Abstract
Background
Birth weight influences profiles of dried blood amino-acids and acylcarnitines in newborn screening. This study aimed to define a more appropriate cut-off value to reduce the false positive rate and the number of recalled patients in newborn screening.
Methods
All babies who underwent newborn screening in our center were included; they were divided into groups by birth weight: 2500–3999 g (comparator group), <1000 g (group 1), 1000–1499 g (group 2), 1500–2499 g (group 3), and >4000 g (group 4). The 0.5th and 99.5th percentiles were used as the cut-off values. Comparisons were done on amino acid and acylcarnitines concentrations between the groups. False positive rate, positive predictive value, corrected false positive rate by birth weights were determined.
Results
Data on a total of 578,287 newborn infants were included in the analysis. The total false positive rate was 0.75%, and positive predictive value 2.89%. The false positive rate was 0.69%, 0.54% and 5.31% in infants with normal birth weight, birth weight of >4000 (group 4) and low birth weight of < 2500 g (groups 1, 2 and 3), respectively. Low-birth weight infants had much higher phenylalanine, tyrosine, methionine, arginine, propionylcarnitine, isovalerylcarnitine and octadecanoylcarnitine concentrations. Free carnitines and palmitoylcarnitine concentrations were lower. After adjusting for birth weight, false positive rate of all indices decreased to 0.53%, and positive predictive value increased to 4.31%.
Conclusions
Amino acid and carnitine concentrations in low-birth weight newborn infants may differ from the normal term newborn infants. The cut-off values of individual metabolites should be adjusted based on birth weight, to reduce false positive rate and increase positive predictive value.
Introduction
With the development of tandem mass spectrometry (MS/MS), more inborn errors of metabolism have been introduced in our newborn screening program. Many previous studies have revealed distinct metabolic profiles in preterm infants.1–6 Gestational age (GA) and birth weight may affect metabolic patterns in preterm infants; thus, establishing cut-off values for metabolites in these infants is important for timely diagnosis and an optimal treatment strategy for patients with inborn errors of metabolism.7–13
Gucciardi et al. 14 found that acylcarnitine concentrations are lower in preterm newborn than in term newborn infants. They established GA-based reference ranges for preterm newborns. Matern et al. 2 and Clark et al. 7 developed cut-offs for amino acids and acylcarnitines in preterm newborns. Up to now, few laboratories have established reference value of amino acids and acylcarnitines for preterm infants. However, this is important as the use of more appropriate cut-offs can avoid false positive or negative results in the MS/MS screening of preterm newborns.
Zhejiang Province, China has approximately 600,000 live births per year. Accordingly the Newborn Screening Centre of Zhejiang Province receives 500,000 to 600,000 blood spot samples per year, of which 10% are from preterm infants. In this study, we aimed to define cut-off values adjusted by birth weight, to improve the false positive and recall rates
Methods
Study subjects and data collection
Written parental consent was obtained from the parents or guardians before sampling for newborn infant screening. This study was approved by Institutional Review Board, Children’s Hospital, Zhejiang University School of Medicine (Rf. 2013-112).
All data were collected before November, 2013. Clinical records with information including gender, reported race, GA, birth weight, birth date and sampling date were checked via the newborn screening database. Dried blood spots (DBS) were taken in all infants at 72 h after birth. In our laboratory, all DBS were analysed by using MS/MS with NeoGram Amino Acid and Acylcarnitines Kits (PerkinElmer, Finland). Acylcarnitines in DBS and plasma were measured using an UPLC-ESI-MS/MS method described previously. 4 The reference values were defined by determining 10,000 normal term and appropriate for GA newborns as per our previous study 13 ; we excluded those premature newborns who were on total parenteral nutrition (TPN). If the results fell outside of the cut-off value, repeat analysis was performed; if the re-analysed results remained abnormal, the infant was recalled for further assessment.
