Abstract
Background
Biomarkers can facilitate the diagnosis of sepsis, enabling early management and improving outcomes. Lipopolysaccharide-binding protein (LBP) has been reported as a biomarker for the detection of infection, but its diagnostic value is controversial. In this study, we assessed the diagnostic accuracy of LBP for sepsis in the emergency department (ED) patients, comparing it with more established biomarkers of sepsis, including procalcitonin (PCT) and C-reactive protein (CRP).
Methods
LBP and other sepsis biomarkers, including PCT and CRP, were measured on admission in 102 adult patients presenting with suspected infection. Classification of patients was performed using the recently updated definition for sepsis (Sepsis-3). The diagnostic accuracy of LBP, CRP and PCT for sepsis was evaluated by using receiver operating characteristic curve (ROC) analysis.
Results
A total of 49 patients were classified as having sepsis. In these patients, median (interquartile range) LBP (41.8 [41.1] µg/dL vs. 26.2 [25] µg/dL), CRP (240 [205] mg/L vs. 160 [148] mg/dL) and PCT (5.19 [13.68] µg/L vs. 0.39 [1.09] µg/L) were significantly higher than in patients classified as not having sepsis (P < 0.001 for all three biomarkers). ROC curve analysis and area under curve (AUC) revealed a value of 0.701 for LBP, similar to CRP (0.707) and lower than that for PCT (0.844) (P = 0.012).
Conclusion
In adult ED patients with suspected infection, the diagnostic accuracy for sepsis of LBP is similar to that of CRP but lower than that of PCT.
Introduction
Sepsis is a common condition handled in the emergency department (ED). Despite advances in medical care and antibiotics, it is still a major cause of mortality. 1 Guidelines by Surviving Sepsis Campaign emphasize the importance of early diagnosis and appropriateness of the administered therapy in the initial hours, with a significant impact on outcomes. 2 Delay in initiating antimicrobial treatment is associated with worse prognosis. 3 Therefore, emergency physicians are encouraged to accurately diagnose, assess the severity and provide adequate treatment in a timely manner.
Biomarkers play a key role in diagnosis of infection and sepsis. Pierrakos and Vincent identified 178 sepsis biomarkers evaluated in 3370 studies, but none has sufficient specificity or sensitivity to be routinely employed in clinical practice. Procalcitonin (PCT) and C-reactive protein (CRP) are widely used, but even these have limited diagnostic accuracy. CRP has poor specificity unless high cut-off levels are used. 5 PCT has been identified as having the highest performance among biomarkers for the diagnosis and prognosis of sepsis, 6 although the evidence presented by Tang et al. 7 does not support the widespread use of the PCT test for sepsis diagnosis.
A growing number of studies suggest that lipopolysaccharide-binding protein (LBP) may play a role in diagnosing sepsis. However, whether this biochemical marker is appropriate for use as a diagnostic tool for sepsis is still uncertain. Two recent meta-analyses concluded that serum LBP has a poor degree of diagnostic accuracy for sepsis,8,9 although the number of included studies may have lacked the statistical power to draw a final conclusion and more clinical studies are needed.
LBP is an acute phase protein of hepatic origin that plays an important role in innate immunity mechanisms. LBP binds to the amphipathic lipid A of bacterial lipopolysaccharide (LPS), a component of the outer cell wall of Gram-negative bacteria, and transfers LPS to CD14 protein. 10 By facilitating binding to the CD14 cell membrane molecules, LBP enhances the sensitivity of macrophages and other cells. Cells expressing membrane-bound CD14, such as endothelial cells, are stimulated to bind a complex of LPS and a soluble form of CD14. 10 CD14 can only bind LPS in the presence of LBP; although LPS is considered to be the main ligand, CD14 also recognizes other bacterial components, including the proteoglycans of Gram-positive bacteria. Thus, besides its role in Gram-negative infections, LBP has a more extended role as a general recognition molecule. 11
To our knowledge, no study has evaluated the diagnostic accuracy of serum LBP for sepsis in an ED setting, although some studies have investigated its ability to predict bacteraemia and to identify patients with infections requiring antibiotics.12,13 In this study, we assessed the utility of serum LBP measured on admission to diagnose sepsis, defined according to Sepsis-3, 14 compared with CRP and PCT.
