Abstract
Background:
The use of biomarkers varies from disease etiognosis and diagnosis to signal detection, risk prediction, and management. Biomarker use has expanded in recent years, however, there are limited reviews on the use of biomarkers in pharmacovigilance and specifically in the monitoring and management of adverse drug reactions (ADRs).
Objective:
The objective of this manuscript is to identify the multiple uses of biomarkers in pharmacovigilance irrespective of the therapeutic area.
Design:
This is a systematic review of the literature.
Data Sources and Methods:
Embase and MEDLINE database searches were conducted for literature published between 2010-March 19, 2021. Scientific articles that described the potential use of biomarkers in pharmacovigilance in sufficient detail were reviewed. Papers that did not fulfill the United States Food and Drug Administration (US FDA) definition of a biomarker were excluded, which is based on the International Conference on Harmonisation (ICH)−E16 guidance.
Results:
Twenty-seven articles were identified for evaluation. Most articles involved predictive biomarkers (41%), followed by safety biomarkers (38%), pharmacodynamic/response biomarkers (14%), and diagnostic biomarkers (7%). Some articles described biomarkers that applied to multiple categories.
Conclusion:
Various categories of biomarkers including safety, predictive, pharmacodynamic/response, and diagnostic biomarkers are being investigated for potential use in pharmacovigilance. The most frequent potential uses of biomarkers in pharmacovigilance in the literature were the prediction of the severity of an ADR, mortality, response, safety, and toxicity. The safety biomarkers identified were used to evaluate patient safety during dose escalation, identify patients who may benefit from further biomarker testing during treatment, and monitor ADRs.
Introduction
According to the United States Food and Drug Administration (US FDA), a biomarker is a characteristic that is measured as an indicator of normal biological processes, pathogenic processes, or responses to an exposure or intervention, including therapeutic interventions 1 ; while there are variations in definition 2 ; undoubtedly, the use of biomarkers has expanded in the last decade, particularly in healthcare. 3 Currently, biomarkers have been categorized based on function into susceptibility/risk, diagnostic, monitoring, prognostic, predictive, pharmacodynamic/response and safety biomarkers. 4 It is therefore understandable that utility of biomarkers is integral to pharmacovigilance, which is the science concerned with the detection, assessment, understanding, and prevention of adverse drug reactions (ADRs) or any other drug-related problems, including the causality detection processes.5-7
Many examples of biomarker informing pharmacovigilance related activities in clinical practice exist, such as the monitoring serum creatinine to prevent the risk of drug induced nephrotoxicity. 8 Although this is a very important area of research, there are limited reviews focused on the reported use of biomarkers in patient safety or pharmacovigilance. Furthermore, the field of pharmacovigilance is continuously evolving and justifies a need to evaluate the latest advancements of biomarker applications. Therefore, the objective of this systematic review is to describe the multiple potential uses of biomarkers in pharmacovigilance processes reported in published literature.
Methods
Study design and data source
This is a non-quantitative systematic literature review conducted using the Cochrane Handbook for Systematic Reviews of Interventions 9 and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 10
The literature search was carried out using the databases of Embase (from 1980 to March 19, 2021) Ovid MEDLINE(R), Epub Ahead of Print, In-Process, In-Data-Review & Other Non-Indexed Citations and Daily (from 1946 to March 19, 2021) to identify the relevant titles and abstracts.
The following key terms were used, guided by standard search syntax: *Biomarkers, Pharmacological OR *Biomarkers/OR*Biomarkers, Tumor; *tumor marker/OR *biological marker/; (biomarker* OR biological marker OR tumor marker); *pharmacovigilance/OR pharmacovigilance; *patient safety/OR patient safety; (*biomarker/OR *biological marker/) AND pharmacovigilance; (*biomarker/OR *biological marker/) AND patient safety. The search was restricted to human, English and years 2010 to 2021. The search strategy is presented in Table S1.
Article selection
Any scientific article that contained information on the use of biomarkers, irrespective of the type of biomarker, in pharmacovigilance and fulfilled the eligibility criteria described below was reviewed. The definition of pharmacovigilance provided by the World Health Organization, which states that pharmacovigilance refers to the detection, assessment, understanding, and prevention of any drug-related problems, was used when evaluating articles for inclusion. 5
Inclusion criteria:
Scientific articles describing the use of biomarkers in pharmacovigilance, irrespective of the therapeutic area.
