Abstract
Background
AnnexinA2 (AnxA2) membrane deposition has a critical role in HB-EGF shedding as well as IL-6 secretion in breast cancer cells. This autocrine cycle has a major role in cancer cell proliferation, migration and metastasis. The objective of the study is to demonstrate annexinA2-mediated autocrine regulation via HB-EGF and IL-6 in Her-2 negative breast cancer progression.
Methods
Secretory annexinA2, HB-EGF and IL-6 were analysed in the peripheral blood sample of Her-2 negative (n = 20) and positive breast cancer patients (n = 16). Simultaneously, tissue expression was analysed by immunohistochemistry. The membrane deposition of these secretory ligands and their autocrine regulation was demonstrated using triple-negative breast cancer cell line model.
Results
Annexina2 and HB-EGF expression are inversely correlated with Her-2, whereas IL-6 expression is seen in both Her-2 negative and positive breast cancer cells. RNA interference studies and upregulation of annexinA2 proved that annexinA2 is the upstream of this autocrine pathway. Abundant soluble serum annexinA2 is secreted in Her-2 negative breast cancer (359.28 ± 63.73 ng/mL) compared with normal (286.10 ± 70.04 ng/mL, P < 0.01) and Her-2 positive cases (217.75 ± 60.59 ng/mL, P < 0.0001). In Her-2 negative cases, the HB-EGF concentrations (179.16 ± 118.81 pg/mL) were highly significant compared with normal (14.92 ± 17.33 pg/mL, P < 0.001). IL-6 concentrations were increased significantly in both the breast cancer phenotypes as compared with normal (P < 0.001).
Conclusion
The specific expression pattern of annexinA2 and HB-EGF in triple-negative breast cancer tissues, increased secretion compared with normal cells, and their major role in the regulation of EGFR downstream signalling makes these molecules as a potential tissue and serum biomarker and an excellent therapeutic target in Her-2 negative breast cancer.
Introduction
Breast cancer is one of the most common cancers affecting the women worldwide in all age groups. In India, cervical cancer was the leading cause of cancer in women till recently. Since 2008 estimated breast cancer incidence has surpassed cervical cancer, which has increased by more than 20%, while mortality has increased by 14%.1,2
Breast cancer is a heterogeneous disease having distinct biological subtypes with many targeted biomarkers having therapeutic implications. Hormone receptor analysis is a standard way in the management of breast cancer patients. oestrogen receptor (ER) positivity predicts response to endocrine therapy such as antioestrogen (tamoxifen) and trastuzumab therapy (Herceptin) for tumours with Her-2/neu over-expression. 3
However ER, progesterone receptor (PR) and Her2-neu are not expressed in up to 40% of women with breast cancer in India.4–6 Tumours which are negative for ER/PR and Her-2 are of great concern nowadays as there are no established biomarkers. Recent studies on molecules like ERβ, 7 osteopontin 8 and PARP inhibitors 9 have raised hopes of getting specific targeted therapy in this phenotype. These triple-negative breast cancers (TNBCs) form a distinct group, because they affect younger patients and are more aggressive when compared with non-TNBC. They are unresponsive to usual endocrine therapies with poor clinical outcome and short survival. There is an urgent need to identify new biomarkers in these Her-2 negative breast cancer patients that can be used as diagnostic tools and provide targets for therapeutic intervention.
Our previous studies show abundant expression of a calcium-dependent phospholipid binding protein Annexin A2 (AnxA2) in TNBC cells and clinical tissue specimens as compared with normal and Her-2-amplified breast cancer.10,11 AnxA2 is upregulated in various tumour types and has multiple roles in regulating cellular functions, including proliferation,11,12 apoptosis,13,14 angiogenesis, 15 cell migration, 11 invasion 16 and adhesion, 17 which are essential for cancer metastasis. It is an intracellular protein in its monomeric form, while the heterotetramer with p 11 proteins is localized on plasma membrane. It has also been shown to be secreted by a non-classical secretory pathway.18,19 AnxA2 associates with membrane EGFR complex and mediates the Src-mediated downstream signalling pathways, and hence it may be a therapeutic target in Her 2 negative breast cancer.19,20 In the extracellular environment, AnxA2 is secreted as soluble and membrane-bound tetramer form. 21
Disintegrin and metalloprotease family protease ADAM 17 is responsible for ectodomain shedding of more than 100 transmembrane proteins including EGFR ligands like transforming growth factor α (TGF α), amphiregulin, and heparin binding epidermal growth factor (HB-EGF). 22 Including these ligands, some soluble receptor domains like IL-6 receptor also get released in a process called induced ectodomain shedding. 23 The phosphorylated AnxA2 at the cancer cell surface interacts with ADAM 17 and induces this shedding process. 24 There are also reports that knockdown of AnxA2 inhibits the IL-6 secretion in prostate cancer cells. 25 The membrane association of AnxA2 can be induced by IL-6 in cancer cells, 26 and IL-6-mediated NF-kB signalling requires intracellular AnxA2 in the development of resistance in pancreatic cancer cells after gemcitabine treatment. 27 The released HB-EGF can bind EGF receptor and in turn induce the secretion of all the three molecules.22,28 Even though AnxA2 can mediate the ADAM 17 protease shedding of many ligands and soluble receptor domains, for our current study, we picked IL-6 and HB-EGF to analyse the autocrine regulation with AnxA2 in Her-2 negative breast cancer.
