Abstract
Objectives
The development and progression of malignancies are closely linked to hypercoagulability. As an early type of lung adenocarcinoma, ground glass nodules (GGNs) have been detected increasingly. Blood Maximum amplitude (MA) and mean platelet volume (MPV) are related to various conditions of hypercoagulability. Therefore, the role of MA and MPV in diagnosing lung adenocarcinoma cancer featured with GGNs was investigated in this case-control study.
Methods
The analyzed data of this study is derived from GGNs patients and healthy individuals in West China (Airport) Hospital Sichuan University. The differences between GGNs patients and healthy individuals were determined by one-way ANOVA, logistic regression or chi-squared test. The accuracy of diagnostic was performed by receiver operating characteristic curve (ROC). The relative mRNA expressions were studied by RT-qPCR.
Results
470 patients diagnosed with GGNs which benign lesions (BN group) are inflammatory and malignant lesions (LC group) are adenocarcinoma in stage IA, and 235 healthy subjects (HC group) were enrolled in this study. Levels of MA and MPV were increased in LC group compared with BN and HC group (p < 0.001, p < 0.001). When we combined MA and MPV, MA and MPV presented a sensitivity (SEN) of 0.809 and a specificity (SPE) of 0.774. And the area under the curve (AUC) increased to 0.871 (0.837–0.900) when confidence interval was 95%.
Conclusion
This study demonstrates that there have been systemic changes in coagulation disorders in the pathogenesis of GGNs. The diagnostic ability to different lung adenocarcinoma cancer featured with GGNs from benign or healthy controls can be improved by the combination of MA and MPV. Maximum amplitude and MPV may be used as biomarkers to detect lung adenocarcinoma cancer featured with GGNs.
Introduction
Lung cancer accounts for nearly half of all cancer-related deaths worldwide 1 Over 40% of diagnosed lung cancer cases are adenocarcinoma 2 Ground-glass nodules (GGNs), as an early type of lung adenocarcinoma, which is defined as hazy lesions on computed tomography (CT) scans that do not obscure underlying bronchial structures or pulmonary vessels3,4 are getting more and more attention in clinical practice. These lesions include both malignant (lung adenocarcinoma and their preinvasive lesions) and benign lesions (focal interstitial fibrosis, inflammation, or hemorrhage) 5 Up to now, neither assistant tests nor recommended biomarkers that can provide a help in the differential diagnosis of GGNs 6 Hence, a better biomarker is essential for identifying which patients with GGNs are at high risk for developing lung adenocarcinoma.
Hypercoagulability plays a highly considerable role in the progression of malignancies7,8 The maximum amplitude (MA), one of thromboelastography (TEG) parameters, by measuring the clot strength in whole blood with a viscoelastic test, can quantify hypercoagulation status and platelet activity 9 Maximum amplitude-identified hypercoagulability has been found to be an independent prognostic factor for lung adenocarcinoma cancer and predictive of TNM Stage10–12 The Mean platelet volume (MPV), a conventional parameter in blood that can be used to determine platelet size and activity. Increasing evidence founded that platelet function and activation may be evaluated by MPV, as a readily blood marker13,14 Mean platelet volume is related to not only a variety of hypercoagulation status but also malignant lung cancer15,16 However, whether MA and MPV can be an available marker to diagnose malignant GGNs is not clear yet. The purpose of the study is to explore the role of MA and MPV in detecting lung adenocarcinoma cancer featured with GGNs when MA and MPV alone or combined.
Patients and methods
Study design
Ethical approval was obtained through ethics committee of the West China (Airport) Hospital Sichuan University (Ethic Approval No.2020-03-07). Written informed consent was acquired from all patients prior to enrollment in this study.