Statistical analysis
All data were analysed with SPSS for Windows software version 13.0
Results
The sample comprised a total of 578,287 newborn infants. For analysis, all the included newborns were divided into five groups by birth weight: 2500–3999 g as the comparator group (
Comparison of results of amino acid profiles between the groups
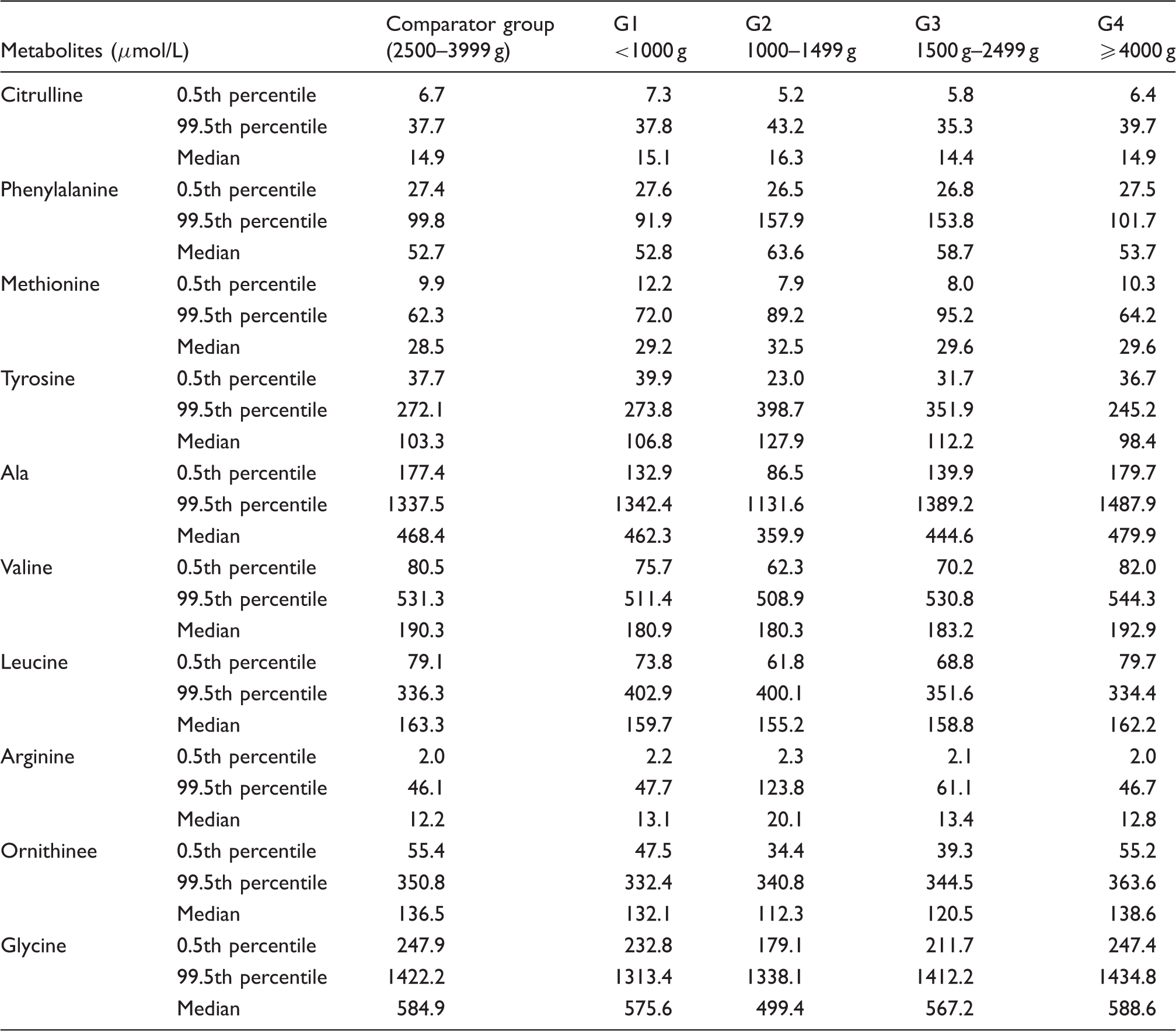
Percentiles and median values for amino acids profiles in infants of different birth weights.