Robust expression of HB-EGF in stroma and endothelium is seen during the progression of various cancers. 28 In cancer microenvironment, this stromal HB-EGF paracrine effect has a major role in cell proliferation, 29 progression and metastasis. 30
IL-6 is involved in the host immune defence mechanism as well as the modulation of growth and differentiation in various malignancies. 31 These effects are mediated by several signalling pathways, in particular the signal transducer and transcription activator 3 (STAT-3). 32 Clinical studies have revealed that increased serum IL-6 concentrations in patients are associated with advanced tumour stages of various cancers and short survival in patients. Therefore, blocking IL-6 signalling is a potential therapeutic strategy for cancer. 33
With this background, we hypothesize that AnxA2 and its downstream molecules like HB-EGF and IL-6 are co-expressed in Her 2 negative breast cancer cells. In the current study, we will be referring TNBC as Her-2 negative, as we have established in our earlier study that, our molecule of interest, AnxA2 and Her-2 are reciprocally regulated and AnxA2 interaction with EGFR has a critical role in the downstream regulation and progression of Her-2 negative breast cancer. We will be correlating the expression of AnxA2 and its downstream molecules HB-EGF and IL-6 in Her-2 negative and Her-2 positive breast cancer.11,12 We also propose to study the secretory status of AnxA2, HB-EGF and IL-6 to confirm the cell line and tissue expression analysis and the same secretory molecules may be helpful as serum biomarkers and excellent therapeutic targets in Her 2 negative breast cancer.
Materials and methods
Sample collection
The serum/tissue samples were obtained from Department of Pathology, SDM College of Medical Sciences, as well as from Karnataka cancer Therapy and Research Institute, Hubli as per established core procedures and Institutional Ethical Board approval.
Study subjects
Serum samples for soluble AnxA2, IL-6 and HB-EGF analysis were collected from 40 breast cancer patients, and 10 study participants were healthy and disease-free. Out of 40, 16 were grouped as Her-2 positive breast cancer and 20 were Her-2 negative breast cancer as identified by the panel of pathologists after the IHC staining. None of the patients and age-matched normal controls was receiving hormone replacement therapy or any cancer therapy at the time of sample collection. Any person who had received any type of therapy ≤ 6 months before the enrolment was disqualified for this study. Five millilitres of blood was collected from each individual and centrifuged at 800 g for 10 min and the serum fraction was separated, aliquoted, and used for the regular screening tests. The spare sample was used for the current study, which was stored at −80℃ in the Central Research Laboratory repository until further use. Collection, preparation, and storage of the blood samples were done by using guidelines set by the ethical committee. Qualified personnel obtained informed consent from each individual participating in this study.
Serum analysis using enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assay (ELISA) assay kits for analytes AnxA2 and HB-EGF were purchased from USCN Life, WUHAN EIAAB SCIENCE CO, LTD, IL-6 kit from Gen Probe Diaclone SAS, France. Assays were performed following kit instructions. Plates were read on an Epoch, Biotek instrument microplate reader with the appropriate baseline correction for each assay. Each sample was analysed in duplicates to minimize experimental errors. With each experiment, one set of standard (purified AnxA2, HB-EGF or IL-6) was included.
Statistical analysis
We have carried out t-test to find out whether breast cancer with different groups varies in mean AnxA2 or HB-EGF concentration separately. We also did a one-way ANOVA model to test, if mean measurements differ across these groups: normal, HER-2 positive or Her-2 negative. Serum IL-6 is a known prognosticator in various cancers. With serum IL-6, we have performed sensitivity statistical analysis comparing serum AnxA2 and HB-EGF to correlate the predictability of these molecules as biomarker for Her-2 negative breast cancer.
Immunohistochemistry
Tissue samples were stained with hematoxylin–eosin to determine the histological type and grade of tumours. More accurate subtyping of these categories could not be assessed without complete gene profiling studies. In the current study, Her-2/neu scores of 2+ were considered positive because this was the accepted classification scheme at the time of clinical treatment. Therefore, Her-2/neu-positive expressing breast cancer subtypes were compared against with those which are negative for all the known biomarkers (TNBC) but called as Her-2 negative. AnxA2, IL-6, HB-EGF and Her-2 protein concentrations in the 45 tissue samples from breast cancer patients, including cancerous tissue and adjacent non-malignant epithelium, were detected using immunohistochemical staining according to the method previously described. 34 In brief, after deparaffinization and endogenous peroxidase blockage, the sections were heated in 0.01 M citrate buffer solution (pH 6.0) in water bath at 98℃ for 20 min, then incubated with the mouse monoclonal antibody to AnxA2 (BD Biosciences, CA, Catalogue no. 610069), IL-6, Cat. No. SC-130326, HB-EGF, Cat. No. SC-74441 (Santacruz Biotechnology, CA) and Her-2 (Invitrogen Cat. No. AH01011) at 1:100 dilution overnight at 4℃, and visualized using 3,3′-diaminobenzidine (DAB) detection kit (Vector labs). The 1:100 dilution was determined to be the best dilution during immunohistochemistry (IHC) optimization. For the negative control, antirabbit and antimouse IgG whole molecule (Sigma–Aldrich) was used at 1:1000 dilution. IHC stained samples were evaluated by two pathologists, and all samples were blinded. Staining intensity of AnxA2, IL-6, HB-EGF and Her-2 in neoplastic cells was graded on a scale of 0 (no staining) to 3+ (strong staining). The AnxA2 and other secretory protein positivity were scored based on the percentage of positive cells: 0 = 0% of stained positive cells; 1, weakly stained tissue or 1–25% of positive cells; score 2 = moderate stained tissue, or 26–50% of positive stained cells; and 3, strongly stained tissue or more than 50% of stained cells.
