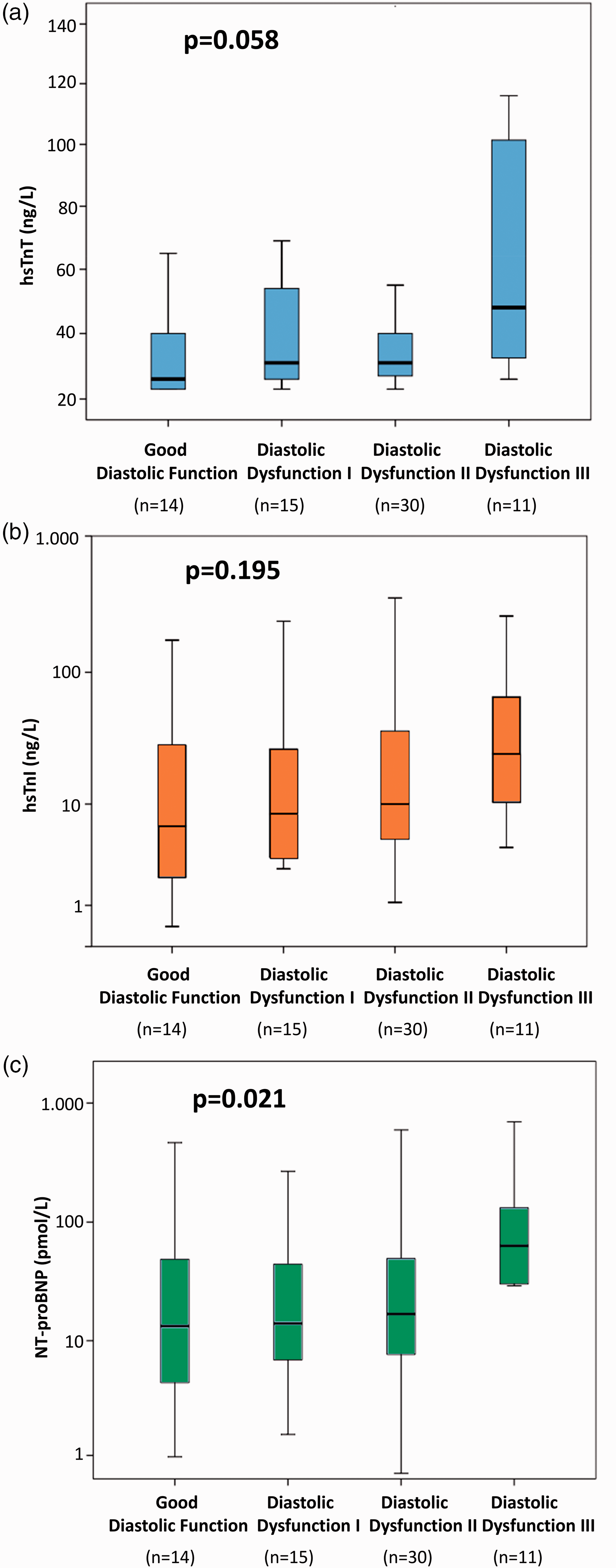
Distribution of hs-TnT (panel A), hs-TnI (panel B) and NT-proBNP (panel C) serum concentrations according to subgroups of diastolic dysfunction. Data are presented as medians with 25th and 75th percentiles (boxes) and 5th and 95th percentiles (whiskers).
Correlation between hsTnT and both baseline characteristics and echocardiographic indices
Univariable correlations between hsTnT and both baseline characteristics and echocardiographic indices are given in Table 5. HsTnT demonstrated a strong relation to serum creatinine (P = 0.0001) and age (P = 0.0001). In this scenario, a significant correlation with the reference biomarker for chronic heart failure, NT-proBNP was also evident in each subgroup of HFpEF (P = 0.0001; range 0.011–0.0001). The relationship between parameters indicating HFpEF and hsTnT can only be described as a tendency (E/A, P = 0.334 and E/E', P = 0.078). Serum concentrations of hsTnT in all patients corresponded with echocardiographic indices describing the LA chamber (LA-diameter, P = 0.003 and LA-area, P = 0.0001). Furthermore, an association to wall thickness as well as the LV endsystolic diameter (LVESD) could be ascertained (LVESD, P = 0.004; LVPW, P = 0.002; IVSD, P = 0.0001). Interestingly, these correlations could only be individually determined in grade II diastolic dysfunction (LA-diameter, P = 0.047; LA-area, P = 0.006; LVESD, P = 0.012; LVPW, P = 0.004; IVSD, P = 0.0001). In all patients, hsTnT corresponded to laboratory values of cell downfall, signs of inflammation as well as GGT (LDH, P = 0.001; CRP, P = 0.039; GGT, P = 0.017).
