Abstract
Background
Plasma viscosity (PV) is used in the investigation of hyperviscosity syndrome. We have evaluated the performance of a new bench-top free oscillation rheometer, ReoRox from Medirox, and established a new reference range.
Methods
Performance of the free oscillation rheometry (FOR) method was evaluated examining within-run and between-run imprecision, duration of measurement and operator simplicity. Furthermore, influence of storage on the PV measurements was assessed and a reference range based on plasma from 585 healthy men and women was established.
Results
Imprecision of the free oscillation rheometer was comparable to other viscometers and performance of the instrument was satisfying. The non-parametrical reference range established was 1.22–1.44 mPa s at 37℃ (2.5th and 97.5th percentiles).
Conclusions
We have established a FOR PV reference range to support clinical decision-making. Furthermore, we found high pre-analytical stability of the samples and an analytical imprecision comparable to other PV methods.
Introduction
Plasma and serum viscosity measurements play an important role in the clinical management of patients prone to hyperviscosity syndrome, e.g. patients with Waldenström macroglobulinemia, multiple myeloma, and leukemia. 1 Furthermore, plasma viscosity (PV) is a risk factor for future cardiovascular events 2 and influences the microcirculation in, e.g. retinal and skin vessels. 3
Traditionally, routine measurement of serum or PV has been performed using a manual capillary viscometer, e.g. the Ostwald viscometer in which the kinematic viscosity is calculated by measuring the time for fluid to flow a predefined distance. The viscosity of the sample is expressed as the amount of time it takes the liquid to travel the distance divided by the time used for the same volume of water expressed in the SI unit Centistokes (cSt). Drawbacks of the manual capillary method is that it is susceptible to possible errors associated with manual stopwatch timing, requires a relatively large sample volume (5 mL) and is fairly time-consuming due to required cleaning and drying of the viscometer between measurements. 4
Several automated or semi-automated capillary viscometers have been introduced for use in clinical laboratories (e.g. Lovis 2000 M/ME from Anton Paar gmbh, Graz, Austria, or DV2TLV from Brookfields, Middleboro, USA). Recently, a new small, robust and portable instrument, ReoRox G2 from Medirox (Nyköping, Sweden) utilizing a patented technology to measure dynamic viscosity of a fluid by free oscillation rheometry (FOR) and which reports the result in the SI unit mPa s was introduced. In the FOR instrument a cup containing the sample is attached to a torsion wire, and the cup is initially rotated around its longitudinal axis and then released. At release, the torsion wire will set the cup into free oscillation and its movement is recorded by an optical detector.
The present study was designed to evaluate the performance of PV measurements by ReoRox G2 in order to assess potential pre-analytical factors influencing its measurement and to establish a reference range for dynamic PV measured by FOR.
Methods
Participants
Plasma samples were obtained from a cohort of 585 healthy adult Danish blood donors at Odense University Hospital (261 females, 324 males, age 17–66 years). Participants were excluded from donating blood if any of the following was present: pregnancy, autoimmune diseases, AIDS/HIV, infectious diseases, cancer, allergy, high (diastolic BP >90 mmHg) or low (systolic BP <110 mmHg) blood pressure, known cardiovascular disease (including thromboses), diabetes requiring treatment, severe skin diseases treated with steroids, ulcer, current use of narcotics or medicine, recent operation/tattoo/piercing or treatment with hormones. Informed consent was obtained from all participants prior to enrolment in the study.
Imprecision
The within-run imprecision was assessed using 10 replicate analyses of plasma samples of two patients and one quality control sample; the between-run imprecision was assessed by analysing these plasma and quality control samples over 20 consecutive days by different operators.
Sample stability
EDTA-plasma from 20 persons was aliquoted and stored at various temperatures and the PV of the stored plasma was compared to the PV of fresh plasma from each person. One aliquot of each plasma sample was stored at 4℃ and re-measured after 20 days, while another aliquot of each plasma sample was stored at −20℃, and, after three months of storage, thawed at 37℃ and re-measured.
Statistical analyses
Statistical analyses were performed using PASW 18 (SPSS) software (IBM, New York). The Kolmogorov-Smirnov test was performed to test for normality distribution. Data were compared by Mann-Whitney U-test for non-parametric data. P values <0.05 were considered statistical significant.
Results
Imprecision
Within-run and between-run assay imprecision determined from aliquots of the same samples.