Cell culture
Human breast cancer cell lines, BT-549, MDA-MB-231, SK-BR-3, MCF-7, MCF-10 A and other cell lines were obtained from National Center for Cell Sciences, Pune, India, and grown in respective medium as prescribed by the supplier.
Total cell extraction and Western blotting
For the expression analysis in different breast cancer cell lines, total protein was extracted and quantitated as described previously. 11 Total protein was separated on 10% Bis-Tris PAGE gel using Tris-Hcl buffer. The antibodies were used against AnxA2 (Mouse monoclonal, BD Biosciences, CA, Cat No. 610069), IL-6, Cat. No. SC-130326, HB-EGF, Cat. No. SC-74441 and GAPDH, Cat. No. SC-166574 from Santacruz Biotechnology, CA. Appropriate secondary antibodies conjugated to horse radish peroxidase (BioRad) were incubated with respective membranes for 2 h at room temperature. The membranes were developed using ECL plus (BioRad), and the image was captured using autoradiography. Immunoblot for GAPDH was considered as internal control for loading.
RNA interference
Small interfering RNAs (siRNA) against AnxA2 and scrambled non-specific siRNA were purchased from Santacruz Biotechnology. To obtain effective silencing of protein expression, we utilized the SMARTpool siRNA reagent, which is a combination of four SMART selection-designed siRNAs in a single pool. Transfection was done following the manufacturer’s instruction using the Santacruz transfection reagent.
Results
Correlation of AnxA2, HB-EGF and IL-6 expression in Her-2 negative breast cancer
The collected tissue samples were analysed for the receptor expression status (Her-2, ER and PR) by IHC. According to the receptor status, the samples were classified as receptor positive (e.g. Her-2 positive) and TNBC. During the tissue collection, brief patient history was also taken like age, sex, tumour size, lymph node involvement, frank or previously treated, family breast cancer history, etc. Further, these tissue samples in various groups like normal, Her-2 positive and TNBC samples were analysed for AnxA2, IL-6 and HB-EGF expression. AnxA2 expression increases with the progression of breast cancer in TNBC cases, as this expression pattern of AnxA2 is already reported in our earlier publications, data are not shown here.11,12 Out of 45 samples, which we have tested, in majority of the cases where Her-2 expression is high, AnxA2 expression is low (Figure 1(b), P = 0.002) except in very few cases of poorly differentiated terminal stage cancer. HB-EGF and IL-6 expression pattern follows AnxA2 protein concentration in these TNBC samples. IL-6 and HB-EGF expression in normal samples is low or near null. The expression pattern of these proteins increases in TNBC with AnxA2 but compared with the intensity of AnxA2 staining, the expression is little low but significantly higher than Her-2 positive cases as shown in Figure 1(a). The expression pattern of HB-EGF associates with AnxA2 (Figure 1(b), P = 0.002); on the contrary, IL-6 expression increases with the progression of the breast cancer rather than associating with any upstream molecular expression in different breast cancer subtypes, which augur the IL-6 involvement in multiple molecular pathways.
