Abstract
We report a 70-year-old female presenting with increased libido and mild but rapid onset virilism. Investigations showed markedly elevated androstenedione and 17 hydroxyprogesterone misdirecting to possible late-onset congenital adrenal hyperplasia. High serum testosterone and oestrogens with suppressed gonadotrophins, however, indicated an androgen-secreting tumour. A normal dehydroepiandrosterone sulphate and elevated inhibins A and B indicated the tumour was ovarian in origin, which was confirmed on pelvic examination and imaging. At laparotomy, a right ovarian sertoliform endometrioid carcinoma was removed, following which the patient developed menopausal vasomotor symptoms and improvement of her virilism. Serum testosterone, oestradiol, inhibins A and B became undetectable, gonadotrophins appropriately increased and 17 hydroxyprogesterone and androstenedione normalized. We propose that inhibins may be of diagnostic value and should be included in investigative algorithms of females with virilization and hyperandrogenaemia, especially if postmenopausal. Androgen-secreting tumours must be excluded before raised 17 hydroxyprogesterone concentrations are used to diagnose late-onset congenital adrenal hyperplasia in females with new-onset virilization.
Introduction
Ovarian sex hormone-secreting tumours are rare and are most commonly ovarian sex-cord stromal tumours.1,2 Of sex-cord stromal tumours, typically Sertoli-Leydig tumours (SLT) secrete testosterone, whereas granulosa cell tumours and thecomas secrete oestrogen. Stromal luteomas, sclerosing stromal tumours and hilus cell tumours may also secrete testosterone, or less commonly oestrogen. 3 Ovarian epithelial tumours usually present with mass effects or genital bleeding but some may secrete oestrogen.4,5 We report an ovarian sertoliform endometrioid carcinoma (SEC), a rare epithelial tumour, presenting with virilization and uniquely secreting both oestrogen and testosterone.
Case report
Biochemical investigations before and after surgery.
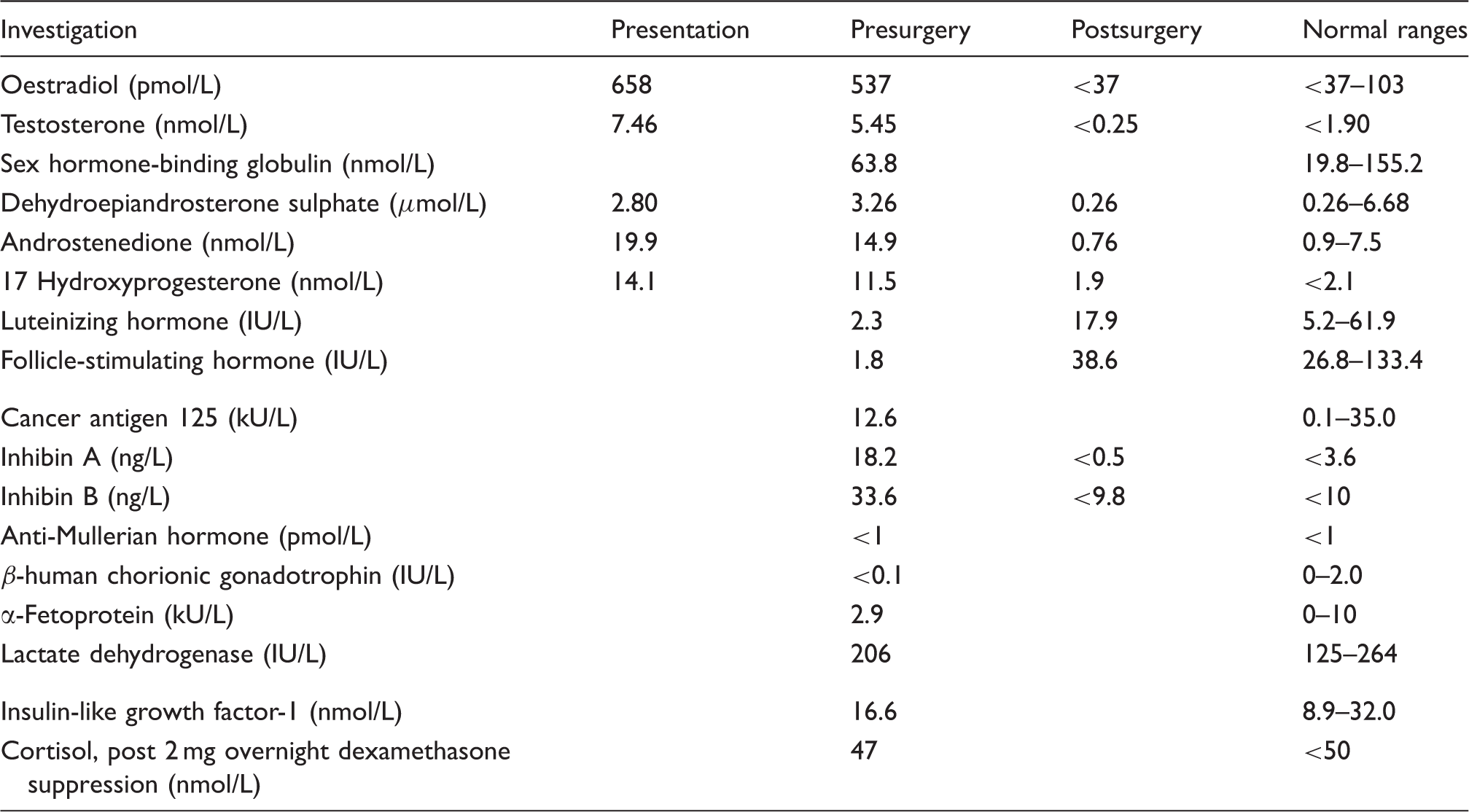
On examination, she was mildly overweight (BMI of 26.3 kg/m2) with greasy skin, mild facial hirsutism and mild acne, but without balding. Further investigations (Table 1) confirmed increased serum concentrations of testosterone, androstenedione, 17 hydroxyprogesterone (17OHP), and 17 β-oestradiol, and showed suppressed gonadotrophins and a normal dehydroepiandrosterone sulphate (DHEAS). Cushing’s syndrome and acromegaly were excluded. A transvaginal ultrasound revealed an 80 mm × 70 mm solid mass in the right adnexa and absence of a uterus, which was confirmed by a CT scan of the abdomen and pelvis, where no other abnormality and no evidence of disseminated disease were reported. Serum inhibins A and B were elevated but other serum tumour markers, including cancer antigen 125 (CA125) were normal. Subsequent pelvic examination confirmed a palpable right adnexal mass, but no clitoromegaly.
At laparotomy, a right ovarian tumour which was stuck to the bladder and right pelvic wall was removed, as was the left ovary. Biopsies of the omentum, peritoneum and a right pelvic lymph node together with peritoneal washings were taken.
