Abstract
Background
Takayasu arteritis is a chronic non-specific inflammatory disease and mean platelet volume can either be decreased or increased during inflammation. However, there are no published data to confirm an association between mean platelet volume and Takayasu arteritis. Our aim was to evaluate the role of mean platelet volume in patients with Takayasu arteritis.
Methods
A total of 119 consecutive patients with Takayasu arteritis and 217 healthy individuals were included in this study. Forty-five Takayasu arteritis patients with active disease were followed with prednisone therapy.
Results
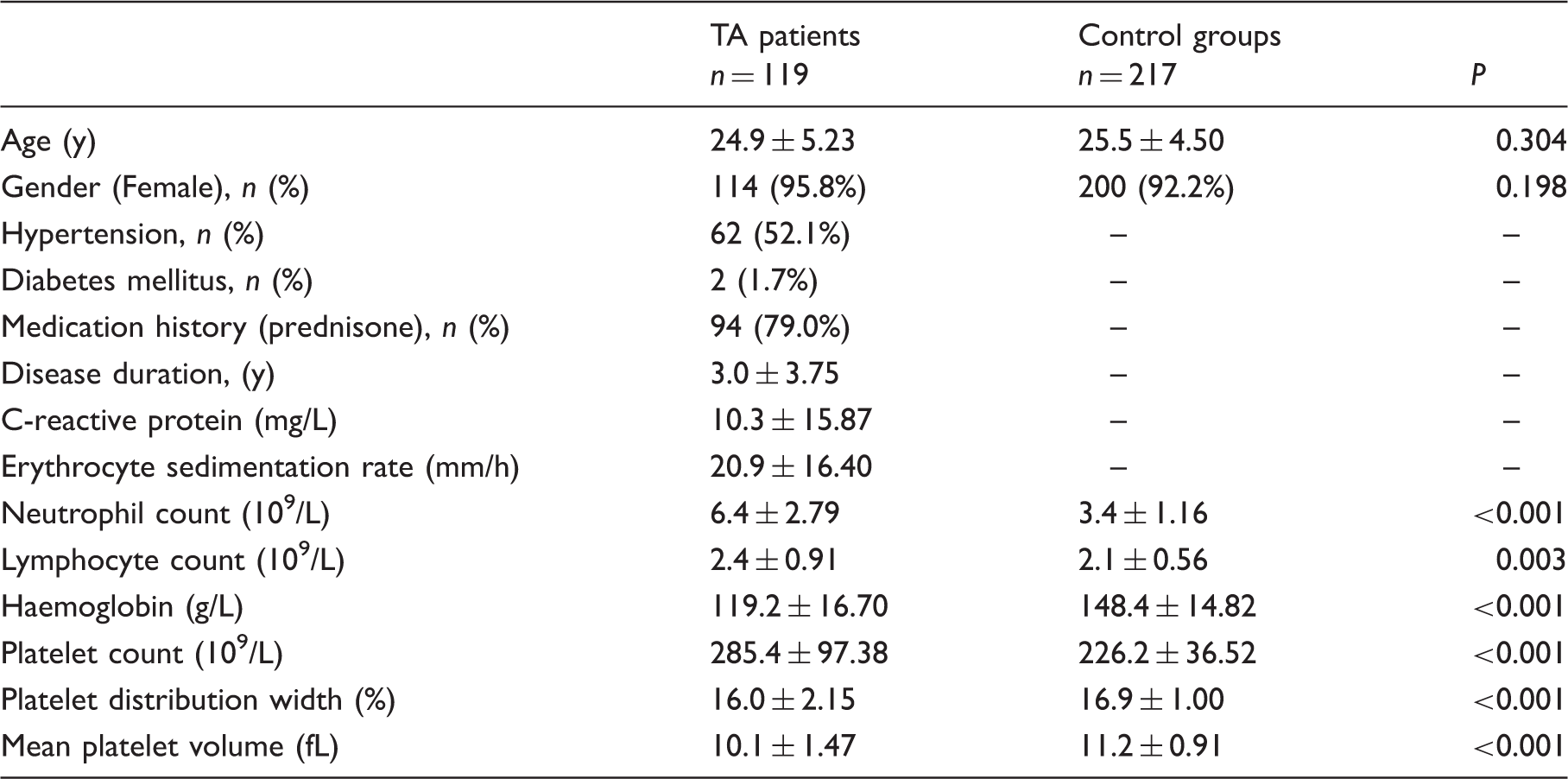
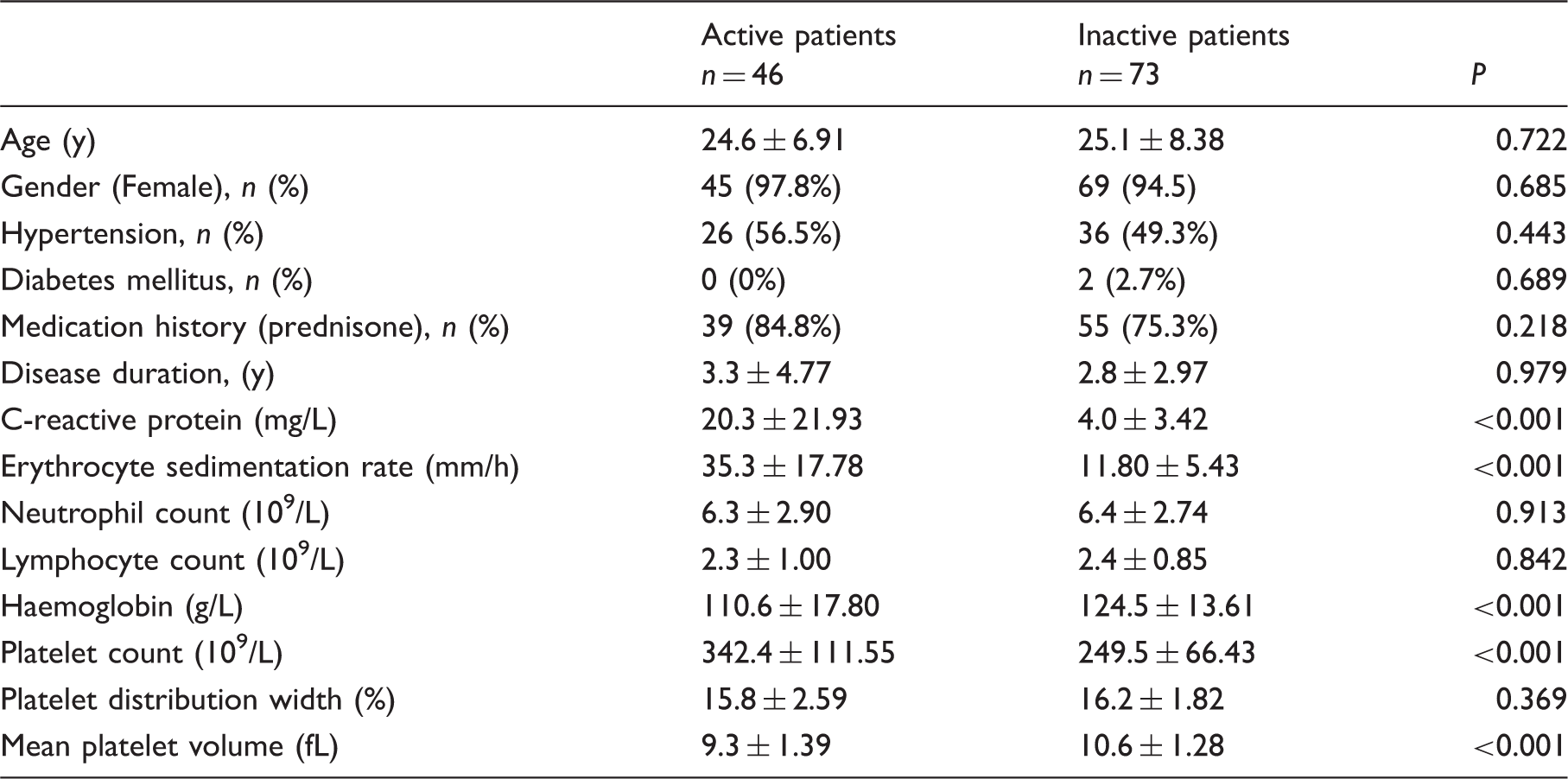
Mean platelet volume of patients was low compared with control groups (10.1 ± 1.47 fL vs. 11.2 ± 0.91 fL; P < 0.001). Mean platelet volume was lower in active Takayasu arteritis than in inactive Takayasu arteritis patients (9.3 ± 1.39 fL vs. 10.6 ± 1.28 fL; P< 0.001). Mean platelet volume values were significantly increased after prednisone treatment (9.3 ± 1.45 fL vs. 10.5 ± 1.29 fL; P < 0.001). Mean platelet volume negatively correlated with C-reactive protein, erythrocyte sedimentation rate, neutrophil count and platelet count (r = − 0.219, P = 0.018; r = − 0.296, P < 0.001; r = − 0.273, P = 0.003; r =−0.486, P< 0.001), and positively correlated with platelet distribution width (r=0.304, P ≤ 0.001) in patients with Takayasu arteritis. An inverse correlation between mean platelet volume and erythrocyte sedimentation rate was observed in active Takayasu arteritis patients (r = −0.406, P = 0.010). In multiple linear regression analysis, mean platelet volume was independently correlated with erythrocyte sedimentation rate in patients with Takayasu arteritis.
