Abstract
Background
Biotin interference in streptavidin-based immunoassays causes widespread analytical distortions that may lead to clinical confusion, inappropriate patient management and, ultimately, adverse events. Its prevalence has increased recently due to the increased use of high-dose biotin therapy in specific patient groups (notably multiple sclerosis) and possibly the general community.
Methods
We have developed a method to deplete biotin from samples using the streptavidin-coated magnetic microparticles that are a component of most susceptible assays.
Results
We show that high concentrations of spiked biotin can be adequately depleted from serum using this approach, and that gross biochemical derangements can be restored to normality. We also show that biotin in samples derived from multiple sclerosis patients receiving 300 mg biotin daily can be adequately depleted to remove associated analytical interference and restore normal results. The method is applicable to competitive and sandwich immunoassays and importantly, because it does not change the volume of the sample, suitable for the measurement of free thyroid hormone assays. Application of the method does not significantly change the precision of measurement, and for the majority of analytes, the accuracy is not substantially altered.
Conclusions
Adopting this method enables laboratories to confirm biotin interference in the appropriate clinical setting. Moreover, it enables laboratories to remove the interference and report accurate and reliable results, without the need for patients to withhold beneficial therapy prior to blood tests. Until the biotin tolerance of susceptible assays is improved, our method gives laboratories a safe alternative for reporting results using streptavidin-based methods.
Introduction
Biotin interference in streptavidin-based immunoassays is an old problem1–3 attracting renewed attention. In the last year, numerous case reports have documented interference in patients for whom high-dose biotin is either an established or emerging therapy, namely in the setting of biotin-responsive inborn errors of metabolism and multiple sclerosis (MS), respectively.4–11 There is also growing concern that the increased availability of over-the-counter ‘mega’-biotin supplements may have introduced the risk of interference into the general population,9,12,13 although the prevalence of this is unknown.
The striking increase in the frequency of reports of biotin interference reflects a genuine increase in prevalence, due to change in clinical practice and patient behaviour, rather than a simple increase in awareness. For laboratories employing assays based on streptavidin-coated microparticles and biotinylated antibodies, this newly expanded threat of biotin interference is significant, and poses high potential for clinical harm. Contemporary challenges for affected laboratories include timely identification of biotin interference in order to prevent the reporting and consequences of erroneous results. This is likely to be multifaceted; however, one aspect is clear: ideally, laboratories need to be alerted to patients receiving high-dose biotin therapy so that appropriate responses can be mounted. These might include using an alternative, non-biotin-affected platform if the option is available within the laboratory, or sending out samples to independent laboratories if safe internal testing is not possible. Withholding biotin for several days prior to testing is another approach that may be used in the non-urgent setting.8,9 In the acute setting, however, these latter approaches are problematic.
There is a tangible need for pragmatic, definitive solutions to the problem of biotin interference. With this in mind, we sought to develop a simple, accessible and reliable protocol that laboratories could use to identify biotin interference and, if possible, eliminate it. We envisaged a method that could be applied to selected samples to confirm biotin interference, rather like scantibodies and polyethylene glycol precipitation are used to investigate the presence of interfering antibodies and macro-complexes. 14 Moreover, our aim was to develop a protocol that could adequately remove the biotin interference and facilitate reportable results on biotin-depleted samples. We have used the problem itself (streptavidin-coated magnetic microparticles) as the basis of the solution.
Materials and methods
Biotin spiking
Biotin was purchased from Astral Scientific (Cat. No. BIODB1001, Lot No. VH20150310CAS; purity 99.5%). Biotin stock solution at 1 ug/uL in 0.01 M NaOH was stored at 4℃ for one month, or frozen at −20℃ for longer term storage. Stock was diluted into working solutions (in distilled water) that were spiked into serum samples to achieve the indicated final concentration of biotin (up to 1000 ng/mL). The volume of spike was minimized to <5% of the final volume in all experiments, and control samples were spiked with the same volume of distilled water to account for matrix effects. Biotin concentrations were chosen based on the available pharmacokinetic data. 15 The validity of these concentrations was supported by reproducing the interference we had observed in our laboratory, in samples from patients with MS receiving high-dose biotin therapy.
Samples
De-identified routine serum samples that would otherwise have been discarded were used for our spiking and depletion experiments. These were either used as single or pooled serum samples. We also used serum samples from patients with MS known to be on 300 mg biotin daily. These patients presented over the last nine months for routine testing at our laboratory. Samples were either used at the time or frozen at −20℃ for later use. The Royal Australasian College of Pathologists Guideline on the use of human tissue and test results in Australia outlines Pathologists’ rights to carry out procedures deemed necessary for diagnostic purposes and quality assurance, among other things. 16 Our work adheres to this guidance.
Assays
The following Elecsys immunoassays (catalogue number in brackets) were run on the Roche Cobas 8000 e602:
For thyroid function tests (TFT), we also analysed samples on the Abbott Architect i2000 using the following immunoassays: TSH (7K62-35), free T3 (7K63-35) and free T4 (7K65-39).