Histology of the tumour showed a predominant sex-cord like pattern with small sertoliform tubules and trabeculae, on a fibromatous background. The cords and tubules stained strongly positive with broad spectrum cytokeratin and epithelial membrane antigen (EMA), and were found to be diffusely ER and PR positive. Staining of tubules was negative for the sex-cord markers, calretinin, inhibin and CD56. The tumour was, therefore, classified as a SEC grade I, FIGO stage 1c with right ovary capsule involvement. Omental, peritoneal and lymph node biopsies and peritoneal washings showed no evidence of malignancy. Closely associated with the tumour was a population of luteinized stromal cells which stained positive with inhibin and calretinin. Omental, peritoneal and lymph node biopsies and peritoneal washings showed no evidence of malignancy.
Following removal of the tumour, the patient developed menopausal symptoms and resolution of the increased libido, greasy skin and acne. Serum testosterone, oestradiol and inhibins A and B became undetectable, gonadotrophins appropriately increased, and 17OHP and androstenedione normalized (Table 1).
The patient was offered and elected to have six cycles of carboplatin chemotherapy, adjunctive to her surgery.
Discussion
SEC, first described in 1982, has histological similarities to SLT, and frequently causes diagnostic challenges. 6 Ovarian endometrioid carcinoma is a surface epithelial tumour, so called because of its similarity to the uterine endometrioid carcinoma, in both morphology and immunohistochemical expression. SEC has the histological appearance of a well-differentiated endometrioid carcinoma with the presence of mucin at apical borders of the tumour cells and integrated sertoliform-like areas. Cytokeratin and EMA stain positively but inhibin and calretinin stain negatively, 7 as described in this patient’s tumour, whereas the reverse staining pattern is seen in SLT.
SEC presents in postmenopausal females with mass effect or atypical genital bleeding, but rarely with endocrine symptoms,7,8 whereas SLT commonly presents in premenopausal females and often with hyperandrogenism.5,6 Generally, endometrioid carcinomas are not associated with sex hormone secretion, although Liang et al. 9 have described an SEC which showed genuine sex-cord differentiation. In our case, these sex-cord immunohistochemical markers were negative, demonstrating absence of true differentiation; the likely source of oestrogen, androgen and inhibin secretion in this tumour was the numerous accompanying non-neoplastic luteinized stromal cells which stained positive for inhibin as previously described. 7 This is consistent with the notion that ovarian tumours may induce the ovarian stromal cells, such as luteinized stromal cells, to secrete hormones. 10 In our case, the patient’s age and tumour were characteristic of a SEC, but her presentation with postmenopausal virilism was rare, and diagnosis of SEC secreting both oestrogen and testosterone is previously unreported.