Conclusions
Our results suggest that mean platelet volume may identify active disease in patients with Takayasu arteritis, and the values of mean platelet volume may help to establish remission of active disease after treatment in Takayasu arteritis patients.
Introduction
Takayasu arteritis (TA) is a chronic non-specific inflammatory disease with considerable morbidity and mortality in the Asian region. Inflammation primarily affects large arteries such as aorta, pulmonary artery and major branch vessels, 1 and idiopathic inflammation in vessel walls leads to cerebral, organ and limb symptoms of ischaemia. 2 In fact, non-specific inflammatory conditions are inchoate compared with the signs and symptoms of vascular involvement in TA patients. 3 Therefore, markers are needed to monitor and estimate inflammatory states, to predict exacerbations and aid early treatment of the disease. Traditionally, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) have been considered as useful laboratory indices in assessing disease activity for patients with TA, despite the lack of sensitivity and specificity. 4 However, in the clinical laboratory, other available and novel inflammatory markers may also potentially provide additional information for the assessment of inflammatory conditions and disease activity in patients with TA.
Mean platelet volume (MPV) that reflects platelet size and platelet function is a calculated laboratory marker by the complete blood cell counter. 5 Accumulating data have shown that MPV is a potential inflammatory index and tends to decrease or increase during inflammation. It has been shown that increased MPV is an independent risk factor in patients with coronary artery disease. 6 Several recent studies have reported that increased MPV is also associated with supraventricular tachycardia, retinal artery occlusion and adnexal torsion.7–9 On the contrary, Gasparyan et al. 10 found that anti-inflammatory treatment had a significant effect on MPV in patients with rheumatoid arthritis, and decreased MPV values have also been demonstrated in some rheumatic diseases such as systemic lupus erythematosus, inflammatory bowel disease, ankylosing spondylitis and polymyositis.11–14 However, there are no published data investigating an association between MPV and TA. In the present study, our aim was to evaluate the role of MPV in patients with TA.
Materials and methods
Patients and controls
A total of 119 consecutive patients with TA in Affiliated Hospital of Youjiang Medical University for Nationalities from January 2014 to December 2015 period (114 females and 5 males, mean age 24.9 ± 5.23 years) were included in this study. All patients with TA were diagnosed according to the American College of Rheumatology classification criteria. 15 Two hundred and seventeen age- and gender-matched healthy individuals served as controls. Patients with cardiovascular disease, infectious disease, hepatic or renal insufficiency, dyslipidemia, malignancy and other rheumatic diseases were excluded. The study was approved by the Ethics Committee of Affiliated Hospital of Youjiang Medical University for Nationalities, and informed consent was obtained from all participants.
Disease activity assessment
Before entering the study, the National Institutes of Health criteria were used to assess the disease activity of patients with TA. 16 Clinically active disease in TA was defined by at least two of the following features: (1) typical symptoms of the disease; (2) elevated ESR with no infection; (3) vascular insufficiency symptoms and (4) typical angiographic characteristics.