Streptavidin-coated microparticle preparation
All Roche Elecsys immunoassay kits contain a streptavidin-coated magnetic microparticle suspension at a concentration of 0.72 mg/mL. In our initial experiments, we used microparticles taken from fresh (unused) reagent packs; however, we found that microparticles could also be salvaged from used reagent packs. The microparticles are present in excess and several mL can be recovered from each used reagent pack when defined by the analyser as ‘empty’. These were pooled, then aliquoted into V-bottomed tubes and stored at 4℃ for later use. The data presented herein were derived using salvaged microparticles from used reagent packs.
Optimization of the method
Our initial experiments were designed to estimate biotin-binding capacity (not stated by the manufacturer) by titration against known biotin concentrations spiked into samples. To our knowledge, there is no readily available method for biotin measurement in Australia; thus, we had to optimize our method by empirical means, without opportunity to measure the biotin concentration of spiked and depleted samples. We conducted dose–response curves to work out the microparticle volume required to deplete a given concentration of biotin from a given volume of sample and restore measured analyte concentrations to those of control (water-spiked) samples. After preliminary studies, we chose 250 µL as our standard sample volume, balancing sample volume availability (possibly limiting) with the requirement for sufficient volume to cover dead volume and several tests. Microparticles were aspirated to a fluid-free, dry weight using a 96-well V-bottomed plate magnet (Alpaqua, Cat. No. 29020) and V-bottomed tubes (Globe Scientific, Cat. No. 5106), the base of which inserted neatly into the magnet. The microparticles naturally sediment upon storage and were not resuspended prior to use, to expedite magnetic separation. The tube was placed in the magnetic device, the microparticles were immobilized to the magnet and the excess fluid was aspirated by manual pipetting. Sample (250 µL) was added to the dry microparticles and incubated for various durations at room temperature, with regular mixing to maximize the interaction between the microparticles and the sample. After incubation, the microparticles were immobilized at the base of the tube using magnetic separation (as above). Optimal magnetic separation time needs to be determined by each laboratory, and is dependent on the strength of the magnet, its surface area, the type of tube used and the volume that the microparticles are suspended in. For our set-up, a 1-min exposure to the magnet was required to adequately immobilize microparticles suspended in 250 µL of sample. After immobilization, the biotin-depleted supernatants were aspirated and analysed for the relevant analyte. Once optimized, the protocol was applied to samples from MS patients on high-dose biotin therapy to ensure that ingested biotin and relevant biotin metabolites could also be adequately depleted. This analysis involved comparison of TFT results measured on the Elecsys and Architect platforms, in samples before and after depletion.
Statistical analysis
The paired Student’s t-test was applied to determine statistical significance. Analyte concentration of biotin-spiked samples that had been subsequently depleted of biotin were compared with control, water-spiked samples. Method precision was evaluated from analysis of routine QC material at each of three levels for each analyte studied in detail. Measurement uncertainty was estimated for the period of study as 2× coefficient of variation (CV; CV = standard deviation (SD)/mean). Expanded uncertainty was calculated as the interval ([mean−2 × SD]−[mean + 2 × SD]). For method correlation between Roche and Architect TSH, free T4 and free T3, linear regression was applied to a series of routine samples from patients who had no known history of thyroid disease, and were anti-TPO and anti-Tg antibody negative.
Results
Optimization of the depletion protocol
Titration of microparticle volume against a fixed amount of spiked biotin.
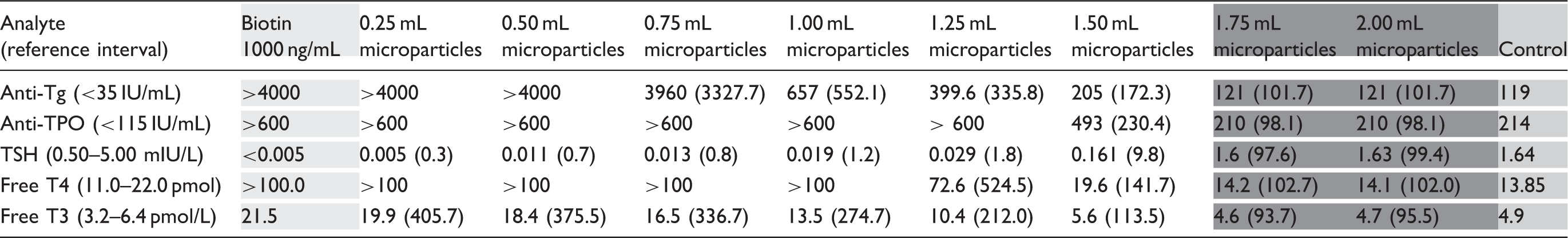
Note: Serum aliquots were spiked with biotin at 1000 ng/mL (pale grey column) before biotin depletion using the indicated volumes of streptavidin-coated microparticles. A volume of 1.75 mL and above was required to remove 1000 ng/mL biotin (a total biotin mass of 250 ng) from 250 µL of sample volume (dark grey columns) and restore results to levels similar to controls (medium grey column). Where appropriate, measured analyte concentration is expressed as a percentage of control, indicated in brackets.
Adequacy of depletion
From our investigation of the varying biotin susceptibility of different assays,
17
we identified anti-TPO and anti-Tg as assays that were highly sensitive to very low concentrations of biotin. We thus reasoned that a depletion protocol that could restore accurate measurement of these analytes would be readily transferable to other analytes and allow removal of biotin to below the tolerance of essentially all assays. As shown in Figure 1, using our biotin-depletion protocol we were able to restore spiked samples (resulting in apparent anti-Tg and anti-TPO antibody concentrations of > 600 and > 4000 IU/mL, respectively) to the very low level of controls.
