Abstract
Background
Turnaround time can be defined as the time from receipt of a sample by the laboratory to the validation of the result. The Royal College of Pathologists recommends that a number of performance indicators for turnaround time should be agreed with stakeholders. The difficulty is in arriving at a goal which has some evidence base to support it other than what may simply be currently achievable technically. This survey sought to establish a professional consensus on the goals and meaning of targets for laboratory turnaround time.
Methods
A questionnaire was circulated by the National Audit Committee to 173 lead consultants for biochemistry in the UK. The survey asked each participant to state their current target turnaround time for core investigations in a broad group of clinical settings. Each participant was also asked to provide a professional opinion on what turnaround time would pose an unacceptable risk to patient safety for each departmental category. A super majority (2/3) was selected as the threshold for consensus.
Results
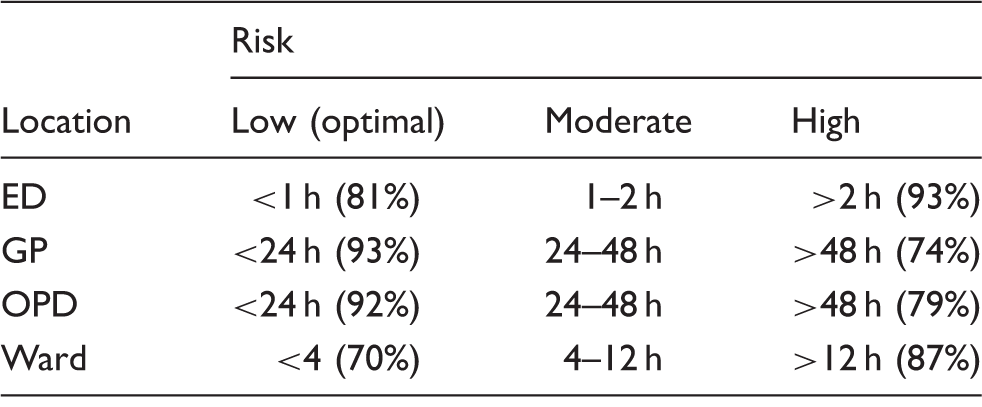
The overall response rate was 58% (n = 100) with a range of 49–72% across the individual Association for Clinical Biochemistry and Laboratory Medicine regions. The consensus optimal turnaround time for the emergency department was <1 h with >2 h considered unacceptable. The times for general practice and outpatient department were <24 h and >48 h and for Wards <4 h and >12 h, respectively.
Conclusions
We consider that the figures provide a useful benchmark of current opinion, but clearly more empirical standards will have to develop alongside other aspects of healthcare delivery.
Introduction
Modern clinical medicine relies on the timely availability of the results of investigations, including those from the clinical biochemistry laboratory. This not only promotes efficient and effective patient management but may be critical to patient safety in mitigating issues where morbidity and mortality is at risk.
Pathology services are an essential component of a patient-care pathway, with individuals’ treatment decisions – and the monitoring of their response to treatment – often dependent on a range of pathology-based tests and investigations. 1 It is therefore vital that the pathology service is of the highest possible quality.
Quality can be defined as the ability to satisfy the expectation of the customer and is enshrined in ISO 15189:2012 standard 4.1.2.2 ‘Laboratory management shall ensure that laboratory services, including appropriate advisory and interpretative services, meet the needs of patients and those using the laboratory services’. 2
Turnaround time (TAT), which can be defined as the time from receipt of a sample by the laboratory to the availability of a validated result, is probably the most accessible measure of the performance of a laboratory service. TAT is readily understood by both service users and providers alike and lends itself to quantitative analysis, an essential prerequisite for evidencing quality and its improvement. The Healthcare Commission 3 found that approximately a third of ward managers reported that at least once a week late or lost pathology reports had an adverse impact on the care or discharge of patients. It is therefore recognized that the only true assessment of performance of a laboratory service is related directly to its effect on outcomes for patients. However, the difficulty of unravelling the laboratory contribution from the many other factors involved has given rise to initiatives such as that of The Royal College of Pathologists in 2013. 4 This proposed a number of key performance indicators (KPIs) of desirable standards of performance all clinical laboratories should aspire to. TAT was included but the difficulty in setting one or more suitable KPI is in arriving at a goal which has some evidence base to support it other than what may currently be a technical possibility.
This raises two important issues around TAT which maximizes support for patient management and when a protracted TAT could be considered a significant risk factor.