Review articles including meta-analyses were reviewed to identify additional relevant primary articles or new uses of biomarkers in pharmacovigilance.
Editorials, commentaries, and opinions were also reviewed to identify additional relevant references.
Limited to articles in the English language only.
No geographical limit.
Exclusion criteria:
Papers that did not fulfill the US FDA definition of a biomarker.
Articles only focused on genomic biomarkers.
Animal & preclinical studies.
Articles with incomplete information (eg, abstracts or posters with no full text and/or specific details on biomarker use).
Letters to editor and notes.
Duplicated articles.
Literature review
The first screening of titles and abstracts was carried out independently by 3 researchers (MS, SD, MGW) using the pre-determined eligibility criteria described above. Any disagreements were discussed in a consensus meeting to select final included articles.
Full articles were reviewed independently by 4 researchers (PY, OA, MM, and SD) using the same predetermined inclusion and exclusion criteria as the ones used for the abstracts. If there were disagreements, a similar process used for the final selection of abstracts/titles was followed, and a consensus meeting was organized to discuss the final inclusion or not of full articles.
All data were extracted using an Excel spreadsheet, where researchers also recorded the reason for any article exclusion. The data extracted included country of study, name of biomarker, use of biomarker, drugs involved, total number of patients included in study, sample size per group, disease, and organs related to biomarkers reported (Table S2). The quality control for the data extraction was conducted by 3 researchers (MS, TJ, and PY) to ensure accuracy of the extracted fields for each article.
The data were analyzed using descriptive statistics (counts, proportions, percentages) and narrative summary. Biomarkers were grouped into categories based on US FDA definition. 4 System Organ Class for Diseases/Conditions Under Study was categorized using the Medical Dictionary for Regulatory Activities (MedDRA) Version 24.0. 11
The detailed documentation of the search and review contributed to build the PRISMA flow diagram (Figure 1). Duplicated abstracts or full papers were also excluded in the final selection.
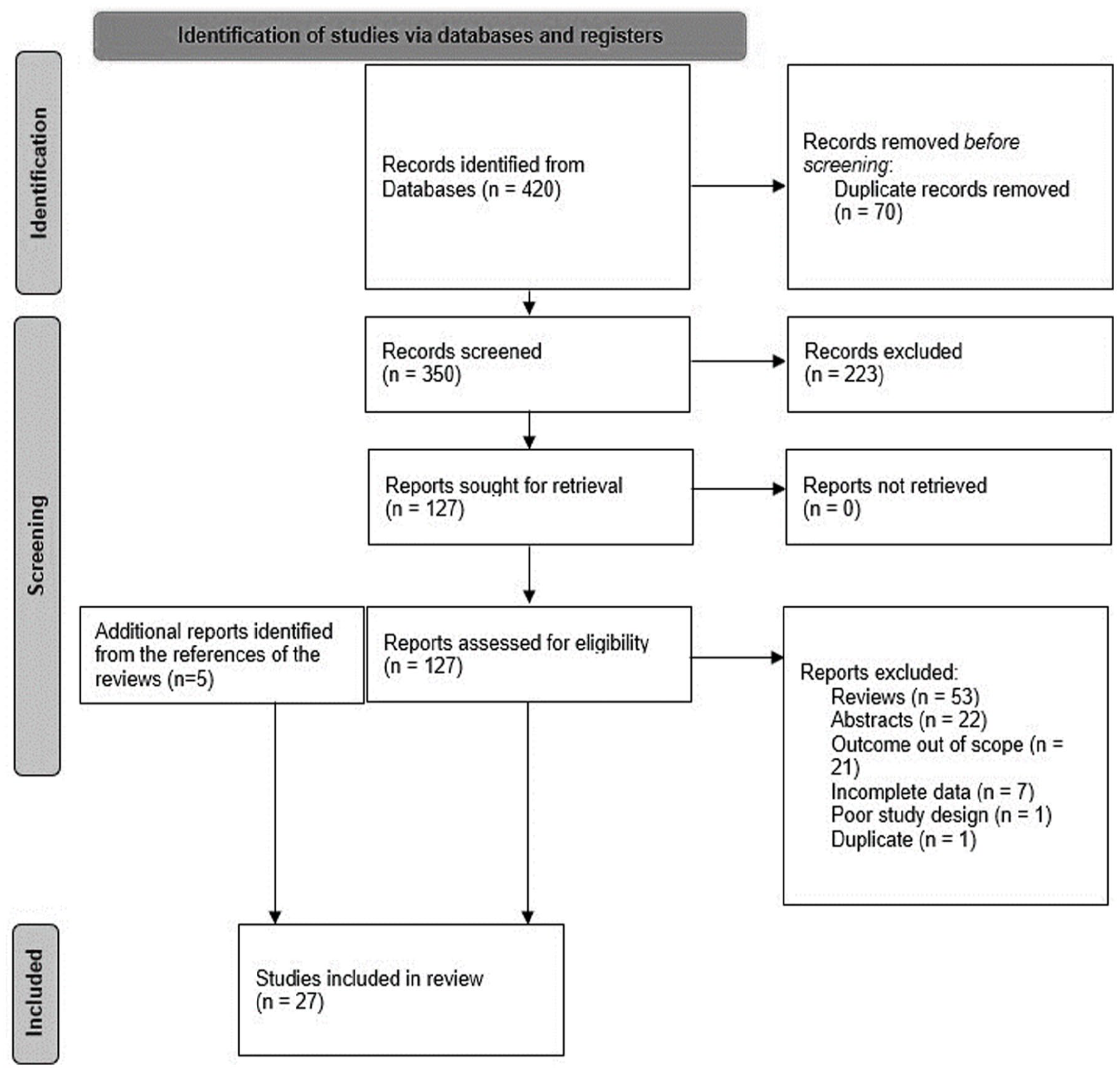
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram showing documentation of the literature search process. 12
Article selection
