Abstract
Background
Human chorionic gonadotrophin is a marker of early pregnancy. This study sought to determine the possibility of being able to distinguish between healthy and failing pregnancies by utilizing patient-associated risk factors and daily urinary human chorionic gonadotrophin concentrations.
Methods
Data were from a study that collected daily early morning urine samples from women trying to conceive (n = 1505); 250 of whom became pregnant. Data from 129 women who became pregnant (including 44 miscarriages) were included in these analyses. A longitudinal model was used to profile human chorionic gonadotrophin, a Cox proportional hazards model to assess demographic/menstrual history data on the time to failed pregnancy, and a two-stage model to combine these two models.
Results
The profile for log human chorionic gonadotrophin concentrations in women suffering miscarriage differs to that of viable pregnancies; rate of human chorionic gonadotrophin rise is slower in those suffering a biochemical loss (loss before six weeks, recognized by a rise and fall of human chorionic gonadotrophin) and tends to plateau at a lower log human chorionic gonadotrophin in women suffering an early miscarriage (loss six weeks or later), compared with viable pregnancies. Maternal age, longest cycle length and time from luteinizing hormone surge to human chorionic gonadotrophin reaching 25 mIU/mL were found to be significantly associated with miscarriage risk. The two-stage model found that for an increase of one day in the time from luteinizing hormone surge to human chorionic gonadotrophin reaching 25 mIU/mL, there is a 30% increase in miscarriage risk (hazard ratio: 1.30; 95% confidence interval: 1.04, 1.62).
Conclusion
Rise of human chorionic gonadotrophin in early pregnancy could be useful to predict pregnancy viability. Daily tracking of urinary human chorionic gonadotrophin may enable early identification of some pregnancies at risk of miscarriage.
Keywords
Background
Sporadic miscarriage is a common complication of early pregnancy, and prospective studies in women trying to conceive have found that approximately only one third of conceptions actually result in a live birth.1–3 It is estimated that 30% of fertilized ovum are lost prior to implantation and a further 30% following implantation but before the day of the missed period (approximately the third or fourth week of gestation). 4 An estimated 15% of conceptions result in early miscarriage, 5 while late pregnancy losses (between 12 and 22 weeks) are less common, occurring in approximately 4% of pregnancies. 6 Early pregnancy loss is associated with substantial healthcare costs, often caused by unnecessary diagnostic tests, hospital admissions, surgical management and follow-up. 7 In addition, early miscarriage can have a profound impact on the social and psychological wellbeing in affected couples. Early pregnancy loss has been reported to cause depression and/or anxiety in almost 20% of affected women, with symptoms persisting for up to three years after the miscarriage and affecting quality of life. 8
Human chorionic gonadotrophin (hCG) is a hormone produced by trophoblast cells in very early pregnancy, thus making it an ideal marker for pregnancy, and its detection is utilized in both laboratory and home pregnancy tests.9–12 During early pregnancy, concentrations of hCG rise exponentially and predictably.11,13,14 The presence of hCG can be detected in the serum of pregnant women around eight days after conception (nine days after the luteinizing hormone [LH] surge); hCG has been reported to be present at a concentration of approximately 10 mIU/mL between nine and 10 days after follicular rupture.15,16 Serum concentrations of hCG then continue to rise during early pregnancy, at a rate of about 50% per day, reaching concentrations as high as 1,000,000 mIU/mL by week 10, after which a decrease to a stable concentration of approximately 20,000 mIU/mL for the remainder of the pregnancy is typically seen.16,17
hCG is also readily detected in maternal urine, where its concentrations show a similar pattern to that observed in serum. 18 The concentration of hCG in the urine of pregnant women 10 days after LH surge has been reported to be approximately 0.93 mIU/mL, and concentrations reliably rise daily until reaching a plateaux at approximately 45 days after conception,14,18 a finding that has been consistently observed across different study populations and ethnic groups.13,19
Pregnancy loss has been defined as the spontaneous demise of a pregnancy that has been confirmed by at least two positive tests for hCG in urine or serum,
20
and clinical studies have confirmed that early pregnancy loss is associated with a transitory rise and fall in urinary hCG concentrations, with the fall in concentrations coinciding with the onset of menstrual bleeding.