Comparison of acylcarnitine concentrations between the groups
Free carnitine and short chain acylcarnitine
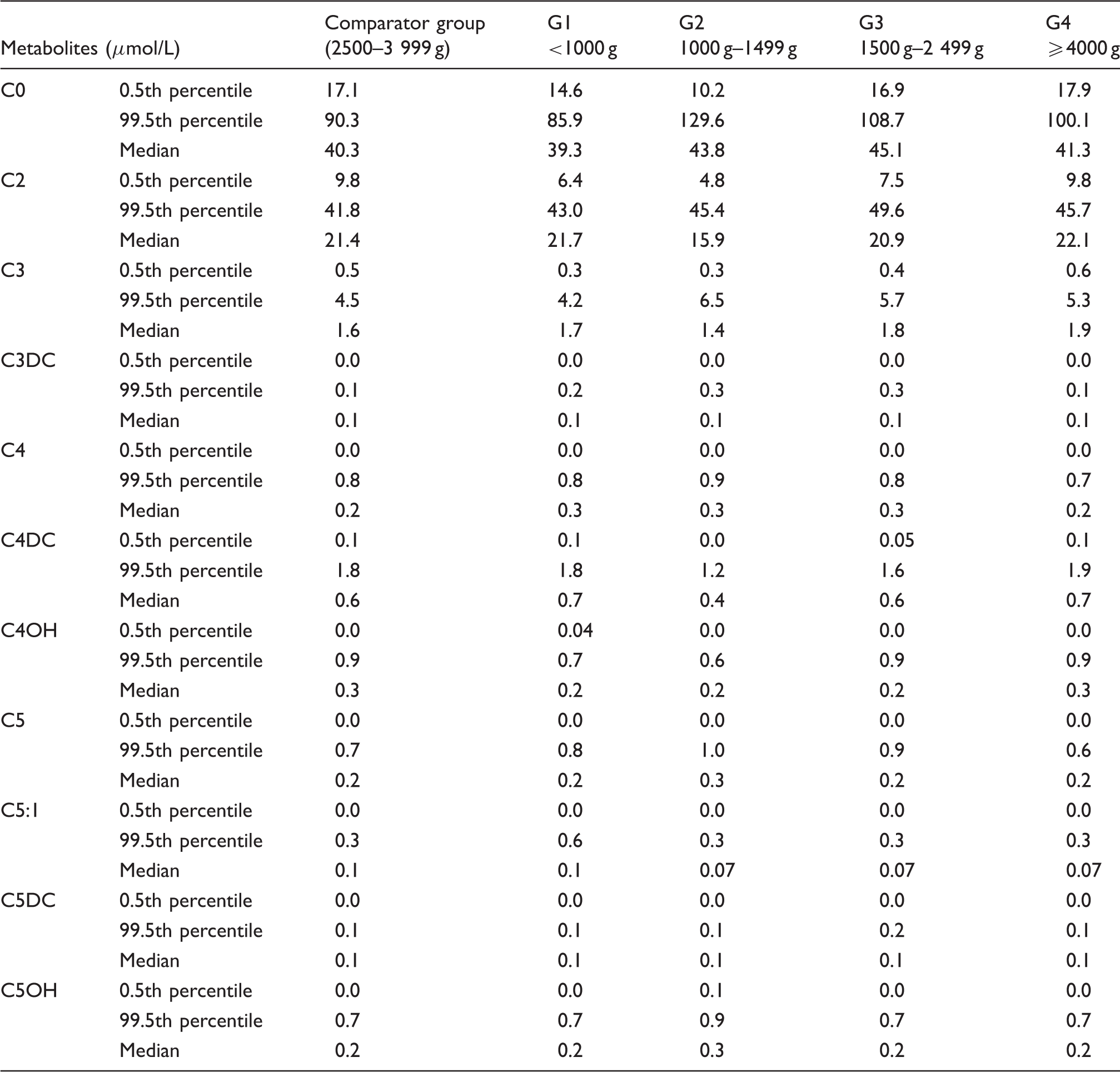
Percentiles and median values for free carnitine and short-chain acylcarnitines in infants of different birth weights.