A case-control study was conducted on patients with primary lung cancer treated and screened for the following eligibility criteria between January 2019 and February 2021. All patients with lung cancer were adenocarcinoma and all benign nodules were inflammatory nodules unless otherwise stated. Inclusion criteria: (1) computed tomography (CT) confirmed ground glass-like pulmonary nodules in the lungs; and (2) primary lung cancer of stage IA or benign lesions were confirmed by histologically or cytologically. Exclusion criteria: (1) Any disease process known to alter coagulation (i.e., genetic haemostatic disorders); (2) Anticoagulant therapy such as warfarin, heparin; (3) Previous treatment for cancer and presence of metastatic lesions. Demographic information (age, sex), stage, tumour histology and clinical data were recorded and anonymized for each patient. Tumor staging was coded bases upon the TNM classification of the Union for International Cancer Control
17
and all patients with lung adenocarcinoma are in stage ΙA. A detailed patient inclusion flowchart is shown in Figure 1. The flow diagram of the study population in this study. GGNs, ground-glass nodules; SCC, squamous cell carcinoma; d, diameter.
Laboratory assays
Each participant’s venous blood (5 mL) was collected in the form of fasting blood and deposited in EDTAK2 anticoagulation tubes. To detect whole blood routine parameters, the blood samples collected above were examined in an Automatic Hematology Analyzer called Sysmex XE-5000 (Sysmex Corp, Japan), including the number of red blood cell (RBC), the number of platelet (PLT), the concentration of hemoglobin (HGB), MPV, Neutrophil count (Neut#) and Neutrophil count ratio (Neut%). Blood samples were centrifuged for 3 min at 3000 × g, and serum samples were tested for biochemical markers in 2 h using a Beckman AU5800 Automatic Analyzer (Beckman Coulter Inc. CA, USA) including alanine transaminase (ALT), creatinine (CR), total protein (TP), globulin (GLO), albumin (ALB) and aspartate aminotransferase (AST). 