Abstract
Whole blood, serum or plasma chloride is almost exclusively measured by potentiometry with an ion-selective chloride electrode which utilizes membrane selectivity to chloride ions. Other anions such as bromide, iodide and thiosulphate can interfere but usually are not present in high enough concentration to cause significant cross reactivity. A patient from our burns unit had serial chloride measurements on a Radiometer ABL800 blood gas analyser. The results were higher in contrast to plasma measurements on the Abbott Architect Ci8200, which were within reference intervals and in line with the patient's pathophysiological status. This indicated a likely interference with the blood gas analyser chloride estimation. The chloride results on the ABL800 for 3rd, 4th and 5th day after the burn accident were 170, 137 and 119 mmol/L. Corresponding plasma chloride results on the Ci8200 were all around 105 mmol/L. Nitrate was found to be markedly elevated in these samples, and the results were 6.7, 4.9 and 1.1 mmol/L, respectively (reference limit < 0.08 mmol/L). To further demonstrate nitrate was the causative agent, pooled plasma spiked with 7 mmol/L of sodium nitrate caused a rise in the ABL800 chloride from 105 to 202 mmol/L. Later we confirmed that the patient was topically medicated with cerium nitrate cream (Flammacerium®, Sinclair IS Pharma, UK) for his burns. In summary, the results clearly indicated nitrate was the interferent with the ABL800 chloride estimation and the source was the topical burns cerium nitrate cream.
Introduction
Ion-selective electrodes (ISEs) are used in clinical laboratories and point-of-care analysers to measure electrolytes including sodium, potassium, chloride and ionised calcium. Selectivity of ISEs depends on the characteristics of the ISE membranes. 1 The two major types of chloride ISEs are solid state silver/silver chloride electrode and liquid/liquid ion-exchange membrane electrode. 2 Flammacerium® (silver sulfadiazine and cerium nitrate, Sinclair IS Pharma, UK) is a topical cream introduced in 1976. It is recommended for extensive burns when early excision is not feasible.3,4
The study aim was to demonstrate that the topical Flammacerium® cream was the causative agent in the chloride overestimation on the ABL800 blood gas analyser.
Clinical case
A previously fit and healthy 11-year-old boy suffered a severe burn injury from a fuel explosion in French Polynesia. He suffered 80% total body surface area full thickness burn and Flammacerium was applied to his burn wounds. Three days after the accident, he was transferred to the National Burns Centre at Middlemore Hospital in Auckland, New Zealand for further treatment. On admission, blood test by Abbott Architect Ci8200 (C8) showed Na 132 mmol/L (135–145 mmol/L), Cl 100 mmol/L (95–110 mmol/L), K 5.1 mmol/L (3.5–5.2 mmol/L), Urea 3.0 mmol/L (3.2–7.7 mmol/L), Creatinine 74 µmol/L (40–80 µmol/L), Albumin 22 g/L (38–52 g/L) and normal liver enzyme tests. Full blood count analysis showed Hb 118 g/L (115–145 g/L) and platelet 78 × E9/L (150–425 × E9/L). Simultaneous venous whole blood analysed by Radiometer ABL800 blood gas analyser (ABL) gave the following results: pH 7.34 (7.36–7.44), bicarbonate 20 mmol/L (20–28 mmol/L) and sodium 130 mmol/L (135–146 mmol/L). However, an unexpectedly high chloride of 170 mmol/L (95–110 mmol/L) was recorded. The chloride results from the ABL and C8 analysers and nitrate concentrations over time are shown in Figure 1.