The literature search resulted in 350 articles which were evaluated for relevancy based on their titles and abstracts. Following the title and abstract evaluation, 127 articles were sought for retrieval. Following the subsequent reading of full texts, 22 articles were selected along with 5 additional relevant articles that were selected from reviewing the references of the 53 narrative reviews.13-39 The reasons for exclusion related to the following: narrative review article without relevant data (n = 53), only the abstract was available with incomplete information (n = 22), outcome was out of scope (n = 21), incomplete data (n = 7), poor study design (n = 1), and duplicated articles (n = 1). The details are documented in the PRISMA chart which can be found in Figure 1.
Sample characteristics
Most articles were from Spain (n = 5), followed by the United Kingdom (n = 4), Germany (n = 3), US (n = 2), Brazil (n = 2), Canada (n = 1), France (n = 1), Finland (n = 1), China (n = 1), Vietnam (n = 1), South Africa (n = 1), Argentina (n = 1), India (n = 1), Australia (n = 1), Switzerland (n = 1), and Netherlands (n = 1). The sample sizes for the biomarker of interest ranged from 1 to 904, although some sample sizes were not reported. Most studies were specific to certain diseases or conditions, and these are noted in Table 1. The potential uses of biomarkers for pharmacovigilance along with their categories are shown in Table 2. The US FDA evaluates the main use of a biomarker to determine its assigned category. The categories of biomarkers were chosen based on definitions from the FDA-National Institutes of Health (NIH) Joint Leadership Council and are shown in Table S3.
Disease or conditions studied in the literature, grouped by System Organ Class.

Potential uses of biomarkers for pharmacovigilance.

Abbreviations: IgG, immunoglobulin; IL, interleukin.
The US FDA evaluates the main use of a biomarker to determine its assigned category. Some biomarkers, particularly in the development phase, are tested with different objectives, but when they are used in “routine care” or in research, the main use of that biomarker determines its category.
Overall potential use of biomarkers in patient safety or pharmacovigilance
There were several different potential uses of biomarkers in pharmacovigilance identified in the literature. Figure 2 shows the distribution of the articles by category of biomarker identified. Most articles involved predictive biomarkers (n = 12), followed by safety biomarkers (n = 11), pharmacodynamics/response biomarkers (n = 4), and diagnostic biomarkers (n = 2). These biomarkers were categorized based on the US FDA guidance on classification of biomarkers. 1 Most common applications of biomarkers in pharmacovigilance were related to: prediction of the severity of an ADR, mortality, response, safety, and toxicity. The specific biomarkers identified within each category are listed in Table 3, which were again categorized based on the definitions provided by the US FDA.

