Abstract
Background
Faecal calprotectin is an inflammatory marker used to triage patients for further investigation with suspected inflammatory bowel disease (IBD). Our current method requires faecal samples be sent to the laboratory, where calprotectin is extracted before analysis. This is a time-consuming, potential bottleneck in the pathway. We have recently evaluated the OC-SENSOR PLEDIA fCAL method that uses the same sampling device as used in some bowel cancer screening and symptomatic colorectal cancer programmes that detect faecal haemoglobin. The below study is a comparison of the OC-FCa method with the BÜHLMANN fCAL Turbo which is used routinely within BSPS.
Method
150 homogenised and 110 non-homogenised faecal samples were loaded into OC-Sampling Bottle 3 and BÜHLMANN CALEX cap sampling devices. The samples were then analysed on their respective systems according to manufacturer’s instructions.
Results
The OC-FCa assay had a mean positive bias of 67.3% (homogenised) and 88.4% (non-homogenised). Homogenised samples showed substantial agreement between the methods for normal (<50 µg/g) and elevated (150+µg/g) risk categories (k = 0.794, k = 0.788, respectively) and moderate agreement for borderline (51–150 µg/g) (k = 0.25) according to the current Berkshire and Surrey Pathology Service (BSPS) guidelines. Non-homogenised samples had none to slight agreement for normal and borderline values (k = 0.02 for both) and moderate agreement for elevated (k = 0.596).
Conclusion
The OC-FCa method is a viable alternative for faecal calprotectin testing, but requires an adjustment to clinical cut-off values due to the lack of standardisation and strong positive bias. A clinical comparative study is required to assess the impact of patients collecting their own samples into the devices, as this may negate any potential degradation samples may exhibit during transit to the laboratory.
Introduction
Faecal calprotectin (f-cal) is a surrogate marker of gastrointestinal inflammation. It is used in the diagnosis and management of inflammatory bowel disease (IBD). irritable bowel syndrome (IBS) and other functional disorders of the gastrointestinal tract often present with similar symptoms to IBD including abdominal pain and a change in bowel habits. F-cal can aid in the differentiation of IBD and IBS and is recommended by NICE guideline DG11 1 for this purpose
It is common practice for patients to collect faecal samples at home into collection containers. These are sent to the laboratory where the calprotectin is extracted and quantitated. Initially, f-cal was determined to be stable at room temperature in faeces for up to 7 days; 2 however, more recent evidence suggests that degradation occurs much sooner. 3
The York faecal calprotectin care pathway 4 has widely been adopted as a model for the most cost effective strategy for faecal calprotectin testing in primary care. This pathway uses defined cut-off concentrations of f-cal to determine risk of IBD and differentiation from IBS. Our local pathway uses the following thresholds to determine risk; patients with a f-cal <50 µg calprotectin/g faeces (µg/g) are considered to be at low risk of IBD; 50–150 µg/g are considered borderline with an intermediate risk of IBD and follow up or a repeat test is recommended; tests with a f-cal >150 µg/g indicates patient likely to have active inflammatory bowel disease and referred to gastroenterology for further assessment. Across the UK, pathways are implemented based on the York pathway and as there is currently no analytical standardisation for faecal calprotectin, different methods can give different results so local thresholds are implemented.
We have recently carried out an analytical evaluation of the OC-FCa latex immunoturbidimetry method by Eiken Chemical Co., Ltd 5 that uses the same collection device and analyser as a currently established haemoglobin method used for colorectal cancer screening for both symptomatic and asymptomatic populations.
Here we present results of a sample comparison of the OC-FCa method on the OC-SENSOR PLEDIA with the established B
Method
Excess portions of anonymised faecal samples for routine calprotectin measurement sent to Berkshire and Surrey Pathology Services (BSPS) were collected. Samples were selected to cover the analytical range of the OC-FCa assay of 20–2720 µg/g. Two cohorts of samples were used, homogenous to assess comparison between methods of samples that should have the same result. Non-homogenised samples to assess if any bias observed on homogenous samples is also observed on native samples. There were 111 homogenised results and 49 non-homogenised results obtained for both methods.