Chloride concentrations of the index patient from Radiometer ABL800 and Abbot Architect Ci8200 versus nitrate concentrations over time. Diamonds: chloride concentration from Radiometer ABL800, squares: chloride concentration from Abbot Architect Ci8200, triangles: nitrate concentration.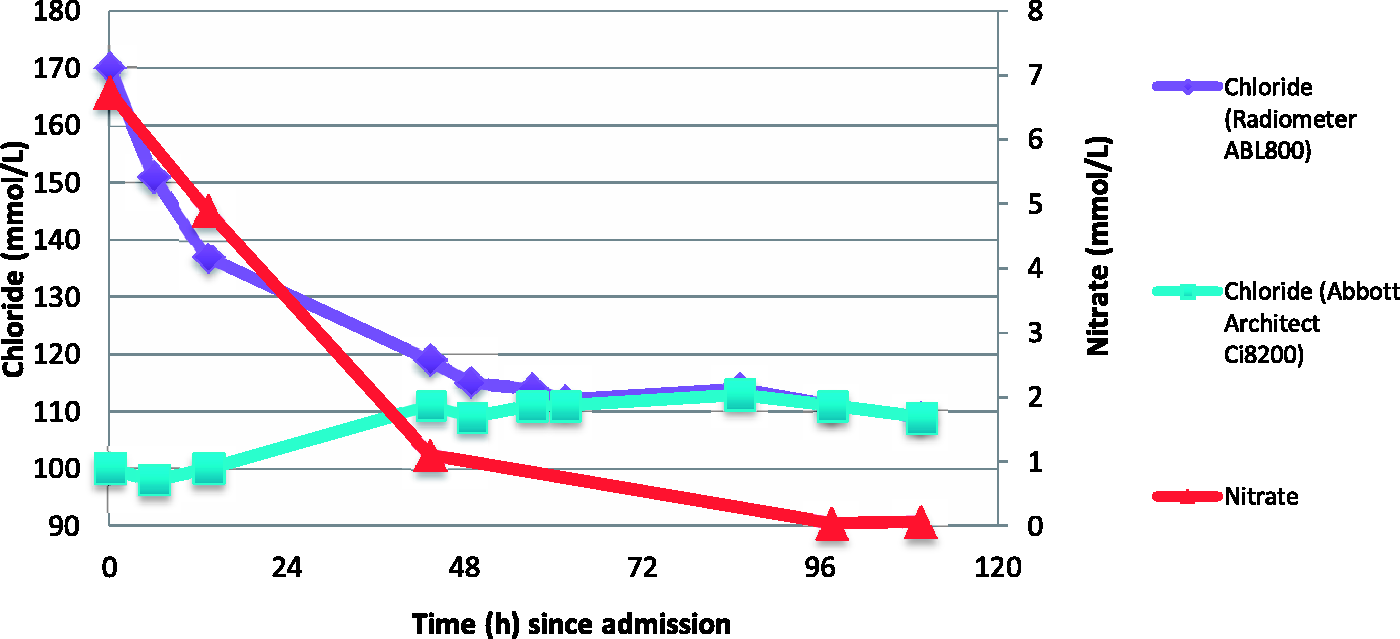
By day six, the nitrate concentrations had decreased to near normal and chloride measured by ABL and C8 became comparable, at 114 mmol/L and 111 mmol/L, respectively. Despite intensive treatment, he developed wound sepsis and passed away on day 10.
Positive interference on ABL but not on C8 ISE chloride electrode was suspected. This interference was further investigated.
Methods
Assaying for some known interferents
The admission and three subsequent blood samples were analysed for the most likely interferents. Plasma bromide, thiocyanate and nitrate were measured by ion-paired reverse phase high-performance liquid chromatography (HPLC) with low ultraviolet detection at 205 nm (all components supplied by Thermo Scientific Sunnyvale CA, USA), and plasma iodine was measured by Varian 820-MS ICP-MS, Varian, Melbourne, Australia.
Interference study
Aliquots from fresh blank plasma pool prepared by collecting left-over plasma from hospitalized patients were spiked with varying volumes of 0.2 mol/L NaNO3 solution, achieving nitrate concentrations of 0, 1.0, 1.5, 2.0, 2.5, 3.3, 4.0, 5.0, 7.1 and 10 mmol/L. The volume of NaNO3 solution added to each sample was ≤5%. Plasma chloride was then measured by C8 and ABL.
These samples were re-assayed on the following day on ABL after chloride ISE membrane change (new lot number), to assess effect of the age of the membrane.
Results
The interferent
Thiocyanate was not detected, and bromide concentrations were normal in all four samples (all ≤0.045 mmol/L; reference limit <0.1 mmol/L). However, while reviewing the bromide chromatogram, an unexpected peak was observed at 7.49 min, which was confirmed to be nitrate (Figure 2).