AnxA2, HBEGF and IL-6 expression in clinical tissue samples. (a) Representative breast cancer tissue image from total 45 clinical samples, showing increased expression status of AnxA2 and HBEGF in triple-negative breast cancer and low staining in Her-2 positive breast cancer. Paraffin-embedded tissue sections were stained with AnxA2 antibody showing 3+ expression (strong membranous staining in >30% cells in Her-2 negative breast cancer tissue section) and HBEGF antibody showing 2+ expression (membrane staining in 0–30% cells) whereas null to low staining was observed in Her-2 positive breast cancer, and thick staining of IL-6 was observed in both the phenotypes. (b) Stacked bar graph showing immunohistochemical staining pattern of AnxA2, HB-EGF and IL-6 with Her-2 in total 45 breast cancer samples.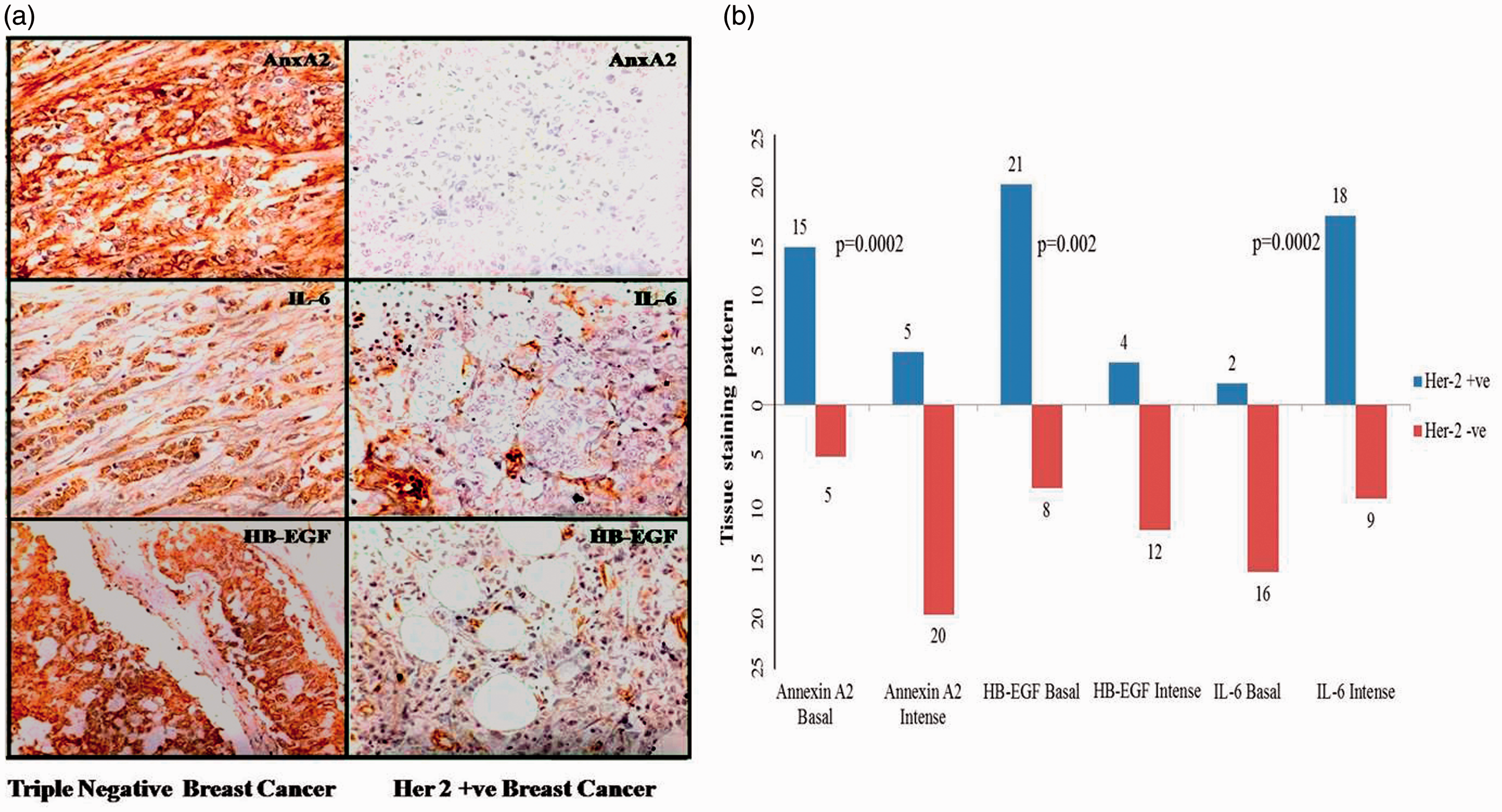
TNBC cells express high membrane localization and secretion of AnxA2
AnxA2 expression is very high in TNBC cells compared with other Her-2 amplified and ER-expressing cells. There are earlier reports suggesting increased localization of AnxA2 in the extracellular surface, and it is implicated in the activation of plasminogen to plasmin. It is believed that plasminogen activation by AnxA2 is critical for ECM degradation and further carcinogenesis. In this particular study, we wanted to verify whether the increased expression of AnxA2 in TNBC mobilizes it to the membrane and its secretion to the extracellular space. Breast cancer cell lines were grown to 60% confluency and were switched to serum free medium for 24 h at which time the medium was collected and concentrated to get the 25% of the earlier volume and analysed for the secreted AnxA2 using Western blot assay (Figure 2(a)). In another experiment, we compared the extracellular membrane localization of AnxA2 in TNBC cells with normal breast epithelial cells. The same number of cells were seeded and grown in respective medium. Once the cells reached 90% confluence, adherent cells were washed in phosphate buffered saline (PBS) twice and minimal amount of versene was added and kept in incubator for 15 min. After the incubation, the versene wash (membrane wash, MW) containing the membrane-associated proteins was collected, cells were washed in PBS and lysed using NP40 lysis buffer. Both versene wash and cell lysate (CL) and secreted AnxA2 in conditioned media (CM) concentrations were screened using Western blot assay. We found that in TNBC cells large amount of AnxA2 is translocated to membrane and importantly this will be present at the extracellular surface (Figure 2(a)). In Her-2 amplified and ER+ cell lines, because of low expression, minimal amount of AnxA2 will be localized in the extracellular surface resulting in decreased secretion. In normal cells (MCF-10 A), even though the expression of AnxA2 is comparable to TNBC cells, membrane localization is very less (Figure 2(b)). In TNBC cells, the secreted AnxA2 shows two bands representing higher molecular weight phosphorylated proteome.