What is the optimum TAT for the efficient assessment of patients?
The RCPath ‘KPI – proposals for implementation’ document was published in 2013 and included a target TAT of 1 h from receipt for 90% of core investigations from emergency departments (ED). 4 (Specified core investigations included LFT, U&E, troponin, FBC.) From April 2015 this was changed to 1 h from sample collection rather than receipt by the laboratory. Beyond this, it is recognized that timeliness and speed are not equivalent and that individual care pathways may require different TATs. Although RCPath does not provide specific targets for other clinical pathways, it advises that TAT categories should be developed taking into account local circumstances and clinical priorities.
What is the maximum TAT above which patient safety is compromised?
The College of American Pathologists lists prolonged TATs as a threat to patient safety. 5 In recognition of the risk posed by suboptimal TAT, the patient safety award 2013 6 has a specific category for initiatives aimed at reducing TATs.
While TAT for critical testing is largely dictated by what is possible given technical and other restraints, the focus must now be on what is considered timely and the point when delays could compromise patient wellbeing. This is particularly pertinent at times when pathology is experiencing significant challenges to quality in the drive to manage scarce healthcare resources. The literature on reasonable or acceptable TAT performance for non-critical testing is scant, with little agreement among clinicians 7 . This survey hopes to begin to address this deficiency.
Aim
To quantify optimal performance for TAT and the levels that TAT may be considered to compromise efficient and effective clinical management of patients.
Methods
Survey design