21
Although, in most cases, the hCG profiles of women with failed pregnancies can be distinguished from those of viable pregnancies when studied retrospectively by experts in this field,
22
it is less certain how the use of hCG concentrations can prospectively predict failing pregnancies. In women undergoing
The aim of this analysis was to determine whether it is possible to distinguish between healthy and failing pregnancies by utilizing patient-associated risk factors and information on the concentrations of hCG in urine in early pregnancy.
Methods
To determine whether it is possible to distinguish between healthy and failing pregnancies in women by using data from daily concentrations of urinary hCG and personal characteristics, a longitudinal model was used to profile hCG and a Cox proportional hazards model was used to assess demographic and menstrual history data. A two-stage model was subsequently used to combine the longitudinal and time-to-events models.
Data source
The data used in this analysis were obtained from a study conducted by SPD Development Company Ltd (Bedford, UK), in which women collected daily early morning urine samples for complete menstrual cycles and for up to 28 days after the day of their expected period if they became pregnant. Women aged 18–45 who were actively trying to conceive were recruited onto the study, and no assessments as to their fertility status were undertaken. Samples were stored at + 4℃ until analysis, unless analysis could not be accomplished within two weeks of receipt, then samples were stored at −80℃. The storage conditions used have previously been validated for maintaining analyte integrity. A range of background information was also collected for each volunteer, including demographics, pregnancy history and menstrual cycle history. Institutional review board approval was obtained from Quorum Review Committee of 30 November 2009, ref number: 2486. All participants provided written consent.
A total of 1505 eligible female volunteers, who were trying to conceive were recruited into the study. Of these volunteers, 250 women became pregnant during the study period, 178 women (71%) had single viable pregnancies, one woman (0.04%) had a multiple pregnancy and 44 women (18%) suffered biochemical/early miscarriage. During the study, 14 women withdrew and 13 women were lost to follow-up.
The data used in this analysis consisted of hCG concentrations and patient information from the 44 biochemical/early miscarriages and 85 randomly chosen single healthy pregnancies. Concentrations of hCG were determined in urine samples using a validated quantitative automated immunoassay system (AutoDELFIA; PerkinElmer, Waltham, USA). 27 Concentrations of LH in these urine samples were also analysed, using a validated quantitative automated immunoassay system (AutoDELFIA; PerkinElmer, Waltham, USA), 17 in order to determine the day of the LH surge (which precedes ovulation by approximately 24 h). 28 Laboratory investigators were blinded to patient clinical information when conducting all analyses. The day of conception for this study is assumed to be the day after the LH surge. 28 The time between the day of LH surge and the first day on which hCG concentration reached 25 mIU/mL (the most common sensitivity of a home pregnancy test) was determined.
Longitudinal modelling
The data consist of multiple samples of urine that were collected daily for a period of time for each volunteer, allowing for a longitudinal modelling approach. The use of longitudinal modelling allows for inferences about the mean response profiles to be drawn, and it also provides the ability to predict individual response trajectories, make inferences about the variability between subjects and make inferences about the dependence between measurements on the same volunteer.
It was hypothesized that volunteers who suffer an early loss will have a different profile to those who miscarry later in the pregnancy or those with a viable pregnancy. How the miscarriage data could be grouped were assessed using visual plots of the data. Longitudinal models allowing random slopes and intercepts for each volunteer were fitted to each of the identified groups; the model also included a random quadratic day term to account for the shape of the profile being curved, and an unstructured covariance structure was assumed.
Survival modelling
A range of background information was collected from women participating in this study, and survival modelling was used to determine if volunteer demographics, pregnancy history or menstrual cycle history had any relation to pregnancy outcome. Time-to-event information was available for those volunteers who miscarried within the sample collection period; volunteers miscarrying after this time were censored at the time the study ended. Kaplan-Meier plots were used to determine the survival function non-parametrically. Time zero (baseline) was defined as the estimated day of missed period.
A Cox proportional hazards model was constructed using a forward selection procedure, proportional hazards were also assessed. Any covariate with a 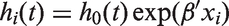
Two-stage modelling
A two-stage model was used to incorporate the results of the longitudinal analysis into the survival model. This model was made up of two components: a longitudinal model of the biomarker (hCG) and a survival (time-to-event) model. The longitudinal model took the structure given below, a linear model with random effects
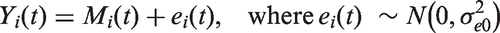
The longitudinal and survival model are linked through the estimated trajectory function
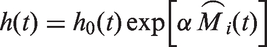
The estimates of the random intercept, slope and quadratic effects for each volunteer were also assessed for inclusion in the survival model, as well as the predicted current value of the longitudinal response at time
Results
Volunteer demographics.

