Abstract
Purpose
Following the analytical phase, the current practice of many hospital laboratories involves the manual verification of all test results followed by the production of the report. However, manual verification is a time-consuming and tedious process. In this paper, we provide a detailed description of how to design autoverification rules for thyroid function test profiles and sex hormones.
Materials and methods
We used DM2 (Data manager 2) to construct the algorithm and build the database for autoverification of thyroid function test profiles and sex hormones, with reference to Boolean logic, Auto 10-A and CLSI’88. The rules consist of checking quality control, instrument error flags, critical values, the analytical measurement range (AMR), the limit range, consistency check and delta check. Firstly, we established the rules in the DM2, collected clinical specimens for validation, then tested the rules in a ‘live’ environment.
Results
Agreement was achieved between manual verification by two senior laboratory personnel and verification using the autoverification rules in 99.78% of the cases. The total autoverification rate for all tests was 77.06%. Following implementation of the rules, the laboratory turnaround time (TAT) was reduced by 54.55% and staffing numbers fell from three to two whole time equivalents (WTE). Statistical analysis resulted in a kappa statistic of 0.99 (
Conclusion
Implementing autoverification rules can reduce TAT, minimize the number of samples that require manual verification and allow for a reduction in staffing numbers. It also allows laboratory staff to devote more time and effort to the handling of problematic test results and contributing to improved patient care.
Introduction
Currently, clinical laboratories are under continual pressure to increase their productivity, through the handling of larger workloads with fewer qualified staff. Following the parallel developments of increased laboratory automation and major developments in information technology (IT), many clinical laboratories in hospitals now utilize two-way communications between laboratory information system (LIS) and analytical instruments.1,2 However, despite this, the speeds by which some test results are released remain compromised by the low efficiency of manual verification, which is performed by one or more senior member of staff, on a single analytical result or on a group of results, in order to ensure that no incorrect results are reported to the hospital information system (HIS) or to clinicians. Autoverification, a process by which clinical laboratory results are released without manual intervention or review,3–9 can overcome this limitation.3,10,11 The Autoverification of Clinical Laboratory Test Result Approved Guideline (AUTO 10-A) was issued by the American Clinical Laboratory Standards Institute (CLSI) in 2006. 9 This guideline provides a basic framework to allow each clinical laboratory to design, implement and validate specific autoverification rules for specific tests according to a laboratory technologist’s demands. The use of such autoverification can enhance the efficiency of the laboratory, reduce budgetary costs and decrease turnaround time (TAT), as well as ensure quality 12 and enable laboratory staff to focus more on potentially problematic test results. 4 Currently, many laboratories worldwide are exploring and implementing autoverification in several areas, including urine analysis,13,14 haematology,15,16 clinical biochemistry,17–20 coagulation,21,22 and clinical immunology. 7 Although there is nearly a 20-year history of autoverification systems that can verify and validate clinical laboratory results and the Auto10-A guidelines have been around for nearly 10 years, there remains a lack of standardization, especially for the algorithms and verification limits, 18 and it remains unclear how to build autoverification rules and parameters.12,22
Although commercial autoverification systems, such as VALAB, are available, their processing algorithms and verifying rules are considered proprietary; therefore, they cannot be modified by the users.10,23 Moreover, commercial software addresses only the most basic levels of autoverification, e.g. the reference interval, instrument alarm and internal quality control (IQC), and none of the commercially available programs can handle complex clinical data, such as testing logical relationships, clinical presentation and clinical history. As a result, clinical laboratories have built their own autoverification systems, such as DNSev™ 24 and LabRespond. 10 This can also be achieved by developing a within-laboratory autoverification system that would work as a part of the LIS or the middleware, which in our laboratory consists of Data Management 2 (DM2), and which was developed by Beckman Coulter.
Thyroid function and sex hormone tests are two examples of common clinically complex tests that are processed in the laboratory. Familiarity with the physiology and pathophysiology of the thyroid gland and the gonads is important to ensure appropriate requesting of the hormones they produce. A number of medications and diseases have been shown to influence the circulating concentrations of these hormones.25,26 Circulating concentrations of sex hormones can also vary with the menstrual cycle and in patients undergoing treatment through assisted reproductive technology (ART). In those women receiving ART, circulating sex hormone concentrations differ from those found in normal or pregnant women,27,28 presenting a challenge for the setting of any autoverification rules.26,29–32 In this study, we present a detailed description of how to construct autoverification rules for thyroid function and sex hormone tests using the DM2 middleware, and describe their subsequent validation and evaluation in the clinic.