1 mL of citrated blood was activated by kaolin in advance, 340 μL of blood mentioned above was re-calcified using 20 μL STARTEM reagent (0.2 M CaCL2) in a specifical plastic cup and the procedure was started in a coagulation analyzer (CFMS LEPU-8800; LEPU Diagnostic Technology Corporation, Beijing, China) to detect MA. Quality-control and calibrator were performed according to the manufacturer’s instructions. All samples were processed within 2 h of venesection.
RT-qPCR
The primer sequences for qPCR.
Statistical analysis
The analysis of the data was performed using SPSS version 20.0 (SPSS Inc., Chicago, IL, USA). Unless otherwise stated, all values are expressed as mean ± standard deviation. Logistic regression, One-way ANOVA test or chi-squared test was used to compare differences between values of different groups. The area under the receiver operating characteristic curve (ROC) was used to assess the diagnostic accuracy of MA and MPV. A significance level of p < 0.05 was considered statistically significant.
Results
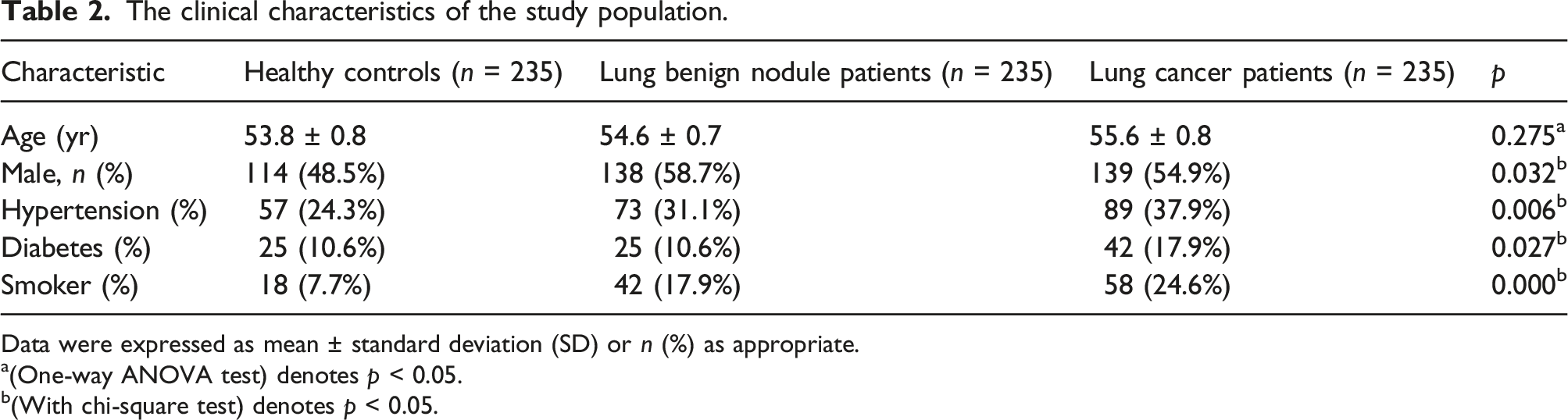
Characteristics of study population
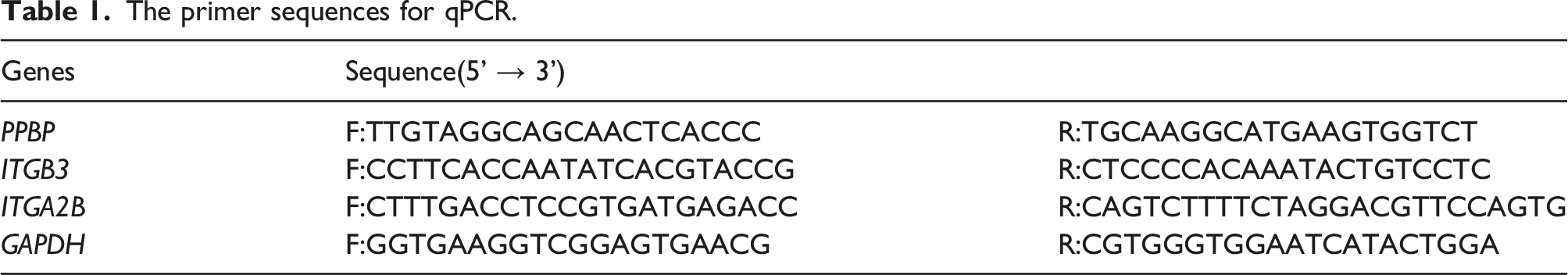
We enrolled a total of 705 participants, including 235 patients with stage IA lung adenocarcinoma (LC group), 235 patients with benign nodules (BN group) and 235 healthy subjects (HC group). Representative computed tomography images of GGNs resected from enrolled patients were shown in Figure 2. All pathological results were confirmed by surgery. The mean age of the three groups (294 women, 411 men) respectively was 53.8 ± 0.8 years, 54.6 ± 0.7 years, 55.6 ± 0.8 years (range, 21–78 years).118 patients had a smoking history. Significant differences were observed among the three groups of patients on sex, hypertension, diabetes and smoking history. Demographic and clinical data are presented in Table 2. Representative computed tomography images of GGNs. The clinical characteristics of the study population. Data were expressed as mean ± standard deviation (SD) or n (%) as appropriate. a(One-way ANOVA test) denotes p < 0.05. b(With chi-square test) denotes p < 0.05.