21
This underscores the importance of studying secreted and membrane localized AnxA2 in TNBC.
AnxA2 membrane localization and secretion in breast cancer cells. (a) HER-2 amplified cell lines SK-BR-3 and HCC-1569; TNBC cell lines HCC-38, MDAMB-231, HCC-1187; and Her-2 null/low with ER+ cell lines MCF-7 and HCC-1500 were grown in their respective media as prescribed by the ATCC. Once these cells became 60% confluent, they were switched to serum-free medium for 24 h at which time the medium was collected and concentrated to get the 25% of the earlier volume and analysed for the secreted AnxA2 using Western blot assay. (b) In another experiment, we compared the extracellular membrane localization of AnxA2 in TNBC cells (MDA MB-231) with normal breast epithelial cells (MCF-10 A) and Her-2 amplified cell line SK-BR-3. Confluent cultures were washed with 0.5 mM EDTA and PBS buffer (Versene, Himedia) for 20 min at 37℃, and the membrane protein fraction in the supernatant was designated EDTA eluate or membrane wash (MW). This membrane wash, respective cell lysate (CL) and media supernatant (CM) were analysed for AnxA2 by Western blot. The Western blot experiments were performed three times, and the representative data were shown in the figure. Simultaneously, densitometric scanning was performed with Western blot data, and the semi-quantitative bar graph shows the respective band intensity of the AnxA2 membrane wash (MW), cell lysate (CL) and conditioned media (CM) of the different cell lines.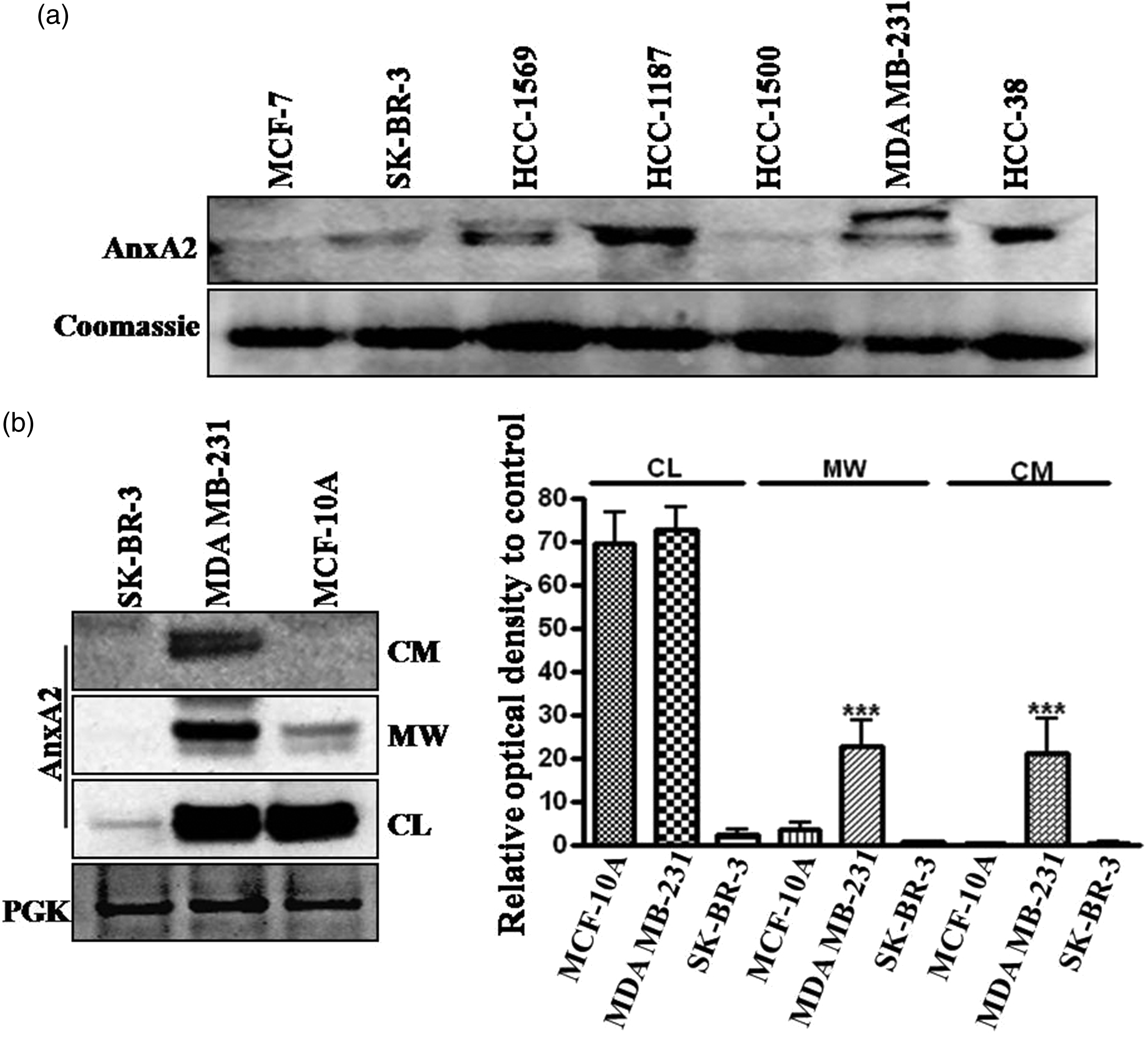
AnxA2 is the upstream molecule for HB-EGF and IL-6
In our earlier study, we have reported that TNBC cell lines like MDA MB-231 and BT-549 express high AnxA2, and in MCF-7 and SK-BR-3, the concentration is very low. In these cell lines, we have also analysed the cellular expression of IL-6 and HB-EGF, which showed similar pattern as that of AnxA2. The cell supernatant also contains very high quantity of HB-EGF and IL-6 with AnxA2 in TNBC cell lines compared with Her-2 and ER positive cell lines as shown in Figure 3(a). The IL-6 differential secretion is not that significant, which is also represented in our serum and tissue concentration as compared with AnxA2 and HB-EGF.