Adequacy of depletion.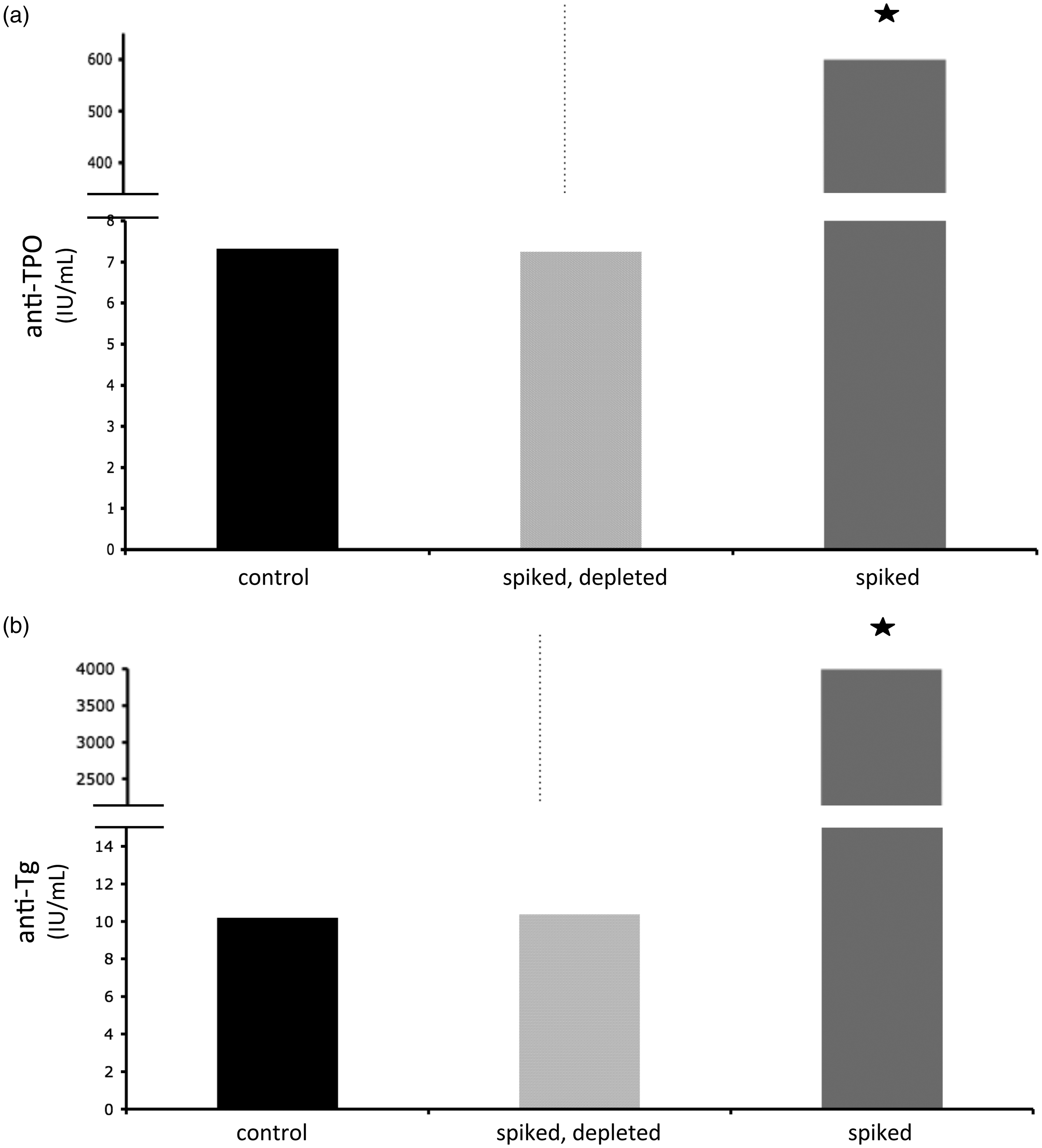
Accuracy and precision
To report results from biotin-depleted samples, the depletion protocol should introduce minimal change in accuracy and precision, and ideally any variation should be within laboratories’ stated measurement uncertainty. The results of TFTs are given in Figure 2. Biotin was spiked into a biochemically euthyroid sample at a concentration of 1000 ng/mL, producing characteristic thyrotoxic-appearing TFT. After biotin-depletion, the results were restored to within 2% of control values for TSH, free T4 and free T3. There was no statistically significant difference between the measured values of the depleted sample versus the control, indicating that the depletion protocol did not significantly change the accuracy of measurement. Control (non-biotin containing) samples were also run through the depletion protocol without substantially changing the measured analyte concentration. For TSH and free T3, there was no significant difference in results. For free T4, while the difference between unspiked-depleted samples and controls appeared to be statistically significant (P = 0.029), the measured ‘difference’ was minor, at approximately 1%. We conclude that, at least for the analytes studied in detail, the depletion procedure does not cause substantial change in analyte measurement. Precision was similarly unaffected by the depletion protocol. As shown in Figure 2, the results for replicate samples that were spiked then depleted of biotin showed similar precision to replicate measurement of the original sample. Our calculated measurement uncertainty for the assays and instruments used for the period of study is included in Supplementary Table 2. For each analyte studied in detail, results were well within our laboratory’s measurement uncertainty.
