Abstract
Background
Measurements of ionized calcium (Ca2+) at concentrations less than 0.3 mmol/L are required for postfilter control in patients who receive extracorporeal circulation with sodium citrate anticoagulation. This study evaluates the stability of the Ca2+ measurements at such concentrations.
Methods
The stability of the Ca2+ measurements was tested by measuring daily the external standard Qualicheck concentration 3 s7950, Radiometer (0.22–0.25 mmol/L) by blood gas instruments ABL800 and ABL90, Radiometer. Two different Ca2+ membrane lots were tested for the usual membrane lifetime of 12 weeks at ABL800 instruments. For the ABL90 instrument, the sensor cassette (with Ca2+ membrane and electrode) was replaced after four weeks as required.
Results
We observed over 40% Ca2+ increase within the usual 12 weeks lifetime of the Ca2+ membrane at the ABL800 instruments. Measurements of Ca2+ at concentrations less than 0.3 mmol/L were within acceptable limits for both ABL800 and ABL90 instruments when Ca2+ membrane and sensor cassette were replaced after four weeks.
Conclusions
For ABL800 instruments, it is necessary to use an extra quality control (<0.3 mmol/L) in addition to the usual quality controls to monitor Ca2+ measurements below 0.3 mmol/L. The acceptable stability of the Ca2+ measurements can be achieved by the Ca2+ membrane and sensor cassette replacement after four weeks. If the usual 12 weeks of Ca2+ membrane lifetime is maintained, it may result in a clinically significant overestimation of Ca2+ by ABL800 instruments.
Introduction
Extracorporeal anticoagulation with sodium citrate is used in critically ill patients with continuous renal replacement therapy. 1 Citrate is administered in the extracorporeal circulation to prevent activation of blood coagulation by binding ionized calcium (Ca2+). The known complication of this procedure is hypocalcaemia. Therefore, monitoring of ionized calcium is of great importance for evaluation of the extracorporeal citrate anticoagulation. Blood samples are usually analysed by point-of-care blood gas instruments. In order to achieve sufficient extracorporeal anticoagulation, the postfilter Ca2+ must be maintained at extremely low concentration (0.25–0.34 mmol/L).1–3 There is no information about stability of Ca2+ measurements at this concentration. Recently, concern has been expressed about the discrepancy of postfilter Ca2+ measurements by different blood gas instruments. 4 The aim of this study was to evaluate the stability of Ca2+ measurements at concentrations below 0.3 mmol/L by blood gas instruments ABL800 and ABL90, Radiometer.
Methods
Ca2+ was measured by the potentiometric method with ion-selective electrodes by ABL800 and ABL90 instruments, Radiometer Medical, Copenhagen, Denmark. The measurement interval for Ca2+ was 0.20–9.99 mmol/L for both instruments.5,6 Altogether four identical ABL800 instruments were used in the study: two of them for real postfilter samples, while two other instruments were used for other routine measurements at the clinical laboratory. Mean bias between different ABL800 instruments was 0.01 mmol/L (95% CI: 0.00–0.02). To evaluate stability of Ca2+ measurements below 0.3 mmol/L, we used the external standard Qualicheck level 3 s7950 (Radiometer Medical, Copenhagen, Denmark) as a quality control.
The assigned value of the external standard was 0.22 mmol/L for ABL800 and 0.25 mmol/L for ABL90. For each measurement, a new ampoule of the external standard was opened and an adaptor was used to connect the original glass ampoule to the Radiometer instrument. The external standard Qualicheck level 3 s7950 was measured once per day at all four ABL800 instruments and one ABL90 instrument. Mean weekly control values were based on these daily measurements.
Two different lots of Ca2+ membranes were tested for the same ABL800 instruments, each lot for the period of 12 weeks. The Ca2+ membranes were replaced after four weeks at two instruments that were routinely used for the postfilter samples, while two other ABL800 instruments had Ca2+ membranes for 12 weeks, which is the recommended lifetime of Ca2+ membrane for ABL800. 5 For the ABL90 instrument, the sensor cassettes were replaced after four weeks, as required. 6
Further, weekly mean deviations from the assigned control value for Ca2+ measurements with two different Ca2+ membrane lots were calculated for ABL800 instruments for weeks 1–12. These means were presented with standard deviations (SDs) to show lot-to-lot variation.