SD: standard deviation.
Women who miscarried were, on average, older than those with viable pregnancies (mean age 32.3 vs. 30.0 years, respectively). Also, the average self-reported cycle length for women who miscarried was shorter 28.7 days, compared with 29.9 days for women with viable pregnancies. However, no between-group differences were found to be statistically significant.
Longitudinal modelling
The profiles of log hCG for women whose pregnancies resulted in a live birth are shown in Figure 1(a). In these women, an initial rise in hCG following conception can be observed, concentrations then continue to rise over the first three weeks of pregnancy and subsequently begin to plateau; had the study continued further into early pregnancy, it is expected that levels would have reached a plateau at around 10 weeks. The hCG profiles for women whose pregnancies resulted in a biochemical loss (loss before 6 weeks, recognized by a rise and fall of hCG)/early miscarriage (loss six weeks or later) are shown in Figure 1(b). In some of these volunteers, the initial rise in hCG concentrations is followed by a sharp drop, believed to correlate to early biochemical loss. Women who miscarried following the end of sample collection showed a rise in hCG up to the first three weeks of pregnancy.
Log hCG profiles in (a) viable pregnancies for women whose pregnancies resulted in a live birth and (b) women whose pregnancies resulted in miscarriage.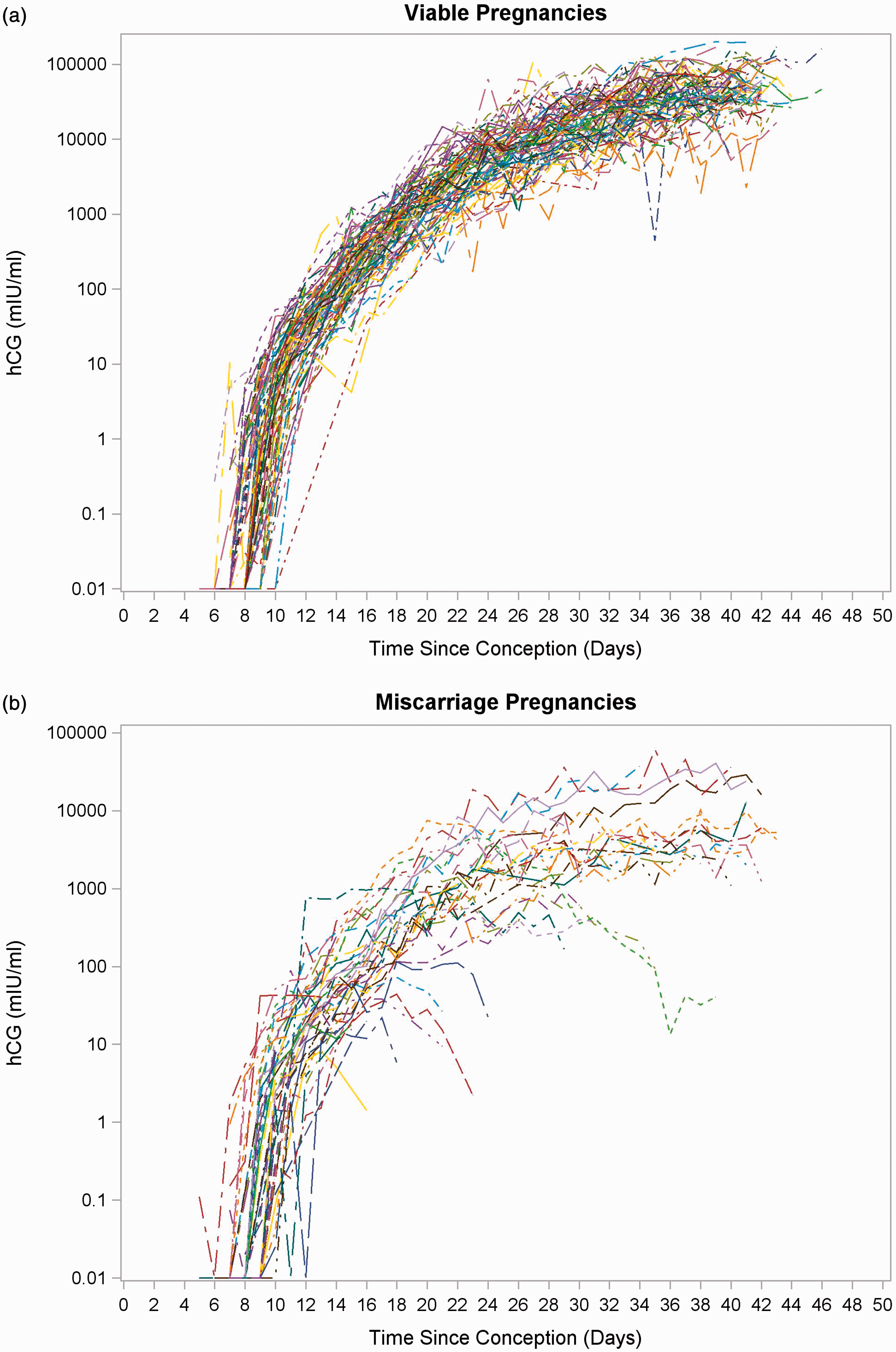
The fixed-effect log hCG profiles for the three groups are shown in Figure 2. The hCG profile for volunteers suffering an early biochemical loss is statistically different from the profiles of women with viable pregnancies or those who went on to suffer an early miscarriage. The profile for women suffering a biochemical loss shows a steep rise in log hCG before decreasing when pregnancy loss occurs, compared with a steadier rise in hCG in the profiles of viable pregnancies and early miscarriages. The profile for volunteers