Medium-chain acylcarnitine
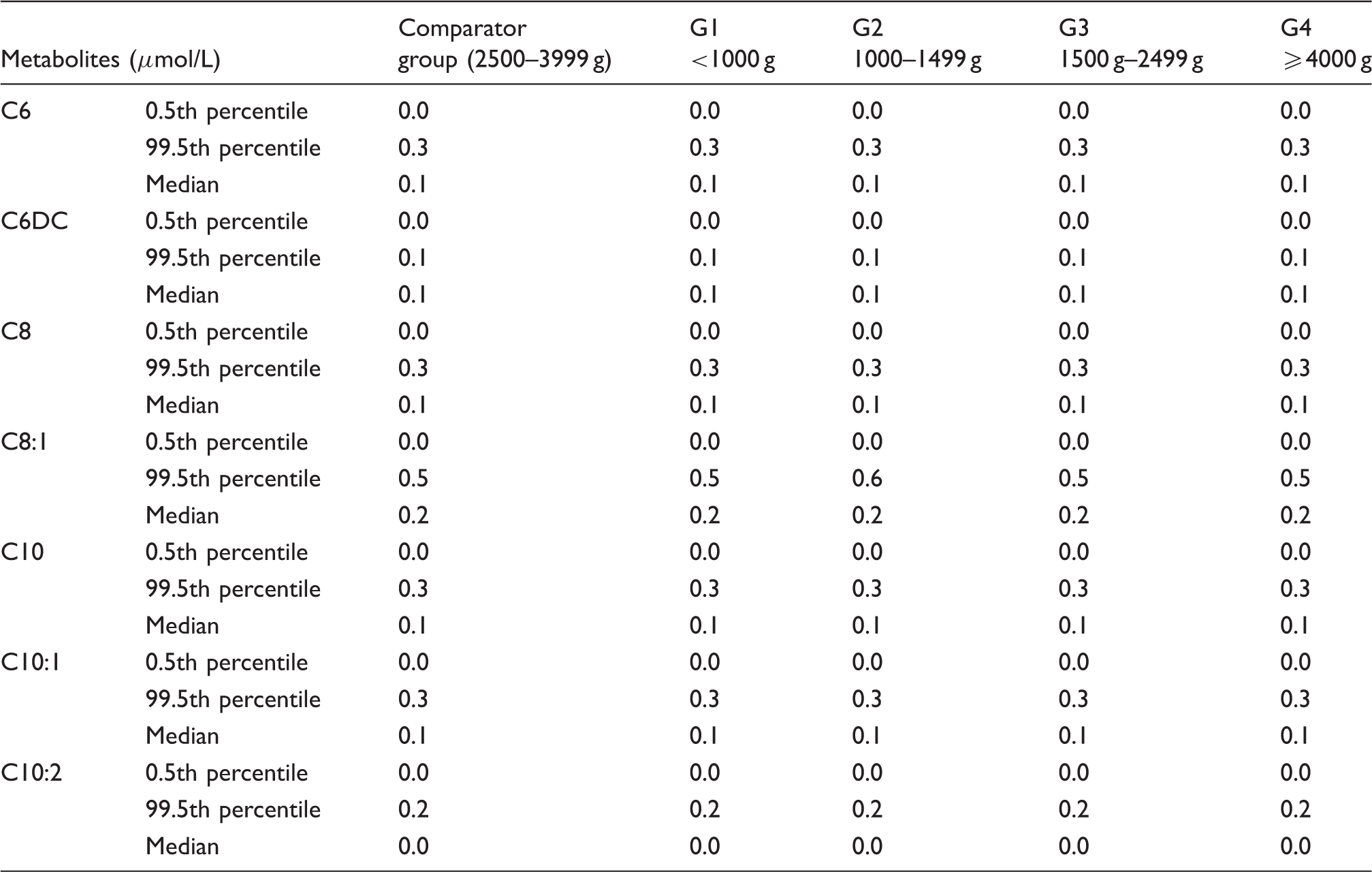
Percentiles and median values medium-chain acylcarnitine profiles of different birth weights.
