Abstract
Background
Henoch-Schonlein purpura is a systemic small-vessel vasculitis that occurs mainly in children. A review of the literature has suggested a correlation between mean platelet volume and several inflammatory disorders. However, to the best of our knowledge, any potential correlation between mean platelet volume and Henoch-Schonlein purpura has not been reported in the literature. Therefore, our study aimed to evaluate the role of mean platelet volume concentrations in patients with Henoch-Schonlein purpura.
Methods
This study included 97 children with Henoch-Schonlein purpura and 120 healthy individuals as controls.
Results
Mean platelet volume concentrations were found to be significantly lower in Henoch-Schonlein purpura patients compared with healthy controls (8.1 ± 0.86 vs. 9.4 ± 0.81, P < 0.001). Similarly, significant negative correlations were observed between mean platelet volume and neutrophil count, platelet count and erythrocyte sedimentation rate in patients with Henoch-Schonlein purpura (r=−0.327, P = 0.001; r=−0.419, P < 0.001; r=−0.255, P = 0.012). Interestingly, mean platelet volume was significantly lower in the acute phase compared with the convalescent phase of Henoch-Schonlein purpura patients (7.8 ± 0.86 vs. 8.3 ± 0.77, P = 0.002). A cut-off value for mean platelet volume was 7.85 with area under the curve of 0.726 to identify acute phase vs. convalescent phase in patients with Henoch-Schonlein purpura. Mean platelet volume was independently associated with Henoch-Schonlein purpura in logistic regression analysis (odds ratio = 0.114, 95% confidence interval = 0.053–0.243, P < 0.001).
Conclusions
Our results suggest that mean platelet volume is inversely associated with disease in patients with Henoch-Schonlein purpura, and mean platelet volume may be a useful marker to identify active disease in Henoch-Schonlein purpura patients.
Introduction
Henoch-Schonlein purpura (HSP) is a systemic small-vessel vasculitis that mainly occurs in children. The main presenting features are purpura, abdominal pain, arthralgia/arthritis and kidney disease. 1 Although HSP tends to be self-limiting in children, adult patients exhibit severe clinical manifestations.2,3 Previous studies have shown that high C-reactive protein levels (CRP), leukocytosis, mean platelet volume (MPV), elevated neutrophil–lymphocyte ratio and platelet–lymphocyte ratio are associated with gastrointestinal bleeding in children with HSP.4–6
MPV reflects the size of platelets and is correlated with platelet function and activation. 7 In addition, MPV has been considered as an inflammatory biomarker in several diseases, such as tuberculosis, chronic obstructive pulmonary disease, urinary tract infection, severe periodontitis and systemic lupus erythematous.8–12 However, to the best of our knowledge, there are no studies in the literature on MPV concentrations in childhood HSP activity. Therefore, the aim of this study was to investigate the role of MPV concentrations in patients with HSP.
Patients and methods
This study included 97 HSP children who were admitted to the First Affiliated Hospital of Guangxi Medical University (Nanning, China) from January 2014 to December 2016. Healthy controls consisted of 120 healthy individuals undergoing routine physical examinations at our hospital. Clinical data and laboratory parameters of the patients were collected in electronic medical records at the hospital. The definition of HSP was according to the European League against Rheumatism/Paediatric Rheumatology European Society (EULAR/PRES) diagnostic criteria. 13 A clinical scoring system was used to identify disease activity in the acute and convalescent phases.14,15 The acute phase was defined as the period when the patient showed clinical features, including purpura, haematuria, arthralgia, and abdominal pain. However, the convalescent phase was defined as the period during which all HSP symptoms had resolved. Patients with immunologic disorders, hypertension, diabetes, hyperlipidaemia, obesity, acute or chronic infectious disease, chronic renal disease, nephrotic syndrome, inflammatory bowel disease and chronic inflammatory disease were excluded from the study. In addition, patients also were excluded if they had received systemic steroid treatment before the blood count analysis. Platelet count, MPV, haemoglobin, neutrophil count, lymphocyte count, Erythrocyte sedimentation rate (ESR) and CRP were retrospectively collected from the medical records of patients. Routine blood tests were performed using an automatic blood cell analysis instrument (COULTER LH750 Hematology Analyzer, Beckman Coulter, USA).