A questionnaire was devised by the authors and the National Clinical Biochemistry Audit Group (Appendix 1). TAT was defined as being the time from receipt to reporting for core investigations (i.e. U&E and LFT). It was agreed that consensus TAT would be defined as at least a two-thirds majority. The questionnaire was then distributed electronically using SurveyMonkey (www.surveymonkey.com) by Association for Clinical Biochemistry and Laboratory Medicine (ACB), to the head of department (HOD) of all known UK biochemistry laboratories listed in the ACB database. Each recipient was asked to report their stated TAT and provide a professional opinion as to what TAT would pose a significant risk to patient safety for the following categories of patient pathways, ED, outpatient (OP), inpatient (IP) and general practice (GP). In addition to the TAT survey, respondents were asked to highlight investigations requested by GP or outpatient department (OPD) which are prioritized and tests which are unavailable to GP. Returned questionnaires had their results collated electronically.
Results
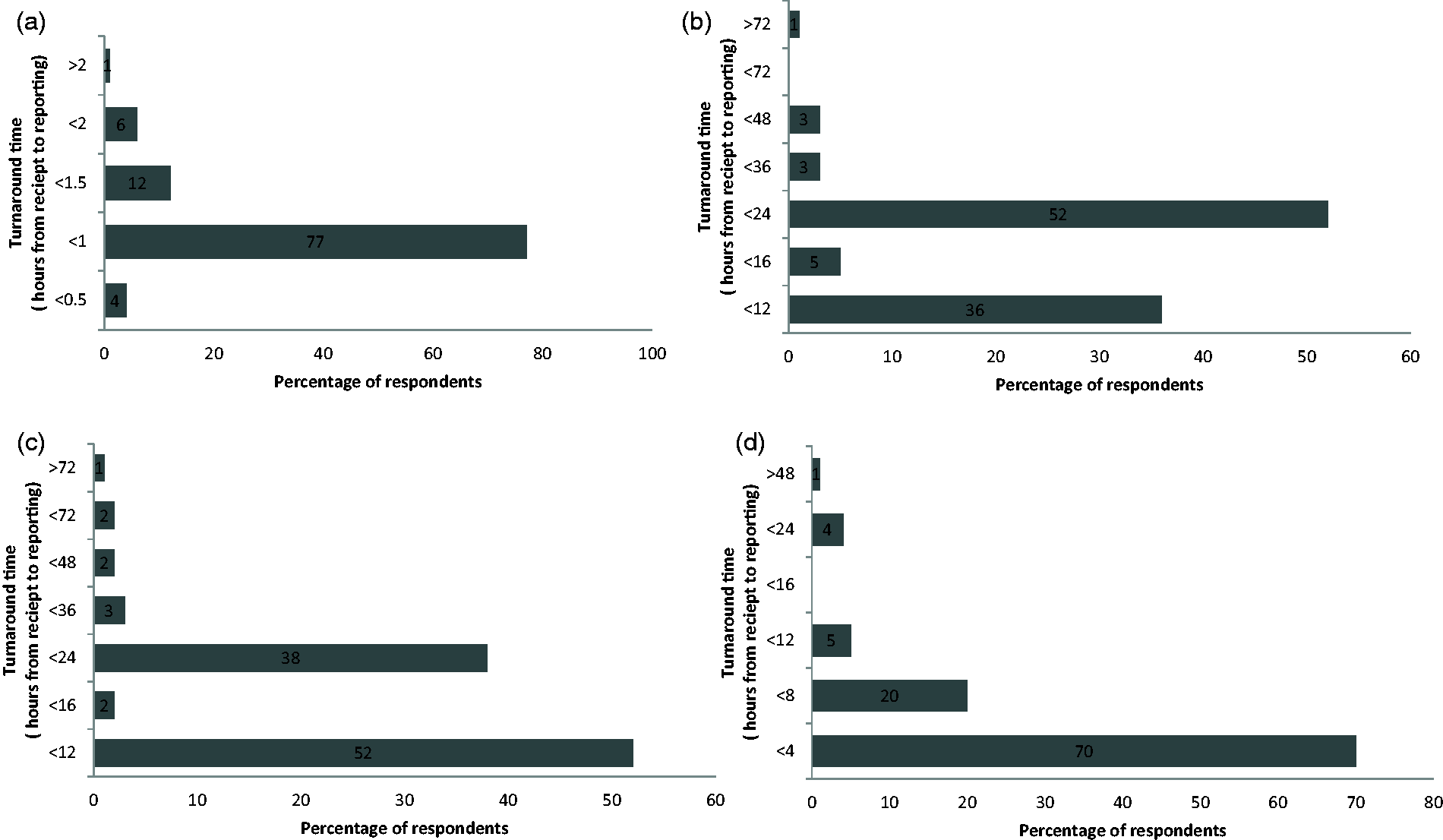
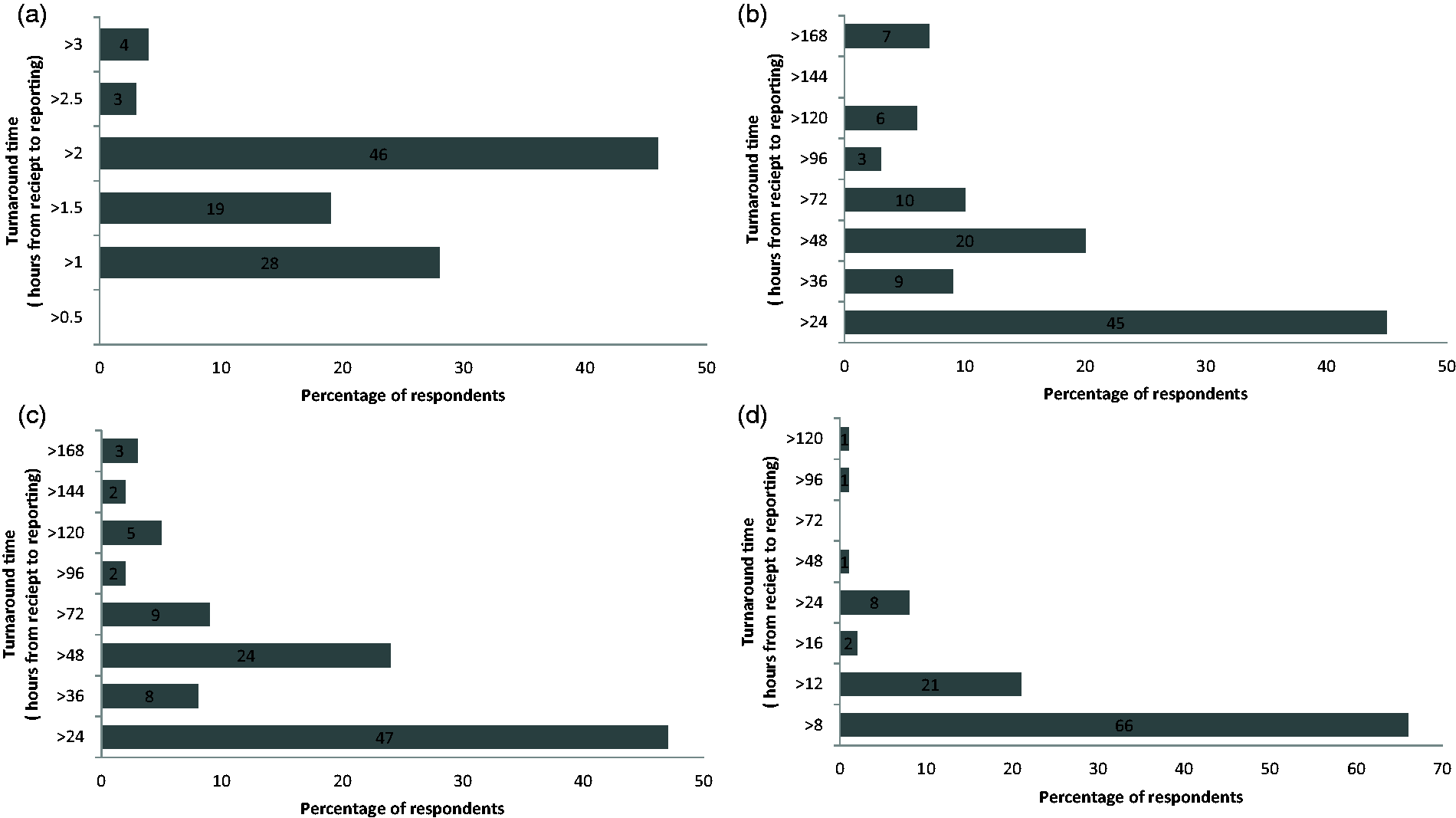
One hundred and seventy-three questionnaires were sent and 100 returned, giving a response rate of 58%. The regional breakdown is illustrated in Figure 1. Figures 2(a) to (d) and 3(a) to (d) show the percentage of respondents with a stated TAT less than the quoted value. The consensus optimal ED TAT was <1 h with >2 h considered unacceptable. The times for GP and OPD were <24 h and >48 h and for inpatients wards <4 h and >12 h. The total percentage for each of the consensus TAT is presented in Table 1. Some 39% listed tests that were prioritized (fast tracked) for GP/OPD and a further 29% stated that they would prioritize if requested and the remaining did not prioritize any testing from GP/OPD; 64% respondents did not restrict any test requests by GP/OPD and the remainder did not offer troponin (30%) or other tests (6%) including salicylate, paracetamol, ammonia and serum human chorionic gonadotropin.