Long-chain acylcarnitine
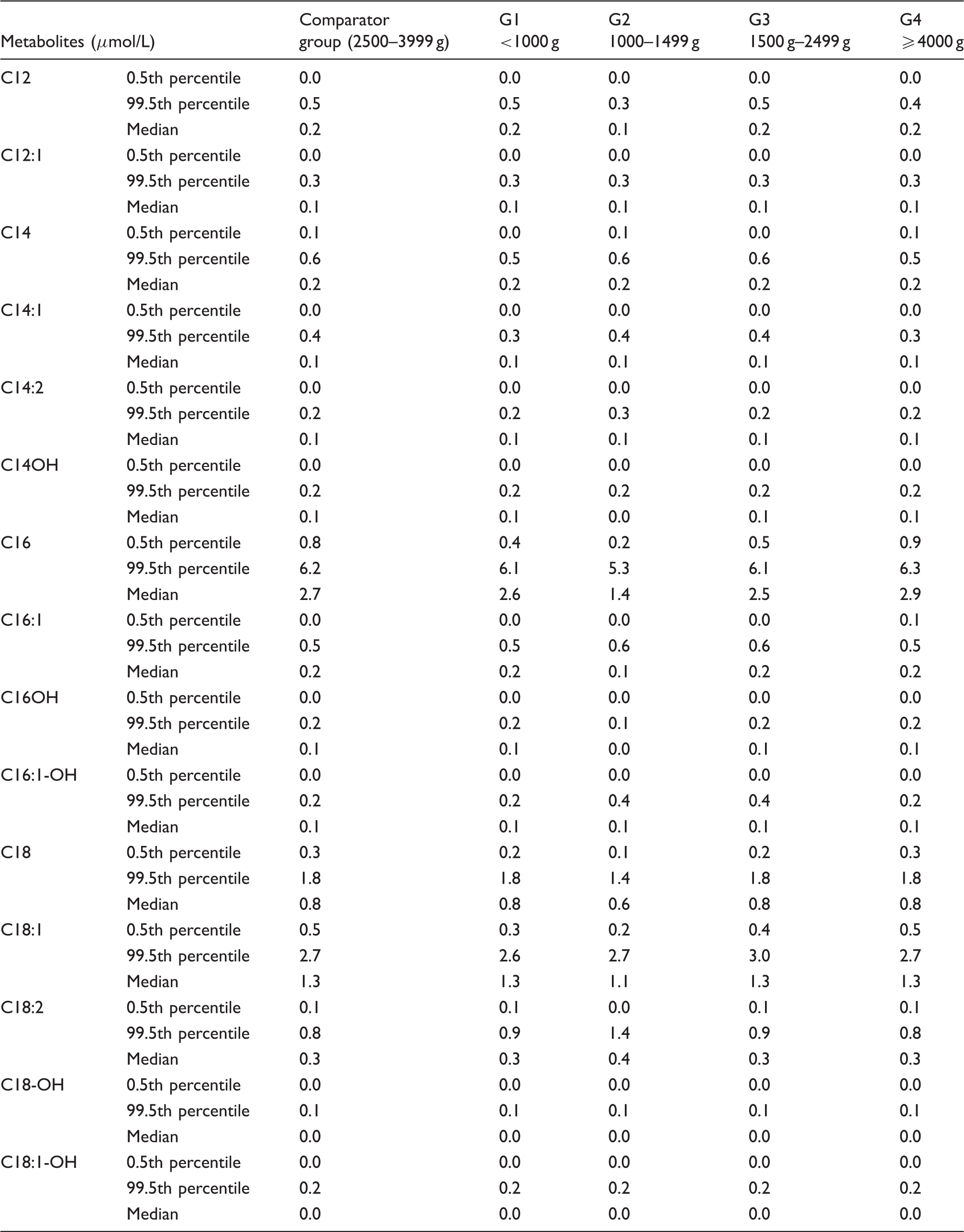
Percentiles and median values for long-chain acylcarnitine profiles of different birth weights.