This study was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University, and informed consent was obtained from all individuals.
Statistical analyses
Continuous variables are presented as mean ± standard deviation or median (interquartile range), and categorical variables are shown as percentages. The Shapiro-Wilk test was used to evaluate data normality. Continuous variables with a normal distribution were compared using the independent student’s t-test and data that were not normally distributed were analysed by the Mann-Whitney U test. The differences in proportions between two groups were analysed by the Chi-square test. The Spearman approach was used to analyse the correlation between two continuous variables. Receiver operating characteristics (ROC) curve analysis was used to measure the performance of MPV. We used multivariate logistic regression analysis to assess independent parameters associated with HSP. Statistical analysis was performed using SPSS 19.0 software (IBM, USA), and P < 0.05 was considered to be statistically significant.
Results
Comparison of demographic and laboratory variables of HSP patients and healthy controls at baseline.
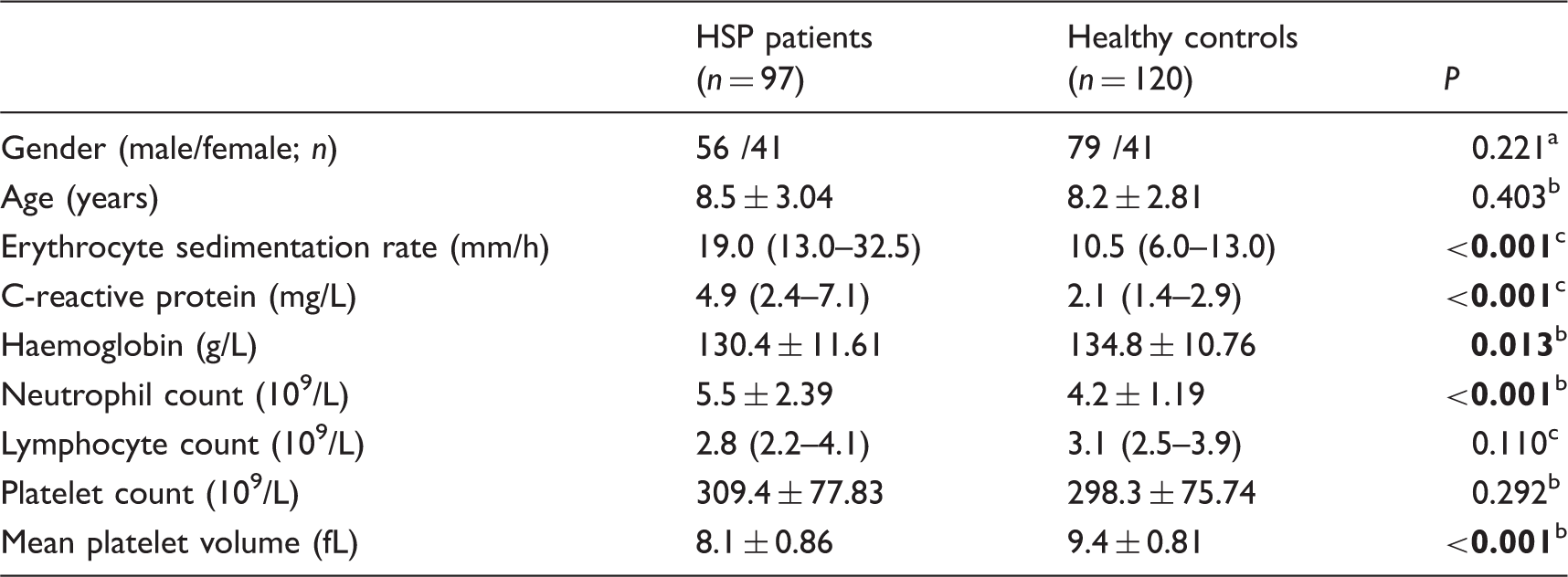
Note: Data are reported as mean ± SD or median (interquartile range).
HSP: Henoch-Schonlein purpura.
