Abstract
Background
Monitoring of treatment for patients diagnosed with congenital adrenal hyperplasia (CAH) can be performed by measuring the concentration of 17α-hydroxyprogesterone (17OHP) in bloodspots collected on filter papers. A method is described here for measuring 17OHP by liquid chromatography tandem mass spectrometry (LCMSMS).
Methods
17OHP was extracted by liquid–liquid extraction and analysed by LCMSMS. The method was validated for sensitivity, specificity, linearity, recovery, ion suppression, precision and bias.
Results
The standard curve was linear from 0 to 400 nmol/L. Intra-assay %CVs were <10 and inter-assay %CVs were <15 over the range 10–200 nmol/L. Limit of quantitation was 6 nmol/L. No ion suppression was detected. The only interfering compound detected was deoxycorticosterone, an intermediate steroid with the same molecular weight as 17α-hydroxyprogesterone. The method was more accurate and precise than an existing radioimmunoassay. There was poor correlation between the two assays.
Conclusions
We have developed a sensitive and specific assay suitable for quantitation of 17OHP in bloodspots. This method performs better than radioimmunoassay and allows smaller samples to be used.
Introduction
17α-hydroxyprogesterone (17OHP) is a progestogen produced in the zona fasiculata of the adrenal glands. It is an intermediate in the steroid metabolic pathway, responsible for the production of glucocorticoids and sex steroids. 1 Measurement of 17OHP is used primarily for diagnosis but also to monitor patients with congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency. 2 Patients diagnosed with CAH can be followed up by measuring 17OHP on filter paper bloodspots. Bloodspots are a very convenient means of monitoring CAH because they allow patients to take samples at home and post them into the laboratory rather than coming into hospital for a whole day to have samples taken at 4-h intervals.
In recent years, steroid analysis has increasingly moved away from immunoassay methods to liquid chromatography tandem mass spectrometry (LCMSMS).3–6 This move has come from increasingly sensitive and affordable tandem mass spectrometers and also from recognition of the poor performance of many immunoassays in measuring steroid hormones.7,8 A desire to replace radioimmunoassays with assays that do not use radioactive material is also a driving factor in laboratories setting up new LCMSMS assays.
While LCMSMS methods for serum steroids have become common, few laboratories use it for bloodspot assays, as in the UK newborn screening for CAH is not undertaken. We therefore set out to develop and validate an LCMSMS method for bloodspot 17OHP to replace the existing radioimmunoassay. 9
Material and methods
Materials
17OHP was purchased from Sigma-Aldrich (Poole, UK) and 17OHP-d8 from CDN isotopes Inc (Quebec, Canada). Tuning solutions of both were made by preparing 1.5
Calibrators and additional material for method validation purposes were made by creating a 400 nmol/L 17OHP whole blood sample. Four hundred microliters of a 10
Working internal standard solution (17OHP-d8) was a 20 nmol/L solution in methanol.
For the radioimmunoassay method (RIA), a Tris buffer was made containing 50 mmol/L trizma base (Sigma-Aldrich), with 0.5% bovine serum albumin and 15 mmol/L sodium azide set to pH 7.5 using hydrochloric acid (Sigma-Aldrich). Antisera for 17OHP was purchased from Guildhay Antisera (Guildford, UK). The tracer, 125I-17OHP was made in-house using 17OHP from Sigma-Aldrich and 125I from MP Biomedicals (California, USA). Sac-cel donkey antisheep/goat solid phase antibody-coated cellulose suspension was from IDSplc (Boldon, UK). Methylated spirit was from Sigma-Aldrich and dry ice from Yara Industrial (Heathrow, UK).
Sample preparation
Bloodspot samples were extracted based upon the method described by Lacey et al.
10
Briefly, bloodspots were punched out using a 5 mm hole punch into 5 mL glass tubes. Deionised water (450
Analysis
Analysis of samples was performed on a Waters Acquity UPLC and Waters TQD tandem mass spectrometer with a Waters BEH C18 column (1.7
The tandem mass spectrometer was operated in multiple reaction monitoring (MRM) mode with the following mass transitions: 17OHP quantifier ion 331.0 >97.2, 17OHP qualifier ion 331.0 >109.1 and 17OHP-d8 339.1 >100.4. The instrument was operated in electrospray positive ionisation mode.
Radioimmunoassay method
The LCMSMS assay was compared against the existing RIA method. For the RIA method, calibrators, QCs and patients’ samples were punched out into 5 mL glass tubes. Five hundred microliters of Tris buffer was added and the samples shaken in a waterbath at 37℃ for 30 min. Three millilitres of diethyl ether were added to the tubes and shaken vigorously for 4 min, followed by centrifugation at 3000
Validation
Method validation followed guidelines set out by John Honour, 2011. 11 All concentrations calculated were based on area under the curve.