Accuracy and precision.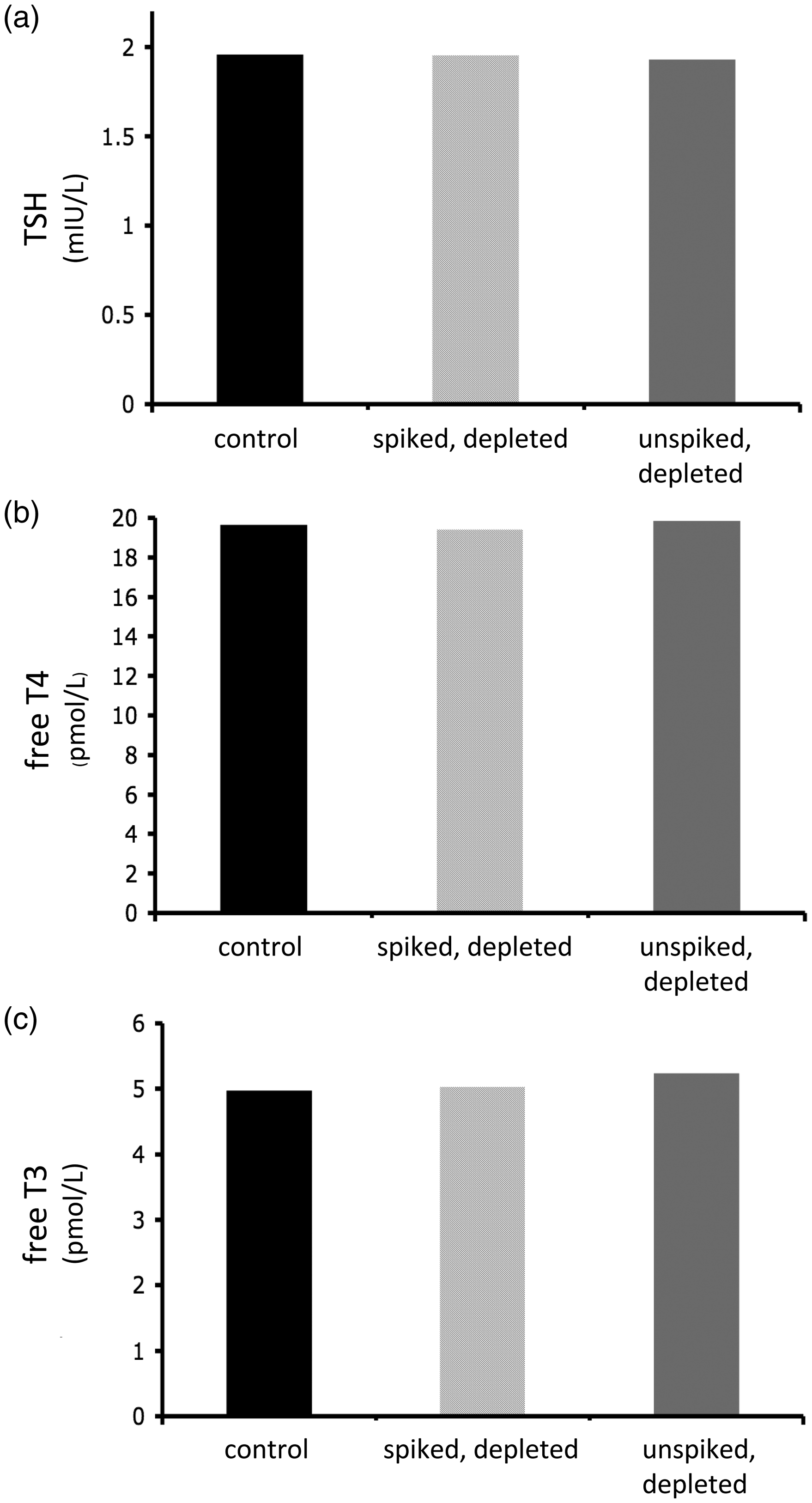
Applicability of depletion protocol to all assays
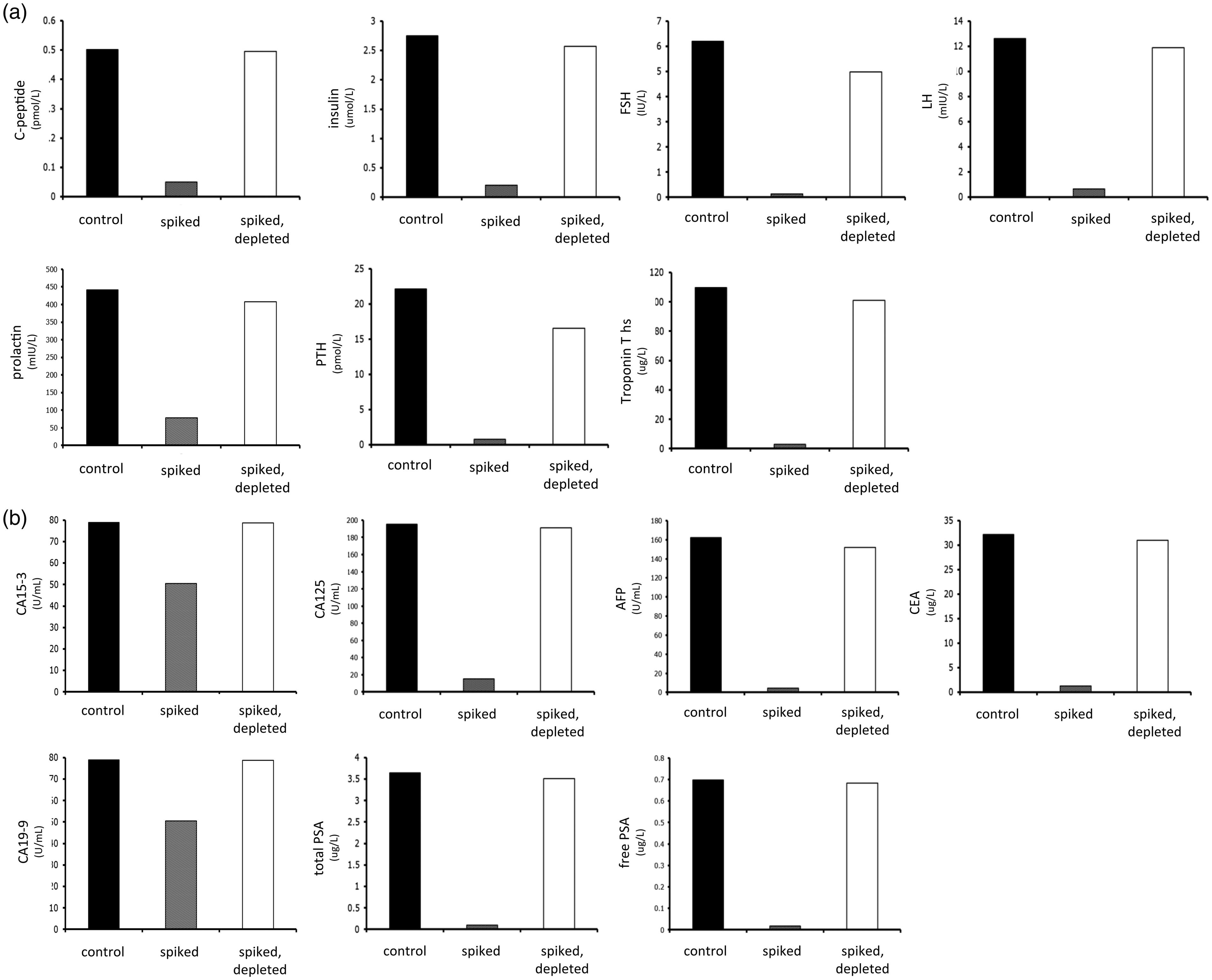
More extensive studies across multiple assays demonstrated that depletion of biotin using this protocol restored the measurement of analytes to levels similar to controls in both competitive (Figure 3) and sandwich immunoassays (Figure 4(a) and (b)). For the majority of the analytes, depleted results were within 95% of the control values. For certain analytes – and these were notably sandwich immunoassays, not competitive immunoassays – the depleted value could only recover 90% or less of the control value. For these analytes, scaling up the microparticle volume did not further increase recovery (not shown), confirming that the problem was not a failure to deplete excess biotin. Nonetheless, in comparison to the spiked results, depletion of biotin led to recoveries very close to control levels for almost all analytes.
Applicability of the method to competitive immunoassays. Serum samples were spiked with 1000 ng/mL biotin (pale grey columns) then subjected to the depletion protocol (white columns). For each analyte tested, depletion of biotin restored measured analyte concentration to a level similar to that of controls (black columns). (a) Applicability of the method to sandwich immunoassays. Serum samples were spiked with 1000 ng/mL biotin (pale grey columns) then subjected to the depletion protocol (white columns). For each analyte tested, depletion of biotin restored