suffering an early miscarriage tends to plateau at a lower log hCG compared with the profile of women with viable pregnancies. The difference between the mean slope in the viable pregnancy volunteers and those who suffered an early biochemical loss is 0.648 (95% confidence interval [CI]: 0.587, 0.709), indicating that the rate at which hCG rises is slower in this biochemical loss group compared with that in healthy pregnancies.
Fixed-effect profiles for log hCG concentrations in women with viable pregnancies and those suffering biochemical or early loss.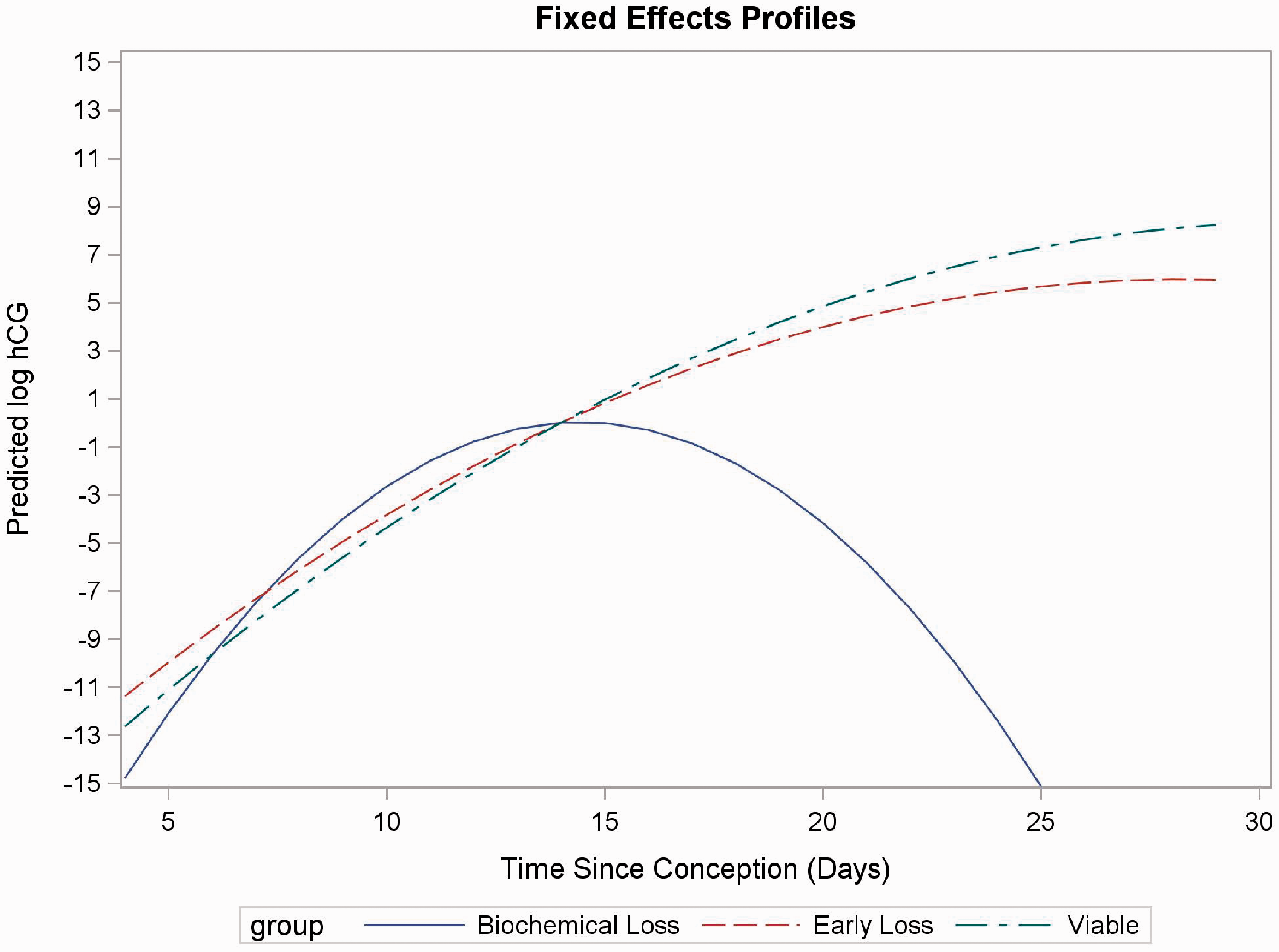
Survival modelling
The Kaplan-Meier estimate of the survival function was estimated (Figure 3), showing that by 5 days after the estimated day of the missed period the pregnancy survival is approximately 91%, reducing to approximately 86% at 18 days after the estimated day of the missed period.
Kaplan-Meier Plot of survival probability by day relative to the day of the missed period.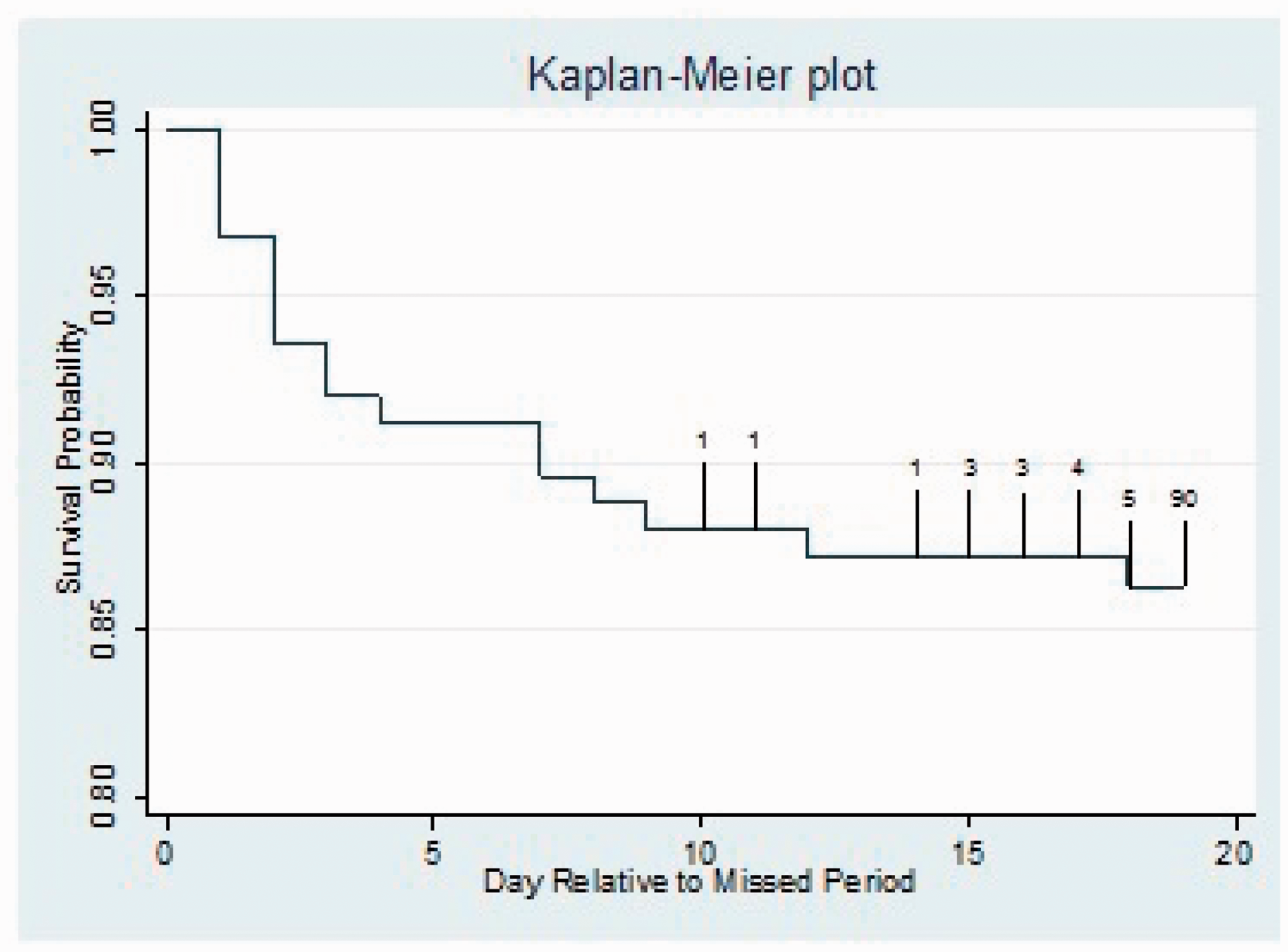