Precision and bias
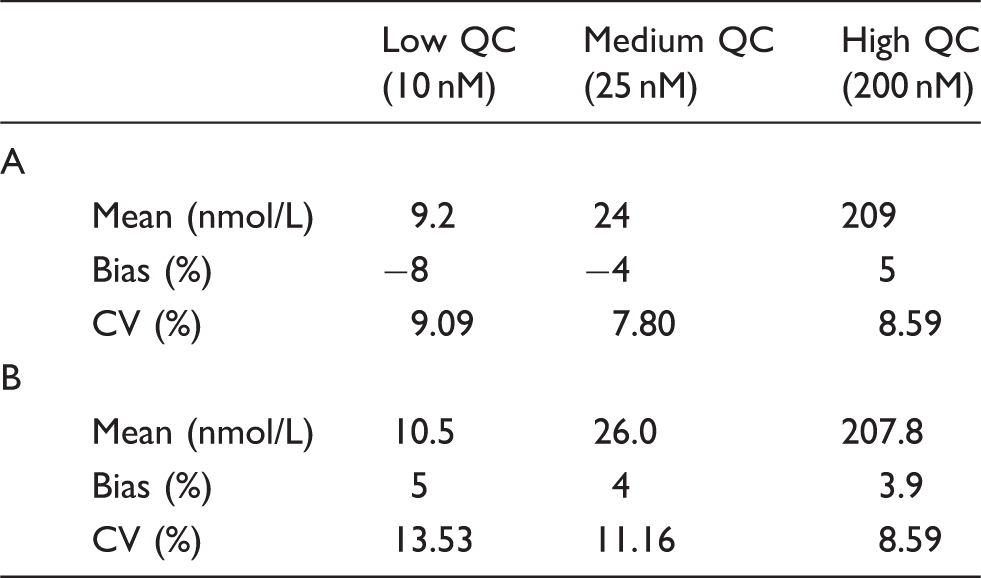
These were assessed by measuring three samples (10, 25 and 200 nmol/L) 10 times each in a random order for intra-assay precision and bias. Inter assay precision and bias was assessed by measuring the three samples five times each on three further days. Acceptance criteria were %CV <10 for intra assay and <15 for inter assay, and for bias by ensuring mean results were within 10% of target values.
Carry over
By measuring the 10, 25 and 200 nmol/L samples for precision and bias in a random order, carry over was also assessed. If carry over was present, then a 10 nmol/L sample analysed following a 200 nmol/L sample would be measured with concentration greater than 10 nmol/L. The assay would be free of carry over if all three sample concentrations were repeatedly measured with a consistent concentration no matter what samples preceded them.
Functional sensitivity
This was determined by double dilution of samples from 12.5 to 3.1 nmol/L. The functional sensitivity was the lowest concentration which had %CV <15 and bias within 10% of target value.
Specificity
Steroids used to determine the specificity of the 17OHP bloodspot assay.
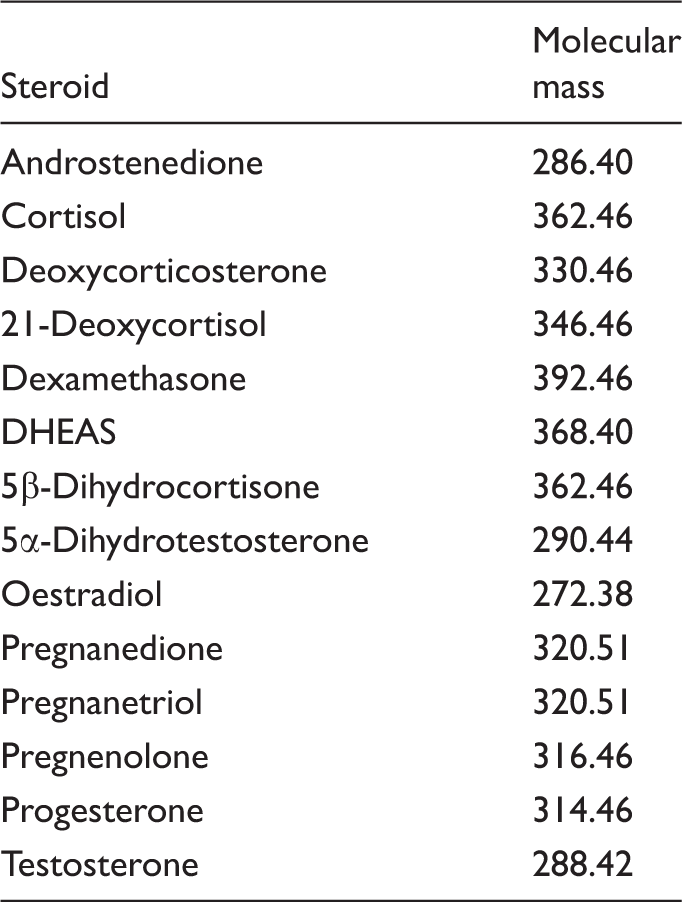
Ion suppression was also assessed to identify whether any compounds interfered at the point of 17OHP elution. This was determined by infusing 17OHP directly into the mass spectrometer while running a blank sample. A change in peak response at the point of 17OHP elution of >10% in either direction would indicate ion suppression or enhancement.
Recovery
This was assessed by analysing samples of known concentration and then re-analysing them after they had been spiked with a known amount of 17OHP. The recovery should be 90–110% and was determined by equation (1)
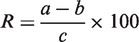
A 25 nmol/L sample was spiked with a further 25 nmol/L 17OHP, 50 nmol/L was spiked to 150 nmol/L, 100 nmol/L was spiked to 150 nmol/L and 200 nmol/L sample was spiked to 300 nmol/L. The 25, 50, 100 and 200 nmol/L blood samples were made as described in the Methods section. Before spotting out the samples, half was aliquoted into a fresh tube and spiked with more 17OHP to make the concentrations described above. All bloodspots were therefore treated the same, going through the full extraction procedure.
Method comparison
The LCMSMS assay was compared with the existing radioimmunoassay. Thirty patients’ samples which had previously been analysed for bloodspot 17OHP by radioimmunoassay were re-analysed by LCMSMS. Calibrators used for the assays were different lots, but made using the same protocol. The number of samples that could be used was low because of the small number of requests for bloodspot 17OHP and because many of the samples were too small to measure by both methods. Results were compared using a Bland–Altman plot. All samples were from CAH patients who were having their treatment monitored.