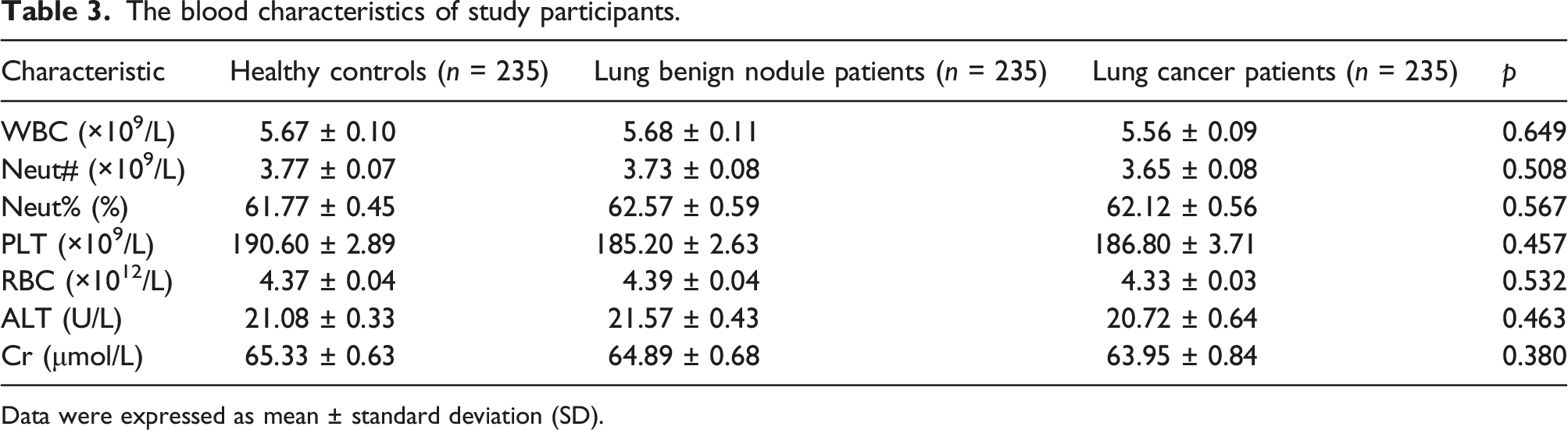
Blood characteristics
We continued to analyze the blood characteristics of the study participants. LC group had significantly higher levels of MA and MPV compared to the BN group and HC group (Figure 3(a) p < 0.001 and Figure 3(b) p < 0.001), but no statistically significant differences were present among the three groups for other indicators (Table 3). And the logistic regression for MA and MPV was conducted to adjust for confounding variables (sex, hypertension, diabetes and smoking history) (as shown in the Table 4). The levels of MA and MPV in LC group, BN group and HC group. Significant differences were observed in MA (a) and MPV (b). The blood characteristics of study participants. Data were expressed as mean ± standard deviation (SD). The logistic regression for MA and MPV.
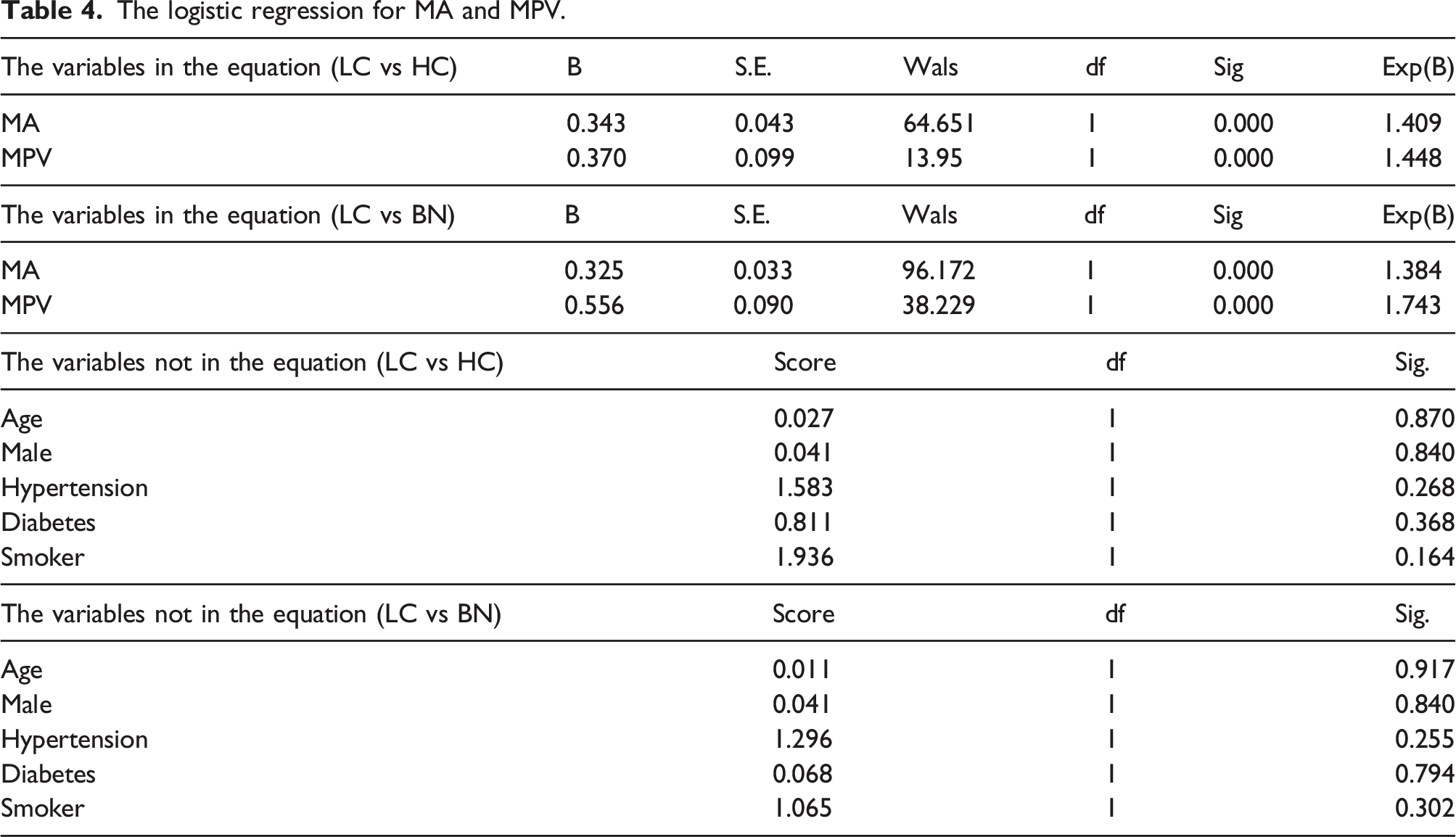
Diagnostic accuracy of MA and MPV
Having observed the levels of MA and MPV were significantly increased in LC group, we went on to explore whether MA and MPV, either alone or combined, could be diagnostic for the identification of malignant GGNs. Subsequently, the diagnostic accuracy of MA and MPV in separating lung cancer from benign subjects was shown in Figure 4 and Table 5. MA presented a sensitivity (SEN) of 0.796 and a specificity (SPE) of 0.744 at the optimal cutoff point of 64.6 mm, and the AUC was 0.853 (0.818–0.884) when confidence interval was 95% (Figure 4(a)). mean platelet volume presented a SEN of 0.400 and a SPE of 0.872 at the optimal cutoff point of 12.2 (fL), and the AUC was 0.705 (0.662–0.746) when confidence interval was 95% (Figure 4(b)). Subsequently, we combined MA and MPV, MA and MPV presented a SEN of 0.809 and a SPE of 0.774, and