Using a Cox proportional hazards model, the following covariates were deemed to be significantly associated with risk of pregnancy loss: maternal age, longest cycle length and time from LH surge to hCG reaching a concentration of 25 mIU/mL (the concentration of hCG that is indicative of a normal pregnancy at approximately three days), 14 which occurs before the time zero. Other covariates added to the model, but found not to be significant, included previous pregnancies/miscarriages, time trying to conceive, usual cycle length, shortest cycle length, difference in longest and shortest cycle length, ethnicity, race, education and occupation. Schoenfeld residuals were used to assess for proportional hazards in the model; these plots indicated that the covariates showed some signs of having non-proportional hazards.
Survival modelling final model estimates.
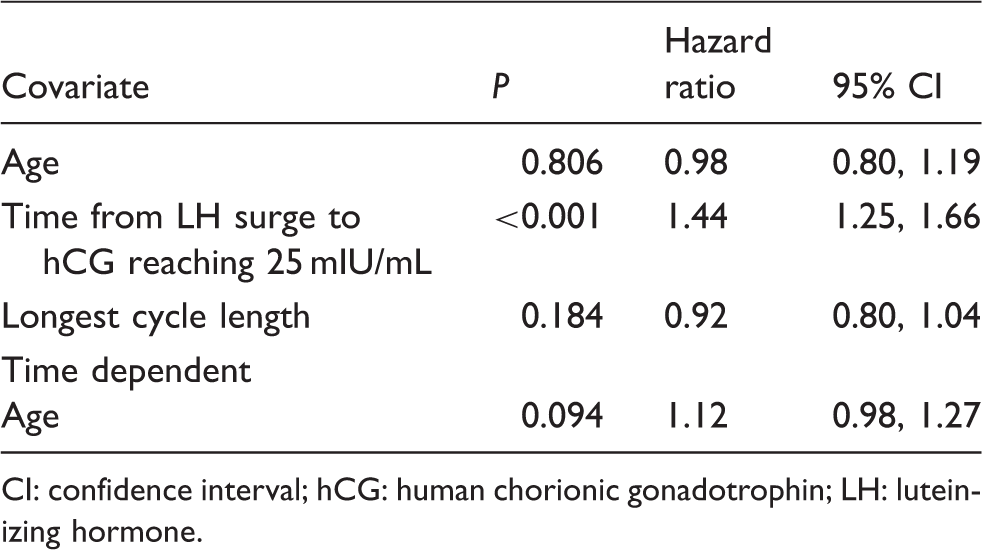
CI: confidence interval; hCG: human chorionic gonadotrophin; LH: luteinizing hormone.