Results
Extraction of the samples produced clear extracts and chromatography was excellent with no peaks detected besides 17OHP. 17OHP and 17OHP-d8 both elute at 1.2 min (Figure 1). The quantifier:qualifier ion ratio was 1.2 with a tolerance limit set of ±20%. Any sample which breached this limit was rejected from the analysis, but this was a rare occurrence, normally due to very low concentration samples.
Chromatography showed good 17OHP peaks with no other peaks detected. These were taken at a concentration of 200 nmol/L.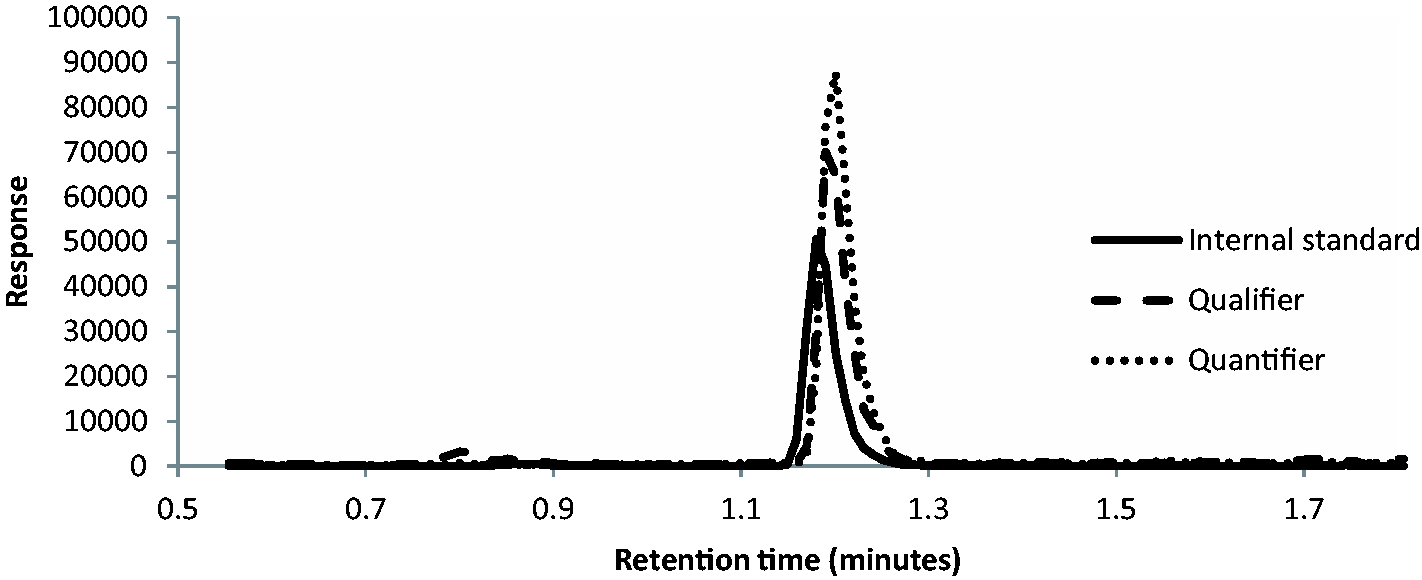
Calibration curves were linear from 0 to 400 nmol/L (Figure 2) with r2 of >0.99. Intra-assay %CV was <10 at all concentrations and measured values were within 10% of target values at all concentrations. Inter-assay performance was also good with measured values within 