All samples were frozen on receipt removed from the freezer and allowed to equilibrate to room temperature for 1 h before processing. One set of samples were homogenised by mixing with single-use wooden stirrers for 1 min. Each homogenised and non-homogenised sample was loaded into an OC-Auto Sampling Bottle 3 and a BÜHLMANN CALEX® Cap device. Homogenised samples required an extra step, and therefore, it was not feasible to analyse the samples on the same day. Samples were stored at 4°C until analysis, within the recommended manufacturer guidelines of both methods. Non-homogenised samples were loaded and stored at room temperature until analysis, as per manufacturer guidelines. Prior to analysis, all samples were mixed and processed according to the manufacturers’ instructions on their respective systems
All OC samples were tested on an OC-Sensor Pledia analyser. Homogenised fCAL Turbo samples were measured on a Roche Cobas 8000 and non-homogenised on an Abbott Alinity where internal comparison studies demonstrated similar results between the two methods. Samples <26 µg/g on the fCAL Turbo were excluded as no numerical result was provided.
Data analysis
The results were visualised using a Bland–Altman plot and the methods were analysed using Wilcoxon signed-rank test. The data was also broken down into categories of normal (<50 µg/g), borderline (50–150 µg/g) and elevated (150+ µg/g) based on the current BSPS guidelines using BÜLHMANN fCAL® Turbo and the methods compared using Cohen’s kappa (k). All data analysis was completed using Analyse-it Software (Leeds, UK) on Excel 2016.
Results
Comparison of outcomes of Fcal Turbo and OC-FCa methods based on current local thresholds.

Discussion
The OC-FCa method shows a clear positive bias when compared to the BÜHLMANN fCAL Turbo. As there is no standardisation for f-cal measurement, the implementation of any new method would require an adjustment of any cut-off levels to maintain the status quo for referrals to gastroenterology and repeat testing. A recent clinical study using the OC-FCa method recommended a cut-off of 600 µg/g which yielded 91.8% sensitivity and 79.3% specificity. 6 If the current BSPS cut-off values and standards are applied then, 11/49 homogenised and 11/13 non-homogenised samples that were in the normal range (<50 µg/g) for the fCAL Turbo method were elevated to the ‘borderline’ risk group when analysed by OC-FCa and would be placed on a different clinical pathway. 10/27 (37%) homogenised and 8/13 (62%) non-homogenised samples that were 51–150 µg/g (borderline) were in the higher risk group of >151 µg/g for OC-FCa which would result in additional referrals directly to gastroenterology rather than monitoring and offering a repeat test. The samples were not loaded directly into the devices by the patients and therefore may not reflect the true calprotectin value at the time the sample was produced. Our previous work has shown a median 80% positive bias for samples collected into extraction devices at home by the participant compared to those extracted in the laboratory from faecal samples sent by the patient. 7 This suggests the true time zero value for f-cal is much higher than the current cut-offs suggest.