Chromatograms showing markedly elevated nitrate peak in the index patient and a normal peak in a normal subject. Top arrow: nitrate peak in the index patient, bottom arrow: nitrate peak in normal subject.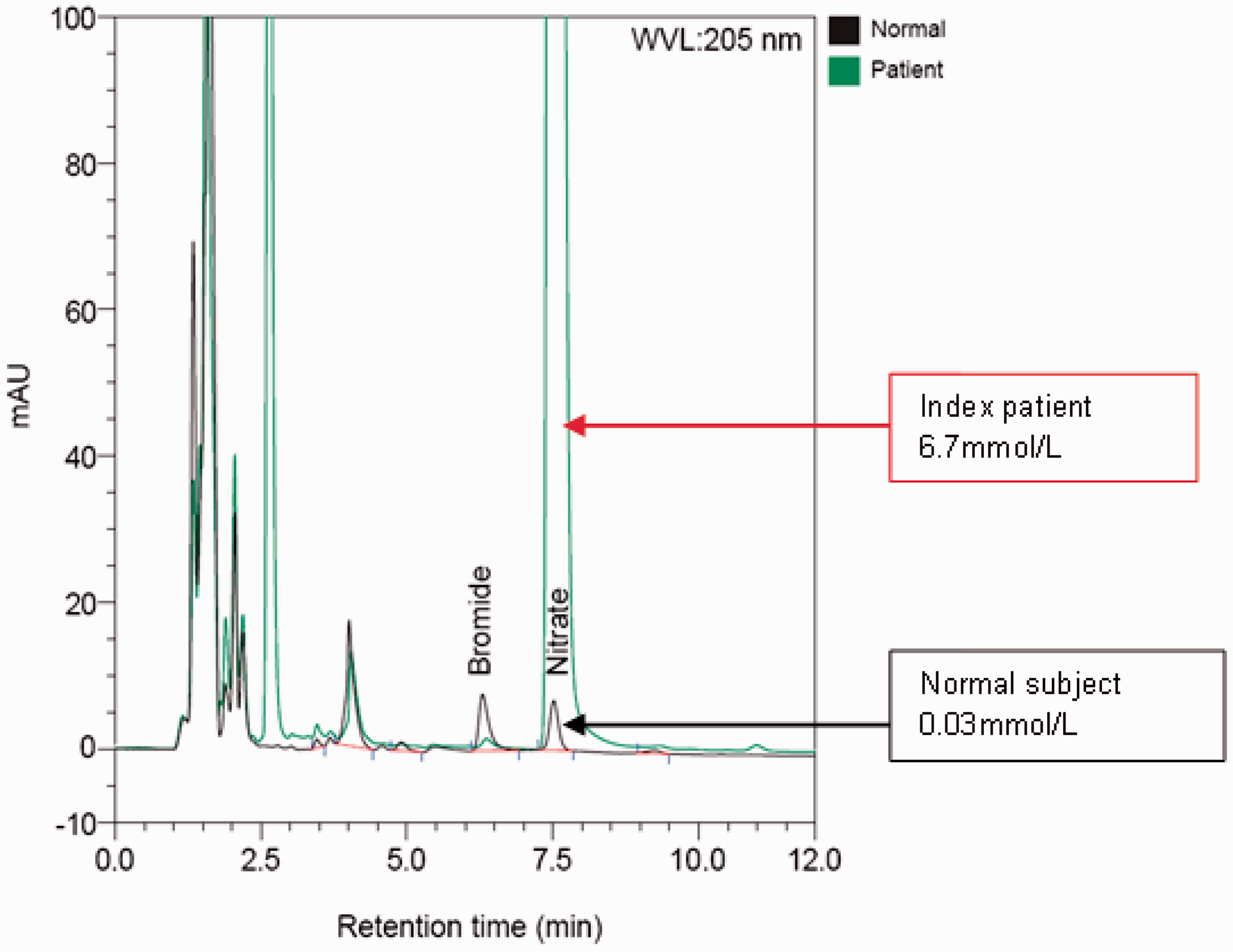
Admission blood (postburn day three) plasma nitrate was 6.7 mmol/L (locally derived upper reference limit <0.08 mmol/L). It gradually decreased to 4.9 mmol/L, 2.3 mmol/L and 1.1 mmol/L in the next three days. The apparent chloride measured by ABL came down in synchrony with the plasma nitrate concentration (Figure 1). Iodine was significantly elevated ranging between 4 and 155 µmol/L (reference interval 0.31–0.72 µmol/L), but the change in iodine concentration did not correlate with the apparent change in chloride concentration. Furthermore, the iodine concentration was not high enough to cause sufficient interference on either ABL or C8. A brief interference study showed 1400 µmol/L of iodine caused a mere 8% increase in apparent ABL chloride but no change in C8 chloride concentration (data not shown).