Two-stage modelling
Two-stage modelling model estimates.
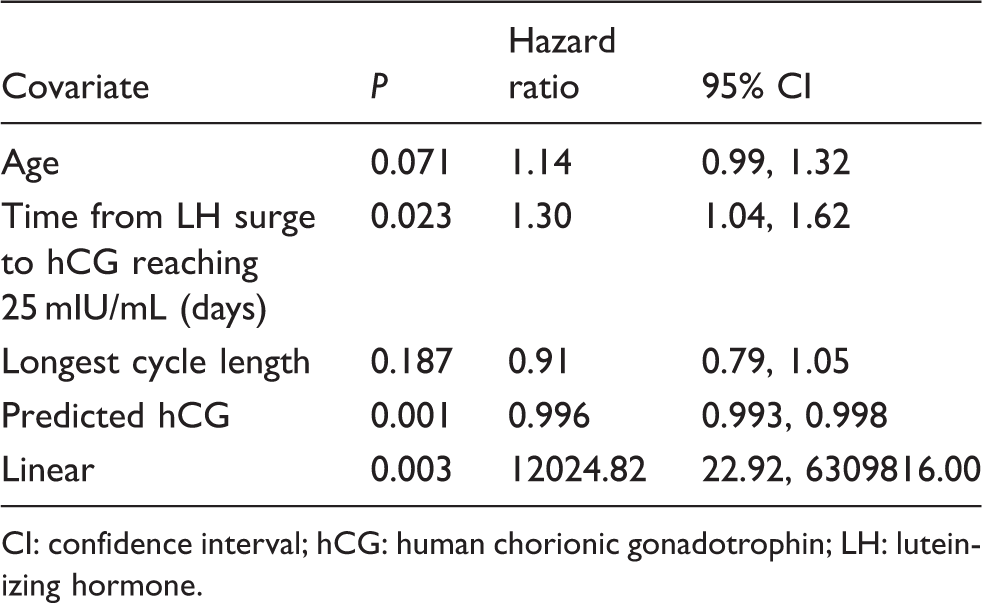
CI: confidence interval; hCG: human chorionic gonadotrophin; LH: luteinizing hormone.
Discussion
The results of this analysis provide new evidence that the risk of early miscarriage can be identified independently by measuring hCG in urine over a period of time and when considered in conjunction with identified risk factors for women. This supports previous studies that have identified that serum hCG also has similar utility in the identification of failing pregnancies.24,25 The hCG profiles differed between women with viable pregnancies, those with early biochemical loss (loss before 6 weeks, recognized by a rise and fall of hCG) and those suffering an early miscarriage (loss six weeks or later). Utilization of the two-stage model in this analysis showed that there is evidence that early biochemical loss and healthy pregnancy groups can be distinguished from one another. However, there is a large variation within the groups; thus, whilst some miscarriages were clearly identifiable from a viable pregnancy at a very early stage, others had an hCG trajectory that did not markedly differ until the loss was in process. This variation presents problems with regard to defining hCG standard cut-off values to determine pregnancy viability. However, it highlights that there is utility in daily tracking of hCG concentrations for individuals to evaluate if a pregnancy is failing. This could potentially have clinical utility for women who have suffered repeated miscarriage, to offer reassurance of a viable pregnancy if hCG concentrations are observed to increase normally, or to alert concern if a pregnancy appears to be failing.
Important covariates of risk of miscarriage identified in this analysis were maternal age, longest reported cycle length and time from LH surge to hCG reaching a concentration of 25 mIU/mL. The concentration of 25 mIU/mL of hCG in urine was chosen because this is the concentration of hCG that is indicative of a normal pregnancy at approximately three days before the expected period.14,29 While spontaneous miscarriages occur with concentrations below 25 mIU/mL,1–3 this concentration is typical for home and clinic urine pregnancy tests; thus, pregnancy is not usually confirmed below this concentration. In particular, the time from the LH surge to hCG reaching 25 mIU/mL was found to be a significant prognostic factor for determining risk of miscarriage. An increase of one day in the time from LH surge to hCG reaching 25 mIU/mL was calculated to increase the risk of miscarriage by 44%. These results support previous work that has indicated that a delay in implantation is associated with risk of early biochemical pregnancy loss and fetal size at 10–14 weeks.29,30 It also highlights that these pregnancies show early signs of being non-viable from conception.