Materials and methods
Instrumentation
All equipment was provided by Beckman Coulter Inc., CA, USA (Power Processor Automated Sample Processing System, UniCel DxI 800 Immunoassay System, Prelink, Centrifuge, Specimen Stockyard, Outlet Rack System).
Methods
All tests were measured using chemiluminescent microparticle immunoassay (CMIA), on a Unicel DxI 800 immunoassay analyser (Beckman Coulter Inc., CA, USA). Thyroid function tests consisted of thyroid-stimulating hormone (TSH), total triiodothyronine (TT3), total thyroxine (TT4), free triiodothyronine (FT3) and free thyroxine (FT4).
Sex hormone tests consisted of: β-human chorionic gonadotropin (β-HCG), progesterone, testosterone, oestradiol, prolactin, follicle-stimulating hormone (FSH).
In addition, all internal quality control serum products were provided by Beckman Coulter Inc., CA, USA.
Software
The DM2 was obtained from Beckman Coulter Inc., CA, USA. The hospital information system (HIS) and Laboratory Information System (LIS) were both obtained from B-Soft Co., Ltd, Hangzhou, China.
Statistical analysis
Analysis of the data was performed using the SPSS statistics software package, version 20.0 for Microsoft Windows (SPSS Inc., Chicago, IL, USA). A
A kappa test was used to evaluate the difference between autoverification and manual verification. The kappa coefficient represents the observed agreement above and beyond that due to chance. The strengths of any observed agreements were as follows: <0.20 bad; 0.20–0.40 common; 0.41–0.60 moderate; 0.61–0.80 strong and 0.81–1.00 very strong.
Methods
This study was conducted in the Clinical Chemistry Core Laboratory of the First Affiliated Hospital of Shantou University Medical College in cooperation with a medical expert from the Department of Reproductive Medicine, the DM2 vendor (Beckman Coulter, CA, USA) and the LIS vendor (B-Soft Co., Ltd, Hangzhou, China). Our hospital is a regional general academic tertiary care referral centre and university affiliated hospital with 67 clinical departments and 1806 beds. In this study, the DM2 was integral to the autoverification rules. We designed them by using Boolean logic according to the CLSI Auto10-A guidelines.
9
We then simulated and validated their performance on the DM2 using historical patient data stored in the database. The details of the rules and the flowchart are shown in Figure 1. The flowchart shows that the rule processes consist of IQC, instrument error flags, critical value, instrument analytical range, limit range, consistency check and delta check.