The deviation of Ca2+ control value >0.05 mmol/L was considered as clinically significant (>20% deviation from 0.25 mmol/L). The definition of the clinically significant drift was based on the Fresenius Medical Care instructions for stepwise adjusting of the citrate anticoagulation, 3 as well as on the recommendations for the drift tolerance limits (±0.05 mmol/L) for the Ca2+ analysis set by the Radiometer Medical, Copenhagen, Denmark.5,6
Results
The stability of Ca2+ measurements below 0.3 mmol/L is presented in the Figure 1. Figure 1 shows the deviations (in %) of the Ca2+ control weekly means from the assigned control value with SDs that reflect lot-to-lot variation for ABL800 instruments (Figure 1). During the first four weeks, all Ca2+ measurements were within the acceptable range and showed the same results for all Radiometer ABL800 instruments. Further, we have observed a continuous increasing of Ca2+ values at the ABL800 instruments. Already after four weeks, we observed a deviation of the control over 0.05 mmol/L from the assigned value. The mean increase of the Ca2+ control values was 28% at the beginning of the 5th week. At the beginning of the 9th week, the increase of the Ca2+ values was over 40% with lot-to-lot variation 5%. The deviation from the assigned control value was 42% at the end of the 12th week with lot-to-lot variation 15%.
The 12 weeks’ stability of Ca2+ measurements at concentrations less than 0.3 mmol/L by ABL90 and ABL800 blood gas instruments. Weekly means were calculated based on daily measurements of the Qualicheck level 3 external standard and then converted to % deviation from the assigned value. Two different Ca2+ membrane lots were tested for ABL800, presented SDs reflect lot-to-lot variation.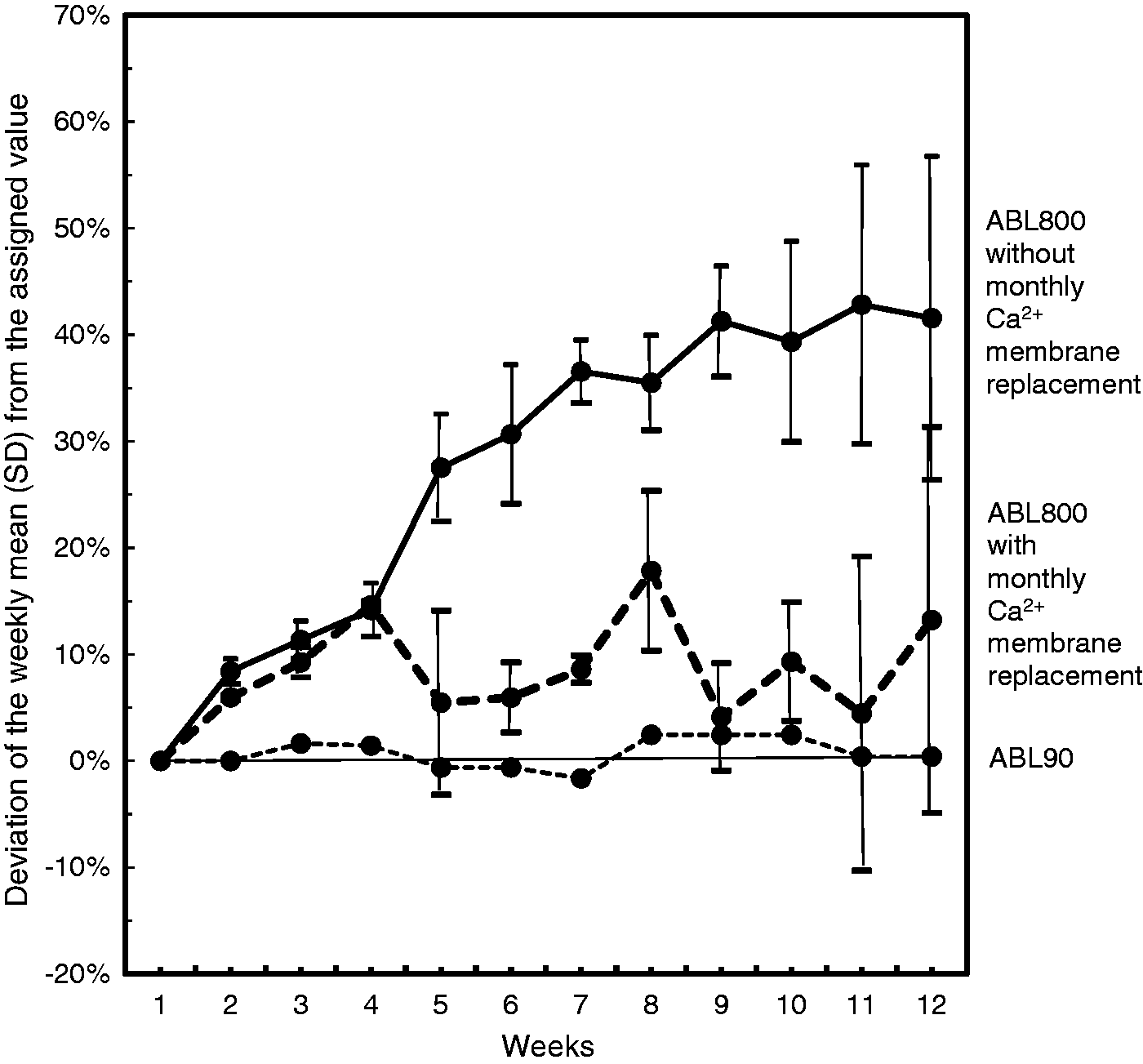
When the same two lots of the Ca2+ membrane were replaced after four weeks at the ABL800 instruments, the control values remained within the acceptable range. The mean deviation from the assigned value was 9%.
The control Ca2+ measurements were more stable when measured by ABL90 instrument. The mean variation was 0.005 mmol/L; the largest control deviation did not exceed 0.01 mmol/L (4% of the assigned control value).