Material and methods
Study design and patients
This observational prospective single-centre study was conducted at the ED of Hospital General Universitario Santa Lucía of Cartagena, Spain, from November to December 2013. Patients with suspected infection, as judged by the referring physician and the requesting of cultures (blood culture and others, according to the suspected source of infection) collected at the time of admission, were included. Exclusion criteria were: (1) age less than 18 years old; (2) evidence of an immunocompromised state (e.g. malignancy), terminal stage of disease and (3) pregnancy.
To classify the patients, the Sepsis-3 recently published definitions were used. 13 Sepsis was defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. Organ dysfunction was identified as an acute change in sequential (sepsis-related) organ failure assessment (SOFA) score of ≥ 2 points or more resulting from infection. Septic shock is defined as sepsis with persisting hypotension requiring vasopressors to maintain mean arterial pressure ≥ 65 mm Hg and having a serum lactate concentration > 2 mmol/L despite adequate volume resuscitation. 14 Classification of patients was determined by the agreement of two physicians blinded to biomarker levels and based on the results of systematic reviews of available clinical, microbiological and radiographic data. If there was discrepancy between these, a third physician made the determination.
Informed consent was obtained from all participants or from their close relatives and the study was approved by the local Ethics Committee.
Blood sampling and laboratory assays
Biomarker levels were measured in blood specimens drawn in the ED at the first medical evaluation. CRP and PCT were measured in the laboratory within 2 h. A serum aliquot was immediately frozen and stored at −80℃ until the end of the study, when all samples were tested for LBP.
Serum CRP concentrations were measured by an inmunonephelometric assay on a Dimension Vista analyzer (Siemens Healthcare Diagnostics, Los Angeles, CA, USA), with a detection limit of 2.9 mg/L.
Serum PCT concentrations were measured on a Cobas e411 analyzer (Roche Diagnostic, Mannheim, Germany), according to the electrochemiluminescence immunoassay measurement principle, with a detection limit of 0.02 µg/L and a functional sensitivity of 0.06 µg/L.
Serum LBP concentrations were measured using a solid-phase, enzyme-labelled chemoluminescent immunometric assay on an IMMULITE 2000 analyzer (Siemens Healthcare Diagnostics, Los Angeles, CA, USA). The assay detection limit, as described by the manufacturer, was 1.2 μg/mL. The measuring range was 1.2 to 170 μg/mL with a functional sensitivity of 1.5 μg/mL.
Statistical analysis
The normality of continuous variables was tested by the Kolmogorov-Smirnov test. Continuous variables are presented as median (interquartile range [IQR]). Comparisons of group differences for continuous variables were assessed by Mann–Whitney U test or Student’s t-test. Categorical variables are presented as an absolute number with the corresponding proportion. The significance of differences in proportions was tested by Chi-square test. In order to evaluate the discrimination ability for the diagnosis of sepsis of the biomarkers, we estimated the area under the curve (AUC) of the receiver operating characteristic (ROC) curves and their diagnostic accuracy for sepsis was compared with the DeLong test. The optimal cut-off value was set for each ROC curve through the Youden Index (corresponding to the maximum of the sum ‘sensitivity + specificity’). Statistical analysis was performed using SPSS v. 20.0 software (SPSS Inc., Chicago, IL, USA) and MedCalc Statistical Software version 13.3.3 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2014). A P-value of less than 0.05 was considered statistically significant.