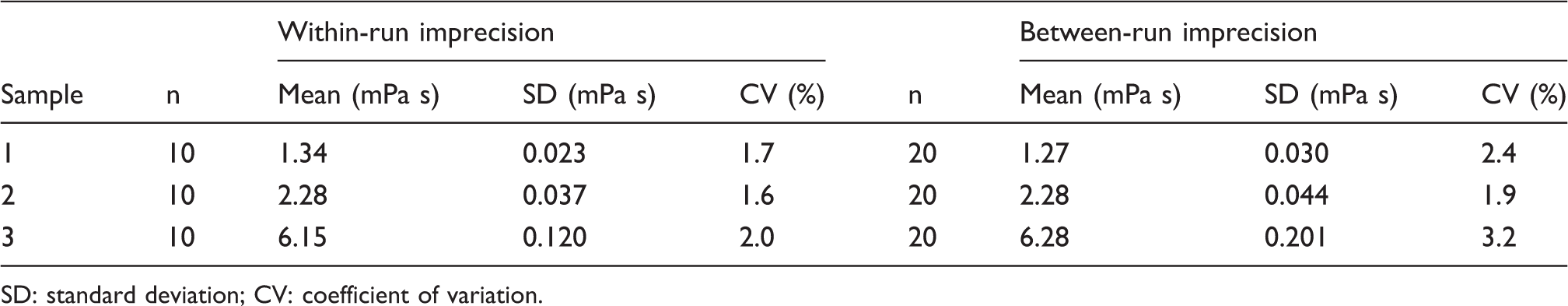
SD: standard deviation; CV: coefficient of variation.
Sample stability
Storage of EDTA-plasma samples at 4℃ for 20 days (P = 0.963), at 20℃ for 48 h (P = 0.801) and at −20℃ for 90 days (frozen and re-thawed) (P = 0.838) did not affect the viscosity measurements significantly.
Reference range
The one-sample Kolmogorov-Smirnov test of normality indicated that the distribution of FOR PV was non-Gaussian (Z = 0.099, P < 0.0001) and positively skewed (skewness = 1.422). Therefore, establishment of the reference range employed a non-parametric approach. The median PV for all 585 subjects was 1.32 mPa s, with the reference range determined as 1.24–1.44 mPa s at 37℃ (2.5–97.5 percentiles). There was no significant difference between genders (Mann-Whitney U-test; P = 0.066).
Discussion
Measurement of PV has potential as a useful biomarker in the assessment of cardiovascular risk.5,6 In contrast to the erythrocyte sedimentation rate (ESR), PV is not influenced by haematocrit, red blood cell aggregation, haemoglobinopathies, or time to analysis, and is therefore recommended as a substitute to ESR in diagnosis and follow-up of hyperviscosity syndrome and rheumatoid arthritis. 7 However, the potential of PV has been limited due to the lack of automated and reliable instruments.
Performance evaluation of the ReoRox G2 FOR instrument is in accordance with the ICSH recommendation for a selected method to measure PV, 8 with operative simplicity, a test duration below 1 min and a low sample volume (400 µL). Disposable sample cups eliminate sample carry-over and need for cleaning steps. A drawback of the FOR method is the high sensitivity to external vibrations requiring that the instrument ideally is placed on a firm bench without any disturbances from surrounding instruments or personnel.
The within-run imprecision for measurements of PV on ReoRox G2 was higher than the within-run imprecision stated in the ReoRox G2 application manual (CV = 1.0%). Nevertheless, the overall imprecision of the method is better than achieved with our manual capillary rheometry method (CV = 4.2%) and comparable to other viscosity instruments. 9
In the present study, dynamic PV values established for a healthy Danish population are similar to or higher than those from other European countries, where PV values in Glasgow (Scotland) and Ausberg (Germany) were 1.327 ± 0.093 mPa s and 1.261 ± 0.067 mPa s for men and 1.318 ± 0.087 mPa s and 1.248 ± 0.066 mPa s for women, respectively. 10 Similarly, in a Spanish study the PV was 1.235 ± 0.061 mPa s for males and 1.236 ± 0.059 mPa s for females. 6 Also, 1.24 ± 0.10 mPa s was found in a healthy German population aged 45–65 years. 11 The slightly higher PV level found in the current study might be due to different exclusion criteria’s, e.g. was plasma cholesterol, lipid levels,5,12 age and smoking 11 and obesity 6 not used for exclusion in this study.
Finally, we found a high pre-analytical stability of the samples making it possible to send plasma separated from the packed erythrocytes by mail at room temperature or to store the plasma frozen for longer periods.
Footnotes
Acknowledgements
We thank Elsebeth Parlev for technical assistance.
Declaration Conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The protocol was approved within the guidelines from the local ethical committee of Region Syddanmark for the use of donor blood for quality assurance experiments.
Guarantor
LP.
Contributorship
LP wrote the paper. MN reviewed and edited the manuscript. EB was involved in protocol development and data analysis. MKC and MB was involved in protocol development and undertook the laboratory studies.