Articles identified per biomarker category.
Specific biomarkers used in each category.

Abbreviations: IgG, immunoglobulin; IL, interleukin.
Use of safety biomarkers in pharmacovigilance
The safety biomarkers identified in the literature have been used in the monitoring of patient safety during dose escalation, the identification of which patients may benefit from further biomarker testing during treatment, and the monitoring of ADRs. Landmesser et al evaluated interleukin-6 (IL-6) and analyzed the effects of inclisiran on hematological and inflammatory biomarkers. 21 The investigators found no significant alterations in tumor necrosis factor alpha or IL-6 in patients taking inclisiran and no adverse effects on inflammation or immune activation during the treatment period, which demonstrates the safety of inclisiran since previous clinical studies using RNA-targeted treatments had reports of immunological and hematological ADRs. 21 Molecular biomarkers were also assessed by Kar et al and safety was evaluated using the plasma concentrations of inflammatory biomarkers such as IL-6 and C-reactive protein, showing the utility of using biomarkers to contextualize safety evaluations. 20
One study evaluated safety biomarkers with a long-term follow-up period to assess renal parameters in patients with chronic hepatitis C who are taking sofosbuvir and found that significant improvement in renal parameters was observed in patients with glomerular filtration rates greater than 45 ml/min/1.73 m2. 25 One of the more complex applications of safety biomarkers was in analyzing sotalol-induced corrected QT prolongation and specific T-wave morphological changes between patients who take oral contraceptives and those who do not. 30 One of the investigators’ findings was that drospirenone was associated with increased sotalol-induced QTc prolongation. 30 One study evaluated C-reactive protein and determined its role in Drug Rash with Eosinophilia and Systemic Symptoms syndrome. 34 Taegtmeyer et al found that C-reactive protein and procalcitonin are elevated in Drug Rash with Eosinophilia and Systemic Symptoms syndrome and can reach levels typically seen in acute bacterial infections, sepsis, or septic shock, highlighting the importance of choosing the proper diagnosis to ensure patient safety. 34 While the studies identified in the literature evaluated a multitude of different applications of biomarkers, they are all related to the potential use of biomarkers in pharmacovigilance.
Matthay et al assessed endothelial injury and inflammation using biomarkers such as angiopoietin 2, IL-6, IL-8, Protein C and receptor for advanced glycation end products to examine association of predefined hemodynamic and respiratory ADRs following the administration of mesenchymal stromal cells to patients with moderate to severe acute respiratory distress syndrome. 23 Although there was some evidence of decreases in biomarkers, the results were inconclusive, since no patients in the treatment group experienced the ADR of interest. 23
Use of predictive biomarkers in pharmacovigilance
Most of the predictive biomarkers identified in the literature were used to identify patients likely to experience inflammatory or immunologic ADRs.13,15,16,22,27,36,37 Mortality as an ADR to highly active anti-retroviral therapy was found to be predicted by hepatic parameters such as alanine transaminase, albumin, alkaline phosphatase, and hemoglobin 10 and in patients treated with mesenchymal stromal cells for steroid-refractory grade II–IV acute graft-versus-host disease, by the biomarkers IL2Rα, tumor necrosis factor receptor 1, hepatocyte growth factor, IL-8, elafin and regenerating islet-derived protein 3α. 35
With more emphasis being placed on reducing the inappropriate use of antibiotics, predictive markers with the potential to limit the overuse of antibiotics is of importance. C-reactive protein point-of-care testing was explored in this way to reduce antibiotic use in patients with non-severe acute respiratory tract infections without compromising patient recovery. 16 A pilot study evaluated the assay of activated partial thromboplastin time and procalcitonin as possible biomarkers to assess the need for empiric antibiotic use in patients in the intensive care unit. 31 Among obese women on levonorgestrel contraceptive, biomarkers of cardiovascular risk included assessment of systolic blood pressure, diastolic blood pressure, high-density lipoprotein, low-density lipoprotein, carotid intima-media thickness, and brachial artery flow-mediated dilation. 39 For neonates, the urinary biomarkers Kidney Injury Molecule-1 was found to monitor for aminoglycoside induced nephrotoxicity. 24
Use of pharmacodynamic/response biomarkers in pharmacovigilance
Pharmacodynamic/response biomarkers can also play a key role in pharmacovigilance. Several studies were identified using response biomarkers,25,28,29,38 with most of the biomarkers being cytokines. Medeiros et al found that patients with chronic hepatitis C and baseline glomerular filtration rates less than 45 ml/min/1.73 m2 did not experience alterations in renal parameters with sofosbuvir therapy. 25 Ranki et al described how ONCOS-102 treatment induced upregulation of PD-L1 in tumors and showed changes in response to interferon gamma. 29 Pannucci et al described how anti-factor Xa can be used to monitor breakthrough venous thromboembolism and adjust enoxaparin dosing. 28 Anti-factor Xa was classified as a pharmacodynamic/response biomarker rather than a safety biomarker because of its main use in assisting in dose selection and measuring a response to products, including the use as a measure of potential harm. 1 The knowledge on these pharmacodynamic biomarkers can be useful in determining or measuring a response to products.