Multivariable logistic regression model for evaluating the ability of hsTnT to identify patients with diastolic dysfunction III.
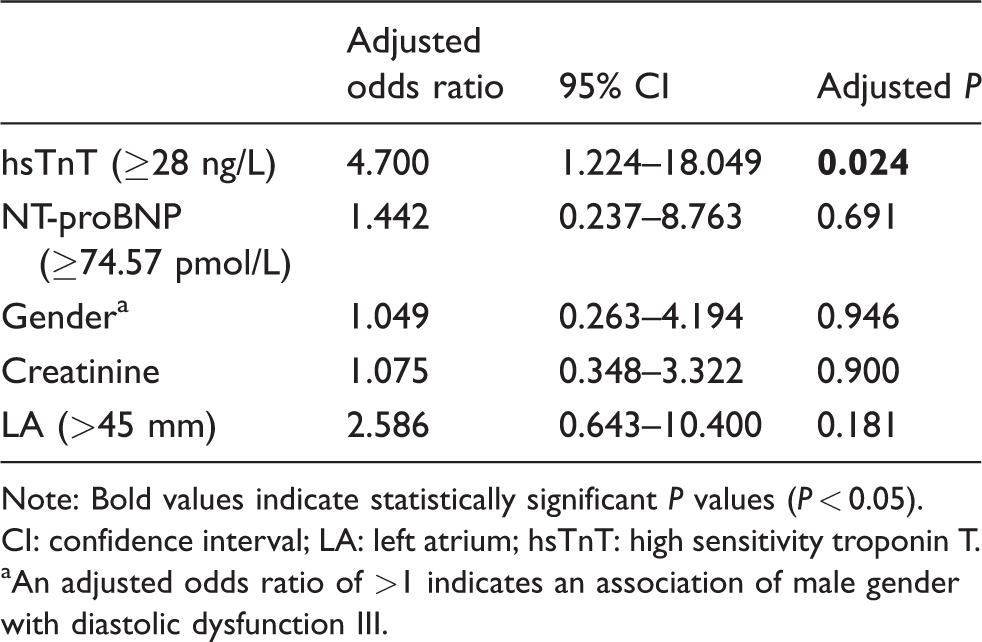
Note: Bold values indicate statistically significant P values (P < 0.05).
CI: confidence interval; LA: left atrium; hsTnT: high sensitivity troponin T.
aAn adjusted odds ratio of >1 indicates an association of male gender with diastolic dysfunction III.
Correlation between hsTnI and both baseline characteristics and echocardiographic indices
Demonstrations of hsTnI association to baseline characteristics as well as echocardiographic indices through a univariate linear regression analysis are outlined in Table 6. A strong correlation with serum creatinine (P = 0.0001) is shown through all groups of HFpEF. A relationship with the reference biomarker NT-proBNP, describing chronic heart failure, could be reproduced in all patients (P = 0.001). Meanwhile, the concentration of correlation between the biomarker and the different subgroups, defined by varying severities of diastolic dysfunction, was insignificant. Surprisingly, concentrations of hsTnI did not correspond with any echocardiographic indices suggesting HFpEF (E/A, P = 0.386 and E/E', P = 0.317). Serum concentrations of hsTnI in all patients corresponded significantly to LVPW (P = 0.013) and IVSD (P = 0.001), whereas this was statistically insignificant across the various HFpEF subgroups. Additionally, there existed a significant positive correlation between the right ventricular volume and patients with HFpEF grade I (P = 0.009). This relationship was not expressed in other subgroups with advanced diastolic dysfunction.