Results
Subject characteristics
A total of 102 consecutive adult patients (median age: 70.5 years (IQR: 19, range: 22–94), 61 males (59.8%) and 41 females (40.2%) consecutively admitted to the ED and fulfilling the inclusion criteria were enrolled. Twenty-three patients (22.5%) had received antibiotic therapy previously; 17 patients (16.7%) required management in ICU and in-hospital mortality was 16.7%. Infection was microbiologically documented in 59 patients (57.8%), and 25 (24.5%) had proven bacteriaemia, being the most common causative agent Escherichia coli (40%).
Characteristics of study population.
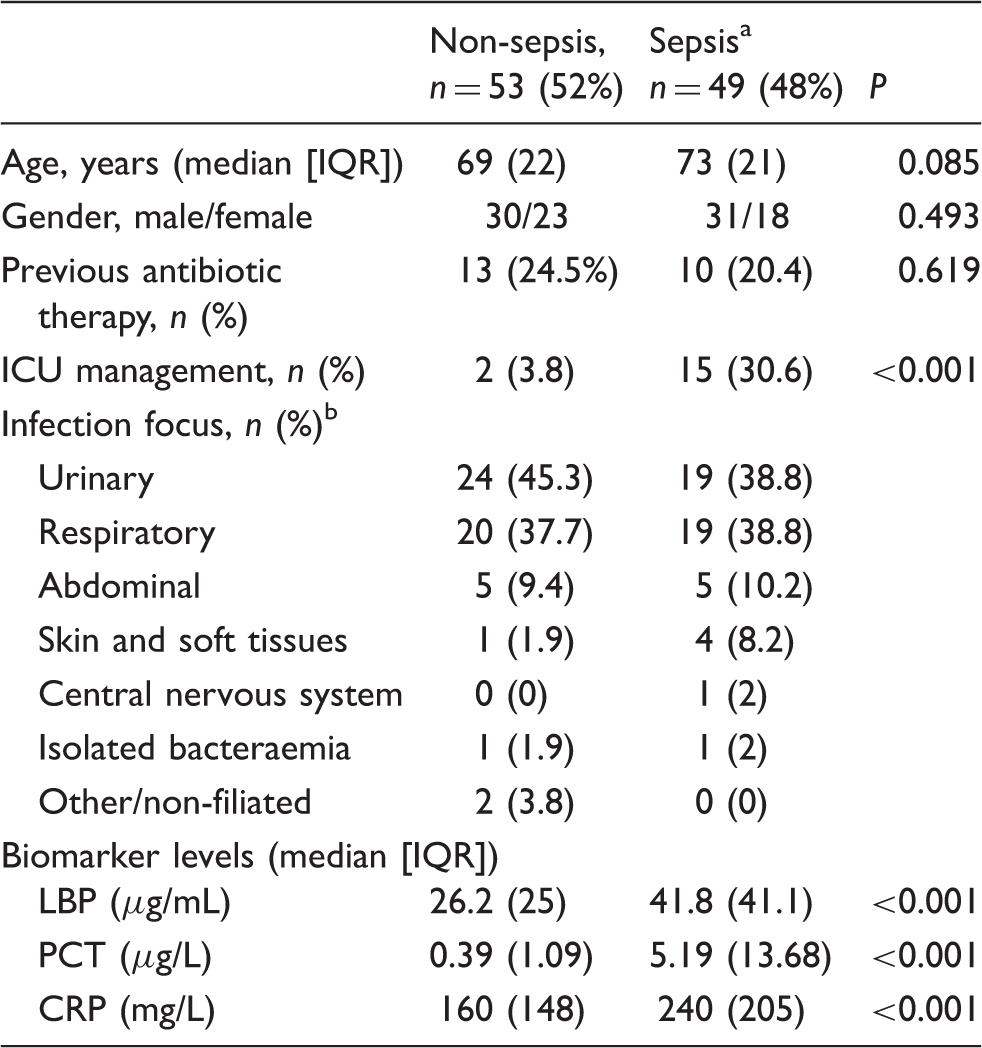
IQR: interquartile range; LBP: lipopolysaccharide-binding protein; CRP: C-reactive protein; PCT: procalcitonin.