Use of diagnostic biomarkers in pharmacovigilance
Diagnostic biomarkers can play a role in pharmacovigilance as well, however more studies are needed to identify their contribution to safety. Hellyer et al evaluated biomarker-guided antibiotic stewardship in suspected ventilator-associated pneumonia but ultimately could not conclude the usefulness of biomarkers in reducing need for antibiotics because compliance with recommendations based on low IL-8 and IL-1β was not maintained. 19 Histological biomarkers were used by Schindewolf et al to diagnose the cause of skin lesion that occurred during low molecular weight heparin therapy. 32 Heparin induced thrombocytopenia is confirmed by skin biopsy showing evidence of lymphocyte and eosinophil infiltration, as well as thrombus formation in dermal vessels. 32 The study used these biomarker characteristics to eliminate low molecular weight heparin as the cause of the skin lesions. 32
Discussion
Many biomarkers are being investigated for potential utility in pharmacovigilance and the most common potential uses of biomarkers in pharmacovigilance in the literature were the prediction of the severity of an ADR, mortality, response to treatment, safety, and toxicity. Since biomarkers are indications of what is happening within a patient’s body, they can be helpful in interpreting their current or future response to a drug therapy. Some biomarkers may be elevated in conjunction with an ADR, while other biomarkers may be indicative of sensitivity to a drug compound. While biomarkers that fall into the safety category have the greatest application to pharmacovigilance, some other categories of biomarkers are related to safety as well. As the field of pharmacovigilance evolves, it is important to monitor advancements in biomarker applications to ensure their use to protect patients. This literature review sought to find recent information and trends in the use of biomarkers in patient safety and pharmacovigilance in general.
Use of safety biomarkers in pharmacovigilance
The safety biomarkers in the literature were used in the evaluation of patient safety during dose escalation, identification of patients who could benefit from further biomarker testing during treatment and monitoring of ADRs. Since many safety biomarkers are non-invasive, the benefits often outweigh the risks when using these biomarkers to assist in safety efforts. For example, IL-6 can be a marker for immune system activation and only requires a blood sample from the patient. 21 These findings from biomarker testing can describe a patient’s response to therapy which may correspond to clinical findings. In dose escalation studies, it can be difficult to assess a patient’s toxicity threshold simply by measuring vitals or performing a physical assessment. Biomarkers can be useful in seeing how the patient’s body is responding to a drug so that treatment can be adjusted accordingly. 1 When biomarkers that are known to correlate to certain drug therapy effects, such as inflammatory biomarkers and inclisiran, this can be helpful in monitoring for adverse effects. 21 This can be critical to safety efforts since there are usually a limited number of ways to promptly know if a patient is experiencing toxicity from a treatment.