measured analyte concentration to a level similar to that of controls (black columns). (b) Applicability of the method to sandwich immunoassays (tumour markers). Serum samples were spiked with 1000 ng/mL biotin (pale grey columns) then subjected to the depletion protocol (white columns). For each analyte tested, depletion of biotin restored measured analyte concentration to a level similar to that of controls (black columns).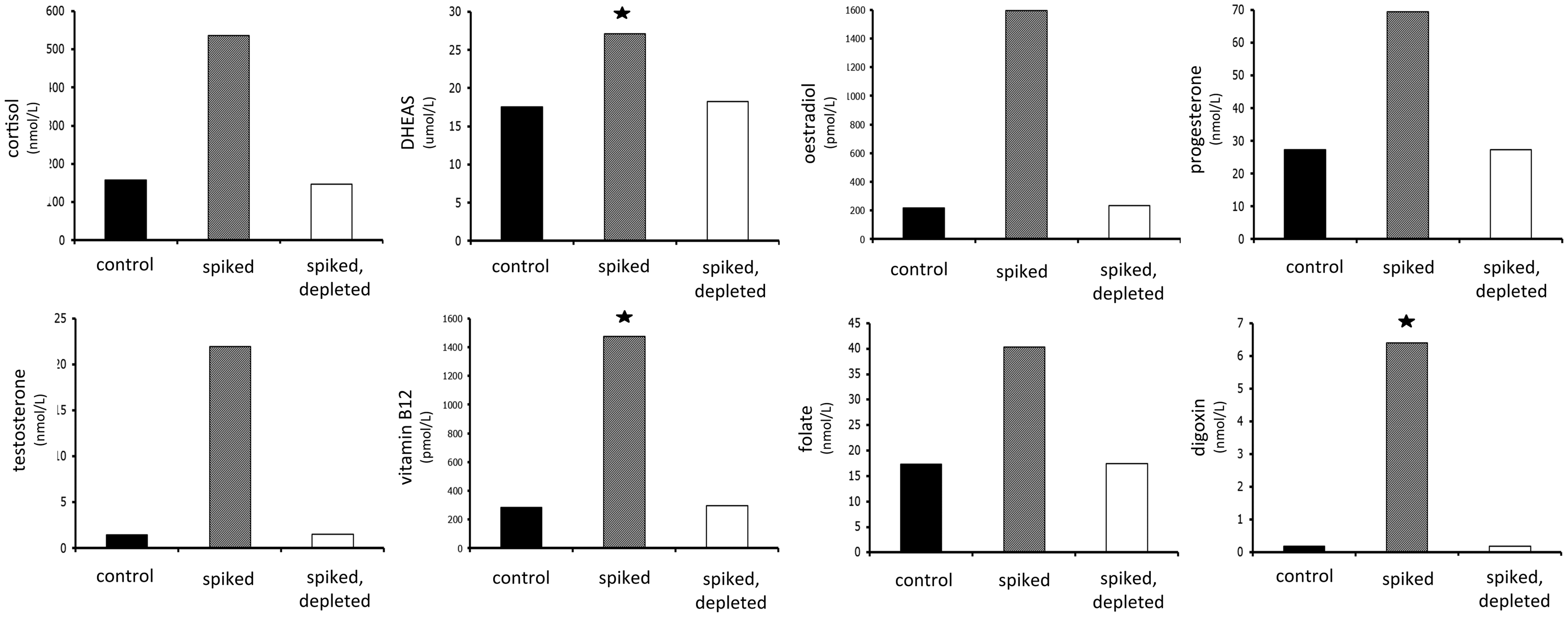
Proof of principle: Depletion of biotin, removal of interference and restoration of results in serum samples derived from MS patients treated with high-dose biotin therapy
Depletion of biotin from serum samples of patients on high-dose biotin therapy.
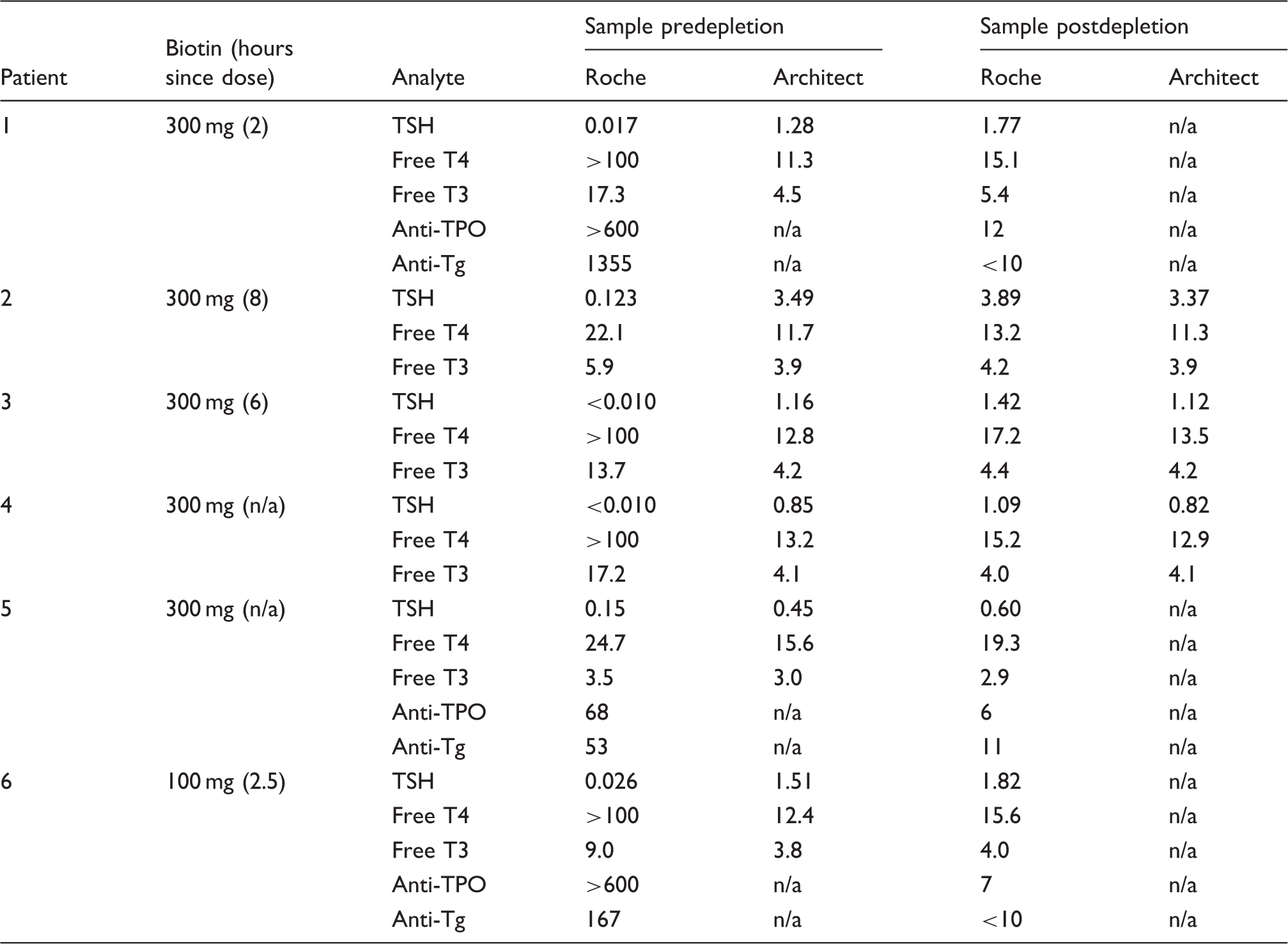
Note: Biotin dose (300 mg daily or 100 mg tds) and time from ingestion (bracketed) is indicated in column two. Biotin was depleted from 250 µL of sample using our protocol as described (2 mL of streptavidin-coated microparticle suspension, 45-min sample incubation). This restored TFTs to biochemically euthyroid status, consistent with results obtained using the (non-biotin-affected) Architect method (method correlations between Roche and Architect are given in Supplementary Data).
Reference Intervals are as follows:
Roche: TSH: 0.50–5.00 mIU/L; free T4: 11.0–22.0 pmol/L; free T3: 3.2–6.4 pmol/L
Architect: TSH: 0.40–4.00 mIU/L; free T4 9.0–19.0 pmol/L; free T3: 2.6–6.0 pmol/L
n/a: not available.
Discussion
Poetic symmetry – The problem becomes the solution