Pearson’s chi-square test.
Student’s t-test.
Mann-Whitney U-test.
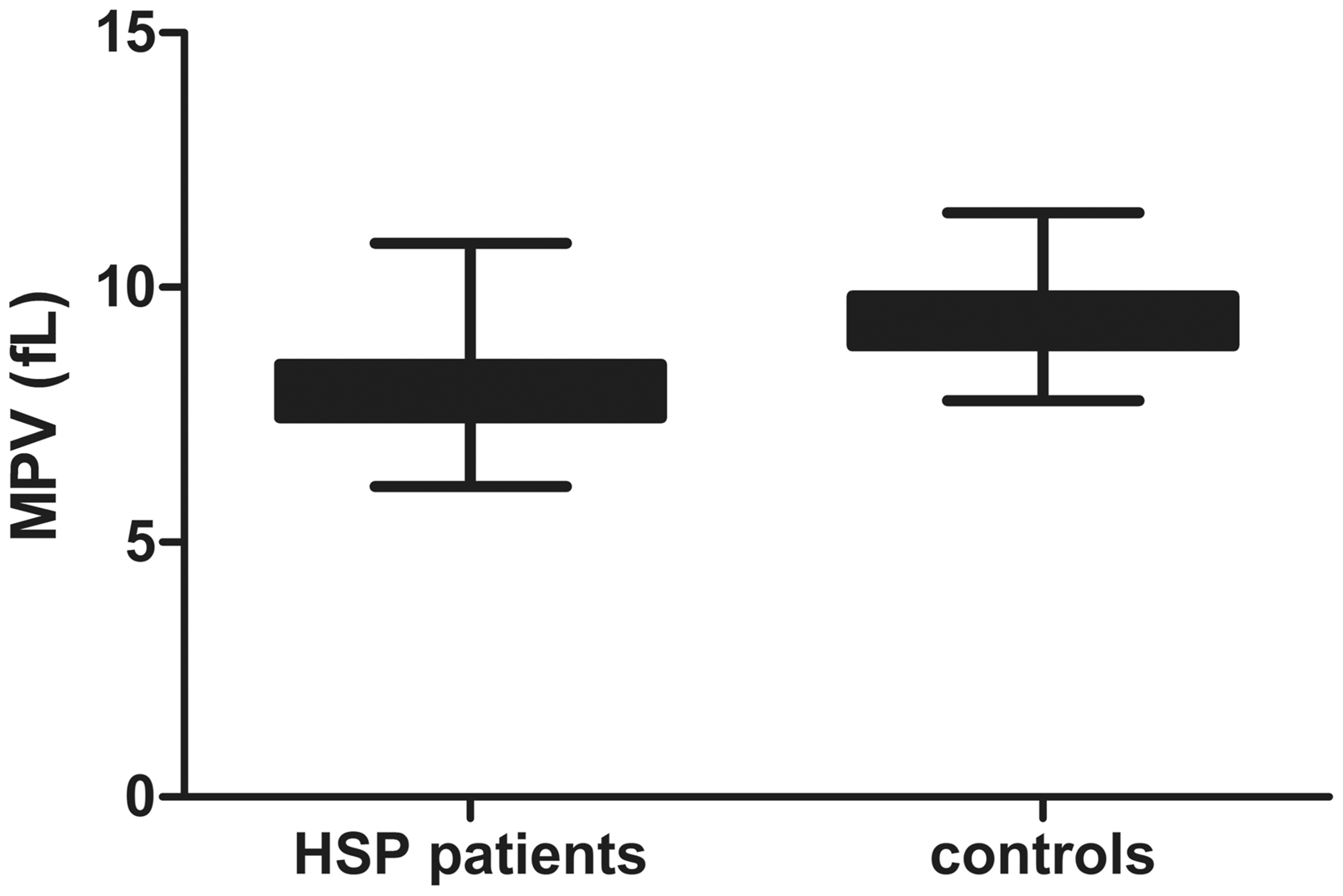
Mean platelet volume (MPV) in children with Henoch-Schonlein purpura (HSP) and healthy controls. P < 0.001, Student’s t-test.