AnxA2, HBEGF and IL-6 protein expression in cell surface of breast cancer cell lines. (a) Her-2 negative MDA MB-231 and ER expressing MCF-7 cell lines were grown for 90% confluence, and the membrane wash was collected by treating EDTA (Himedia) for 20 min at 37℃. The cell surface eluates were concentrated and immunoblotted for AnxA2 using a rabbit anti-AnxA2 antibody, HBEGF and IL-6 antimouse antibodies. To normalize for loading, an SDS-PAGE was performed with identical concentration of protein and the gel was stained with coomassie brilliant blue. A 50 kDa coomassie stained band was used as protein loading. Data presented are representative of three independent analyses. (b) Her-2 negative MDA MB-231 cells were transfected with non-specific siRNA or AnxA2 siRNA. Cells were collected at different time points after transfection, lysed and analysed by Western blotting with AnxA2, HB-EGF and IL-6 antibodies. The GAPDH was used as loading control. The experiments were repeated three times with identical results. (c) EGFR activation by EGF increases AnxA2 expression with HB-EGF and IL-6 in MCF-7 cells. MCF-7 cells were serum starved for 12 h and treated with EGF (100 ng/mL) for different time intervals. AnxA2, HB-EGF and IL-6 expression were analysed by Western blot. Representative data were shown after performing the experiments three times.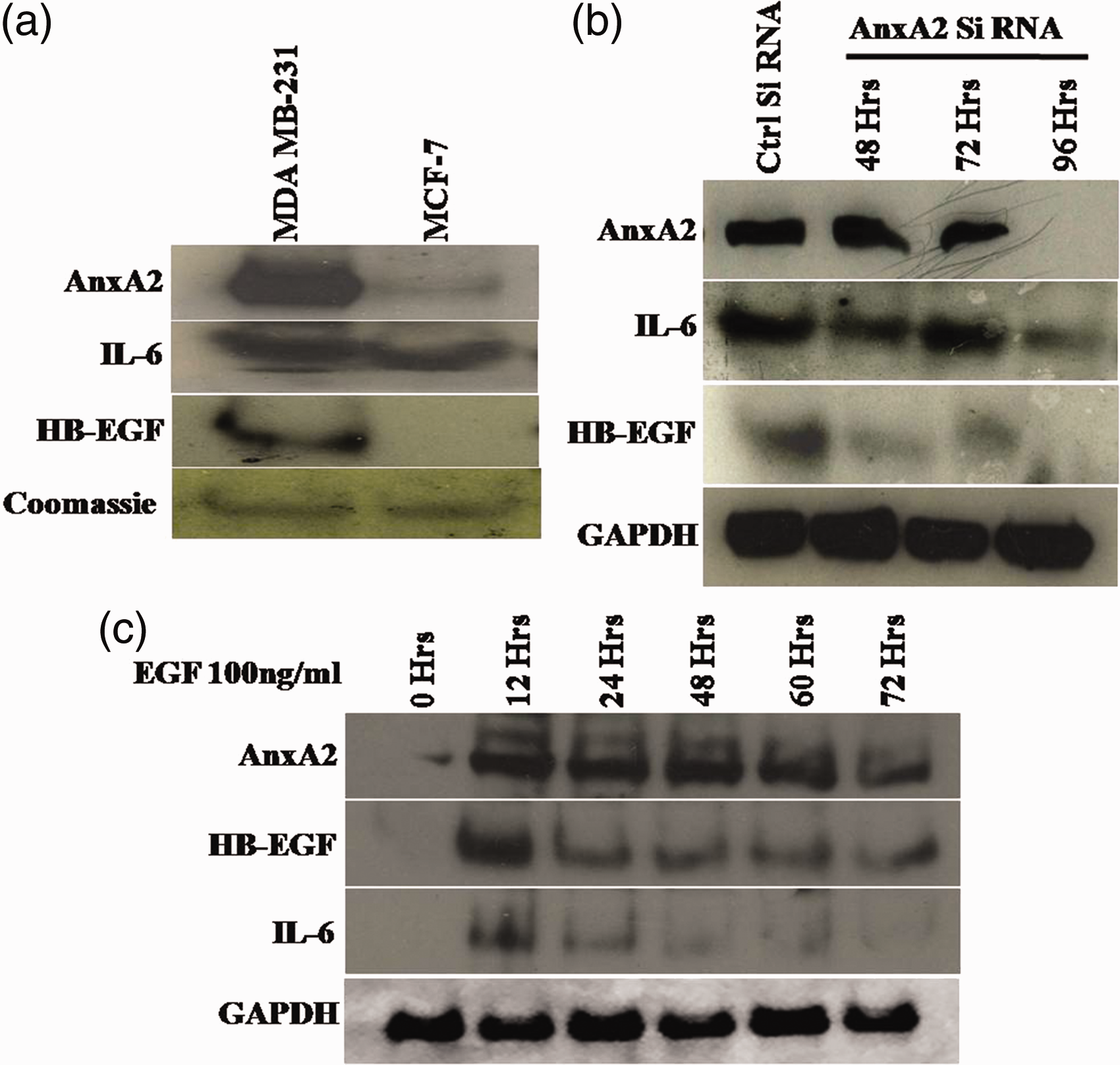
The membrane association of AnxA2 and its interaction with EGFR, further its role in the regulation of EGFR downstream signalling made us to verify whether IL-6 and HB-EGF are the downstream molecules of AnxA2. siRNA-mediated AnxA2 downregulation decreased the expression of both IL-6 as well as HB-EGF as shown in the Figure 3(b). The relative decrease in the expression of HB-EGF is more prominent compared with IL-6 as IL-6 expression is regulated by multiple pathways, and HB-EGF release is directly dependent on phosphorylated AnxA2 association with ADAM-17.23,24 On the other hand, EGF-mediated induction of AnxA2 also augmented the expression of IL-6 and HB-EGF, which is shown in Figure 3(c).
