Abstract
Background
We investigated the in vitro effects of various phospholipids as emulsifiers on the hydrolysing activities of lipoprotein lipase (LPL) Arg243His against triolein as substrate. LPL Arg243His, identified in a patient with hyperchylomicronaemia, displays severely diminished activity for triolein when emulsified with Triton X-100.
Methods
Lipolytic activities of plasma obtained by heparin injection from a homozygous patient with LPL Arg243His were analysed using triolein emulsified with phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS), phosphatidylinositol (PI), lysophosphatidylcholine (LPC), or Triton X-100 as substrates.
Results
The hydrolysing activities of the patient’s plasma for triolein emulsified with PC, PE, PS, PI, LPC and Triton X-100 were 9.22 ± 1.06 μmol/ml/h/ngLPL, 2.94 ± 1.60 μmol/ml/h/ng LPL, 3.72 ± 1.63 μmol/ml/h/ng LPL, 3.40 ± 1.20 μmol/ml/h/ngLPL, 3.72 ± 1.96 μmol/ml/h/ngLPL and 7.80 ± 4.48 μmol/ml/h/ng LPL, respectively. Thus, the specific activities of the patient’s LPL determined with triolein emulsified with PC were significantly higher than those with PE, PS, PI or LPC as emulsifiers. Relative to the activities of normal plasma measured with PC, PE, PS, PI and LPC as emulsifiers, the mutant’s activities were 49.1 ± 5.2%, 44.1 ± 5.7%, 31.7 ± 12.6%, 19.2 ± 6.9% and 23.8 ± 11.3%, respectively. Using PC, PE, PS, PI and LPC as emulsifiers, the mutant’s activities for triolein-lipolysis relative to normal were significantly increased in comparison to the relative activity measured with the classical emulsifier, Triton X-100 (12.9 ± 6.7%).
Conclusions
Impaired triolein hydrolysis by LPL Arg243His was partially ameliorated by triolein emulsification with phospholipids. The in vitro analysis of triolein hydrolysis using various phospholipid emulsifiers may be useful for the further understanding of impaired LPL function.
Introduction
Lipoprotein lipase (LPL) is a key enzyme which hydrolyses triglycerides (TG) in circulating lipoproteins, and the dysfunction of the enzyme causes severe hyperchylomicronaemia, often accompanied by acute pancreatitis. 1 Various mutations in the LPL gene have been reported to cause decreased hydrolysing activities, decreased protein production, or both. Possibly, one of the mechanisms underlying the deterioration of hydrolysing activities in such patients may be the disturbance of substrate recognition for subsequent catalytic reactions by the abnormal LPL.
We have previously reported a patient homozygous for LPL Asp204Glu, the hydrolysing activity of which was remarkably decreased for very low density lipoprotein-TG and Triton-X-100-emulsified triolein. 2 However, the observed lipolytic activities against triolein emulsified with phosphatidylethanolamine (PE) or phosphatidylserine (PS) were not disturbed, as the activities were increased 1.5- to 2-fold compared with the Triton-X-100-emulsified triolein. 2 This suggested that the diminished activity of the patient’s LPL was ameliorated by modifications of the phospholipid (PL) composition(s) in the substrates.
Recently, we have reported a patient with homozygosity for LPL Arg243His. 3 Using glycerol tri[1-14C]-oleate as substrate, the mutant LPL activity has previously been shown to be severely disturbed, although the circulating protein concentration was not much decreased.4,5 Here, we have investigated the effects of three PLs as emulsifiers on the triolein-hydrolysing activities of LPL Arg243His.