Multivariable logistic regression model for evaluating the ability of hsTnI to identify patients with diastolic dysfunction III.
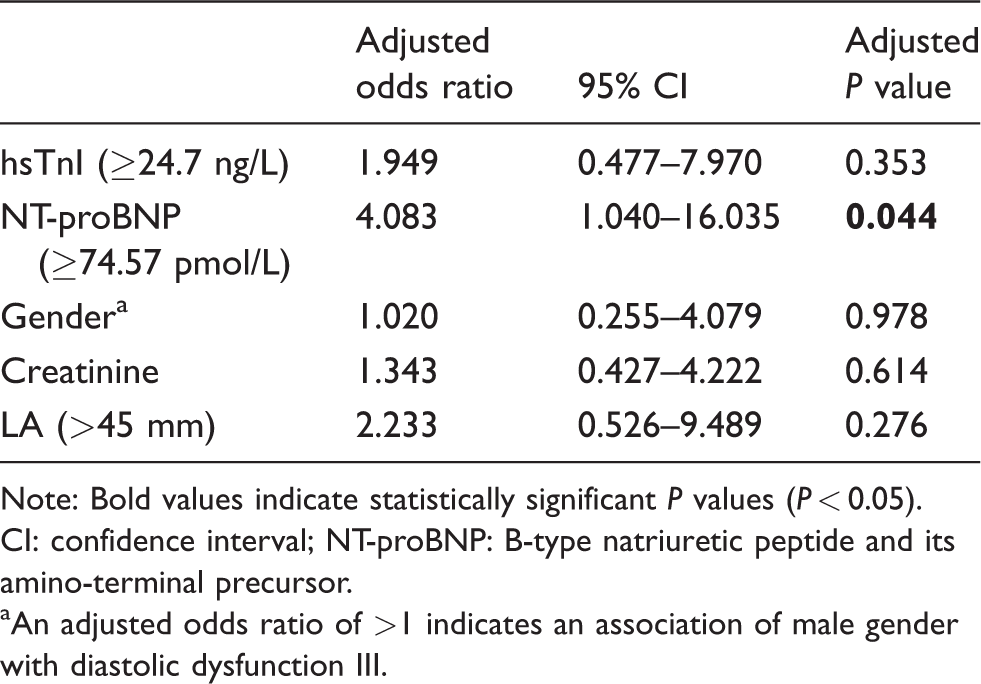
Note: Bold values indicate statistically significant P values (P < 0.05).
CI: confidence interval; NT-proBNP: B-type natriuretic peptide and its amino-terminal precursor.
aAn adjusted odds ratio of >1 indicates an association of male gender with diastolic dysfunction III.
hsTnT discriminates patients with diastolic dysfunction grade III
Receiver-operating characteristic (ROC) analysis revealed that hsTnT were able to identify patients with HFpEF grade III (AUC 0.737; P = 0.013) (Figure 2(a)). NT-proBNP as the reference biomarker also detected such characteristics, admittedly with a greater AUC (AUC 0.798; P = 0.002). Additionally, the combination of hsTnT and NT-proBNP showed no improvement in the diagnostic accuracy of diastolic dysfunction grade III (AUC 0.767; P = 0.005). The AUC for hsTnI discriminating diastolic dysfunction grade III was 0.667 (P = 0.08) (Figure 2(b)).