Laboratory parameters
Clinical characteristics of all patients were extracted from the medical records. Laboratory data were recorded at baseline in patients with TA. Fasting blood samples were collected to measure laboratory parameters within 2 h, including neutrophils, lymphocytes, haemoglobin, platelets, platelet distribution width (PDW), MPV, CRP and ESR. Haematological parameters were determined by an automatic blood cell analysis instrument (Beckman Coulter Inc, LH750, USA). ESR and CRP were measured by immunonephelometry and the Westergren method, respectively. All tests were performed on one instrument in a single clinical laboratory, and the reference range for MPV is 7.4–12.5 fL in this laboratory.
Statistical analysis
All statistical analyses were performed by SPSS 16.0 statistical package (SPSS Inc., Chicago, IL, USA). Continuous variables and categorical variables were presented as mean ± standard deviation (SD) and percentages, respectively. We used Student's t-test or Mann–Whitney U test to compare continuous variables, while Chi-square test was used to analyse categorical variables. Correlations between two continuous variables were examined by the Spearman approach. Multiple linear regression analysis was used to identify the association between MPV and other laboratory parameters. To identify active TA in all patients with TA, a cut-off value was calculated by the receiver operating characteristic curve. Paired t test was used to compare laboratory indices before and after treatment. Results were defined as statistically significant if P<0.05.
Results
Patient and control characteristics
Demographic characteristics and laboratory parameters of Takayasu arteritis (TA) patients and controls.
MPV and TA activity
Demographic and laboratory parameters between active patients and inactive patients.
MPV and treatment
Laboratory characteristics of active patients before and after treatment.

MPV and correlation analysis
The concentrations of mean platelet volume as dependently variable in multiple linear regression analysis.
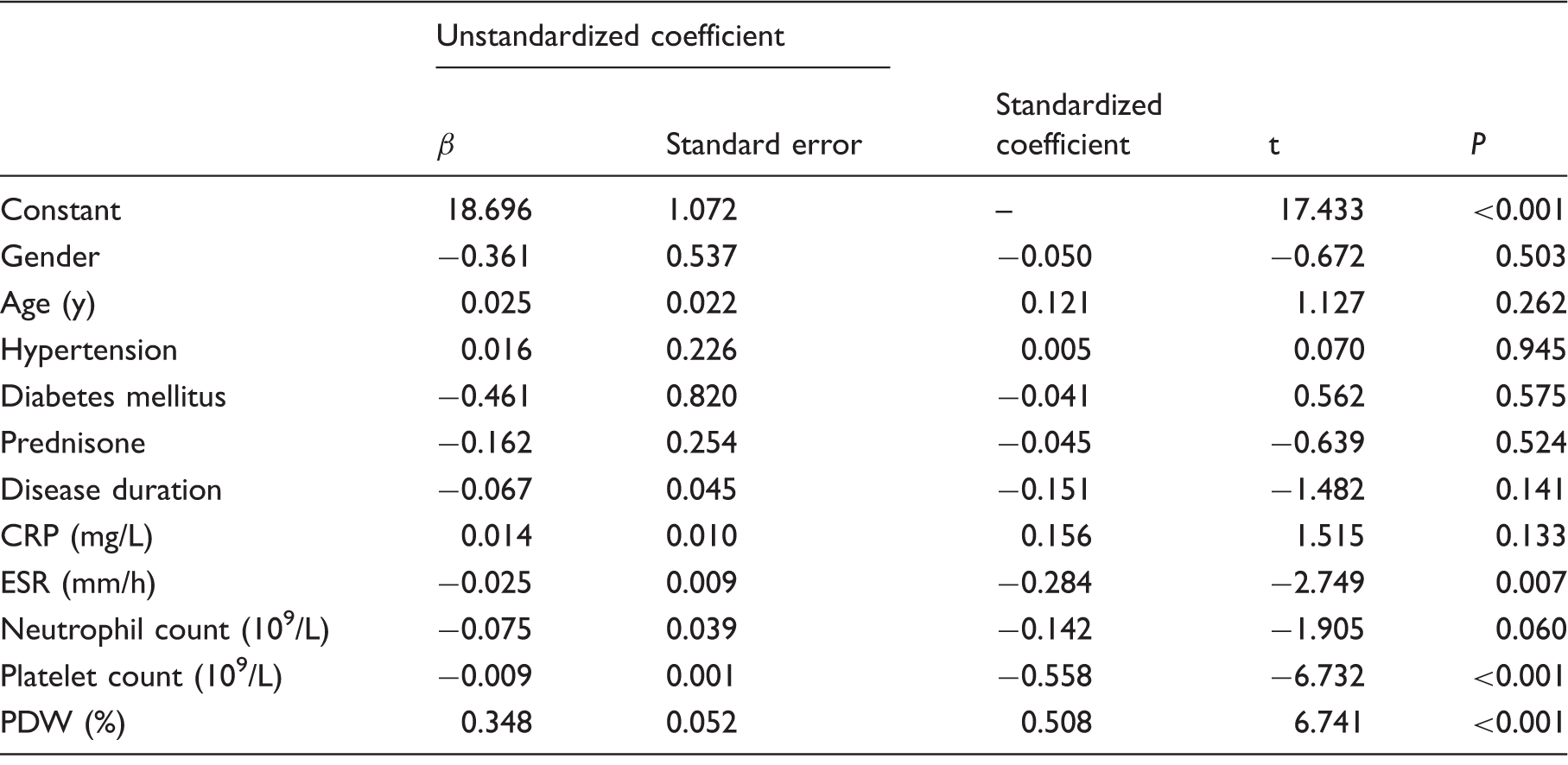
CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; PDW: platelet distribution width.