The finding that maternal age is a risk factor for pregnancy loss in this analysis is in agreement with previous studies5,31–33 and serves to both corroborate the other findings in this analysis and confirm the accuracy of the techniques used in this analysis. The association between longest reported cycle length and risk of miscarriage is not as strong as the previously discussed co-variates and is possibly an indicator of reproductive age, as cycle lengths have been observed to change in length in women approaching menopause.34,35
In this analysis, the difference between the longest and shortest cycle lengths was not a statistically significant covariate, in contrast to a previous study. 36 In this study, cycle lengths were reported by volunteers and are thus subject to recall error; further studies with confirmed accurate cycle length data might therefore be of interest. Further possible limitations of this study include the fact that urinary hCG concentrations were not normalized using creatinine to account for the concentration of the sample, and this may have some impact on the results of the analysis. Unfortunately, corresponding creatinine concentrations were not available for these women, and thus it was not possible to undertake this analysis. However, creatinine adjustment has been found to be unnecessary for the determination of a specific hormone on a given day, and creatinine has been observed to introduce an error in older women, due to an observed decline in creatinine clearance with age.37,38 Furthermore, in the clinical situation of women presenting with a suspected failing pregnancy, or monitoring pregnancy viability, urinary tests used do not typically correct for creatinine. Thus, the results for creatinine correct values would not relate as accurately to the clinical situation. Another limitation is the fact that the collection of urine in this study only continued for approximately 28 days postconception; thus, it was not possible to analyse the hCG profile in women who miscarried after this time and compare them with viable pregnancies to see if they could be distinguished. Furthermore, a number of women were missing hCG measurements for some of the study days. Other concentrations of hormones not analysed in this study could similarly be indicative of viable pregnancies or biochemical loss/early miscarriage, e.g. progesterone. Therefore, studies looking at the concentrations of these hormones and risk of miscarriage would also be of interest. A further limitation of this study was that the two-stage modelling approach was applied in two stages, in which the longitudinal and time-to-event models were fitted and then combined. A joint modelling approach to fit the longitudinal model and survival model within one procedure can reduce bias in the overall effects. 39
Conclusion
In conclusion, this analysis shows that an increase of one day in the time from LH surge to hCG reaching 25 mIU/mL increases the risk of miscarriage by 29.7%, thus showing that the hCG rise in early pregnancy can be indicative of pregnancy viability. Daily tracking of urinary hCG could enable early identification of miscarriage in some cases. This could potentially have clinical utility for women who have suffered repeated miscarriage, to offer reassurance of a viable pregnancy if hCG concentrations are observed to increase normally, or to alert concern if a pregnancy appears to be failing. Use of urine samples for longitudinal hCG assessment is likely to be far more acceptable than using serum samples and is likely to reduce any associated cost and discomfort burdens.
Footnotes
Acknowledgements
Dr Debra Scates from integrated medhealth communication (imc) provided medical writing assistance in the development of this manuscript, supported by SPD Development Co., Ltd. R. Evans, S. Newman, C. Shreeves, D. Jeans, D. Roughley, K. Bradford provided clinical trial conduct, sample preparation and measurement and data management. KRA and MJC were partially supported by the National Institute for Health Research as senior investigators (NF-SI-0512-10159) for their contributions to this study.
Declaration of conflicting interests
SJ and LM are employees of SPD Development Co., Ltd (Bedford, UK) funded this study. MZ has received consultancy from SPD Development Co., Ltd, not relating to this study. KA and MC have no competing interests to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted by SPD Development Company Ltd.
Ethical approval
Institutional review board approval was obtained from Quorum Review Committee on 30th November 2009, ref number: 24861.
Guarantor
SJ.
Authors’ contributions
LM conducted the statistical analysis and mathematical modelling, drafted and reviewed the manuscript. SJ developed the sample collection protocol, managed the clinical study and reviewed the data and manuscript. MZ reviewed the data and manuscript. KA and MC provided support with the statistical analysis and reviewed the data and manuscript.