Diagnosis may have been delayed in our case by the patient’s previous hysterectomy, since the most common endocrine manifestation of oestrogen-secreting tumours in postmenopausal females is abnormal uterine bleeding. 2 Postmenopausal loss of libido is common and of complex aetiology, but is considered to be due to declining oestrogen and testosterone concentrations as the ovaries fail 11 and improves following androgen replacement. 12 A striking symptom in our case was increased postmenopausal libido, particularly as the patient was only mildly virilized. We, therefore, suggest that new-onset increase in libido in postmenopausal females may indicate hyperandrogenism, and should be investigated accordingly.
At presentation, the patient had markedly elevated 17OHP concentrations, considered diagnostic of late-onset CAH, 13 which may present in the postmenopause. 14 Ovarian androgen-secreting tumours in children, however, have been misdiagnosed as CAH based on elevated 170HP concentrations. 15 Souto et al. 16 reported a patient with an ovarian luteoma presenting with increased testosterone and 17OHP concentrations and virilization, which corrected following tumour removal. They suggested that increased 17OHP was due to intratumoral 21 hydroxylase deficiency 16 even though this enzyme is not normally present in ovarian tissue. Turra et al. 17 reported a virilization in a 15-year-old girl due to a testosterone-secreting SLT who had normal baseline, but exaggerated 17OHP and androstenedione responses to gonadotrophin-releasing hormone, which normalized following tumour resection. We therefore postulate that in our case, greatly increased tumour-associated production of testosterone may lead to relative deficiency of 17 β-hydroxysteroid dehydrogenase resulting in increased circulating androstenedione and 17OHP. We also, therefore, advocate that androgen-secreting tumours must be excluded before attributing raised 17OHP concentrations to CAH in patients with hyperandrogenaemia, particularly in those with new-onset virilism.
Suppressed serum gonadotrophins, as in this case, are typical of androgen-secreting ovarian and adrenal tumours, which may help differentiate them from other causes of postmenopausal hyperandrogenism1,14 in which gonadotrophins are usually not suppressed.1,18 Although of limited sensitivity, the normal serum DHEAS in this case made an adrenal androgen-secreting tumour unlikely.1,19
Serum inhibins A and B are well-recognized ovarian tumour markers.20,21 Sex-cord tumours, particularly granulosa cell tumours, almost always have increased inhibins, especially inhibin B, but normal CA125. Ovarian epithelial tumours, including endometrioid, serous and clear cell and undifferentiated tumours have elevated CA125 but usually normal inhibins. Ovarian mucinous tumours frequently have raised CA125 and/or inhibins. 20 In investigating virilized females with increased androgens, the presence of raised inhibin A and particularly B would be supportive of an ovarian tumour.
Although absent in investigative algorithms,1,14,22 we suggest inhibins should be included in the investigative protocols of virilized females with hyperandrogenaemia, since they may be of diagnostic value, particularly in postmenopausal females where they should be undetectable. Inhibins may also have a role in detecting recurrence of androgen-secreting ovarian tumours, since they may recur even though they are very rarely malignant. 21
The raised testosterone, suppressed gonadotrophins, elevated inhibins and normal DHEAS suggested an ovarian tumour which was confirmed on pelvic examination, pelvic imaging and at laparotomy.
In summary, we suggest that measurement of serum inhibins A and B may be usefully included in guidelines for the investigation and management of postmenopausal hyperandrogenaemia, as they are of diagnostic value and may have a role in detecting recurrence. This case report also emphasizes the importance of excluding androgen-secreting tumours before ascribing raised 17OHP concentrations to diagnose late-onset CAH in females with new-onset virilization, and highlights increased postmenopausal libido as a presenting feature of hyperandrogenaemia.
Footnotes
Acknowledgements
We thank Dr R Ganesan, Birmingham Woman’s Hospital, for her histology opinion.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written consent was obtained from patient.
Guarantor
RG.
Contributorship
RG conceived the case report. JLS, RG and NS researched the literature. JLS wrote the first draft of the manuscript. NS, RG and DJM contributed to the data. All authors reviewed and edited the manuscript, and approved the final version of the manuscript.
