Abstract
Background
Reporting critical results in a timely manner is a crucial role of clinical laboratories. Traditionally, these results were reported using the phone or fax system. However, there are now other modes of communication for this reporting. Quality improvement in any organization is driven by detection of errors and benchmarking against peers. In the case of critical result reporting, there are few current widely used Benchmarking schemes.
Methods
The Roche Clinical Chemistry Benchmarking Survey in 2019 added questions about critical result reporting including the mode of communication and turnaround time key performance index. This survey includes over 1100 laboratories from 20 countries.
Results
The survey revealed a range of communication strategies with phone calls still the commonest followed by email. The key performance index for most laboratories was less than 10 min.
Conclusion
Benchmarking can provide key information for quality improvement activities, particularly pre- and postanalytical.
Introduction
Inefficient clinical handover or communication with clinicians at critical steps in the total testing cycle is a significant source of delay and subsequent potential patient harm. 1 To improve the timeliness of direct contact with the requesting doctor, laboratories have started using mobile phones and SMS messaging. To determine the extent that this technology was adopted in different countries in the Asia Pacific Region, Roche Diagnostics added some questions to their 2019 Clinical Chemistry Laboratory Benchmarking Survey. 2
This Survey is a biennial online program that was designed to collect information on three key areas of laboratory practice with a focus on but not limited to clinical chemistry and immunoassay quality, speed and cost.
The survey started in 2011 with 181 laboratories in 12 countries and to date, has included 3500 laboratories from more than 20 countries. In the latest 2019 survey (July 2019–November 2019), questions relating to pre-and postanalytical processes were added. In this letter, we report on some of the findings.
The survey asked the following questions about critical result reporting:
What is your critical result reporting time key performance index (KPI)? How does your laboratory notify the clinician of critical test results?
Results
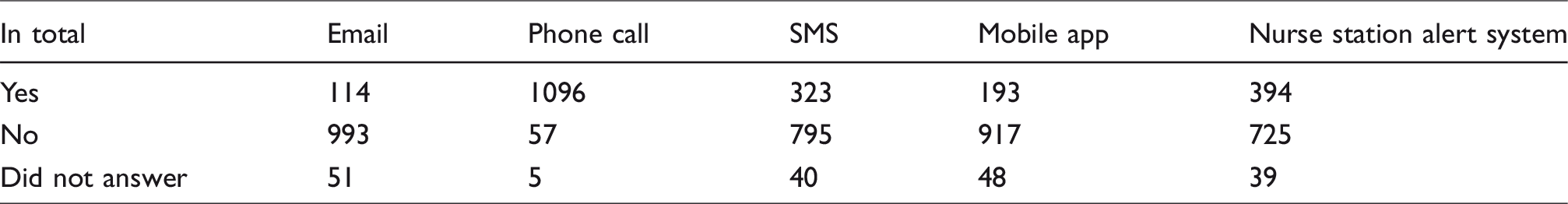
In total, 909 out of the 1158 participants in the July 2019 survey provided data on critical result reporting time KPI, with the remaining 249 reporting they do not have such KPI. The responses to these questions are given in Table 1, and Figures 1 and 2.
Summary of all survey responses.
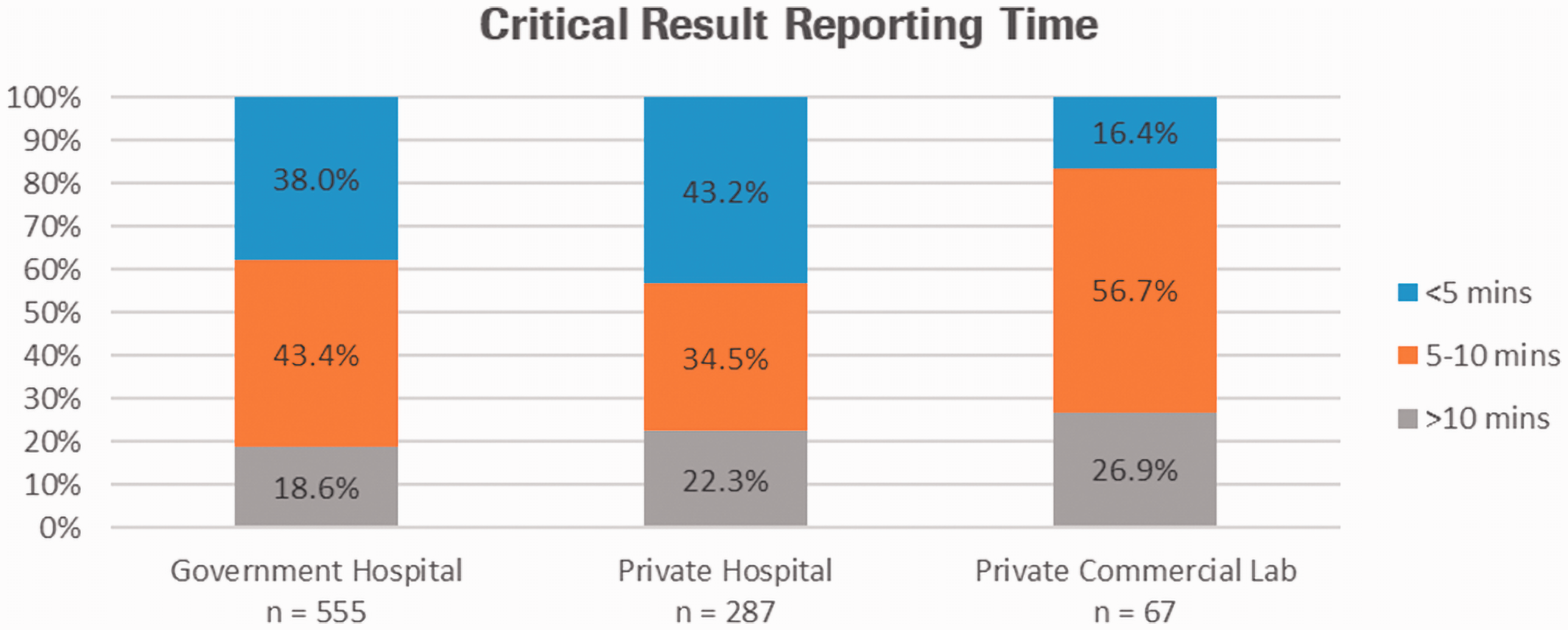
The critical reporting time KPI by laboratory category.
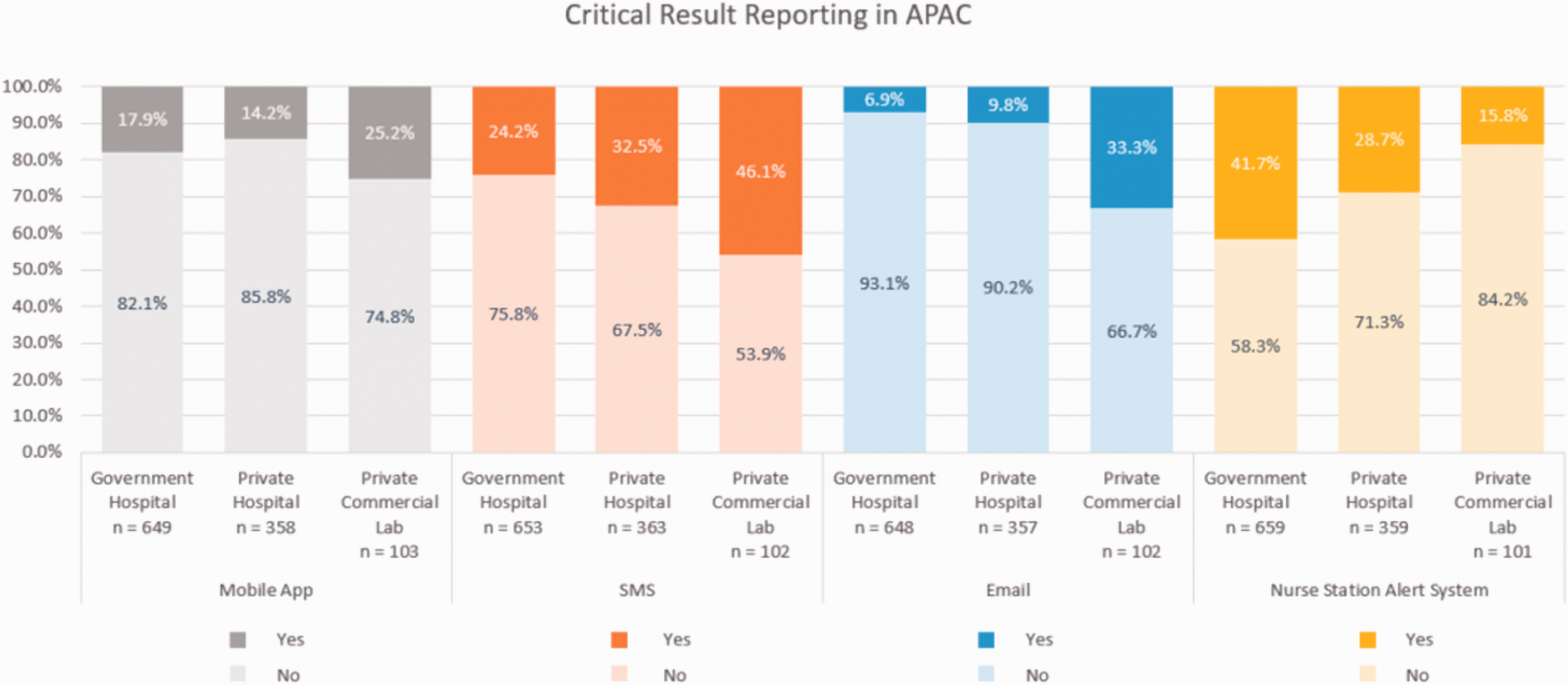
Methods of communication of critical results to referring doctors by laboratory type.