Flowchart of autoverification rules for thyroid function profiles and sex hormone tests.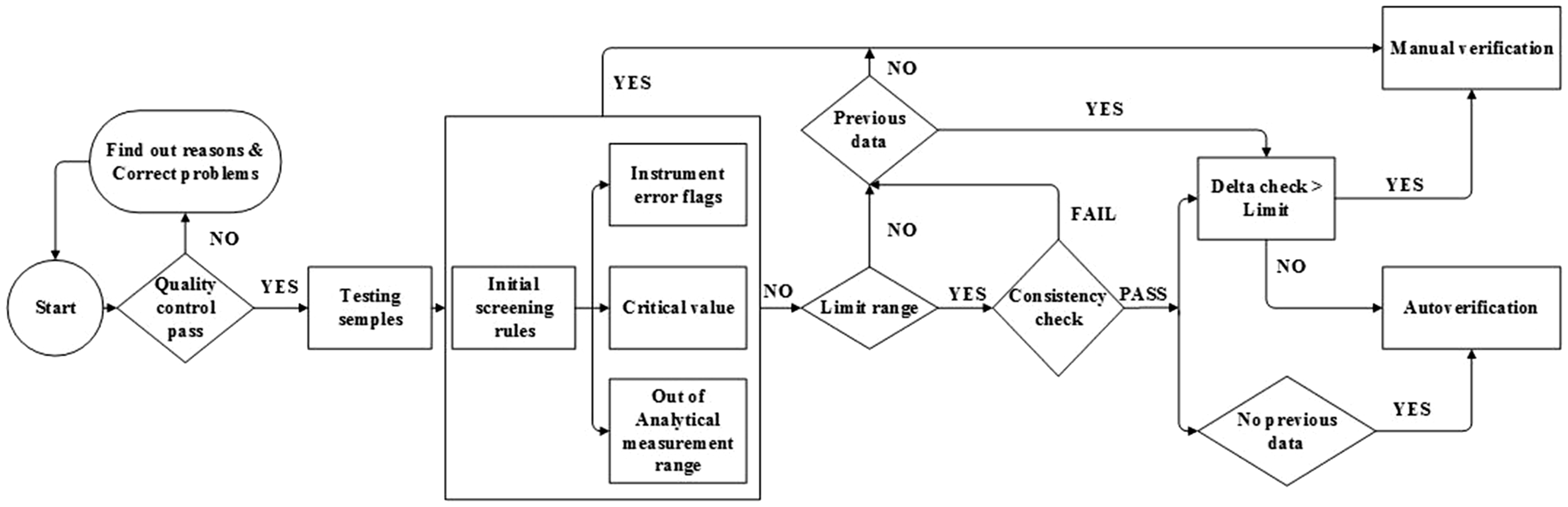
Quality assurance
Our laboratory routinely uses IQC and takes part in external quality assessment. Daily IQC results were transmitted from the analyser to the DM2 and LIS and were evaluated using Levey-Jennings charts 33 and Westgard quality control multirules.34,35 In addition, we also used the ‘moving average’ method as an additional quality assurance (QA) method to help ensure the quality of the results. This involves the collection of patient results over a period of 20 consecutive days. From these results, a mean result is determined, and a warning limit is calculated, which is the mean ±2SD, together with an action limit of the mean ± 3SD.36 For example, in the case of FT4, over one period of 20 consecutive days, the warning limits (mean ± 2SD) were 10.72 and 13.96 pmol/L.
Instrument error flags
The instrument will give alerts when there are problems with the reagents, barcode, samples or mechanical failure, e.g. in the event of reagent crystallization or sample clots forming.
Critical value
The critical values or medical decision levels were determined locally and were based upon those described by Statland et al. 37 The critical values used in our hospital were as follows: β-HCG > 180,000.00 IU/L, oestradiol > 9177.50 pmol/L, LH > 40.00 IU/L, FSH >70.00 IU/L, testosterone < 1.04 nmol/L, prolactin >2120.00 mIU/L, FT3 > 40.00 pmol/L, FT3 < 1.50 pmol/L, FT4 > 22.00 pmol/L, FT4 < 3.50 pmol/L, TT3 >10.00 nmol/L, TT3 < 0.50 nmol/L, TT4 > 380.00 nmol/L, TT4 < 6.50 nmol/L, TSH > 80.00 mIU/L, and TSH < 0.02 mIU/L. Results that were outside the range of the critical value required verification by a technologist, and those within the range passed and continued to the limit check and delta check.
Instrument analytical range
The analytical measurement ranges were: β-HCG, 0.50–1000.00 IU/L (1000.00–200,000.00 IU/L following dilution); progesterone, 0.32–127.20 nmol/L; testosterone, 0.35–55.50 nmol/L; oestradiol, 73.00–17,621.00 pmol/L; prolactin, 5.30–4240.00 mIU/L; FSH, 0.20–200.00 IU/L; LH, 0.20–250.00 IU/L; TSH, 0.01–100.00 mIU/L; T4, 6.40–386.00 nmol/L; FT4, 3.20–77.20 pmol/L; T3, 0.20–12.30 nmol/L; FT3, 1.40–46.00 pmol/L. Results out with the analytical range generated a warning flag and required sample dilution prior to reanalysis.
Limit range
Limit ranges of all the 12 tests.