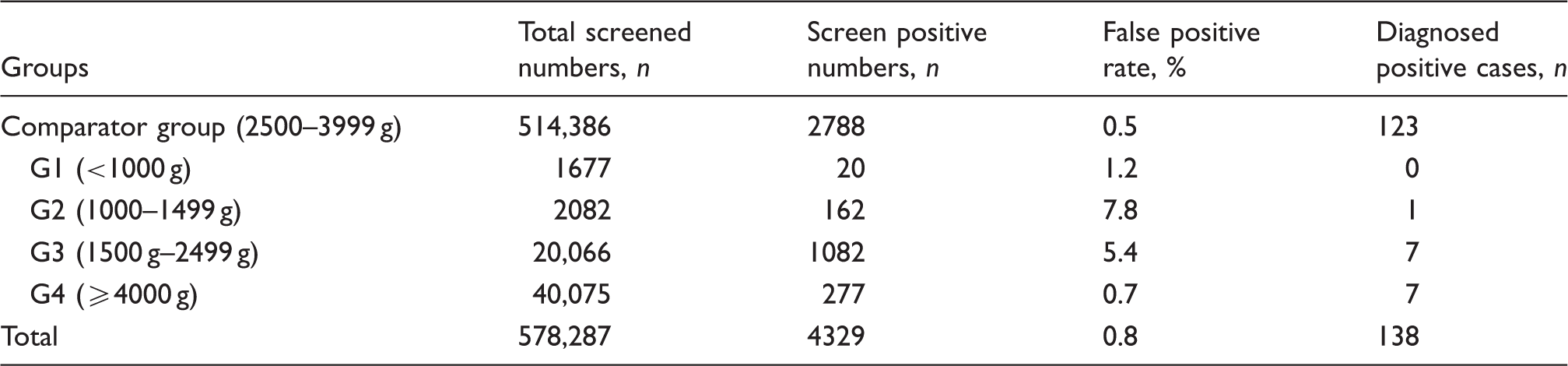
False positive results
Screened infants number, false positive rates and diagnosed cases in different birth weight groups.
Discussion
Our study provides a birth weight-corrected amino acid and acylcarnitine concentrations in newborns. Most low-birth-weight cases are premature infants who have different metabolic profiles from term infants; our study showed that metabolites were significantly different across the groups categorized by birth weight.
Amino acid metabolic disorders
The median value and 99.5th percentiles of four amino acids including phenylalanine, methionine, arginine and tyrosine were higher in the low birth weight groups than those of 2500–3999 g control birth weight group. The results were similar to previous reports. Mandour et al. 15 found all amino acid concentrations except valine and proline were much higher in the very low and low birth weight infants than those in the normal birth weight infants. Wilson et al. 16 found that five amino acids (arginine, leucine, ornithine, phenylalanine and valine) were higher in the preterm infants than in the term infants; due to the high 99.5th percentiles of these four amino acids, the total FPR for the four indices was 27.21%; however, based on the corrected cut-off value by birth weight, the percentage reduced to 20.22%. Because of delayed maturation of hepatic function, premature infants had metabolic alterations which may cause significantly elevated phenylalanine, tyrosine and methionine concentrations.15,17,19 Leucine/isoleucine and valine were also elevated in premature infants, possibly reflecting increased dependence on the substrates for gluconeogenesis and ketogenesis. 19 Amino acid supplementation may distort the blood amino acid profile; very low birth weight infants may receive high concentrations of amino acids from TPN administration in conjunction with immature metabolism, 20 combined low amino acids absorptivity of the renal tubules of premature infants.19,20 Only glycine concentration in the low birth weight newborns was slightly lower than that of the normal birth weight newborns in this study. The possible reason maybe that the demand for glycine is high in preterm infants with critical illness or when given supplemental oxygen. 17
Organic acid metabolic disorders
Our study found that the 99.5th percentiles of C3, C3DC, C5 and C5OH in low birth weight infants were higher than normal birth weight infants; these indices were more likely to generate false positive results. Most of them occur in premature and low birth weight newborns. Based on the corrected cut-off value by birth weight, the percentage reduced to 16.94%. Votava et al. 9 also found that FPRs of C3, C4, C5 and C5OH in the low birth weight infants were significantly higher than in the normal birth weight infants; this condition may be related to nutritional state and reduced activity of corresponding metabolic enzymes. Gucciardi et al. 14 found that short- and medium-chain acylcarnitine esters (C4, C5, C6 and C8) concentrations of venous and arterial umbilical blood were increased in very low birth weight newborns. Most organic acid metabolic disorders were caused by branched-chain amino acid metabolic disorders. An excess of branched-chain amino acid supplementation may induce abnormal accumulation of organic amino acids.22,23 Tonazzi A et al. 23 suggested that short- and medium-chain acylcarnitine esters concentrations were increased in very premature infants due to an increased transplacental transfer from the maternal circulation.