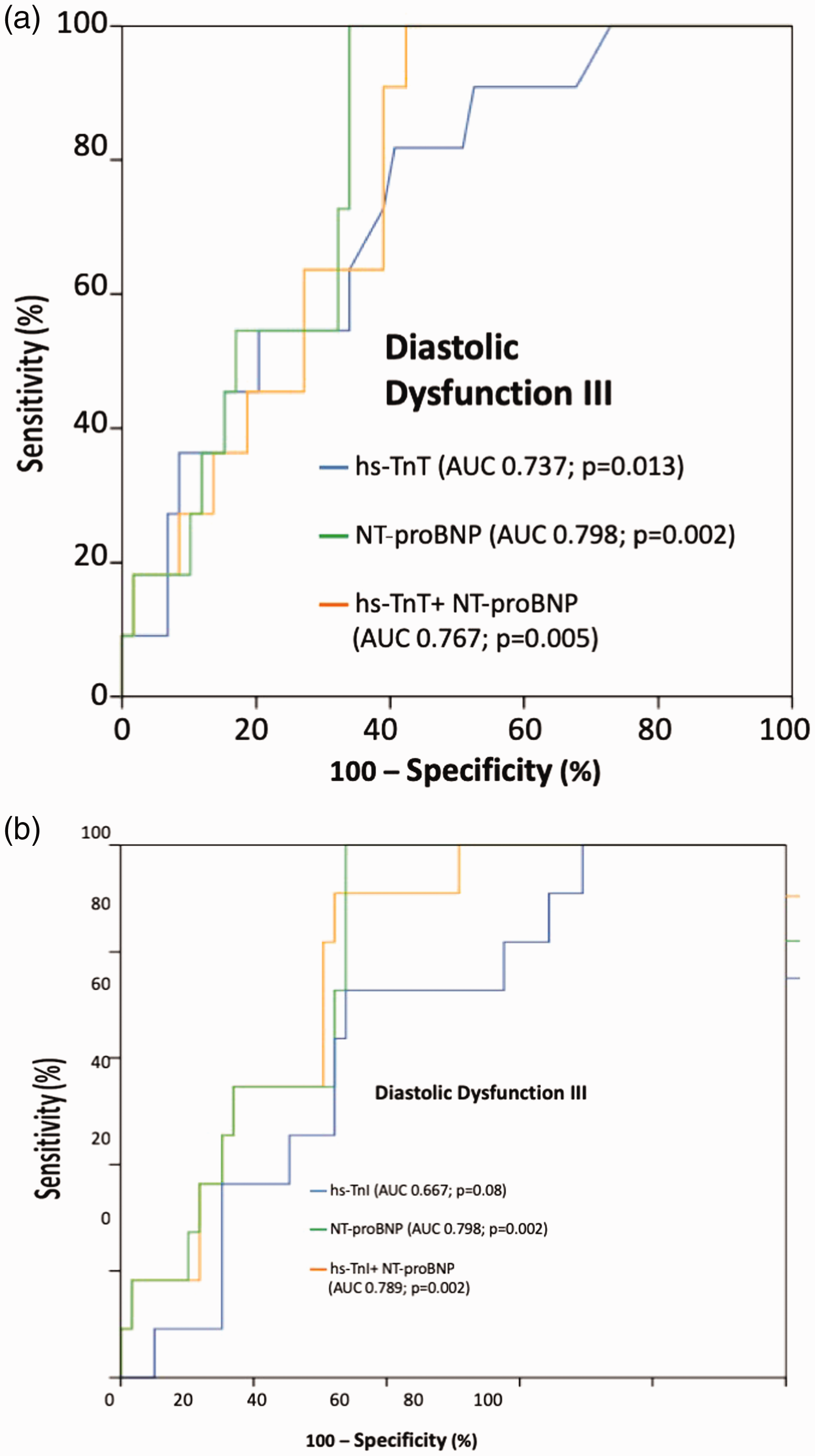
Receiver-operating characteristic curves (ROC) revealing valuable discrimination of patients with diastolic dysfunction III by hsTnT (panel A) and by hsTnI (panel B).
Multivariate logistic regression models for evaluating diastolic dysfunction grade III with hsTnT
Multivariate logistic regression models for log-transformed hsTnT were adjusted according to its strong correlation with baseline characteristics such as gender, age, creatinine, NT-proBNP concentrations and echocardiographic indices such as the left atrial diameter (Table 5). After stepwise backward analysis, results indicated that the probability of diagnosis with diastolic dysfunction grade III in patients with hsTnT concentrations >2.8 ng/L increased almost fivefold (adjusted odds ratio [OR] = 4.7, 95% CI: 1.224–18.049, P = 0.024). Patients with serum concentrations of NT-proBNP > 74.57 pmol/L were not significantly associated with HFpEF grade III in this model (adjusted OR = 1.442; 95% CI 0.237–8.763, P = 0.691).