Including sepsis (n = 45) and septic shock (n = 4).
According to suspicion judged by physician on ED admission.
Comparison between ‘non-sepsis’ and ‘sepsis’ groups
There were no significant differences between the two groups regarding the demographic characteristics, previous antibiotic therapy and infection focus, although the requirement of ICU management was higher in patients with sepsis. Serum LBP, CRP and PCT were significantly higher in the sepsis group (Table 1 and Figure 1).
Median (boxplots) values of CRP, PCT and LBP.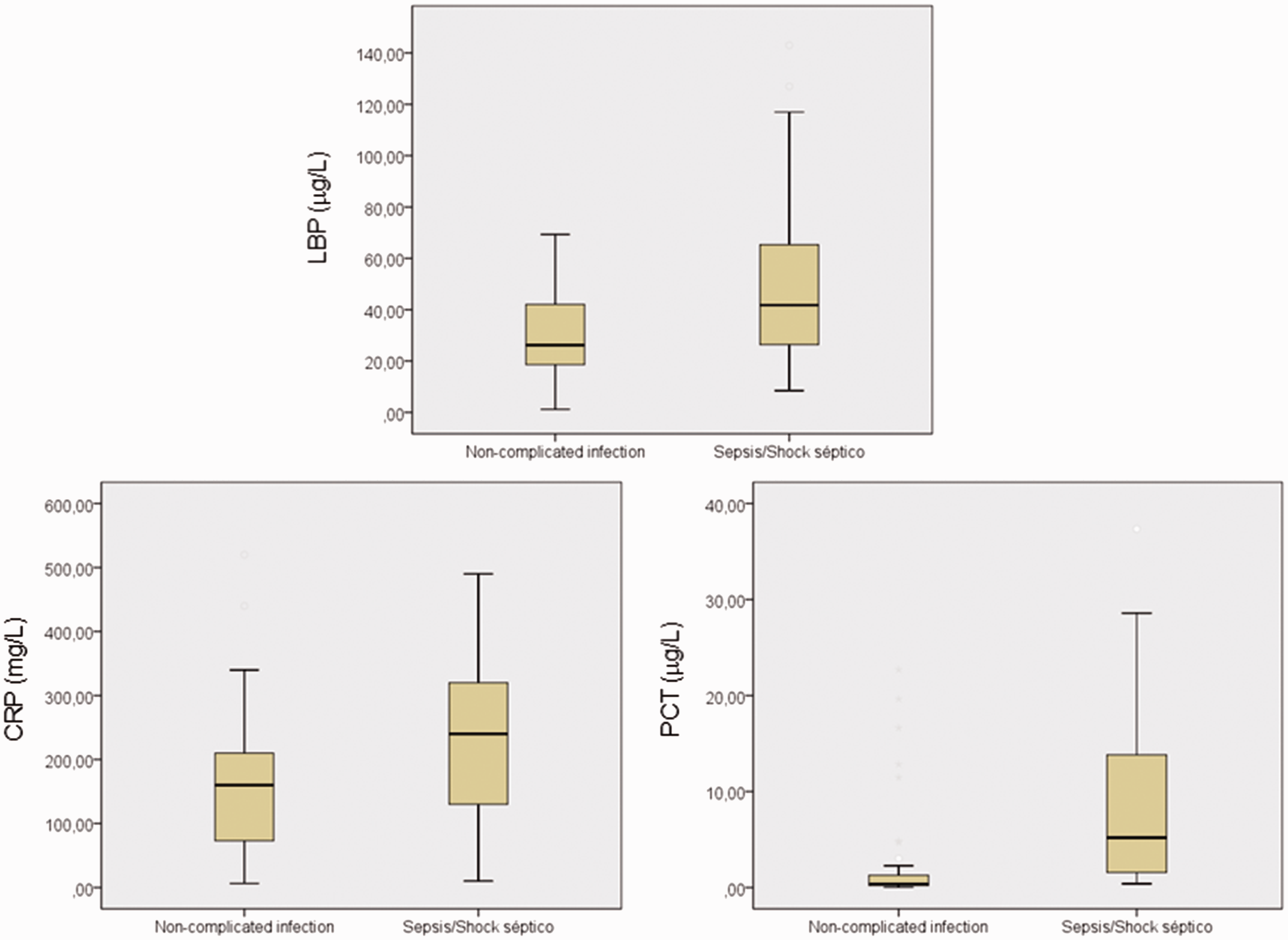
Diagnostic significance: ROC analysis
Performance of the studied biomarkers for sepsis.