Interference study
Sodium nitrate caused a linear positive interference on ABL (r2 = 0.99) with four-month-old chloride membrane but no interference was observed on C8. With a new chloride membrane, similar interference was present on ABL (r2 = 0.98) but to a lesser degree (Figure 3).
Effects of sodium nitrate on chloride results from Radiometer ABL800 and Abbott Architect Ci8200. Squares: Radiometer ABL800 with four-month-old Cl ISE membrane, triangles: Radiometer ABL800 with new Cl ISE membrane, diamonds: Abbott Architect Ci8200.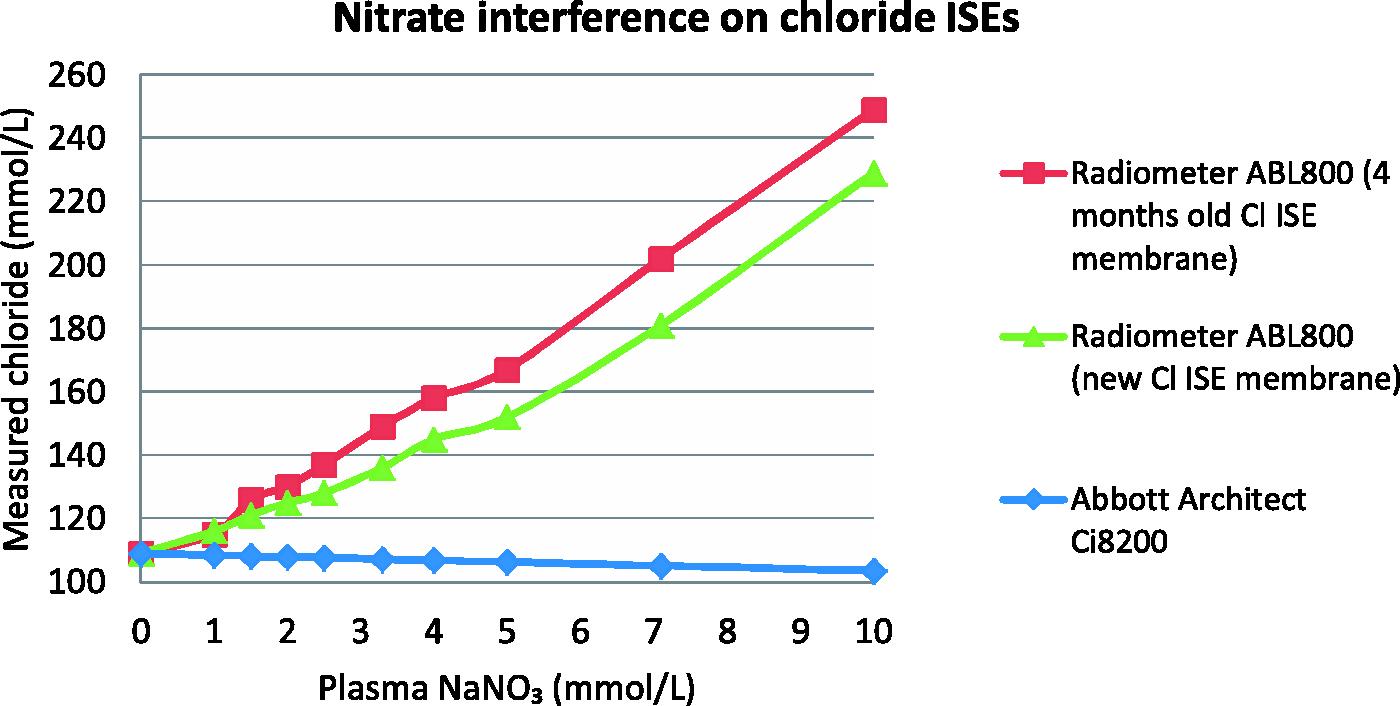
Discussion
Source of the nitrate
Endogenous production of nitrate after extensive burn injury was considered as a putative cause of elevated nitrate. Plasma nitrate is known to increase in burns patients due to increased nitric oxide synthase activity following burn-associated inflammation. However, it is unlikely the main contributor in this case, as the plasma nitrate seen in our patient at 1–6 mmol/L is about 50–300 fold higher than what has been reported in the literature from burn patients.5,6
Exogenous sources were also considered. We excluded exposure to ammonium nitrate during the explosion, smoke-inhalation injury or intravenous/topical usage of nitrate in our hospital. However, further enquiries to the hospital in French Polynesia confirmed the use of a topical cream containing cerium nitrate and silver sulfadiazine (Flammacerium®) prior to transfer. Flammacerium was applied to his burn wounds in 1 cm thickness twice on the day of injury and on day 3 before the transfer.