Biomarkers in the safety category have also been used in long-term settings which demonstrates the potential value of biomarkers in conditions such as chronic hepatitis C. 25 It is important to note that some biomarkers can be elevated in multiple different conditions, so a distinct diagnosis is not always possible from biomarkers alone. For example, C-reactive protein can be elevated in both Drug Rash with Eosinophilia and Systemic Symptoms syndrome and septic shock. 34 However, biomarkers such as C-reactive protein can still be important in narrowing down conditions and ADRs. Westwood et al was a systematic review that evaluated the utility of procalcitonin testing to identify those who may have a bacterial infection in the intensive care unit and found that procalcitonin could be an effective biomarker when used to guide discontinuation of antibiotics for sepsis, but it is not clear if the costs and effects are directly related to the procalcitonin testing. 40 Earlier discontinuation of an unwarranted therapy can keep patients safe. Overall, safety biomarkers have the potential to offer clinical guidance and safety assessments in an efficient manner based on the effectiveness of biomarker use presented in the literature.
Use of predictive biomarkers in pharmacovigilance
Predictive biomarkers can also contribute to pharmacovigilance despite not being in the safety category of biomarkers. Certain biomarkers such as albumin and alkaline phosphatase can be predictive of mortality in disease states such as human immunodeficiency virus. 14 These biomarkers are commonly measured in lab tests and can be used to construct a score for predicting short-term mortality. 14 Predictive biomarkers can also be used to reduce unnecessary antibiotic use, thereby eliminating the potential for unnecessary ADRs in patients. This category of biomarkers also has been used to optimize clinical outcomes which can keep patients safe by avoiding additional therapies.
Use of pharmacodynamic/response biomarkers in pharmacovigilance
Pharmacodynamic/response biomarkers have also been investigated for potential utility in pharmacovigilance. These biomarkers can indicate the body’s response to a drug. The same drug may have different effects in one patient when compared to another patient due to differences in these biomarkers. Shu-Feng Zhou identified that some genetic mutations are associated with specific ADRs and can act as pre-warning biomarkers after using genetic testing. 41 By understanding the role of biomarkers in this setting, the best and safest therapies can be chosen for each individual patient. It is important to note, however, that the extent to which genetic factors contribute to drug toxicity depends on the gene effect in addition to several non-genetic factors (eg, drug-drug interactions). 42
Use of diagnostic biomarkers in pharmacovigilance
Diagnostic biomarkers can also be vital to pharmacovigilance. Schindewolf et al found that the risk for delayed-type hypersensitivity differs considerably between individual patient cohorts and the data collected can help to tailor anticoagulatory treatment individually and to increase patient safety. 32 While some of the methods used with diagnostic biomarkers need further validation, they may be a promising tool in determining patients with a disease. By appropriately identifying the disease status of a patient, proper therapy can be initiated which avoids the potential for inappropriate treatments which may have unnecessary side effects. This is important because antibiotic resistance continues to be prevalent in healthcare settings and can make it more difficult to adequately treat a patient.
Limitations
There are some limitations to this study. There are limited reviews on the use of biomarkers in patient safety and pharmacovigilance, especially in the monitoring and management of ADRs. Most of the literature investigated biomarkers for potential utility in pharmacovigilance although, they were not directly tested in routine safety surveillance activities. Therefore, more studies are needed to determine the utility of these biomarkers in routine pharmacovigilance. Techniques associated with the development of genomic biomarkers, biomarker testing, techniques of biomarkers associated with mutations, insertions, deletions, or rearrangements in deoxyribonucleic acid or ribonucleic acid were not the main focus of this article and were therefore excluded from the review. However, genomic biomarkers will be reviewed in a future manuscript.
Conclusion
Safety, predictive, pharmacodynamic/response, and diagnostic biomarkers have been investigated for potential utility in pharmacovigilance and continue to be evaluated. These biomarkers have been used in the prediction of the severity of an ADR, mortality, response, safety, and toxicity. The benefits of using biomarkers in pharmacovigilance include the ability to assess safety in ways that may not be possible with other techniques such as imaging, the minimally invasive techniques required to assess and interpret biomarkers, and the abundance of information from biomarkers that can subsequently be used to guide therapy and ensure safety in patients.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719231164528 – Supplemental material for The Use of Biomarkers in Pharmacovigilance: A Systematic Review of the Literature
Supplemental material, sj-docx-1-bmi-10.1177_11772719231164528 for The Use of Biomarkers in Pharmacovigilance: A Systematic Review of the Literature by Maribel Salas, Maxine Gossell-Williams, Priyanka Yalamanchili, Sameer Dhingra, Marina A Malikova, Omar Aimer and Toluwalope Junaid in Biomarker Insights
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