Fatty acid metabolic disorders
Carnitine exists in two forms: free carnitine and acylcarnitine. Endogenous carnitine synthesis is limited in the newborns due to low concentrations of γ-butyrobetaine hydroxylase, which is the final step of enzyme catalyzing in carnitine synthesis (from γ-butyrobetaine to carnitine). 24 Studies also found that due to decreased tissue storage and the placental carnitine transfer occurring during the third trimester, preterm newborns are particularly at risk of carnitine deficiency.18,25 Our results found that the 0.5th percentile of free carnitine (C0) in the low birth weight infants was lower than that in the normal birth weight infants, and that the 99.5th percentile of C0 was higher in low birth weight infants. The results were consistent with the findings of Mandour et al. 15 The low C0 in the premature and low birth weight infants resulted in an increased FPR for primary carnitine deficiency in screening. The proportion of recalled infants due to false positive C0 result accounted for 11.56%, but after correction for birth weight reduced to 8.03%. However, several studies have found that premature infants had higher C0 concentration.21,25
After birth, nutrients are absorbed through the gastrointestinal tract. We found no changes in the concentrations of medium-chain acyl carnitines which was inconsistent with findings of Meyburg and Mandour that C6, C8, C10:1 and C18:1-OH concentrations were increased.6,14 Medium-chain triglycerides (MCT) digestion depends on salivary and intestinal lipase with no requirement for bile salts. Long-chain triglyceride (LCT) digestion depends on pancreatic lipase and bile salt chylomicrons; due to the low activity of pancreatic lipase in conjunction with relatively low concentrations of cholic acid and bile salt concentrations, premature infants have limited ability to absorb and digest fat. Our results found that 0.5th percentiles of C16, C16:1, C18 and C18:1 increased with birth weight, which increased the FPR of long-chain fatty acid metabolic disorder; after correction, the FPR reduced from 3.27% to 2.7%. The carnitine palmitoyltransferase system is an essential step in the beta-oxidation of long-chain fatty acids. Carnitine palmitoyltransferase I is a mitochondrial enzyme responsible for the formation of acylcarnitines by catalyzing carnitine and the acyl group of a long-chain fatty acyl-CoA. Therefore, insufficient carnitine palmitoyltransferase I and reduced free carnitine in preterm infants may affect long-chain acylcarnitine concentrations.
Our study found that the FPR and PPV of the low-birth weight groups were higher than those of the normal control group. The number of positive cases and FPR were reduced, and PPV increased significantly by using the adjusted cutoff value of indices by birth weight. We suggest that it is important for each newborn screening laboratory to define corrected cut-off value for preterm newborns.
The limitation of this study was that we defined the corrected cut-off value based only on birth weight. The optimal cut-off value may require a more complex model based on birth weight, GA, and other variables. Further research is needed to establish a model based on multi-variables for premature infants.
In conclusion, amino acid and carnitine concentrations were correlated with birth weight. The adjusted cut-off values by birth weight could be used to increase the specificity of screening and reduce FPRs of diagnosing metabolic disorders in our newborn screening programme.
Footnotes
Acknowledgements
We would like to thank Hua-Qing Mao, Yan Yao and Cheng-Gang Wang for their assistance and guidance in this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Parental consent was obtained for this study. This study was approved by Institutional Review Board, Children’s Hospital, Zhejiang University School of Medicine (Rf. 2013-112).
Guarantor
WXW.
Contributorship
YLL and HXW researched literature and conceived the study. ZY and YJB were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. YLL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