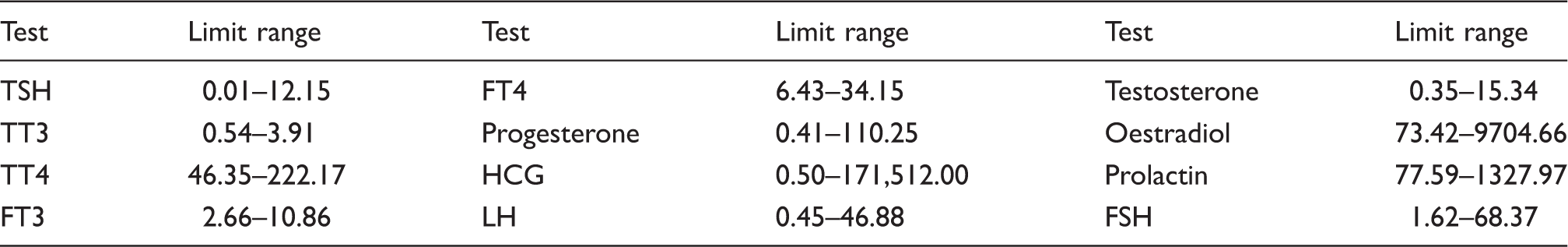
FT3: free triiodothyronine; TT3: total triiodothyronine; TT4: total thyroxine; TSH: thyroid-stimulating hormone; FSH: follicle-stimulating hormone; LH: luteinizing hormone; FT4: free thyroxine; HCG: human chorionic gonadotropin.
Consistency check
Given the nature of acute illness and the fact that clinical test results fluctuate frequently, it was very difficult to perform a consistency rule check. Only some tests had a consistency check established based on clinical and practical diagnostic criteria. The hypothalamic pituitary-gonadal (HPG) and hypothalamic-pituitary thyroid (HPT) axes are negative feedback control systems. Therefore, there are some necessary relationships between the circulating concentrations of the hormones, e.g. elevated T4 or T3 concentrations associated with a decreased TSH concentration, in patients with toxic diffuse goitre. It was necessary that any consistency rule took account of the interrelationships of all of the hormones, and consequently, if any results violated the consistency check, test results were unable to be sent to the HIS. For example, if the test result for TSH was lower than 0.34 mIU/L, with a FT3 < 6.00 pmol/L or a TT3 < 2.73 pmol/L and FT4 < 14.40 pmol/L or TT4 < 157.40 pmol/L; or TSH > 0.34 mIU/L and FT3 > 6.00 pmol/L or TT3 > 2.73 pmol/L, the report was intercepted as a manual verification (MV) report and could not be sent to the HIS.
Delta check
The delta check compares current test results with previous results to identify those results that differ by more than a defined amount.39,40 Using the delta check rule enables the identification of changes in the test results beyond their expected variation, e.g. due to changes in a patient’s clinical status or due to preanalytical errors.41,42 We therefore believe that all test results that have previous test values must be verified by a delta check. 40 The parameters that have previously been used include the delta difference, rate difference, rate percent change, cumulative incremental weighted index43–45 and delta percent change. 46 In this study, we chose the rate percent changes, which were calculated as the current value subtracted from the previous value and then divided by the previous value. We chose a time interval as less than seven days based on discussions of the biological and pathological functions of the thyroid gland and gonads with physicians in the Department of Reproductive Medicine. In addition, we suggest that if the rate of change of the concentrations of the hormones was within the set range in the previous seven days, the result could pass the delta check. The set ranges were: ±50%/7 days for TSH, TT3, FT3, TT4, progesterone, LH, testosterone, prolactin and FSH; ±70%/7 days for FT4 and oestradiol and ±80%/7 days for HCG.
Patient diagnostic information
Patient information associated with tests report results.