the AUC rose to 0.871 (0.837–0.900) when confidence interval was 95% (Figure 4(c) and (d)). Together, these results suggest that in comparison with either MA or MPV alone, the combination of the two can provide better diagnostic value to differentiate malignant GGN patients from benign or healthy persons. ROC curves of the diagnostic presentation MA and MPV, alone or coupled. (a) MA, (b) MPV, (c) and (d) MA + MPV. Diagnostic value of MA and MPV for predicting lung cancer featured with GGNs.

The relative mRNA expression of ITGA2B and ITGB3
To elucidate whether there was involved in more activated platelets in LC group, we aimed to detect the relative mRNA expression of ITGA2B and ITGB3 genes. ITGA2B and ITGB3, encoded αIIbβ3 protein which assures platelet activation and aggregation, presenting conformational changes from a bent to an extended and open form. The results of RT-qPCR showed that the mRNA levels of ITGA2B and ITGB3 were both significantly increased in LC group (Figure 5, p < 0.01). Therefore, these results demonstrate that more activated platelets participate in lung adenocarcinoma cancer patients featured with GGNs. The relative mRNA expression of ITGA2B and ITGB3 in LC group, BN group and HC group. Data are presented as mean ± SD.
Discussion
Increasing evidence indicates that stage IA lung adenocarcinoma has distinctly different prognoses compared with benign GGNs18–20 Moreover, the diagnosis of lung adenocarcinoma is essential for selection of a surgical plan and clinical management 21 Although several studies have reported that early-stage lung cancer is mainly presented as persistent GGNs at CT 22 Until now, neither CT tests nor recommended biomarkers that can accurately identify early-stage lung adenocarcinoma from benign GGNs. Our present study gives the first report to demonstrate that MA combined with MPV could be used as a promising diagnostic-marker for the identification of lung adenocarcinoma characterized by GGNs.