We have developed a protocol that uses the streptavidin-coated microparticles of Roche Elecsys assays to deplete biotin from serum and thereby remove biotin interference from affected samples. Our method is simple, accurate and reproducible, and allows depletion of extremely high concentrations of biotin that have been spiked into serum samples. It also allows removal of ingested biotin and its (interfering) metabolites from serum samples of patients treated with high-dose biotin therapy. This method can be used to confirm the presence of biotin interference in suspected samples. More importantly, this method in most cases permits the reporting of results from biotin-depleted samples without significant change in precision and accuracy. It therefore equips laboratories with a reliable, timely option of managing biotin-affected samples and reporting accurate results independently, without the need for external testing. This is particularly important in the acute setting or in the context of high clinical concern regarding an abnormal/unexpected value. The depletion protocol introduces delay of less than an hour and thus enables reasonable turnaround time.
Until now, for patients on high-dose biotin therapy, the two certain means of preventing interference were either avoiding susceptible, streptavidin-based platforms or withholding biotin for a sufficient period prior to blood tests.8–10 The former seems hardly possible, due to the widespread use of susceptible platforms around the world. These include Roche, Vitros, Beckman Access, Siemens Dimension and IDS iSYS. The latter is also problematic: the exact duration of biotin withdrawal is not known, and likely varies for different tests. 17 In some patients 8 and for certain highly biotin-sensitive tests, a longer period may be required to clear biotin to sufficiently low concentrations. Active depletion of biotin by the laboratory negates the need for the patient to withhold biotin, which is both more convenient for the patient and clinically sound. Early case reports and a recent randomized control placebo trial point to clinical efficacy of high-dose biotin therapy in a subset of patients with progressive MS.18,19 It seems preferable that patients are not obliged to withhold a potentially efficacious drug for many days prior to every blood test. The success of the depletion approach, however, rests on the imperative that laboratories are informed of biotin therapy so that depletion can be carried out prior to analysis and reporting results. Ideally, clinicians managing patients on high-dose biotin therapy are well aware of the potential for biotin interference. We hope that the recent flurry of case reports encourages clinicians to indicate biotin therapy on request slips. Failure to provide this crucial information at the initial point of testing exposes laboratories, clinicians and patients to the risk of erroneous results. Both identification of the problem and provision of the solution require close communication between clinicians, laboratories and pathologists, and transparent notification of high-dose biotin therapy.