The correlation analysis was conducted among MPV and other variables in HSP patients. The results showed that MPV was inversely associated with neutrophil count (r=−0.327, P = 0.001). Similarly, MPV also had a significant negative relationship with platelet count (r=−0.419, P < 0.001) and ESR (r=−0.255, P = 0.012, Figure 2). Moreover, MPV was significantly lower in the acute phase compared with the convalescent phase of HSP patients (7.8 ± 0.86 vs. 8.3 ± 0.77, P = 0.002), as shown in Table 2. In addition, ROC curve analysis was used to identify acute phase vs. convalescent phase in patients with HSP; the sensitivity of 68.5% and specificity of 70.7% for MPV were observed in the study with a cut-off value of 7.85 and area under the curve of 0.726.
The correlation between mean platelet volume (MPV) and Erythrocyte sedimentation rate (ESR) in children with Henoch-Schonlein purpura (HSP). Comparison of demographic and laboratory variables in Henoch-Schonlein purpura (HSP) patients with acute and convalescent phase. Note: Data are reported as mean ± SD or median (interquartile range). Pearson’s chi-square test. Student’s t-test. Mann-Whitney U-test.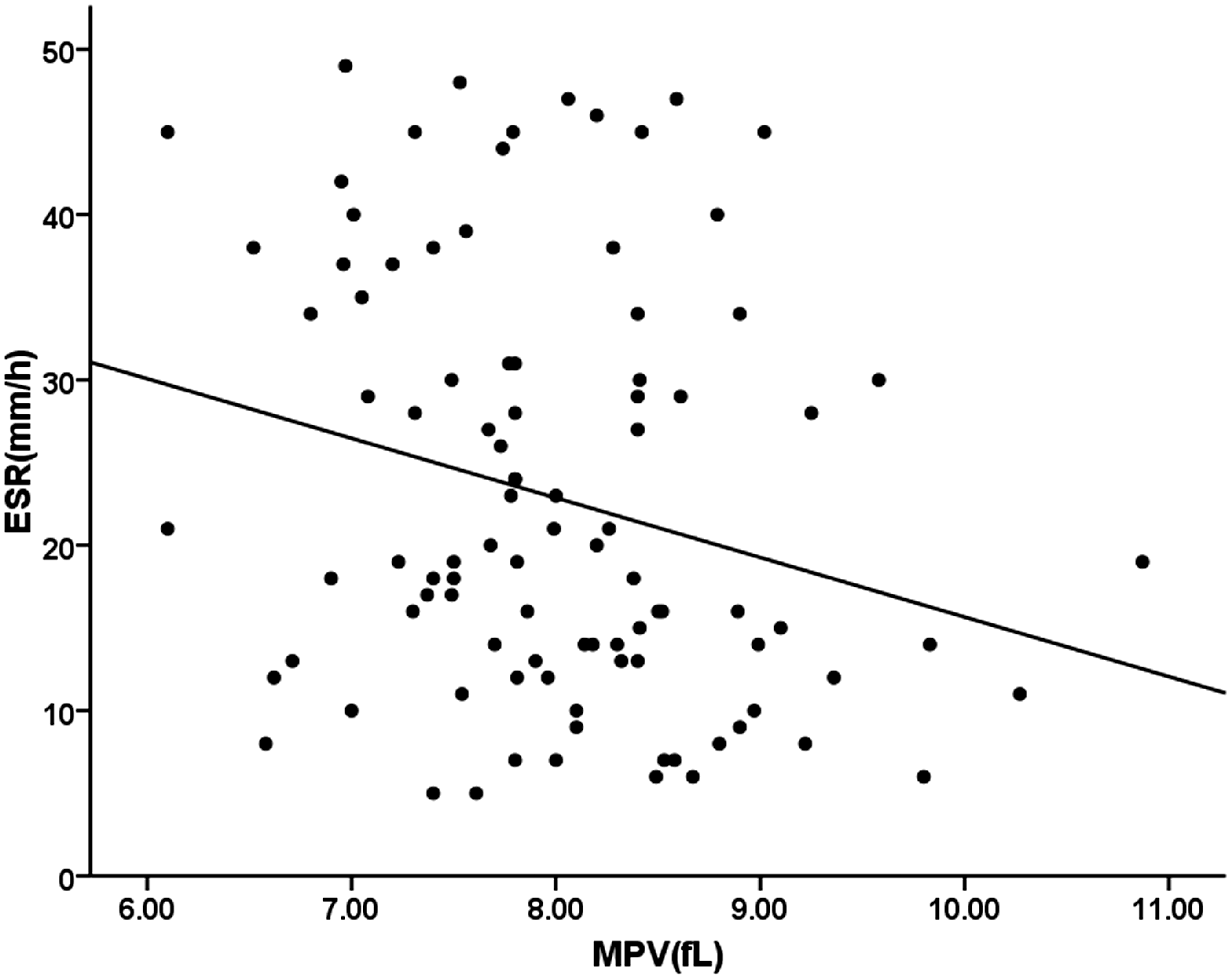
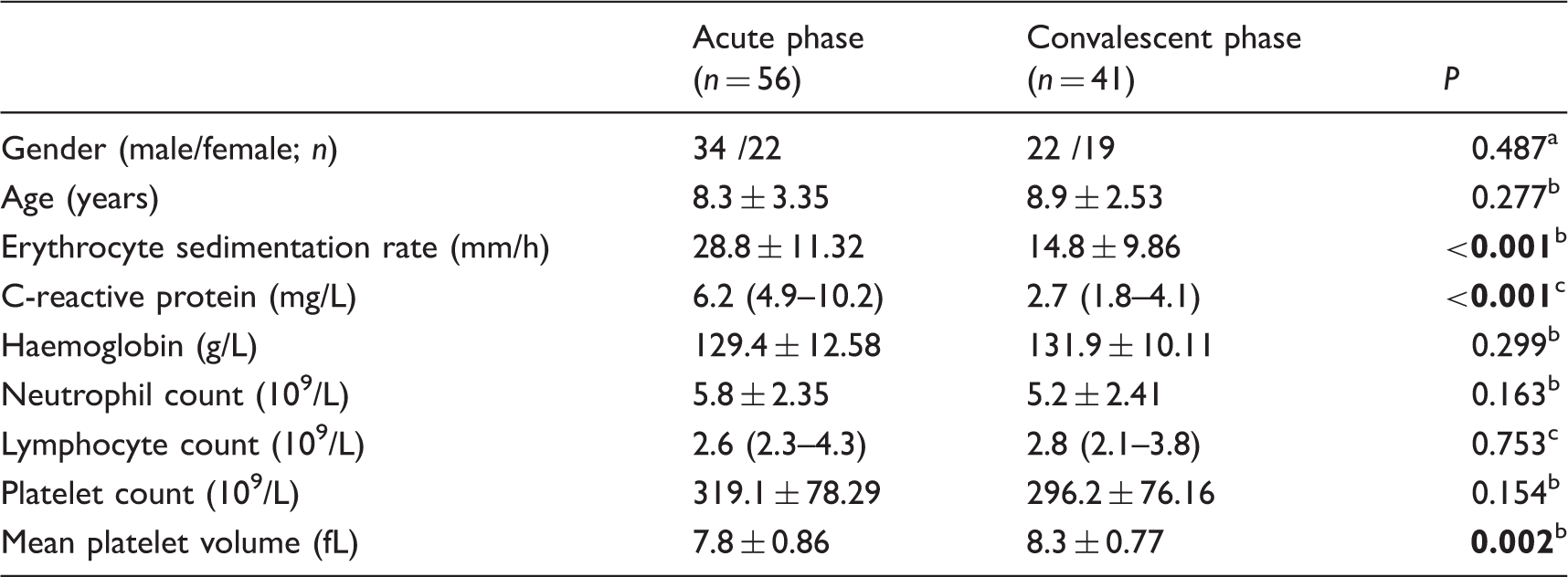
Multivariable analysis between mean platelet volume and patients with Henoch-Schonlein purpura.