Serum screening of AnxA2, IL-6 and HB-EGF in breast cancer
Comparison of serum AnxA2 concentrations determined by ELISA method between healthy volunteers and patients of different phenotypes of breast cancer.
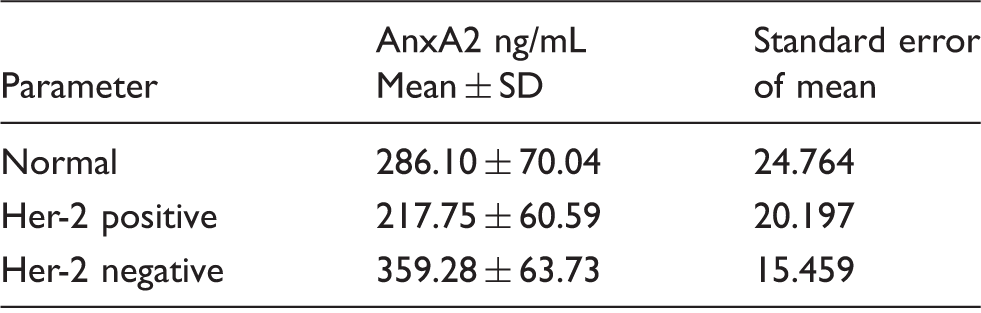
Note: AnxA2 concentrations in normal, Her-2 negative and positive.
Her-2 negative and normal: P < 0.01.
Her-2 negative and Her-2 positive: P < 0.0001.
Her-2 positive and normal: P < 0.05.
Soluble secreted HB-EGF concentrations determined by ELISA method between healthy volunteers and patients of Her-2 negative and Her-2 positive breast cancer.

Note: HB-EGF concentrations in normal, Her-2 negative and positive.
Her-2 negative and normal: P < 0.001.
Her-2 negative and Her-2 positive: P < 0.20.
Her-2 positive and normal: P < 0.01.
Serum IL-6 concentrations in breast cancer subtypes, Her-2 negative breast cancer and Her-2 amplified breast cancer compared with healthy volunteers.

Note: IL-6 concentrations in Normal, Her-2 negative and positive.
Her-2 negative and normal: P < 0.01.
Her-2 negative and Her-2 positive: P < 0.10.
Her-2 positive and normal: P < 0.0001.
Discussion
Breast cancer incidence has surpassed cervical cancer in last few years in India. The identification of different agents against hormone receptors has reduced the mortality rate in these women. This therapeutic approach has been proved futile in Indian scenario, as Indian women present aggressive phenotypes with Her-2 amplification or basal breast cancer. Her-2 amplified breast cancer initially responds to Herceptin treatment but later-on develop resistance. In basal breast cancer, all the known biomarkers are negative leaving the only option of chemotherapy. Early detection and understanding the mechanism of cancer progression in these phenotypes will enable in extending the survival ability of these patients.