Most participants were Roche customers, although the survey was sent to many sites. All participants have access to their benchmarking report via the Roche platform (labinsights.com).
A 'critical' risk result is determined by the laboratory. Still, it means a result requiring immediate medical attention and action because they indicate a high risk of imminent death or significant patient harm. 3 Thus, it needs to be effectively communicated as soon as possible by the fastest method available to the laboratory. This is the final step in the laboratory testing process following authorization. The critical result turnaround time KPI is the time taken by the laboratory after final authorization to contact with the ward/referrer.
There were three-time intervals given for reporting a critical result. Approximately 40% of public or private hospitals reported a KPI of 5 min, with a similar percentage reporting 5–10 min. Not surprisingly perhaps, most private laboratories had a reporting KPI of 5–10 min. The method of reporting was broad and utilized many modes, the most common in private laboratories being emails, SMS in private hospitals and nurse station alert in government hospitals. Of the participants, 534 (46.1%) laboratories only utilize one form of communication to report critical results; 344 (29.7%) laboratories utilize two; 255 (22.0%) laboratories utilize three and, 25 (2.2%) laboratories did not answer the question or do not report critical results.
Discussion
Reporting a critical result rapidly to the correct clinician is one of the essential roles a laboratory has. 4 , 5 The method used will be different depending on whether the clinician is onsite or not, and this probably is why the different types of laboratory use other processes. Each of the methods used has some disadvantages. These include getting the attention of the clinician in an appropriate timeframe and the laboratory having acknowledgement that the report has been received.
There is no standard set for the turnaround time for reporting a critical result. However, it would be assumed that this should be as fast as possible. The range of KPIs set by the laboratory for these results does vary, which represents an opportunity for improvement.
Omdahl 6 defines benchmarking as an ongoing process of measuring and improving the performance of a company's products, services or processes against those considered to be the best in the industry.
Although laboratory benchmarking schemes exist at the local level, there are only a few global schemes, none of which is as large as the Roche scheme. 7 , 8 Laboratory benchmarking approaches such as Q-probe studies which use one-off surveys cannot compare improvements in performance over time. 9 While studies that describe advances in critical result reporting KPIs such as time to reporting by the adoption of instant messaging applications have been reported, 10 , 11 we are unaware of benchmarking programmes in this space. This is the first survey on the Roche scheme that surveyed aspects of critical reporting. While there is variation in the critical result reporting time as well as the method used for reporting, the majority of laboratories used one or more forms of instant messaging applications. Further surveys on the Roche benchmarking scheme could be refined to clarify improvements in critical reporting time KPI relative to uptake in instant messaging applications and to gather more detailed information on this aspect of the laboratory process.
This research provides an overview of aspects of critical result reporting, which would be valuable to laboratories regardless of the analytical system in use. Limitations of this research include being the first survey to include data on critical risk reporting KPIs, it only gives a snapshot in time and is not able to assess improvements in individual laboratories. Other limitations include not acquiring specific information relating KPI targets to actual performance and whether or not instant messaging applications made a difference to the achievement of KPI targets. The variation in KPI targets may be due to different patient population or laboratory setting and should ideally be set by consensus agreement with the clinicians that the laboratory serves.
There are limitations with the survey data. The survey is a benchmarking exercise designed to collect key data from a significant sample of laboratories. It is not possible in this survey to determine the impact of the various communication strategies or reporting time KPIs in improving patient safety. In terms of a quality improvement process, laboratories could see that other similar sites can achieve a shorter turnaround time, and hence change their KPI to match ‘best practice’.
Conclusion
In summary, the Roche scheme provides an ongoing (growing) large sample of benchmarks that can be used by participants to improve their performance and the performance of individual countries in many aspects of laboratory practice including critical risk reporting. Benchmarking can provide key information for quality improvement activities, particularly pre- and postanalytical.
Footnotes
Acknowledgements
We would like to acknowledge Roche Diagnostics Asia Pacific for providing the Survey data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
TB.
Contributorship
TB conceived the idea to publish. TB, SM, WW analysed the data; WW prepared the figures; TB wrote the first draft. All authors reviewed and edited the manuscript including revisions and approved the final version of the article.