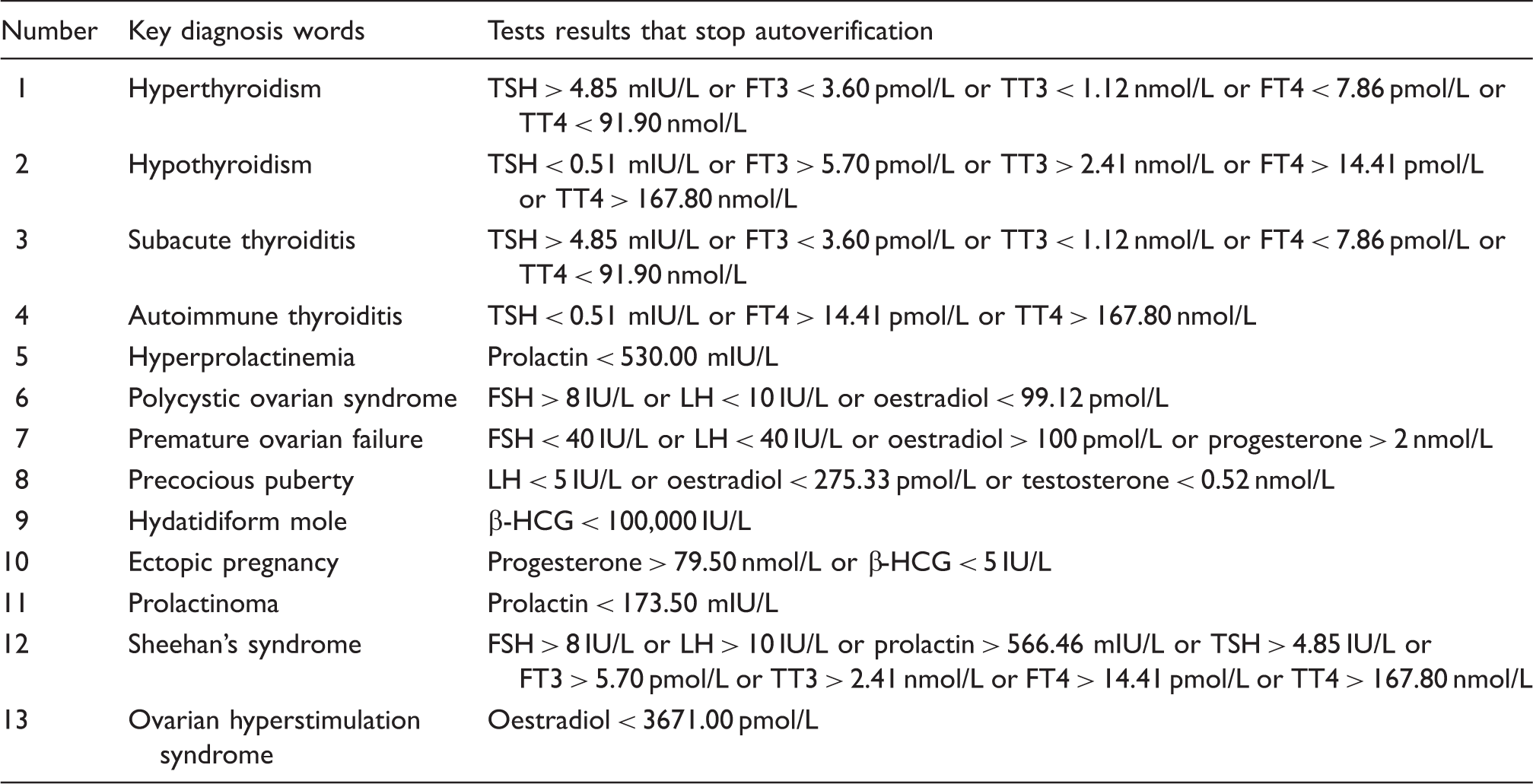
FSH: follicle-stimulating hormone; LH: luteinizing hormone; TSH: thyroid-stimulating hormone; FT3: free triiodothyronine; TT3: total triiodothyronine; FT4: free thyroxine; TT4: β-HCG: β-human chorionic gonadotropin.
Autoverification rules and actions.

AMR: analytical measurement range.
Validation methods
To validate whether the autoverification rules and their settings in the DM2 were able to meet our requirements and be implemented, we used electronically simulated cases, special samples and historical data, and the entire validation process was based on the recommendations in the CLSI Auto10-A. 9
Electronically simulated case validation
Electronically simulated cases were used to verify that the programmed autoverification rules followed the expected logic and achieved the anticipated outcome. One case for each rule was previously programmed. The total number of cases was 1063. A total of 538 (50.6%) cases passed autoverification and 525 (49.4%) failed. The electronically simulated cases that passed the autoverification rules were reviewed in order to ensure that they matched the expected outcome.
Special sample validation
Special samples included the abnormal proficiency test samples and more than 50 patient samples, containing low and high concentrations of each analyte. Most of the test results fell outside the acceptable range of the critical value, instrument analysis and limit check. These tests results were chosen to verify the efficiency of the autoverification rules and the reliability of the reports. The validation results demonstrated that there were no errors in the autoverification rule use, which indicated that the reports generated using the rules were accurate.