Survey response rate per region as a percentage. (a) Target turnaround times for urgent/emergency department. (b) Target turnaround times for general practice. (c) Target turnaround times for outpatient department. (d) Target turnaround times for wards/inpatients. (a) Turnaround time for urgent/emergency department which in the professional opinion of those surveyed poses an unacceptable risk to patient safety. (b) Turnaround time for general practice which in the professional opinion of those surveyed poses an unacceptable risk to patient safety. (c) Turnaround time for outpatient department which in the professional opinion of those surveyed poses an unacceptable risk to patient safety. (d) Turnaround time for ward/inpatients which in the professional opinion of those surveyed poses an unacceptable risk to patient safety. Total percentage of respondents reporting a TAT within the consensus target. ED: emergency department; OPD: outpatient department.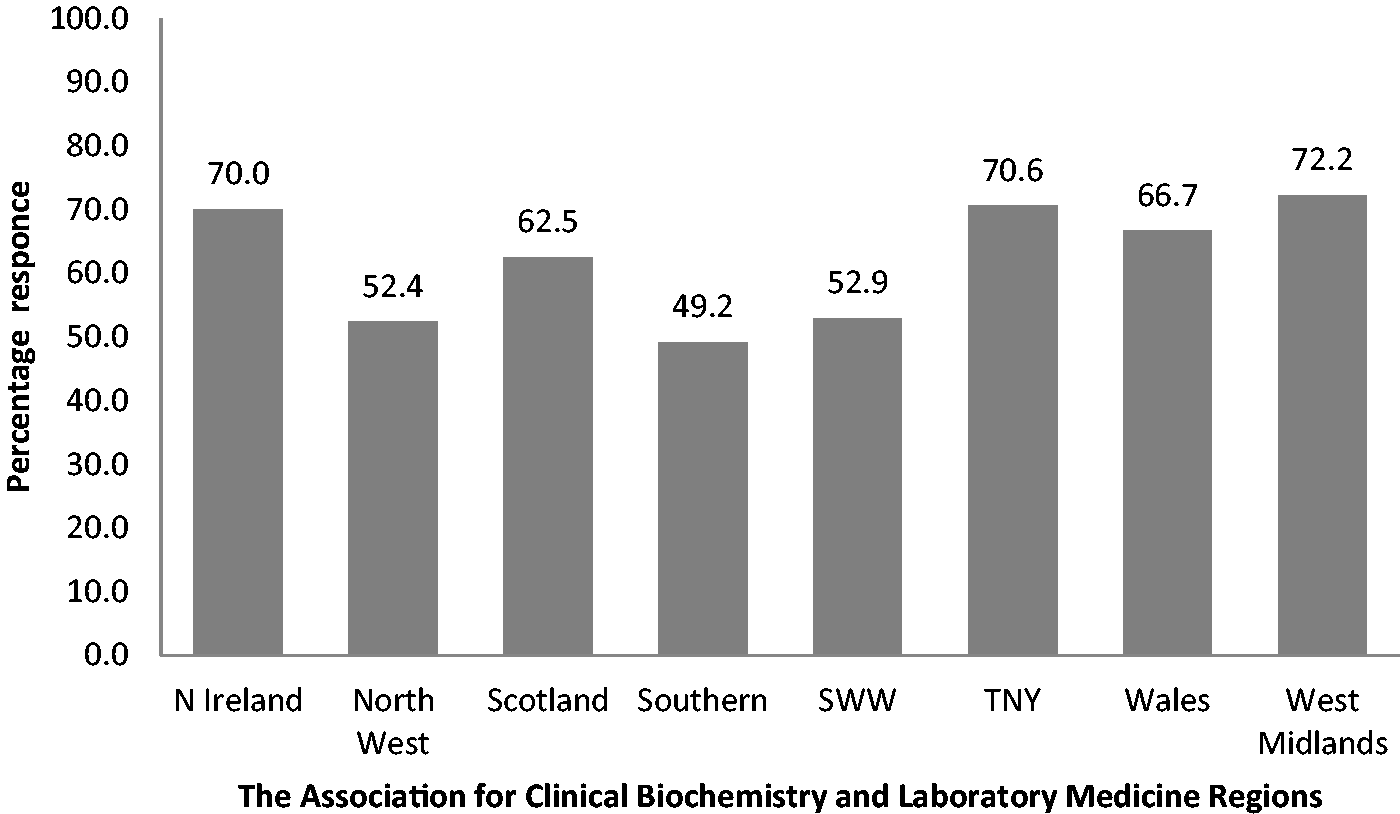
Discussion
Pathology is still enduring an unprecedented period of change and reform stimulated by the Lord Carter report
1
which combined with austerity has the potential to have a detrimental impact on the quality of service. A recent RCPath publication on the reconfiguration of pathology services
8
states:
The only ‘real' test of the quality of a medical laboratory service is its effect on patient outcomes. Anything else is a surrogate measure. Direct measurement of effect on outcomes is rarely possible, so surrogate measures have to be used, but their limits must be understood and a suitable spread of measures is essential.