5% of target values at all concentrations and %CV <15 at all concentrations (Table 2). Functional sensitivity of the assay was 6 nmol/L. No carry over was detected.
Calibrators from 0 to 400 nM show good linearity. A shows the intra assay data, B shows the inter assay data.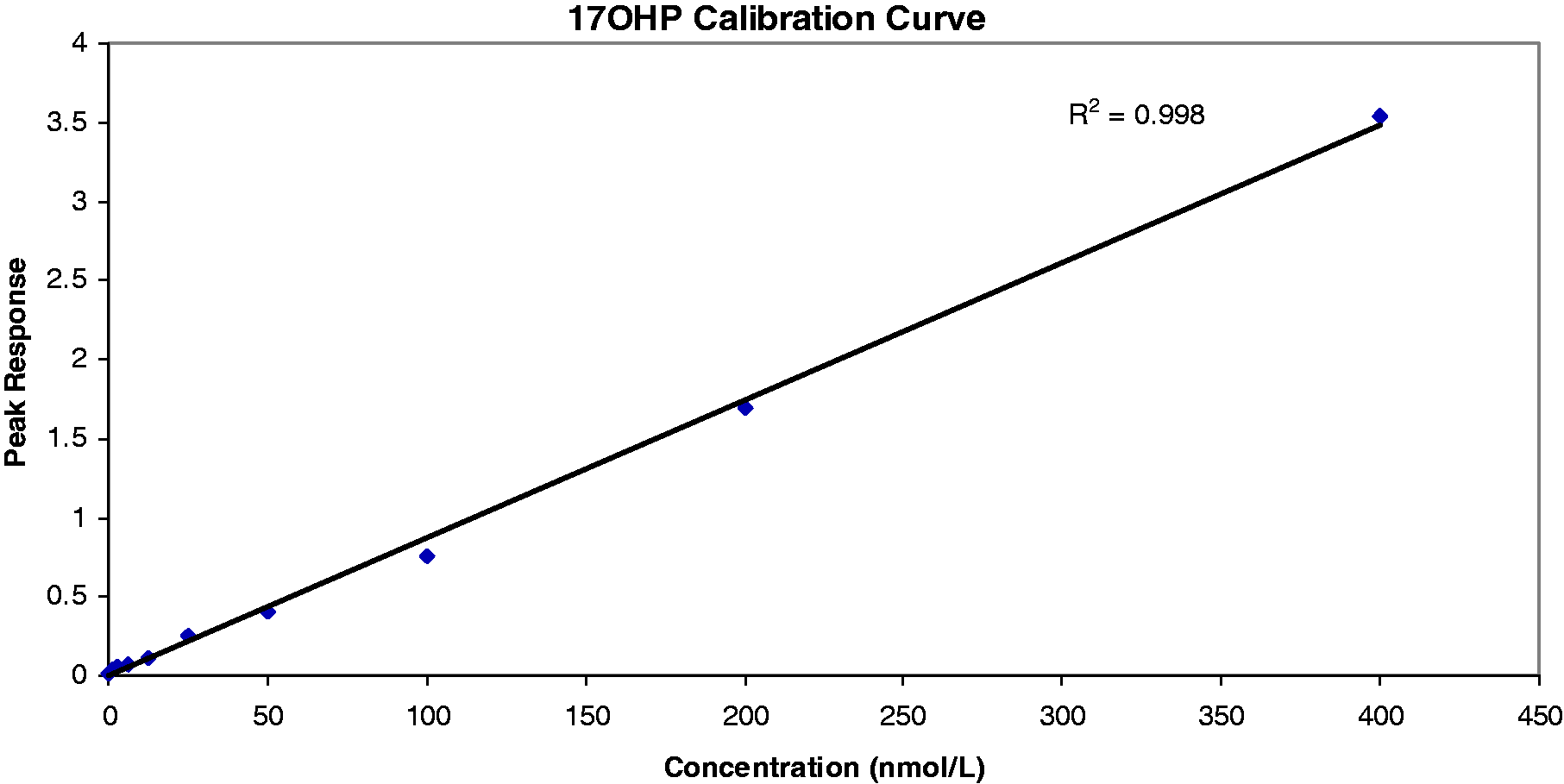
Ion suppression studies showed no interference at the point of 17OHP elution (Figure 3).
The dashed line at 1.21 min shows the point at which 17OHP elutes. At this point, the peak response is steady showing no ion suppression.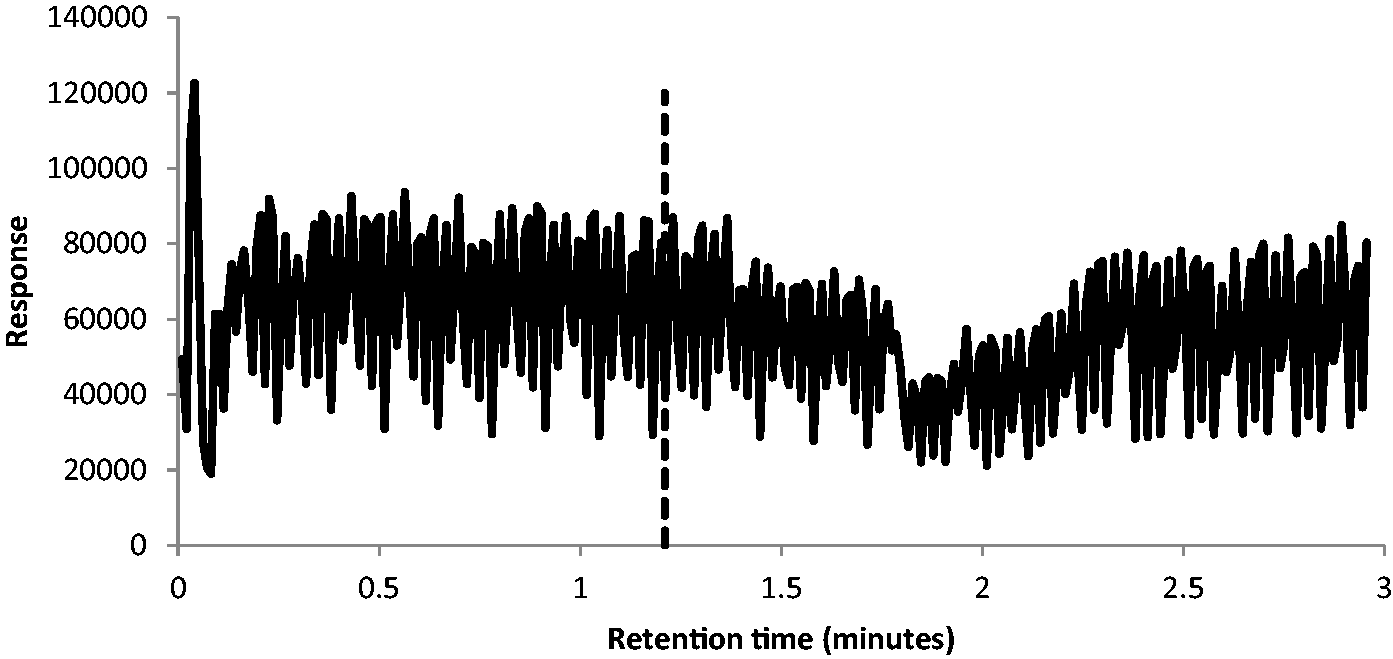
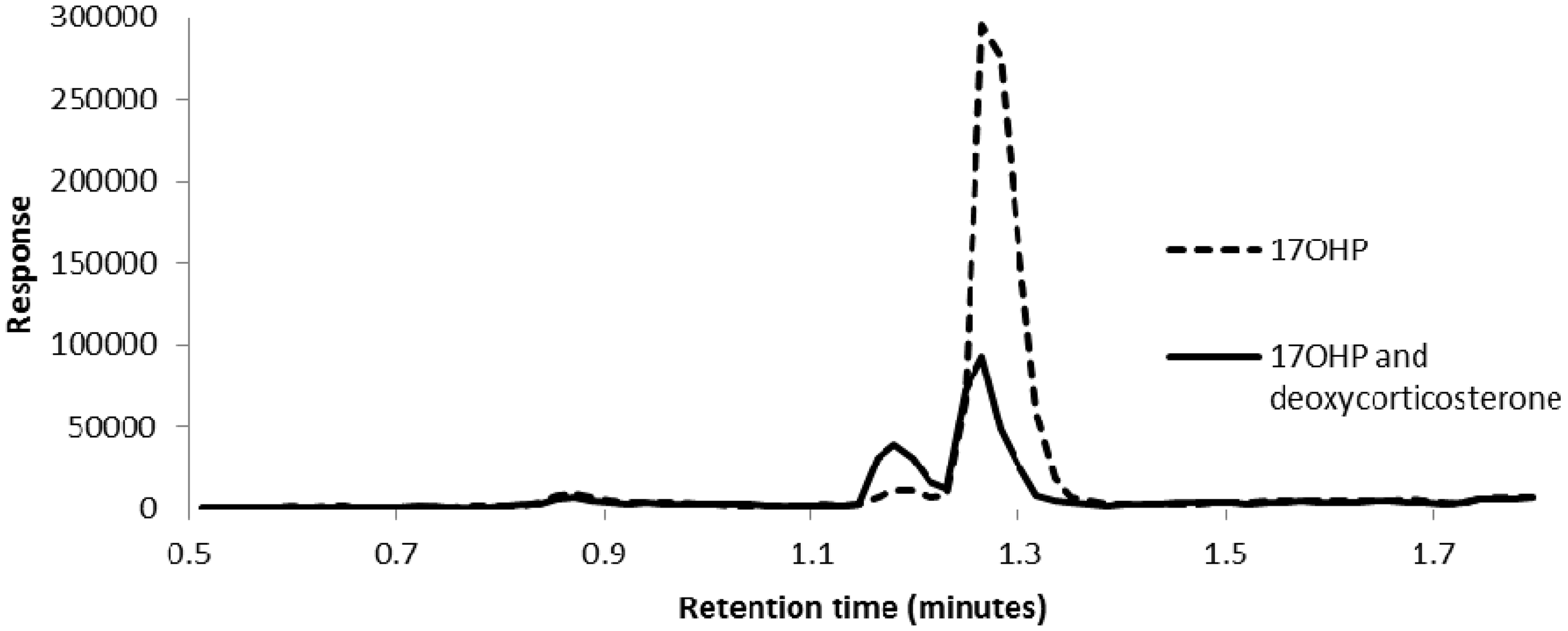
Quantitation of samples with and without spikes of other steroids showed no difference in chromatography or concentration measured (data not shown). Deoxycorticosterone was investigated separately because it had the same