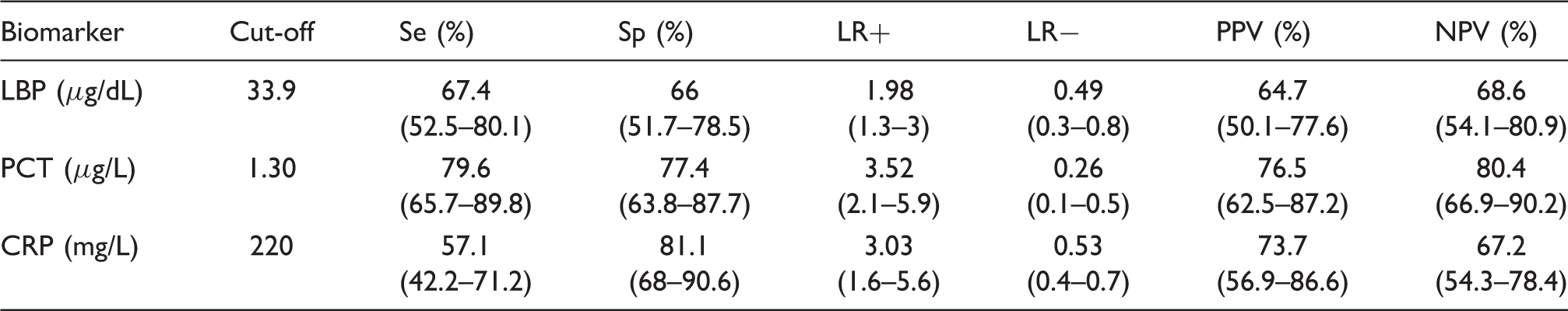
Se: sensitivity; Sp: specificity; LR+: likelihood ratio positive; LR-: Likelihood ratio negative; PPV: positive predictive value; NPV: negative predictive value; LBP: lipopolysaccharide-binding protein; PCT: procalcitonin; CRP: C-reactive protein.
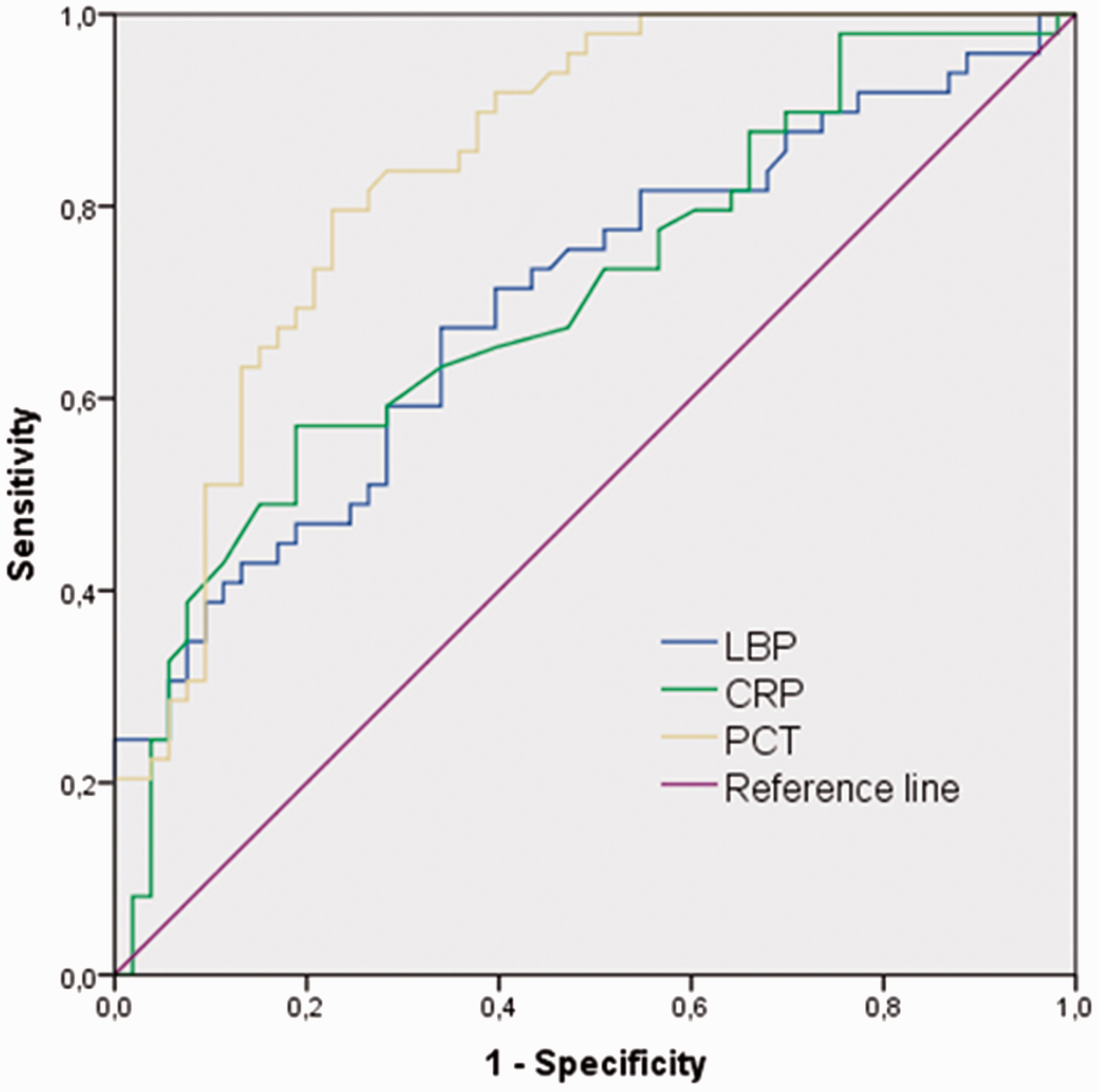
ROC curve analysis on the diagnostic value of LBP for sepsis compared with traditional markers of sepsis.
Discussion
Biomarkers are tools to assist clinicians to make decisions in clinical practice, especially in some populations, such as elderly, in which the incidence and short-term mortality of the infectious diseases have increased significantly in recent years 15 and the clinical manifestations are often non-specific and variable. 16 However, owing to the limitations of currently available biomarkers, such as CRP and PCT, 4 further efforts are needed to identify the novel biomarkers of infection. Previous studies have demonstrated the utility of LBP as a predictor of infection in patients with febrile neutropenia, 17 and of bacteraemia in children with community-acquired infection 1 and for diagnosing of late-onset neonatal sepsis18,19 and febrile urinary tract infection in children. 20
In this study, we have analysed the value of LBP, measured on admission, for diagnosis of sepsis in patients admitted to the ED with suspected infection, compared with CRP and PCT. The main finding is that the diagnostic accuracy for sepsis of LBP is similar to that of CRP but lower than that of PCT.
In our study, PCT was the biomarker that best discriminated between the presence or absence of sepsis. The optimal threshold calculated for diagnosing sepsis (1.2 ng/mL) was higher than 0.5 ng/mL, commonly used in clinical practice, probably due to the differences in the updated sepsis definition, including organ dysfunction. This cut-off is similar to that reported by Liu et al. (1.435 ng/mL) for distinguishing severe sepsis, 21 similar term to the new definition for sepsis.