The RCPath recently published a set of ‘KPIs 4 and the Association for Clinical Biochemistry and Laboratory Medicine has published a set of Quality Indicators for the clinical laboratory’. 9
Although timeliness of results reporting has not been a major focus in clinical laboratories, 10 there is increasing pressure from users to report results rapidly. This emphasis stems from the 4-h emergency care standard, 11 and an assumption that pathology TAT is a key factor in achieving the standard. Even though there are only sparse data, timeliness in reporting of laboratory results undoubtedly affects clinician and patient satisfaction as well as length of hospital stay 12 and could be a significant patient safety issue.
In the literature TAT can be defined by the different start and endpoints, e.g. brain to brain, sample collection to report, sample collection to therapeutic action, lab receipt to lab report. 7 The RCPath has indicated that by 2015, all laboratories are recommended to report TAT as sample collection to report. 4 This delay is in recognition that at present the infrastructure is not yet in place to allow accurate logging of sample collection data. Therefore, it seemed prudent for the purpose of this survey to define TAT as lab receipt to lab report. TAT can be measured at a macrolevel, e.g. population served: IP, OP, ED, primary care (GP) or at a microlevel, e.g. individual tests such a troponin or international normalized ratio. It was agreed to conduct the survey at the macrolevel to establish consensus minimum thresholds for specific population categories. This does not preclude any individual laboratory setting more exacting goals for subcategories, e.g. a particular patient pathway or test following consultation with the local stake holders.