Additionally, the routine Ca2+ quality control (S7745, Radiometer) was measured at the same time at these instruments. This control had higher Ca2+ assigned values: 0.51 mmol/L for ABL800 and 0.37 mmol/L for ABL90 (data not shown). The ABL90 control had little deviation, mean CVa was 0.7%. The ABL800 control has shown a tendency to increase, but remained within acceptable range during 12 weeks. The deviation from the assigned value was less than 0.05 mmol/L. Mean CVa was <3% for all ABL800 instruments. Thus, the routine Ca2+ quality control for the ABL800 has not reflected the drift of Ca2+ measurements at lower concentrations.
Discussion and conclusions
ABL800 instruments were not stable in measuring Ca2+ at concentrations less than 0.3 mmol/L when Ca2+ membrane was used for the usual lifetime of 12 weeks. Ca2+ measurements at ABL90 were more stable compared with the ABL800 instruments. For ABL90 instrument, the sensor cassette was replaced after four weeks, as required. Based on our data, we recommend to replace Ca2+ membrane on the ABL800 instruments after four weeks. Otherwise, it may result in a clinically significant overestimation of Ca2+ at concentrations below 0.3 mmol/L by the ABL800 instruments. It is alarming, because postfilter Ca2+ evaluation in patients with citrate anticoagulation during extracorporeal circulation requires precise Ca2+ measurements at these concentrations. The drift that we observed in quality control measurements may reflect possible Ca2+ overestimation in the real postfilter samples. Another analytical problem is a definite lot-to-lot variation of the Ca2+ measurements at these concentrations. It is necessary to use an extra quality control in addition to the usual quality controls to monitor stability of Ca2+ measurements below 0.3 mmol/L.
Footnotes
Acknowledgments
The authors would like to thank the Department of Laboratory Medicine, University Hospital of North Norway for technical support for the project. The authors also wish to thank laboratory technicians Morten Rafdal, Veronica Salamonsen, Birgitte Jakobsen and Henriette Lind for technical assistance with this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study has received internal funding from the Department of Laboratory Medicine, University Hospital of North Norway.
Ethical approval
The Department of Laboratory Medicine, University Hospital of North Norway has general approval from the Norwegian Ethical Committee to perform quality control studies.
Guarantor
MA.
Contributorship
Both authors researched the literature and planned the study. RJ was responsible for the laboratory analyses. MA wrote the manuscript. Both authors reviewed and edited the manuscript and approved the final version of the manuscript.