Multivariate logistic regression models for evaluating diastolic dysfunction grade III with hsTnI
Results from the correlation analysis dictated that the log-transformed hsTnI multivariate logistic regression model be adjusted to gender, age, creatinine, NT-proBNP and LA-diameter (Table 5). Noteworthy, patients with hsTnI > 24.7 ng/L were not significantly associated with diastolic dysfunction grade III (adjusted OR = 1.949, 95% CI: 0.477–7.970, P = 0.353), whereas NT-proBNP as the reference biomarker revealed a fourfold higher risk for HFpEF grade III with NT-proBNP-concentrations > 74.57 pmol/L (adjusted OR = 4.083, 95% CI: 1.040–16.035, P = 0.044).
Discussion
The present study evaluates the association of high-sensitivity troponin I and T in patients with compensated HFpEF being assessed by transthoracic echocardiography. It was demonstrated that hsTnT revealed to be a reliable biomarker reflecting all stages of diastolic dysfunction, especially in discriminating the severe stage III. Noteworthy, hsTnI did not reveal consistent significant associations with the stages of diastolic dysfunction, whereas the reference biomarker NT-proBNP reflected diastolic dysfunction as comparable to hsTnT. Patients in this study cohort were all in compensated stages of diastolic dysfunction, and the results were proven both in univariate as well as multivariable statistical models.
While many studies have analysed the role of biomarkers in the diagnosis of heart failure, most research has focused on its role in HFrEF. The potential use of biomarkers other than BNP and NT-proBNP, which could define the prognosis in HFpEF has been poorly researched. Perhaps, the complex underlying pathophysiology contributing to the development of HFpEF has compounded to this problem. Early research work attempting to explain the HFpEF syndrome suggested a systemic component, rather than the influence of local myocardial pathologies as in the case of HFrEF. 1 An examination of specific target points commonly associates the origins of HFpEF with inflammation, fibrosis and myocardial stress. 20
The results of the present study have been highlighted. Firstly, hsTnT was able to discriminate patients with severe diastolic dysfunction. This relevance could be increased by combining the measurements with NT-proBNP, while the pure NT-proBNP measurements remained the most significant. To explore the implications of this positive relation between diastolic dysfunction and hsTnT, the different attributes of the protein must be considered. TnT as a subunit of the troponin complex is an integral protein in the contractile apparatus in striated muscle cells. After forming a union with calcium (Ca), the complex binds to tropomyosin and thereby releases it from the myosin head, resulting in muscle contraction. 21 Injuries and loss of permeability in the cell membrane cause increased serum concentrations of TnT. 22 Many studies have shown that TnT is an important marker in diagnosing an acute coronary syndrome (ACS) and chronic systolic heart failure.10,11,23,24 However, the identified significant correlation found in this study has led to examine its role in and relation to the origin and development of diastolic dysfunction.
One of the most common features emphasized in HFpEF patients is arterial hypertension. The increased arterial blood pressure could facilitate the release of TnT, an indicator of cell decay, in the event of cardiomyocyte damage in this syndrome. 25 The causal chain is set in motion by the resulting LV hypertrophy leading to diastolic wall stress and thereby a compromised coronary microvascular function. This in turn causes vascular reorganization and a decreased tolerance for ischaemia. The consequent inadequate supply of oxygen effects cardiomyocyte damage and scarring, which again increases diastolic wall stress creating a vicious cycle. 25
This pathological chain of events finds support in our study, suggesting significant correlation between echocardiographic indicators of LV hypertrophy and TnT measurements in patients with severe diastolic dysfunction. While studies highlighting the correlation of hsTnT to HFrEF and HFpEF found a stronger association of hsTnT to patients with systolic heart failure,11,26 this study provides evidence of a significant relationship between hsTnT and diastolic dysfunction grade III. It suggests some myocardial damage in the severe stages of diastolic dysfunction, which can be explained through the vicious cycle outlined above. However, due to the lower level of correlation, this does not affect the previous assumptions that HFpEF constitutes a systemic or metabolic illness rather than a myocardial-specific condition such as HFrEF.