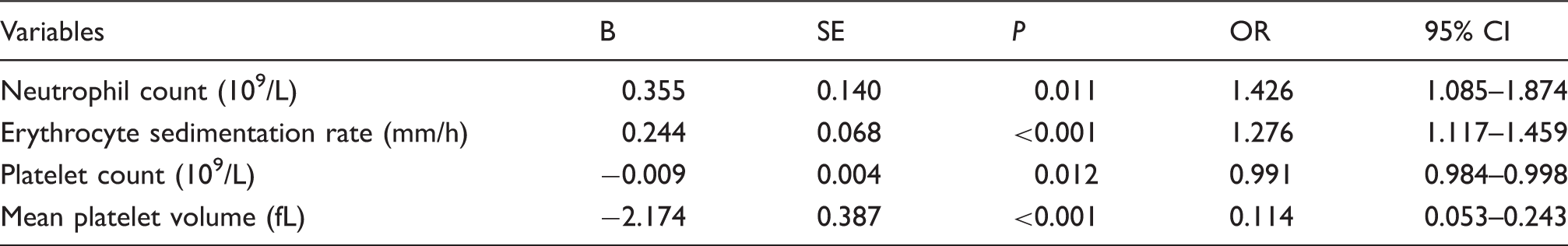
OR: odds ratio; 95% CI: 95% confidence interval.
Discussion
In this study, lower MPV values were observed in children with HSP than healthy individuals, and there was a significant inverse correlation with ESR. Interestingly, MPV concentrations were lower in HSP patients in the acute phase when compared with those of patients in the convalescent phase, suggesting a potential role for MPV in monitoring disease activity in HSP patients.
In the past few years, it has been revealed that MPV plays an actual role in inflammatory disorders. Very recently, Lee et al. reported elevated MPV values in children with acute pyelonephritis compared with lower urinary tract infections. 10 Arpaci et al. 16 also demonstrated that MPV concentrations were higher in acromegalic patients. It has been shown that increased MPV is associated with peritonsillar abscess, retinopathy in type 2 diabetes mellitus and rheumatoid arthritis.17–19 Similarly, increased MPV has been seen in patients with ankylosing spondylitis, hip osteoarthritis and central serous chorioretinopathy.20–22 Conversely, lower MPV concentrations have been reported in patients with acute mania, acute pancreatitis and venous thromboembolism.23–25 Moreover, Wang et al. reported a relationship between decreased MPV and severe periodontitis, suggesting that MPV may be a useful inflammatory marker reflecting the periodontal disease. 11 These observations indicate that MPV could be regarded as an important marker in assessing several inflammatory diseases. Why are MPV levels low in HSP patients in the acute phase? The answer is not yet clear. However, recent evidence suggests that several inflammatory cytokines, such as tumour necrosis factor-alpha (TNF-α), interleukin-6 (IL-6) and interleukin-8 (IL-8), have been found to be increased in HSP patients.26,27 Further investigation suggests that inflammatory cytokines are closely related to platelet activation in the pathophysiology of diseases and the overproduction of inflammatory cytokines activates megakaryocytes to release more small-sized platelets from the bone marrow into the peripheral blood. 28 Even more, there is an evidence that large platelets in high-grade inflammatory diseases are consumed at a greater rate of inflammation sites than small platelets. 29 Therefore, a possible explanation for the low MPV concentrations in children with HSP would be that HSP patients with high-grade inflammation may have increased consumption of large platelets.
In the present study, however, several limitations exist. Firstly, it should be noted that this study used only a relatively small sample size, especially of HSP patients between the acute and convalescent phases. Secondly, MPV concentrations in active HSP patients before and after treatment were not evaluated. However, our results suggest that lower MPV concentrations are associated with disease activity in patients with HSP, and therefore, MPV may be a useful marker to identify active disease in HSP patients.
Footnotes
Acknowledgements
We thank MogoEdit for their English editing service.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Ethics Committee of the First Affiliated Hospital of Guangxi Medical University approved this study (REC number: 201501005).
Guarantor
WNM.
Contributorship
WCL, LJM and XHL researched literature and conceived the study. WNM was involved in protocol development and gaining ethical approval, YZL and LQQ collected the data. ZY performed data analysis and XS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