molecular mass as 17OHP and tuning the molecule showed it had identical parent and daughter ions (Figure 4). Chromatography showed that deoxycorticosterone and 17OHP are only partially resolved, such that 17OHP could not be reliably quantified when deoxycorticosterone was present (Figure 5).
Tuning of 17OHP and deoxycorticosterone showed almost identical mass profiles for both molecules. The parent ions (a – 17OHP; c – deoxycorticosterone) show the primary ion at m/z 331 and two smaller parent ions at 332 and 333, which indicate the presence of naturally occurring carbon 13 (either one or two isotopes). The daughter ion scans are also almost identical (b – 17OHP; d – deoxycorticosterone). 17OHP has eluted at 1.27 min (dashed line). The solid line shows that 17OHP (25 nmol/L) and deoxycorticosterone (10 nmol/L) at 1.18 min are not base line separated.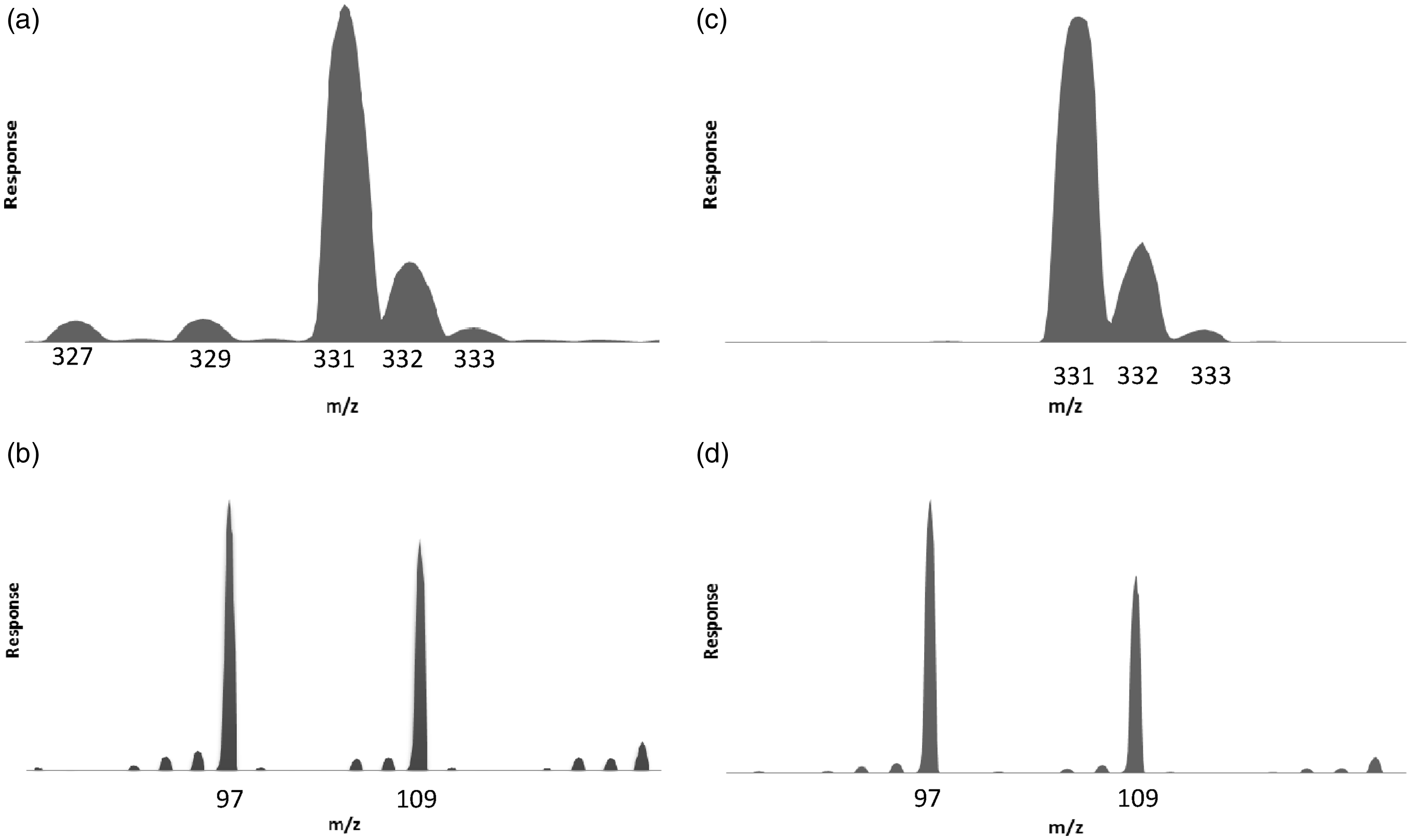