All samples were stored frozen at −20°C until an adequate number of samples within the required concentration categories were collected. We do not have any data regarding the time between sample collection and analysis, and it is therefore possible that different degradation products may be present in the samples. Both homogenised and non-homogenised samples were loaded to both devices consecutively to minimise any differences (Figure 1). Evidence of positive bias of OC-FCa versus fCAL Turbo methods displayed as Bland–Altman plots. (a) Homogenised samples (mean relative difference 67.3%, 95% CI: 79.5–214.1%), (b) non-homogenised samples (mean relative difference 88.4%, 95% CI: 149.0–325.8%).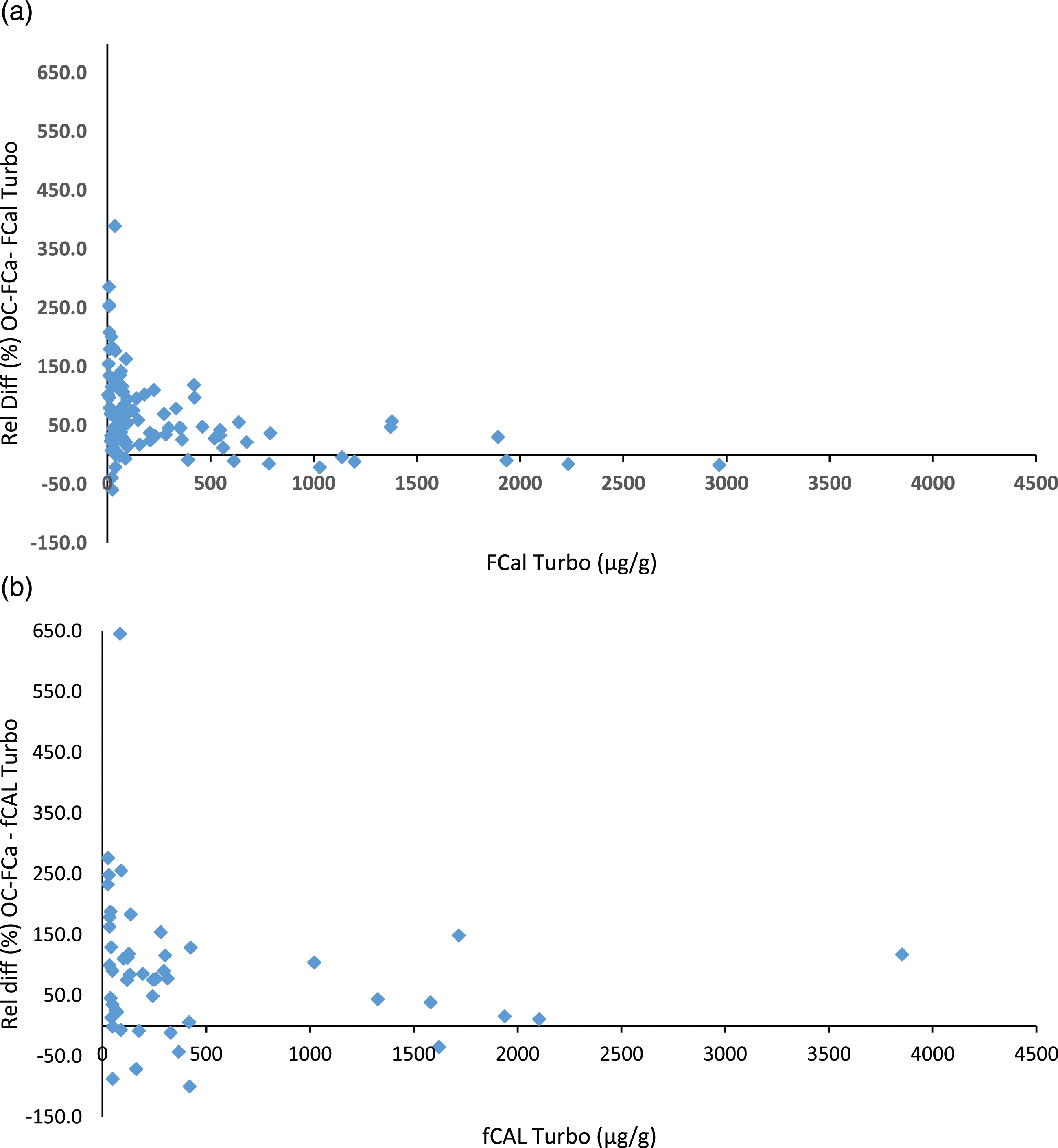
The OC-FCa method is a potential alternative fcal assay. The benefit of which is that direct collection in to faecal collection devices is established for faceal collection in colorectal cancer screening and symptomatic programmes. The advantage for the laboratory is that no extraction process is required and received samples can be placed directly on to analysers. This would reduce workload within the laboratory and improve turnaround times.
Footnotes
Acknowledgements
We would like to thank Eiken Chemical Co., Ltd., Tokyo, Japan and Mast Diagnostics Division, Bootle, Merseyside, UK for supplying the analysers and consumables. BSPS for their assistance with sample collection and analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
SO.
Contributorship
All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