Methods
The history including laboratory and imaging data of the 66-year-old patient with LPL gene homozygous Arg243His, who had suffered from severe hyperchylomicronaemia with recurrent pancreatitis has been described in Suzuki et al. 3 In summary, her concentrations of serum TG, total cholesterol and HDL-cholesterol were 2942 mg/dL, 221 mg/dL and 16 mg/dL, respectively. She was homozygous for a missense mutation, Arg243His, in the LPL gene. For the measurement of LPL protein and activity, plasma was collected 10 min after intravenous heparin (30 units/kg) injection from the patient and normal subjects (n = 3). The patient’s LPL protein concentrations before and after heparin injection were 52 ng/mL (reference range, 45–63 ng/mL) and 90.8 ng/mL (reference range, 168–284 ng/mL), respectively.The activities of LPL and hepatic triglyceride lipase (HTGL) against modified substrates were measured as described. 2 Briefly, for the preparation of PL-emulsified substrates, 10 mg of triolein (glyceryl trioleate) were mixed with phosphatidylcholine (PC) (0.1 mg), PE (0.1 mg), PS (0.1 mg), phosphatidylinositol (PI) (0.1 mg), or lysophosphatidylcholine (LPC) (0.1 mg) in chloroform:methanol (1:2) and evaporated under N2 gas. After addition of 1.5 mL of 1 M Tris-HCl (pH 8.4), the mixture was sonicated for 10 min on ice. Fifty microlitres of the reaction mixture were used as PL-modified triolein for the measurement of LPL activity, or with triolein alone emulsified with Triton X-100. The triolein-hydrolysing activity was expressed as micromole fatty acid released per 1 ng of LPL. The results are expressed as means ± standard deviation (SD). Comparisons were assessed for significant differences by paired Student’s t-test, and the values of P < 0.05 were considered to be statistically significant. All statistical analyses were performed using the ‘Stat View 4.0’ statistical analysis package (Abacus Concept, Inc.).
Results
Hydrolysing activities in the patient’s plasma after heparin injection for triolein emulsified with PC, PE, PS, PI, LPC and Triton X-100 were 9.22 ± 1.06 μmol/ml/h/ng LPL, 2.94 ± 1.60 μmol/ml/h/ng LPL, 3.72 ±1.63 μmol/ml/h/ng LPL, 3.40 ± 1.20 μmol/ml/h/ng LPL, 3.72 ± 1.96 μmol/ml/h/ng LPL and 7.80 ± 4.48 μmol/ml/h/ng LPL, respectively. Thus, the activities for triolein emulsified with different phospholipids were not equivalent, i.e. the activity with PC was significantly higher than that with PE, PS, PI or LPC (Figure 1(a)). Relative to the activities of normal plasma measured with PC, PE, PS, PI and LPC as emulsifiers, the mutant’s activities were 49.1 ± 5.2%, 44.1 ± 5.7%, 31.7 ± 12.6%, 19.2 ± 6.9% and 23.8 ± 11.3%, respectively. Using PC, PE PS, PI or LPC as emulsifiers, the mutant’s activities for triolein-lipolysis relative to normal were all significantly increased in comparison to the relative activity measured with the classical emulsifier, Triton X-100 (12.9 ± 6.7%) (Figure 1(b)).