Previous studies have reported that lung cancer is frequently associated with a variety of coagulopathies and thromboembolic disorders 12 Lung cancer itself can increase coagulability. Tumor cells also release procoagulant microparticles into the circulation, which may also trigger the persistent hypercoagulability 23 as discussed by Nie et al. 24 In this study, we found that MA and MPV were obviously higher in lung adenocarcinoma cancer patients featured with GGNs compared to benign and healthy controls. Recently, MA, one parameter of TEG, was consistently used to evaluate the function of platelets and has been found to be predictive of risk of thrombosis 25 Mean platelet volume, another parameter proposed as an indicator of platelet size and activity, is associated with a variety of established risk factors, cardio- and cerebrovascular disorders prone to arterial and venous thromboses, suggesting a relation between an increase in MPV and the risk of hypercoagulability16,26 Therefore, our results suggest that more activated platelets may be involved and there is an increased risk of hypercoagulability in the pathogenesis of lung adenocarcinoma cancer featured with GGNs.
Platelets, a major component of the developing thrombus, are actively involved in the process of blood coagulation27,28 Patients with metastatic cancer also have increased platelet aggregation and activation29,30 It should be pointed out that blood coagulation, initiated by tissue factors, culminates in the generation of thrombin and fibrin which were involved in activated platelets 31 αIIbβ3, encoded by ITGA2B and ITGB3 genes, assures platelet activation and aggregation. Platelets activation presents conformational changes from a bent to an extended and open form, that allow fibrinogen or other adhesive proteins to bind and crosslink platelets32–34 In parallel, the upregulated mRNA expressions of ITGA2B and ITGB3 were also observed in our real-time PCR results. In a word, the involvement of the ITGA2B and ITGB3 system is important not only for platelets aggregation and activation but also for lung adenocarcinoma cancer, inflammation and many key biological processes34,35
This study showed that when using MA and MPV as the cutoff, the SEN and SPE for differentiating lung adenocarcinoma cancer from benign GGNs were 81% and 77%, and the AUC was 0.871. Furthermore, the combination form of MA and MPV can improve the diagnostic value compared to MA and MPV alone showed by 0.705 of the AUC. It should be noted that the combination of MA and MPV do not have adequate SEN and SPE to diagnose GGNs in terms of clinical utility. The possible reason is that lung adenocarcinoma cancer featured with GGNs has different histological types, and there are many genetic and epigenetic changes present in cancer cells, making it difficult for our biomarkers to overlay all cancer-related changes.
However, our study has several limitations. First, it was a case–control study conducted by a single-institution. We lacked a sufficient sample size. 470 cases of GGNs were diagnosed as benign or early stage I lung adenocarcinoma at postoperative pathologic analysis in the enrolled population. And power calculation was not done for estimation of sample size selected for this study. Further validation of the current findings requires a larger prospective study and power calculation involving multiple-institution. Second, the subjects enrolled were patients, in regardless of pathologic subtypes and growth patterns, who underwent surgery in the thoracic surgery department, selection bias might have occurred. Third, the aim of this study was to investigate the diagnostic value of MA and MPV in differentiating malignant from benign GGNs. However, no prognostic information was available regarding recurrence or death.
Conclusions
Therefore, compared with MA or MPV alone, the combination of MA and MPV, as biomarkers, can improve the diagnostic ability to distinguish lung adenocarcinoma cancer featured with GGNs from benign GGNs patients. Although further studies must be carried out to confirm these findings, this investigation suggests that there is a tendency toward hypercoagulability in the pathogenesis of lung adenocarcinoma cancer featured with GGNs and the combination of MA and MPV may be clinically useful for the early diagnosis of lung cancer.
Footnotes
Acknowledgements
The authors thank the reviewers for their precious comments and all the volunteers who participated in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of Medical Science Foundation of Chengdu (No. 2021104).
Informed consent
Written informed consent was obtained from all subjects before the study. Written informed consent was acquired from all patients.
Trial registration
Not applicable.
Data availability statement
On reasonable request, the corresponding author will provide the data sets generated during and/or analysed during the current study.
Ethical statement
The authors are responsible for the accuracy and integrity of all parts of the work, and any questions related to those parts of the work must be investigated and resolved. The study was approved by the ethics committee of the First People’s Hospital in Shuangliu District/West China Airport Hospital, Sichuan University (Ethic Approval No. 2020-03-07) in accordance with the Declaration of Helsinki (as revised in 2013). Written informed consent was acquired from all patients.