Previous studies have evaluated the diagnostic accuracy of LBP for sepsis. In the ICU setting, including critically ill patients, the performance of this biomarker was moderate, with AUC ranging from 0.66 and 0.74,22–24 lower than PCT, and optimal cut-offs close to 30 µg/mL. In the ward setting, LBP also had moderate performance, with AUC ranging from 0.63 to 0.78.25,26 In two recently published meta-analyses, the diagnostic accuracy of LBP was moderate and lower than that of PCT. AUC values for diagnosis of sepsis were 0.688 and 0.71, 9 respectively, similar to the AUC observed for LBP in the current study (0.701). Caution must be excercised in comparing results obtained in our study with these studies, for two reasons: (1) the criteria for classifying patients was different (in our study, sepsis was defined according to Sepsis-3, whereas in the previous studies the aim was to analyse its ability to differentiate non-infectious systemic inflammatory response syndrome (SIRS) from sepsis 2 and (2) the performance of sepsis biomarkers may vary among different clinical settings, such as the ED, ICU and ward. 27
Our study has several important strengths. First, this is the first study evaluating the utility of LBP, compared with other traditional biomarkers, for diagnosis of sepsis in an ED. Two previous studies have analysed the utility of this biomarker for other purposes in the same setting. Guille-Johnson et al. 12 concluded that LBP and other biomarkers as PCT and IL-6 did not provide additional information on antibiotic decision. Interestingly, in that study, LBP could not identify patients developing severe sepsis during the first 24 h at the hospital. In a second study performed in ED patients, the predictive value of LBP for bacteraemia was significantly lower than that of PCT (AUC: 0.80 [95% CI: 0.75–0.84]). 13 Second, this is the first study evaluating the diagnostic accuracy of LBP for sepsis when the Sepsis-3 definition is used to classify the patients.
Our study has also some limitations. First, it was a single-centre study with a small sample size, so the results may not be applicable to other settings. Second, we have assessed the diagnostic accuracy for sepsis at the time of sampling and we cannot draw conclusions about the value as a predictor for later development of sepsis or the impact of serial measurements. Finally, in our study, ‘quick SOFA’ (qSOFA), the recommended score in Sepsis-3 definition (as a simple bedside tool in out-of-hospital, ED, and ward settings to identify adult patients with suspected infection who are likely to have poor outcomes and as a prompt to consider possible sepsis), 14 was not calculated. However, in a recent retrospective study, in patients admitted with suspected infection in the ED, Brabrand et al. 2 reported, for the suggested cut-off of ≥2, a moderate performance, with an AUC of 0.737 for predicting adverse outcomes, slightly lower than the AUC for PCT in our study (0.844).
In conclusion, the results in our study confirm the conclusion of two recent meta-analyses,8,9 in which LBP had weak sensitivity and specificity, according to the setting. That of Chen et al. 8 included studies in which LBP was evaluated to differentiate non-infectious SIRS from sepsis, and concluded that it may not be practically recommended for clinical utilization. 8 Similarly, and using the Sepsis-3 definition, the results in our cohort indicate that LBP has a moderate degree of diagnostic accuracy for sepsis, lower than that of the PCT, commonly measured in clinical practice for diagnosing sepsis.
Footnotes
Acknowledgments
The authors acknowledge Siemens Healthcare Diagnostic for providing the assays for LBP. Siemens Healthcare Diagnostic did not participate in the protocol development or interpretation of the results.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the ethics committee of Hospital Universitario Santa Lucía, Ref. EO 2013-28.
Guarantor
LGG.
Contributorship
LGG and MDA designed the study. EJS, AHH and PET recruited the subjects for the study. SRA, RJS and AOF compiled the clinical data and categorized the subjects. LGG and MDA analysed the data and wrote the manuscript.