Recovery experiments.
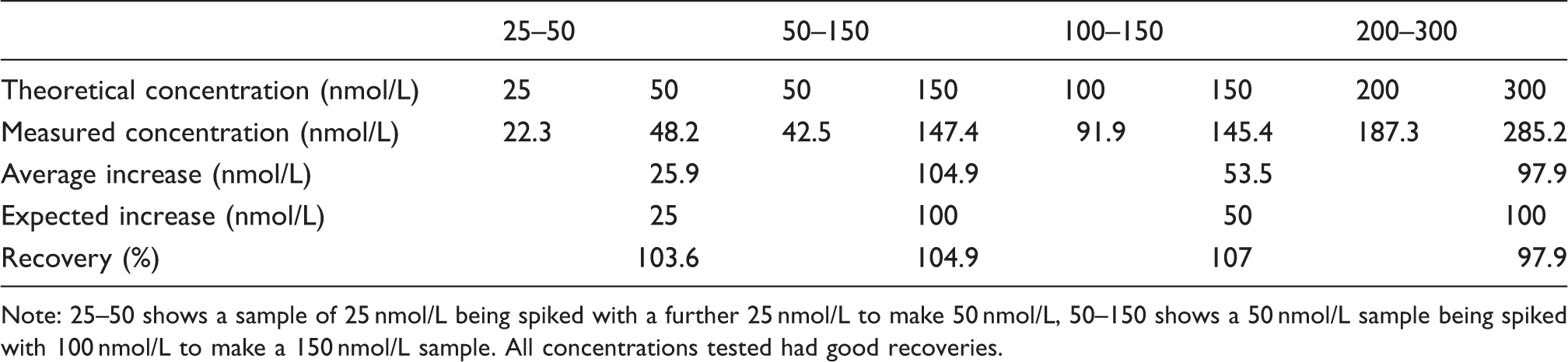
Note: 25–50 shows a sample of 25 nmol/L being spiked with a further 25 nmol/L to make 50 nmol/L, 50–150 shows a 50 nmol/L sample being spiked with 100 nmol/L to make a 150 nmol/L sample. All concentrations tested had good recoveries.
There was very poor correlation between the radioimmunoassay method and LCMSMS (Figure 6). LCMSMS results were on average 18% lower than radioimmunoassay; however, the difference between results covered a very large range from LCMSMS being 67% lower to 22% higher than radioimmunoassay. Of the 30 samples analysed, 11 had an undetectable concentration of 17OHP by both methods and 1 had >400 nmol/L by immunoassay and 340 nmol/L by LCMSMS. These results are not shown in Figure 6.