Biotin pharmacokinetics
Biotin is rapidly absorbed from the gastrointestinal tract by carrier-mediated uptake. 20 Its oral bioavailability is high and peak plasma concentrations occur within approximately 1.5 h of ingestion. 21 Current data, though not extensive, suggest average maximal biotin concentrations of approximately 400 ng/mL after 100 mg doses when taken in the absence of food. The carrier-mediated uptake of biotin does appear to be saturable, and 300 mg doses do not treble the achieved plasma concentration, rather generate average peak biotin concentrations of just over 800 ng/mL. 15 Estimates of biotin clearance vary; however, most recently, an elimination half-life of between 7.8 and 18.8 h has been calculated for high-dose biotin. 15 Excretion of biotin is predominantly renal, and biotin as well as its major (bisnorbiotin, biotin sulfoxide) and minor metabolites (bisnorbiotin methylketone, biotin sulfone) can be detected in human urine. 21 Biotin metabolites that preserve the ureido ring are capable of binding streptavidin, though with reduced affinity than native biotin. 22 They are expected to cause interference with streptavidin-based assays, though to a lesser extent than native biotin. As shown herein, we have successfully removed biotin interference from numerous serum samples derived from MS patients treated with high-dose biotin therapy, demonstrating that our depletion protocol removes biotin and its streptavidin-binding metabolites. Our depletion protocol can adequately deplete biotin at concentrations of 1000 ng/mL. In the general community, biotin is available as an over the counter supplement in doses of up to 5 and 10 mg. Though a potential source of analytical interference, these concentrations are far less than those achieved in high-dose biotin therapy, and our protocol is well capable of depleting these lower biotin concentrations and removing associated interference.
Potential limitations and future investigations
Several areas for further investigation require mention, one of which relates to uncertainties about plasma biotin concentrations in different settings. In renal failure, the half-life of biotin increases and it is possible that higher plasma concentrations of biotin may be encountered in this setting, which may necessitate scaling up of our method in order to adequately deplete biotin to below the tolerance of every assay. Similarly, given the possibility of pharmacokinetic variability in biotin peak concentration, it is possible that in certain patients the method may not completely deplete biotin and may require scaling up. In our experience, from analysis of the degree of TFT derangement in numerous patients, we do not believe we have encountered a sample exceeding 1000 ng/mL biotin.
We have paid particular attention to the accurate determination of free thyroid hormones in biotin-depleted samples. As shown, our method allows accurate and precise quantification of free T4 and free T3. Because of the equilibrium between free and total hormones, these assays are most sensitive to matrix changes. The accuracy achieved postdepletion supports our contention that adsorbing samples to streptavidin-coated microparticles in general does not cause gross matrix effects that distort the measurement of TFTs.
However we cannot exclude subtle changes in matrix. Moreover, in certain individuals, we do expect marked, idiosyncratic matrix effects to occur. In particular, our protocol is expected to deplete antistreptavidin antibodies. These are recognized as interferents in streptavidin-based assays, though their prevalence is unknown. 23 Like biotin, these antibodies cause falsely low results in sandwich immunoassays and falsely high results in competitive immunoassays. While we anticipate that our method would eliminate this interference by depleting these antibodies through adsorption to streptavidin-coated microparticles, the presence of these antibodies may confound interpretation of biotin depletion efficacy/accuracy because of matrix effects. Biotin antibodies have also been identified,24,25 with a Finnish study reporting a 3% incidence of IgM antibiotin antibodies in adults. 24 These antibodies also cause interference with streptavidin-based immunoassays by binding to biotinylated antibodies. In terms of our depletion protocol, by binding to either endogenous or spiked biotin in serum, the presence of these antibodies might have uncertain effects. Finally, we must point out that certain immunoassays, exclusively sandwich immunoassays, failed to recover accuracy to within 95% of the control value. Further studies are required to determine the nature of this effect.
Several technical points must be noted. Our method requires manual handling, and care and attentiveness are necessary for accurate results. Analysis of paired control samples, unspiked and spiked with a known concentration of biotin, is required for each depletion to ensure adequacy of biotin removal. We currently recommend samples spiked with 1000 ng/mL; if, in future, it is shown that patients achieve higher serum concentrations of biotin, the method may require scaling up and higher controls may need to be used. Important sources of error include inadequate time to immobilize the microparticles prior to aspirating the sample, and contamination of the aspirated sample with magnetic microparticles during manual pipetting. For accuracy, it is crucial that all fluid is removed from the microparticles prior to sample addition. Residual fluid introduces dilution error, and in the case of free T4 and free T3 in particular, distorts free:total hormone equilibrium and, thus, measured values. Commercial devices are available that facilitate aspiration of the magnetic microparticle suspension to dry weight.
Changes in clinical practice and patient behaviour have each increased the prevalence of high-dose biotin use, and significantly altered the risk profile of streptavidin-based assays. Manufacturers must respond to this increased risk and improve the biotin tolerance of their assays. Presumably this will occur in future, although it is unlikely to occur promptly. Until more robust methods are developed, our depletion method allows laboratories the means to both identify biotin interference and remove it. It therefore equips laboratories with the capacity to report accurate and reliable results despite biotin therapy, and thus protect patients from the harm that may ensue from biotin interference.
Footnotes
Acknowledgements
We are grateful to the scientists and staff of Melbourne Pathology for assistance and support in carrying out this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This research was conducted in accordance with the Helsinki Declaration as revised in 2013.
Guarantor
CT.
Contributorship
CT conceived the study, designed the protocol, performed the experiments, reviewed the literature, performed the data analysis and interpretation and wrote the first draft of the manuscript. KS contributed to the literature review, was involved in analysis and interpretation of data, provided critical revision of the manuscript and supervised the project. ZL and TY were involved in data interpretation and execution of the project. All authors reviewed and edited the manuscript and approved the final version.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