Validation using historical data
The DM2 went live in the Clinical Chemistry Core Laboratory in 2013. All clinical chemistry test results of patients, from October 2013 to the present, were stored in the DM2 computer, which allowed us to validate the rules by using this historical data. The total number of cases, from October 2013 to December 2014, with thyroid and gonad hormones analysis in the DM2 were 47,448 and 23,983, respectively. All cases had been manually verified and revised by skilled laboratory specialists in the LIS. These cases were used to represent the same distribution and type of cases that are received by the laboratory, and they provided an estimate of the quality of autoverification. This validation process which uses many test results should uncover problems with both the autoverification rules and electronic equipment that occurred infrequently prior to autoverification. We had to ensure that those test results reported by autoverification were consistent with the clinical status of the patients. The use of historical data indicated that the rate of autoverification was 57.56%. In addition, the system and rules ran well and did not demonstrate any error flags.
Results
According to the CLSI Auto10-A Guideline, 9 autoverification rules must be validated by using actual patient results before go-live. To confirm whether the autoverification rules were reliable, we established the rules in the DM2, and collected and assessed patient test results, of which there were 77400, produced during the calendar year 2015. The validation was performed by requesting that two senior technologists, who specialized in thyroid and gonadal disease and who worked in cooperation in the Clinical Chemistry Core Laboratory, verify the results in triplicate.
Passing rate of whole items.
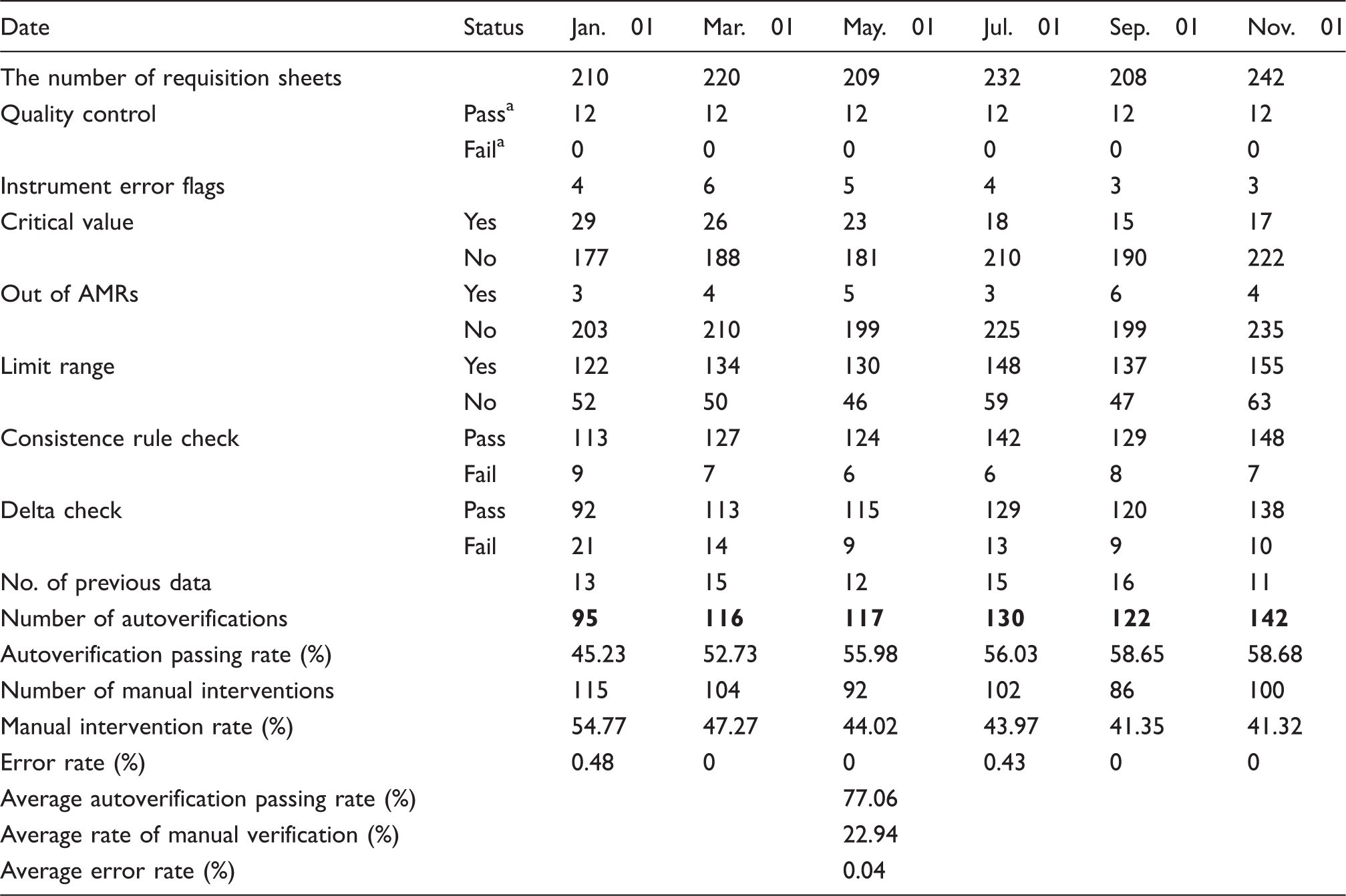
AMRs: analytical measurement ranges.