Bland–Altman plot comparing radioimmunoassay with the LCMSMS method.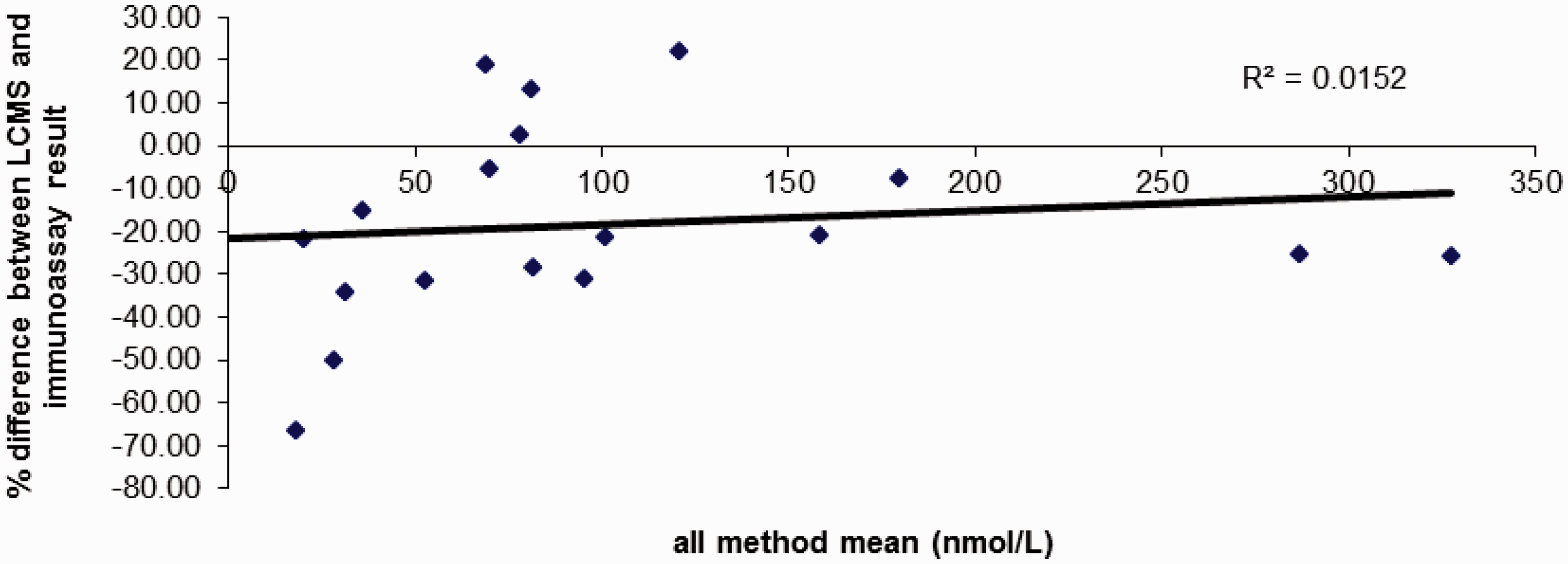
Discussion
The development of this 17OHP bloodspot method was based upon the work published by Lacey et al. 10 17OHP and 17OHP-d8 were detected using electrospray positive ionisation using MRMs 331.0 > 97.2, 331.0 > 109.1 and 339.1 > 100.4. Peaks obtained were of good shape and had high signal:noise ratio even at the limits of analytical sensitivity. The method proved linear from 0 to 400 nmol/L with an r2 greater than 0.99. Recovery was between 90 and 110%. No interference was detected except from deoxycorticosterone.
Deoxycorticosterone is a little studied, intermediate hormone in the adrenal steroid metabolic pathway. 1 It is an intermediate for aldosterone synthesis lying between progesterone and corticosterone. The precise biological function of deoxycorticosterone is unknown, but it is known to have some glucocorticoid and mineralocorticoid activity. 12 Deoxycorticosterone is not secreted from the adrenal glands in large amounts so plasma concentrations are low compared to other adrenal hormones, approximately 150–300 pmol/L.13–15 Due to this very low concentration in healthy people, there are not likely to be many occasions when this steroid is detected. The bloodspot 17OHP assay is designed for monitoring CAH patients, 90–95% of whom will have 21-hydroxylase deficiency. 1 21-hydroxylase catalyses the conversion of progesterone to deoxycorticosterone, therefore in the patient population being monitored deoycorticosterone concentrations will be low. This explains why during the validation of this method and since being in routine use, deoxycorticosterone has not been detected in any patients’ samples.
It is important to be aware of this interference, however, because in patients with the rarer 11β-hydroxylase deficient CAH, deoxycorticosterone concentrations are likely to be higher than in the healthy population and there is potential for interference. There has also been a report of a deoxycorticosterone secreting adenoma where concentrations were measured up to 30 nmol/L. 16 At this concentration, there would certainly be interference with 17OHP quantitation.
In rare situations such as these, 17OHP can still be differentiated from deoxycorticosterone; however, quantitation is inaccurate. Other means of monitoring these patients would need to be used, for example measuring 17OHP by radioimmunoassay (although this may still have significant interference), monitoring patients’ symptoms or using the cortisone:cortisol ratio as a surrogate marker for efficacy of treatment. 17
Lacey et al. reported the detection of ghost peaks in their internal standard. 10 We detected an additional peak at 2.37 min, but the run time of 3 min was sufficient to elute everything from the column. In the initial workup of the method, extended run times up to 6 min were used during which time no further peaks were detected, and no ghost peaks suggesting carry over were detected on subsequent samples (data not shown). The 6-min run and the 3-min run finally used were identical for the first 2.75 min, but differed in the length of the wash step. On this basis, the run time was reduced to 3 min to speed up the assay. Lacey et al. suggest that the contaminant came from the filter paper used for the bloodspots, but do not say the origin of the filter paper. The contaminant they detected may be specific to that paper. Additionally, we used an UPLC system with a flow rate of 0.45 to 0.6 mL/min compared to 0.25 mL/min used by Lacey et al. The increased flow rate will push any contaminants through quicker, so the additional peak we see at 2.37 min may be the same as seen by Lacey et al.