While previous studies revealed a significant role of TnI in cardiac relaxation, this study does not find a significant increase of hsTnI serum concentrations in patients with diastolic dysfunction grade III. 27 Furthermore, measurement of hsTnI in addition to the known biomarker NT-proBNP does not produce an increased significance to the diagnostic and prognostic value. To contextualize this finding and draw conclusions towards the conditions related to the origins of HFpEF, the characteristics of the protein itself need to be outlined. TnI is the inhibitory subunit of the contractile troponin complex in striated muscles. During the systolic phase, when the intracellular (IC) Ca concentration rises, TnI binds to the regulatory troponin C (TnC) unit. TnI inhibition gets lifted and the muscle contraction is activated. Consequently, IC-Ca concentrations and proteins influencing IC-Ca-balance regulate the cardiac contraction and relaxation cycle. Several studies have demonstrated that TnI has a discrete status in the dynamics of cardiac contraction–relaxation as it desensitizes the contractile apparatus to activation by Ca.21,27 As a result, a sufficient diastolic function is only to be achieved by adequate Ca and TnI concentration. 27
A previous study has referred to decreased TnI concentration in the hearts of elderly patients, 27 essentially supporting the lack of significance shown in the present cohort. A different study argued that TnI mutations leading to loss of function is associated with the genesis of diastolic dysfunction.23,27 These findings, along with other studies that confirm a higher incidence of HFpEF in elderly patients, suggest a parallel hypothesis. The decreased concentrations of TnI and the higher incidence of HFpEF in the elderly could indicate a possible relationship existing between the pathophysiology of this syndrome and the reduced TnI concentrations.
General research on TnI supports a case against cell decay as a major factor causing HFpEF, as in this scenario, there would have been some measurable increase in the TnI concentrations. However, considering the occurrence of increased TnT concentrations, the possibility of the generally decreased TnI concentration in the elderly cohort needs to be considered masking the levels of cell decay and thereby distorting an evaluation.
Study limitations
The validity of this study is restricted by the small size of the collective. Furthermore, patients screened for this study usually presented themselves with heart failure symptoms in various stages of HFpEF and underwent hospital treatment. Even though all echocardiographic examinations were executed according to the ESC guidelines, some level of discrepancy must be taken into account. Accordingly, the level of the AUCs was rather moderate, and an overinterpretation needs to be avoided. No serial biomarker measurements were performed in this study. The present results need to be re-evaluated by larger prospective echocardiography studies, additionally to prove the potential for a causal effect in between hsTnT and HFpEF.
Conclusions
HsTnT as a biomarker in the diagnosis of HFpEF reveals significant diagnostic value as a biomarker to classify patients with profound HFpEF, whereas hsTnI serum concentrations did not add information for the stages of HFpEF.
Supplemental Material
Supplemental material for The association of high-sensitivity cardiac troponin I and T with echocardiographic stages of heart failure with preserved ejection fraction
Supplemental Material for The association of high-sensitivity cardiac troponin I and T with echocardiographic stages of heart failure with preserved ejection fraction by Julia Hoffmann, Michael Behnes, Uzair Ansari, Kathrin Weidner, Philip Kuche, Jonas Rusnak, Seung-Hyun Kim, Michele Natale, Nadine Reckord, Siegfried Lang, Ursula Hoffmann, Thomas Bertsch, Marc Fatar, Martin Borggrefe and Ibrahim Akin in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Thomas Bertsch performs reagent evaluation studies for Roche diagnostics. All other authors declare that they do not have any further conflict of interest.
Funding
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Medical Ethics Committee II of the Ruprecht-Karls-University Heidelberg, Medical Faculty of Mannheim, University Hospital Mannheim (Study Id 2014-615N-MA. NCT Number NCT03074253).
Guarantor
IA, MBe.
Contributorship
JH, MBe and UA conceived the study, participated in its design and coordination, participated in data analysis and interpretation and helped to draft and revise the article for important intellectual content.
KW, PK, JR, MN, NR and S-HK participated in the study design and coordination, data acquisition and helped to draft the article for important intellectual content.
SL, UH and MF participated in the study design and coordination, as well as data analysis and revised the article.
TB and MBo conceived the study, participated in its design and coordination, participated in data analysis and interpretation and helped to draft and revise the article for important intellectual content.
IA conceived the study, participated in its design and coordination, participated in data analysis and interpretation and helped to draft and revise the article for important intellectual content.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