Hydrolysing activities of the patient’s plasma and normal subject’s plasma after heparin injection for triolein emulsified with various phospholipids (a) and the activities (%) relative to normal (b). Each activity of plasma was measured in a mixture containing 0.5 μg of apoCII, 20% BSA and triolein emulsified with various phospholipids or Triton X-100. The values represent means ± standard deviations (n = 3). The statistical differences were analysed by Student’s t-test. P < 0.05.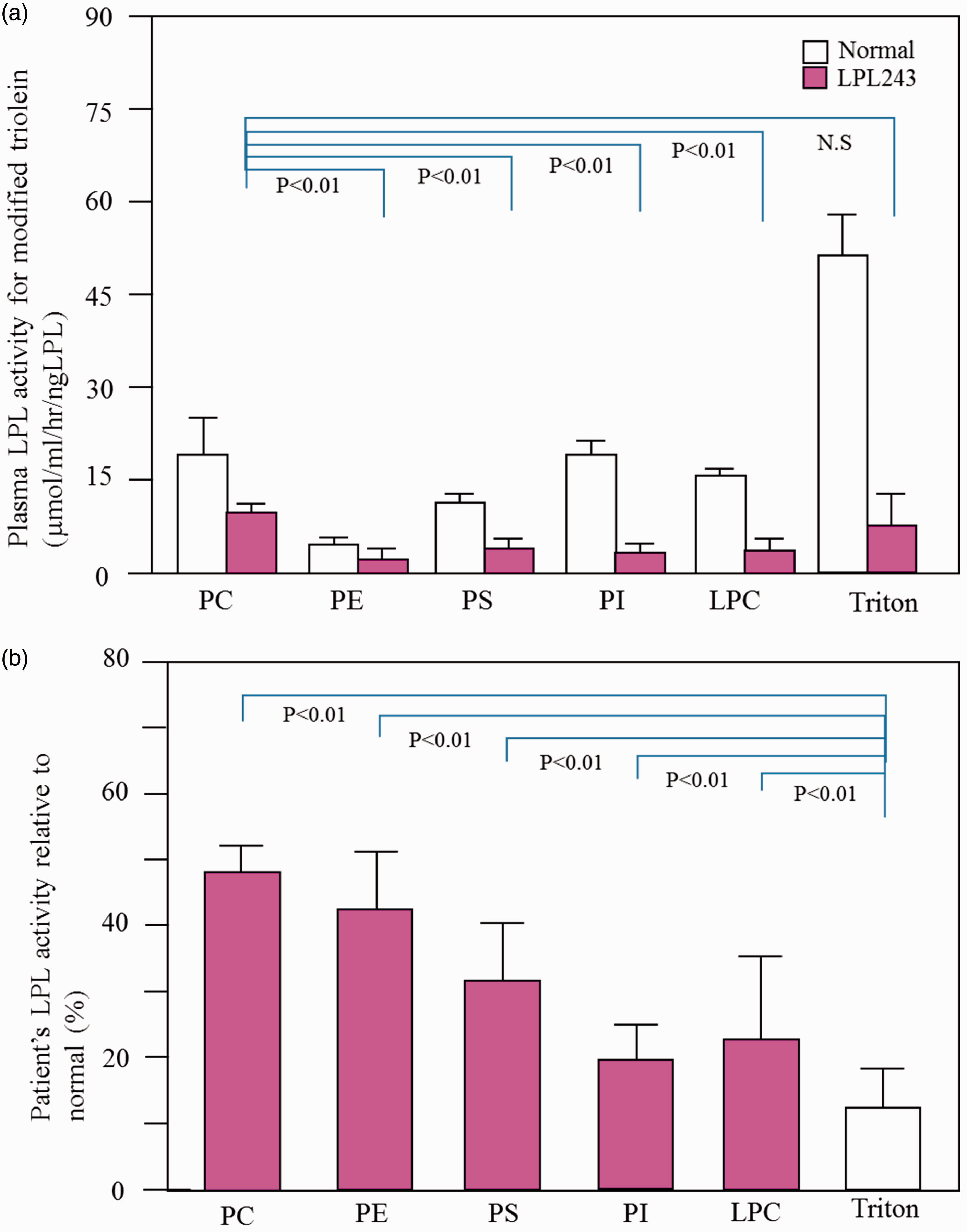
Discussion
Hyperchylomicronaemia in patients with LPL Arg243His has been reported in Japanese and Caucasians.4,5 The patients’ plasma LPL activities have been measured against glycerol tri[14C]-oleate after heparin injection as well as from the media of the cultured cells transfected with mutant cDNA, and the activities were shown to be severely decreased, despite almost normal protein concentrations.4,5 Here, we reveal that the activities of LPL Arg243His for triolein emulsified with PC were increased when compared with those emulsified with PE, PS, PI or LPC, and notably, the mutant’s activities relative to those of normal plasma measured with PLs as emulsifiers were all significantly increased when compared with the relative activity measured with Triton X-100. The hydrolysing activity of LPL Arg243His revealed different properties not only from that of normal LPL but also from LPL Asp204Glu (see Figure 1(b) and also Murano et al. 2 ).
By analogy with the crystallographic structure of human pancreatic lipase, Arg243 of LPL is predicted to be near the proposed His241 of the catalytic triad, and the substitution has been suggested to be significant for the catalytic activity. Furthermore, the 22 amino acids long loop (‘Lid’) spanning from residues 245 to 265 has been proposed to cover the catalytic site of LPL and to be critical for the recognition of substrate surface. Thus, although the Arg243His substitution constitutes a subtle change with a single methylene addition, the mutation may also disturb the recognition function of the Lid located near the affected amino acid. In this context, the disturbed hydrolytic activity, particularly against triolein emulsified with PC, associated with the mutation near the Lid region seems to be mechanistically different from that by the Asp204Glu mutation, which may impair the function of the surface recognition site, but not of the catalytic site of the enzyme. 2 The present study suggests that the analyses of hydrolysing activities of LPLs with different mutation sites against trioleins emulsified with various phospholipids may contribute to the understanding of the complex surface recognition and the subsequent catalytic process of the compromised enzyme.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported, in part, by Japan Health and Labour Sciences Research grant for primary hyperlipidaemia to HB.
Ethical approval
The ethics committee of Toho University Sakura Medical Center (No. 2014-071).
Guarantor
HB.
Contributorship
TY, TM and HB performed all data analysis, and TM, WJS and HB wrote the first draft. TS, SS and HK have done a study on a patient as reported in literature, 3 and processed the samples. All authors reviewed the manuscript and approved the final version of the manuscript.