The emphasis of the RCPath KPI and accreditation is increasingly focused on TAT requirements defined by the users. It could therefore be argued that a weakness of the current survey is that it seeks the opinion of HODs rather than users. Unlike previous publications on this topic, this survey extends beyond the traditional definition off a TAT and for the first time attempts to establish maximum TAT. It was felt by the authors that the HOD were best placed due to their knowledge of their users TAT expectations, analyte stability, clinical utility, biological variability and patient tolerance to provide informed responses.
This national survey revealed a high degree of agreement in relation to the optimum TAT for the ED and ward (IP) categories with 77% and 70% of respondents reporting a TAT target of <1 h for ED and <4 h for IP.
For GP and OP categories, there was a dichotomy of opinion. For GP samples, 36% and 52% reported a target TAT of <12 and <24 h, respectively. For OP samples, 52% and 38% reported a target TAT of <12 and <24 h, respectively. The consensus for ED is not surprising in the context of the well-published RCPath KPI for ED TAT of 1 h. At first glance, it seems remarkable that four laboratories have set a TAT of 30 min, yet when viewed in the context of the proposed aspiration of a 1 h TAT from collection to report by 2015, an analytical TAT (i.e. from receipt to report) of 30 min is probably where laboratories need to be to have a realistic chance of achieving this ambitious new target. The fact that only 4% of respondents are confident in setting a 30 min TAT confirms that a 1 h from collection to report, while achievable, may be difficult for many labs to meet for logistical reasons.
Prolonged TATs are well recognized as posing a significant risk to patient safety. 5 Although the principles of target and maximum standards have been adopted by other specialities, e.g. emergency medicine have used a 4-h wait but no more than 6 h, 11 we are not aware of similar maximum standards in pathology. Considering that this was a novel concept, it was surprising the degree of consensus amongst heads of departments. Respondents (46% and 66%) stated a maximum TAT of no greater than 2 and 8 h for ED and wards, respectively. Twenty-four hours was considered the maximum TAT for GP and OPD by 45% and 47% of respondents, respectively.
A combination of a high degree of consensus and the two-thirds majority threshold has resulted in very credible recommended TAT reported in Table 1.
Where any maximum TAT is exceeded, the risk to patients increases particularly if individual results are close to or at the threshold for critical result communication. 13 By definition, this is a level which requires clinical intervention without delay if serious risk to patient morbidity or even mortality is to be averted. The delay may mean that a result below the given threshold may in fact have reached that threshold by the time it is reported. The RCPath has published advice for pathologists regarding critical result communication out of hours in response to incidents where patients were harmed by a delay in result communication. 13
Urgent requests are received from non-ED areas at a low frequency and the ED TAT of<1 h should apply. A number of variables impact on the laboratories compliance, including test repertoire especially tests such as troponin which has an extended analysis time and identifying urgent requests amongst the routine workload. Among health professionals, there is a debate as to whether best practice for the management of patients requiring urgent assessment should involve urgent bloods or immediate admission to ED.
So as to inform the reader of current practice, the authors included additional questions in the survey on the repertoire and the mechanism of prioritizing urgent requests from GP.
We established that almost 70% of respondents will prioritize specific GP tests or will do so on request. Only 36% limit the test repertoire, and the majority of these were related to the availability of troponin testing.
With the well-published pressures on ED, it is not surprising that the vast majority of laboratories provide urgent bloods rather than direct ED referral. A minority of laboratories restrict availability of troponin. Both these factors, due to their impact on a laboratory’s ability to comply with TAT, should be considered when comparing TAT KPI published on laboratory quality dashboards as recommended in the pathology quality assurance review. 14
Conclusion
In conclusion, we consider that the figures provide a useful indication of the standards of performance which are appropriate across the broad group of clinical settings. For the first time, we present data based on the opinion of senior professionals in clinical biochemistry indicating a TAT threshold beyond which there is a significant risk to patient safety. We believe that the finding of this national survey provides a useful benchmark of current opinion which will inform the efforts of the professional bodies such as RCPath and the ACB in developing appropriate performance indicators. We recognize that more empirical standards of laboratory performance are desirable alongside those for other aspects of healthcare delivery.
Footnotes
Acknowledgements
This survey was undertaken under the auspices of the National Clinical Biochemistry Audit Group, assisted by the chair of the NI audit group Ms. Elizabeth McClean.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
DMK.
Contributorship
DMK conceived the survey. DMK and PA designed, analysed the data and wrote the first draft. DMK and PA reviewed and edited the manuscript. All authors approved the final version of the manuscript.