QC results for thyroid function profiles and sex hormone tests. Bold numerals represent the number of results that passed the autoverification rules.
The autoverification rate for all thyroid function profiles and sex hormone tests ranged from 65.35% to 77.06% (Table 4 ). The autoverification rate for thyroid function profiles and sex hormone tests implemented in the Clinical Chemistry Core Laboratory increased over the period of 12 months from 65.57% to the current overall rate of 86.78% and from 52.78% to 63.70%, respectively.
A high percentage of results with critical values were autoverified. The average rates of results exceeding the critical value, limit range and delta check were 7.21%, 25.20% and 10.97%, respectively.
The reported laboratory TAT was reduced by 54.55% (from 66 to 30 min), and the time interval from completion of analysis to verification was reduced by 61.80%. The staffing numbers were reduced from three to two whole time equivalents (WTE).
Evaluation of the benefits
Degree of agreement between the autoverification rules and two senior technicians.

Degree of agreement: <0.2 poor; 0.2–0.4 fair; 0.41–0.6 moderate; 0.61–0.8 good; 0.81–1.0 very good. bHighly significant difference, indicating the agreement between the autoverification rules and two senior technicians was not caused by accident.
Discussion
Autoverification is a process of using computer-based rules to undertake the initial validation of test results without manual intervention. Data that fall outside the set rules should be reviewed by the laboratory technologists and those that pass the set rules can be directly released by the computer.6,7,9,20,48 Although the autoverification of clinical tests results is an essential tool with which to increase the accuracy and efficiency in clinical laboratories,6,48 there is relatively scant information on its practical applications within a clinical laboratory setting.18,20 In this paper, we described the establishment and application of autoverification in a busy clinical chemistry core laboratory.
Thyroid function and sex hormone tests were chosen for the initial evaluation of the rules because they are tests that have a complex, but defined relationship with other system functions. Furthermore, they have clearly defined reference intervals and well-defined cut-off values for certain clinical abnormalities, and they represent the main bulk (85%) of hormone cases in our laboratory.
As shown in Figure 1, the autoverification rules utilize the following parameters: (1) IQC check; (2) initial screening rules check (including instrument error flags, critical value and out of analytical measurement range); (3) limit range; (4) consistency check; (5) delta check. The IQC check confirms whether each item falls within the acceptable range before the autoverification rules start; otherwise, the verification procedure will be halted according to the preconditions for autoverification. 9 The IQC system should be integrated with the autoverification rules, e.g. when IQC failure occurs, the autoverification rules of the given test items will be automatically stopped to enable a qualified person to analyse the reasons for the failure and correct the problems. Then, once IQC is within limits, the autoverification rules would be restarted.
The critical value is important for enabling physicians to make a diagnosis. To the best of our knowledge, it is unusual to find published data on the critical values for autoverification. Our process allows the autoverification of critical value to proceed as long as no other rules are violated. In addition, critical values still need to be communicated to the clinical locations from which the requests originated. However, the communication of these results by the laboratory is facilitated by clinicians often having access to results, e.g. following their display on ward-based terminals, that have undergone autoverification, prior to any telephone call being made.