Discussion
To the best of our knowledge, this is the first study that investigated the potential role of MPV in patients with TA. Our study revealed that MPV was decreased in TA patients compared with controls. Similarly, MPV was low in patients with active TA when compared with those of patients with inactive disease, and the concentrations MPV were increased in patients with active TA after receiving prednisone treatment. Interestingly, in multiple linear regression analysis, a significant correlation between MPV and ESR was found in patients with TA.
Recently, Sert et al. 17 suggested a relationship between decreased MPV and Reye-like syndrome in children. Lower MPV values have been reported in patients with venous thromboembolism, retinal vein occlusion and brain injury.18–20 Blood tests are routinely undertaken during diagnosis and follow-up for rheumatic diseases. MPV as a component in routine blood tests has no additional cost for patients. We demonstrated decreased MPV values in patients with active disease compared with patients with inactive TA, and found MPV was increased in patients with active disease after undergoing prednisone treatment. This suggests an underlying role of MPV in assessing disease activity and distinguishing remission of active disease for TA. Recent advances in the clinical laboratory have developed the role of platelets in various pathological conditions such as inflammation, immunity and thrombosis. 21 MPV has been considered to be an important marker in assessing platelet volume and platelet activity. 22 Several inflammatory cytokines, including interleukin-3 (IL-3), interleukin-6 (IL-6) and tumour necrosis factor, have considerable effects on haematopoiesis and are primarily responsible for secondary thrombocytosis,23,24 which promotes megakaryocytopoiesis and releases platelets into peripheral circulation. The regulatory process in fact is programmed to activate platelets under physiological and pathological conditions. 25 Indeed, these inflammatory cytokines have been found to be increased in patients with TA,26,27 wherein excessive production of inflammatory cytokines stimulates megakaryocytes to release more small-sized platelets from the bone marrow into the blood circulation. 25 Moreover, there is evidence that large-sized platelets in peripheral circulation are greatly consumed at inflammatory sites. 25 In fact, high-grade inflammation tends to increase consumption of large-sized platelets in some rheumatic diseases, 25 which may be associated with decreased MPV values in patients with TA.
There are several limitations to the current study. First, a small sample size should be noted, especially for active TA patients with prednisone therapy. Second, further investigation with a larger cohort is needed to establish whether the diagnostic performance of MPV is able to be improved for identifying active vs. inactive disease in TA patients. Third, platelet count and function may be affected by glucocorticoids in patients with active disease post-treatment. Finally, the correlation between MPV concentrations and disease classification in TA patients was not evaluated. However, our results suggest that MPV may be a marker to identify active disease in patients with TA, and the values of MPV may help to distinguish remission of active disease after treatment in TA patients. These finding should be further investigated in a larger cohort to verify our findings.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Ethics Committee of Affiliated Hospital of Youjiang Medical University for Nationalities (REC number: 201501005).
Guarantor
YFP.
Contributorship
YFP researched literature, conceived the study, performed data analysis and wrote the first draft of the manuscript. JG was involved in protocol development, gaining ethical approval. JG and YBD contributed to data collection. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