Current research involving breast cancer focuses on validating the secretory autocrine molecules in the differential diagnosis of different breast cancer subtypes and in the therapeutic targeting. In our previous studies, we have demonstrated the role of EGFR in the progression of Her-2 negative breast cancer. We have identified the critical role of phospholipid-binding protein AnxA2 in the EGFR-mediated downstream signalling and in the further progression of breast cancer.10,11 In the present study, we are validating the role of soluble secreted AnxA2 in the autocrine regulation, and also to correlate with other soluble ligands like HB-EGF and cytokine IL-6. HB-EGF is preferentially expressed in tumours expressing EGFR. EGFR-mediated intracellular signalling controls the functions required for growth, migration and proliferation. EGFR ligands located on the cell membrane are proteolytically cleaved and released upon action of various stimuli including another growth factor interaction, cytokine action, ultraviolet light, etc. HB-EGF is synthesized in its precursor form pro-HBEGF. EGFR activation is induced by soluble HB-EGF, which is released by ectodomain shedding in a paracrine manner.22–24, 35–39
Present study reports abundant expression of EGFR ligand HB-EGF and critical EGFR interacting molecule AnxA2 in Her-2 negative breast cancer tissue samples. EGFR downstream inflammatory cytokine and NF-kB pathway activating ligand IL-6 increase with progression of breast cancer, but phenotype-specific expression was not found. In TNBC, membrane expression and secretion of soluble AnxA2 and HB-EGF are more, whereas IL-6 expression and secretion are seen even in ER-positive MCF-7 cells, which go well with clinical tissue expression pattern. Current study data demonstrate that the suppression of AnxA2 downregulated HB-EGF and IL-6 expression in TNBC cell MDA MB-231. ER-positive cell line MCF-7 cells, usually express low HB-EGF and AnxA2, when we induced AnxA2 expression in these cells which intern augmented HB-EGF and IL-6. According to these lines of evidence, it is plausible that AnxA2 expression and its interaction with EGFR at the membrane surface is a must to regulate downstream autocrine factors HB-EGF and IL-6. 24
The cellular expression, membrane deposition and proteolytic release of these autocrine factors to the cancer microenvironment are high in Her-2 negative breast cancer phenotypes. To validate the possibility of these soluble factors as useful differential diagnostic biomarkers in breast cancer subtypes, we measured soluble HB-EGF, AnxA2 and IL-6 concentrations in serum samples obtained from healthy volunteers and patients of various breast cancer subtypes. We demonstrated a statistically significant difference between these autocrine factors (HB-EGF, AnxA2 and IL-6) of healthy volunteers and patients in different breast cancer subtypes. Furthermore, HB-EGF and AnxA2 are increased in Her-2 negative breast cancer compared with Her-2 positive subtypes. Whereas IL-6 concentration does not show any significant difference between these breast cancer subtypes. However, further analysis with more clinical samples will be able to demonstrate the statistically greater correlation between these autocrine factors in breast cancer subtypes.
It was also reported that with HB-EGF proinflammatory cytokines such as TNF-α, IL-1β, IL-6 are released and these together activate NF-kB pathway. This cytokine autocrine loop is regulated by the transcriptional activity of NF-kB.40,41 In our previous work, we have shown that AnxA2-EGFR interaction is the upstream of NF-kB pathway and also can regulate IL-6 and IL-8.11,42 EGFR transactivation in TNBC cells mediated by AnxA2, HB-EGF and IL-6 may result in NF-kB activation leading to increased expression of Bcl-2 cell proliferation proteins, tissue catabolising matrix metalloproteinases, uPA and angiogenic VEGF and also further autocrine regulation of same growth factor ligands and chemokines.40,41 The present study data support that Her-2 negative breast cancer clinical tissue specimens express AnxA2, which is more membranous and HB-EGF expression pattern associates with AnxA2. On the other hand, IL-6 expression is not phenotype specific, but expression increases with the progression of breast cancer. An increased concentration of serum IL-6 in some patients with lung cancer has been reported previously and shown to be part of an inflammatory response. Patients who exhibited autoantibodies to annexins I and II had significantly higher IL-6 serum concentrations than any other comparison groups, which suggest that host factors such as cytokines may affect the immune response to potentially antigenic proteins. The autocrine regulation was confirmed by prior finding that IL-6 treatment of A549 resulted in an increased expression and translocation of these annexins to the membrane surface. In turn, an increase in membrane-associated annexins I and II may enhance immunogenicity. 26
To conclude, the cell line data show that AnxA2 and HB-EGF are expressed more in cancer cell membrane, which supports our hypothesis that more ectodomain shedding in these cells release more soluble HB-EGF and AnxA2 to the cancer microenvironment which later enters into systemic circulation further increasing the blood concentration of these ligands in the patient samples. The specific expression pattern of AnxA2 and HB-EGF in TNBC cells increased secretion compared with normal cells, and their major role in the regulation of EGFR downstream signalling makes these molecules as a potential tissue and serum biomarker and an excellent therapeutic target in Her-2 negative breast cancer.
Footnotes
Acknowledgments
The authors would like to acknowledge Mr. Harikrishna Ellanki for technical help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Indian Council of Medical Research (ICMR) ad hoc grant; IRIS No. 09390 to Praveenkumar Shetty.
Ethical approval
The study was conducted following informed consent and was approved by the Ethical Committees of SDM College of Medical Sciences & Hospital, Dharwad, Karnataka, India (Reg. No. ECR/683/INST/KA/2014).
Guarantor
PKS.
Contributorship
Conceived the idea and designed the experiments: PKS. Helped in clinical sample collection and correlated the clinical relevance to the study: BRP, RM, VH, USD. Performed the experiments: PKS, VSP, AB, LCD, SPK. Analysed the data (Pathology): PKS, AB, USD. Analysed the data (Statistical analysis): PKS, VSP, AB. Wrote the paper: PKS, VSP. Supervised the overall study: PKS.