LCMSMS showed a poor correlation with radioimmunoassay. In samples up to 100 nmol/L, the concentration determined by LCMSMS ranged between 67% lower and 22% higher than the immunoassay value. The majority of this large variation is likely due to imprecision in the radioimmunoassay method, rather than LCMSMS. The two methods used the same calibrators; however, samples were run up to one year apart. The length of time between analyses is not thought to be a problem because the stability of bloodspot 17OHP is very good. Various groups have published stability data, quoting one year at room temperature, 18 two years 19 or even up to a decade. 20 Stability was therefore not a factor in explaining the discrepancy in results. It is likely that the radioimmunoassay results are significantly less accurate than LCMSMS, due to cross-reactivity of the antibody with other steroids as is seen with testosterone assays. 21 The radioimmunoassay is an in-house method, with an unknown cross-reactivity. 9 However, the precision and accuracy for it is poor with inter-assay bias of 19.3% and %CV of 27.42 at 10 nmol/L and 7.2% and 17.86% for 200 nmol/L bloodspots. This %CV and level of inaccuracy combined with the imprecision and inaccuracy of the LCMSMS method can account for the high level of discrepancy between the results.
At the time of setting up this assay, in-house samples were made to be used for internal quality control because no other option was available to our knowledge. Since then we have found that the Centres for Disease Control and Prevention in the USA (CDC) runs a quality assurance programme for newborn screening which includes bloodspots for quality control and proficiency testing. These third party bloodspots are now used for internal quality control and the proficiency testing program is used as an EQA scheme.
A limitation of this method is that the calibrators and QCs were not measured with a different assay to check the concentrations. The concentrations used for target values of the QC and calibrators were based on the enrichment of 17OHP free blood with 17OHP. There are no reference methods for bloodspot 17OHP analysis, so it is difficult for laboratories to properly validate the calibrators and QC material made in house. One solution would be to make up calibrators and QC in whole blood and send the blood for analysis by a validated isotope dilution or GCMS method before spotting the blood to a filter paper. This was not done for this method; however, a sample swap with another laboratory in the process of validating a LCMSMS method for bloodspot 17OHP analysis and the data from the proficiency testing scheme allow us to be confident that the method is working well (data not shown).
Laboratories making a method such as this, where no commercial calibrators are available, must ensure that the blood used to make calibrators does not contain a significant concentration of 17OHP. Concentrations of 17OHP in a healthy male are extremely low; the reference range calculated by Kushnir et al. 5 of 0.8–4.2 nmol/L is below the analytical sensitivity of this assay. While there is a detectable peak of 17OHP in the ‘blank’ sample, it is of such low concentration as to make negligible difference to the calibrator values and quantitation of the patients’ samples. The 17OHP concentration of the ‘0 standard’ is calculated as <1 nmol/L. The error introduced by this is not sufficient to impact the results, particularly when dealing with values up to 200 nmol/L.
Measuring bloodspot 17OHP by LCMSMS is a vast improvement for laboratory medicine. The LCMSMS method has brought improved sensitivity, specificity, accuracy and precision to the bloodspot test giving more reliable results to patients and doctors, allowing improved monitoring of disease. The improved sensitivity of LCMSMS allows smaller bloodspots to be used for analysis. This is beneficial because the quality of bloodspots received by laboratories is often very poor, so the smaller the punched bloodspot the more chance there is of obtaining a viable spot for analysis. Quality of 17OHP bloodspots (Does the blood cover the entire spot? Has the blood soaked all the way through the card? Is the spot “patchy” with areas of lots of blood and more sparse areas?) is a particular issue because the bloodspots are taken by patients or guardians who may lack the knowledge to create a good spot or are reluctant to repeatedly draw blood from themselves or their child and so will create poor quality bloodspots. Using smaller bloodspots means less is rejected due to insufficient sample, thus preventing re-bleeding of patients.
LCMSMS is increasingly becoming the method of choice for steroid analysis. This is partly because steroids are relatively easy to measure and the assays can be multiplexed to measure many steroids simultaneously. 22 This method could be extended to include other forms of CAH rather than just 21-hydroxylase deficiency. 17-hydroxypregnenolone, 11-deoxycortisol and corticosterone could theoretically all be measured using this method, allowing monitoring and diagnosis for other forms of CAH. It could also be extended for use in other conditions; for example, cortisol monitoring for patients treated with hydrocortisone for adrenal insufficiency or testosterone monitoring for patients on treatment with exogenous testosterone could be run at the same time as 17OHP. This would save time for the laboratory and also be more convenient for the patient as a smaller blood sample could be taken at home, and no visits would be required to the GP practice or hospital phlebotomy department for venesection.
A LCMSMS method is presented here for measuring 17OHP in bloodspots. The method is sensitive, specific and removes the need for radioactive material. The extraction method combined with liquid chromatography and mass spectrometry removes potential interfering compounds, although deoxycorticosterone can still interfere in rare instances. This method should prove useful for any laboratory needing to provide a service monitor patients with CAH.
Footnotes
Acknowledgements
Thanks to Craig McEwan for helping train SJS to use the mass spectrometer and Erin Mozley for proof reading the manuscript.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
Annie E Armston.
Contributorship
SJS performed the analyses, analysed the data and wrote the first draft. AEA reviewed and edited the paper. AEA, PC and JD provided assistance with analysing the data. All authors approved the final version of the manuscript.