Because test results fluctuate substantially during acute illness, it was difficult to perform a consistency check on each item, and only portions of the test items were confirmed based on practical and clinical diagnostic criteria. By using the consistency rule check, we would avoid most of the base errors. However, the delta check could help us quickly find changes in a patient’s condition and identify any preanalytical errors.
We chose the total agreement between the two professional reviewers as a benchmark to maximize patient safety. A similar method was applied in the VALAB system.
11
There was a significant degree of consistency between our autoverification rules and the manual verification undertaken by two senior technicians (kappa value = 0.99,
Most publications describe autoverification rules as an approach to shortening the TAT in reporting test results, through reduction in manual intervention, which in turn can facilitate staffing reductions in clinical laboratories.4,20 In our laboratory, the implementation of the rules resulted in the staffing WTE numbers in the Clinical Chemistry Core Laboratory falling from 8 to 6. Furthermore, because results that undergo autoverification are released immediately, the TAT of patient reports was dramatically reduced in comparison to those obtained previously with manual verification.
Our experience has suggested that the TAT is the parameter that is the most affected by the introduction of autoverification rules. However, this improvement was reduced when comparing the average TAT values for the year. Our reduction in the TAT was 54.55%. This fall in improvement was due to the simultaneous increase in the average daily workload for hormone analysis in the clinical chemistry core laboratory from 51,367 samples to 77,400 samples (a 150.68% increase). However, we believe that the TAT will further improve following the full utilization of all the established programmed rules. McFadden 49 indicated that with the application of autoverification rules, there could be up to a 44% savings in time and labour capacity for the laboratory staff. We found that the autoverification rate obtained using historical patient data was 77.06%; this is close to that of the VALAB system which has a mean autoverification success of approximately 50% to 90%. 11 However, VALAB was applied on a wider range of tests. Our success rates are lower than that of the DNSev™ system, which shows a verification rate of approximately 80%. 24 These differences could be explained by differences in the studied group of tests, patient presentations and laboratory equipment used. In our study, the autoverification rates of sex hormone single tests were lower. For example, the autoverification rates of oestradiol and LH were 76.65% and 72.97%, respectively. We initially wanted to include menstrual status and pregnancy in a limit range. However, due to time constraints, this was not possible and it is our aim to develop this area in the future when time permits.
Because autoverification rules are relatively new to laboratory test result reporting, the scientific literature on the subject is limited. In this study, we found that the greatest benefit following the implementation of the rules is in the consistency of the test results, as previously observed.4,7,20 In contrast, despite the advantages of autoverification, potential disadvantages could include an increase in false-negative results and the reporting of erroneous results due to analytical interference, such as when a partial or tiny clot is not detected by the instrument. Mechanical errors of this kind, although few, remain unavoidable. Our study also has some limitations. The limited connectivity between the LIS and HIS resulted in the disabling of any rules associated with any patient clinical information or drug history, which prevented us from taking full advantage of the programmed rules. Although we could make a connection with the keywords of the diagnosis, this work-around approach is insufficient. For the time being, we can only overcome this drawback by registering the previously established comprehensive patient sheets.
In conclusion, we developed and implemented a rule-based autoverification, according to Boolean logic and the guidelines of ‘Design of Algorithms’ in the Auto-10A document that utilizes clinical tests data and other parameters. The autoverification rate of the rules was comparable with that obtained using commercially available software for other systems. The rules that we designed can shorten the TAT, reduce manual data entry, and decrease the probability of errors associated with human review, as well as, minimize the number of samples requiring technologist intervention. However, there was a small sample size in the current study and the implementation time was short (only three months). In the future, we will continue implementing autoverification rules for thyroid function profiles and sex hormones, and add other tests to construct a more complete expert system.
Footnotes
Acknowledgements
We thank Yongxin Qiu, from Beckman Coulter Inc., for computer and DM2 technical support and Linli Fang and Yingqiu Zu, the senior technicians in the Clinical Chemistry Core Laboratory, for result verification. Additionally, we thank Dr. Lin, a professor of Shantou University Medical College (SUMC), for his review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
JL and YC.
Contributorship
All authors reviewed and edited the manuscript and approved the final version of the manuscript. Due care has been taken to ensure the integrity of the work.