Cerium nitrate cream is used in burns patients when early excision and grafting cannot be performed. It causes transformation of burn eschar into a leathery yellowish crust, allowing postponement of excision and grafting. 4 This product is mainly marketed in European countries, and it is not available in New Zealand.
Nitrate interference on chloride ISE
A paper by Chianea et al. 7 described similar cases – they observed some patients with third degree burns had unexpected hyperchloraemia when measured by their blood gas analyser (ABL 700, Radiometer). However, unlike our case, it also interfered with their automated analyser chloride ISE (Cobas Integra 800, Roche Diagnostics) albeit to a lesser degree. They also demonstrated that the interference is from nitrate, from Flammacerium treatment.
ABL uses liquid/liquid ion-exchange membrane electrode for chloride, which contains a chloride ion carrier in the membrane. 8 Selectivity of this type of ISE depends on the ion hydration energy and the lipophilic character of the ion, hence all ions that have hydration energy higher than or equivalent to chloride are potential interferents. Blood samples containing significant amount of anions, e.g. bromide, iodide, thiocyanate, thiosulphate, salicylate, bicarbonate and nitrate can cause positive interference on chloride. The degree of interference tends to vary, depending on the analyser and the electrode system set up.1,9
Another type of chloride ISE used in clinical laboratory is solid-state Ag/AgCl electrodes, 2 and is used by Abbott Architect. 10 These chloride ISEs suffer from positive interferences from iodide, bromide and thiosulphate, as the solubility product of silver chloride is less than that of silver bromide, silver iodide and silver thiosulphate.10,11 However, unlike ion-exchange membrane electrodes, this type of ISE is not susceptible to interference from organic anions such as salicylate, bicarbonate or nitrate. It is worth mentioning that even if Abbott Architect uses ion-exchange membrane electrodes, the degree of interference observed on C8 would have been much less than ABL, as C8 incorporates indirect method which has a predilution step. Predilution step would have diluted the interferent, causing less degree of interference.
We found that nitrate interference on ABL chloride electrode was linear to at least 10 mmol/L. The degree of interference seen in our patient correlates well with what was observed in vitro from the spiking experiment. Plasma nitrate of 6.7 mmol/L in the patient caused 70 mmol/L increase in ABL chloride which matches with the finding that spiking 7 mmol/L NaNO3 caused 98 mmol/L increase in ABL chloride from the in vitro study.
Users of Flammacerium should be aware that ABL chloride measurements can be falsely elevated from systemic absorption of nitrate.
Conclusion
The patient’s falsely high whole blood chloride by ABL was due to abnormally high plasma nitrate concentration, due to topical application of cerium nitrate during burn treatment. We demonstrated differing susceptibilities to nitrate interference on ABL and C8 chloride measurements.
Footnotes
Acknowledgements
We wish to thank Mark Wiggins, Burns specialist nurse at Middlemore Hospital for his contributions into this case; Pam Rowe and Barry Palmer for their analytical support.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
It was not possible to obtain informed consent from the patient or patient’s carers in this case despite every effort being made. The Journal has agreed to publish the paper based on its educational value.
Guarantor
LYH.
Contributorship
LYH performed most of literature search, bench work, data analysis and wrote the manuscript. GAW initially identified the nitrate interference using HPLC. WWC researched literature and was involved in method development. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
