Abstract
Background
Anthracycline drugs are effective anticancer agents, but their optimal use is limited in many patients by the associated cardiotoxicity, even at designated safe doses. As conventionally sensitive cardiac troponin-I assays fail to reliably quantify concentrations of cardiac troponin-I below 30 ng/L, we investigated the potential role of high-sensitive cardiac troponin-I in the detection of subclinical cardiomyocyte injury in patients treated with anthracycline agents.
Methods
Serial high-sensitive cardiac troponin-I concentrations were assessed in 84 patients, receiving anthracycline-containing (n = 38) and non-anthracycline-containing (n = 46) regimens. Results were assessed for change from pretreatment levels and evaluated according to unisex and gender-specific 99th percentiles (25 ng/L and M: 34 ng/L, F: 16 ng/L, respectively).
Results
A significant increase in high-sensitive cardiac troponin-I was observed in the anthracycline cohort following five cycles of treatment, with the greatest change correlating to an absolute δ increase of 30.7 ng/L in the early-dose group (early-dose group: P < 0.0001, late-dose group: P < 0.01 and continuous-dose group: P < 0.0001). Doxorubicin dose did not correlate directly with high-sensitive cardiac troponin-I concentrations (Spearman r < −0.22). No significant changes in high-sensitive cardiac troponin-I were reported among the non-anthracycline cohort with all measurements below the 99th percentiles.
Conclusions
Treatment with anthracycline-based chemotherapeutic regimen demonstrated significant elevations of high-sensitive cardiac troponin-I, indicative of subclinical cardiomyocyte damage. This study demonstrates a role for high-sensitive cardiac troponin-I in evaluating those patients where cardiotoxicity is a concern and a potential future role as a biomarker in optimizing cardioprotective treatments in patients receiving anthracycline therapy.
Introduction
Anthracyclines, mainly doxorubicin, are widely used chemotherapeutic agents in the treatment of solid organ tumours, haematological malignancies and soft tissue sarcomas. However, cardiotoxicity remains a drawback in their use.1,2 Anthracyclines mediate their action through intercalation of DNA, free radical generation and inhibition of topoisomerase IIα. 3 Anthracycline-mediated oxidative stress, free radical generation and individual genetic variants are factors considered causative of cardiotoxicity. 4 In addition, a role for topoisomerase IIβ present in cardiomyocytes in doxorubicin-induced myocyte injury has been hypothesized. 5 Such toxicity can have a serious impact on the cardiovascular health of cancer patients which can culminate in complications including ventricular dysfunction, cardiomyopathy and heart failure.2,6,7
It is recognized that anthracycline-mediated cardiotoxicity which often develops in the early stages following treatment initiation8,9 is commonly reversible with appropriate medical intervention, while cardiotoxicity presenting following treatment completion is largely irreversible, suggesting early detection is essential to allow for the prevention or attenuation of cardiac damage. 10 These clinical scenarios present challenges which draw upon the expertise of both cardiologists and oncologists to offer an effective means of cardiac damage monitoring during chemotherapy. 11
While echocardiography (ECHO) is commonly used to detect anthracycline-mediated cardiac damage, it has limited sensitivity in detecting subtle subclinical changes in cardiomyocytes which are indicative of cardiac injury. Since cardiac troponin-I (cTnI) has good specificity in identifying cardiac injury, it has been investigated for its potential in detecting and assessing risk of anthracycline-mediated cardiotoxicity.12–14 Early studies of such toxicity among patients treated with chemotherapeutic agents, including anthracyclines, utilized conventional cTnI assays which have provided compelling indications of a correlation between elevated cTnI concentrations and cardiac impairment. 15 This association has been reinforced with evidence of decreases in left ventricular ejection fraction (LVEF) following anthracycline chemotherapy. 16 Elevated cTnI concentrations quantified using ‘ultrasensitive’ assays and a strong association with peak systolic longitudinal myocardial strain have also been recognized among patients treated with anthracycline-containing regimens, who later developed cardiotoxicity. 17 The high-sensitive cardiac troponin-I (hs-cTnI) assay demonstrates a superior analytical range with a limit of detection (LOD) of 1.2 ng/L versus the 30 ng/L achieved by the conventional cTnI assay, allowing the quantification of previously undetectable concentrations of cTnI.
In this study, we report the clinical utility of serial hs-cTnI assessment in the detection of subclinical anthracycline-mediated cardiomyocyte injury in a patient cohort prior to and during chemotherapy treatment cycles.
Methods
Study design
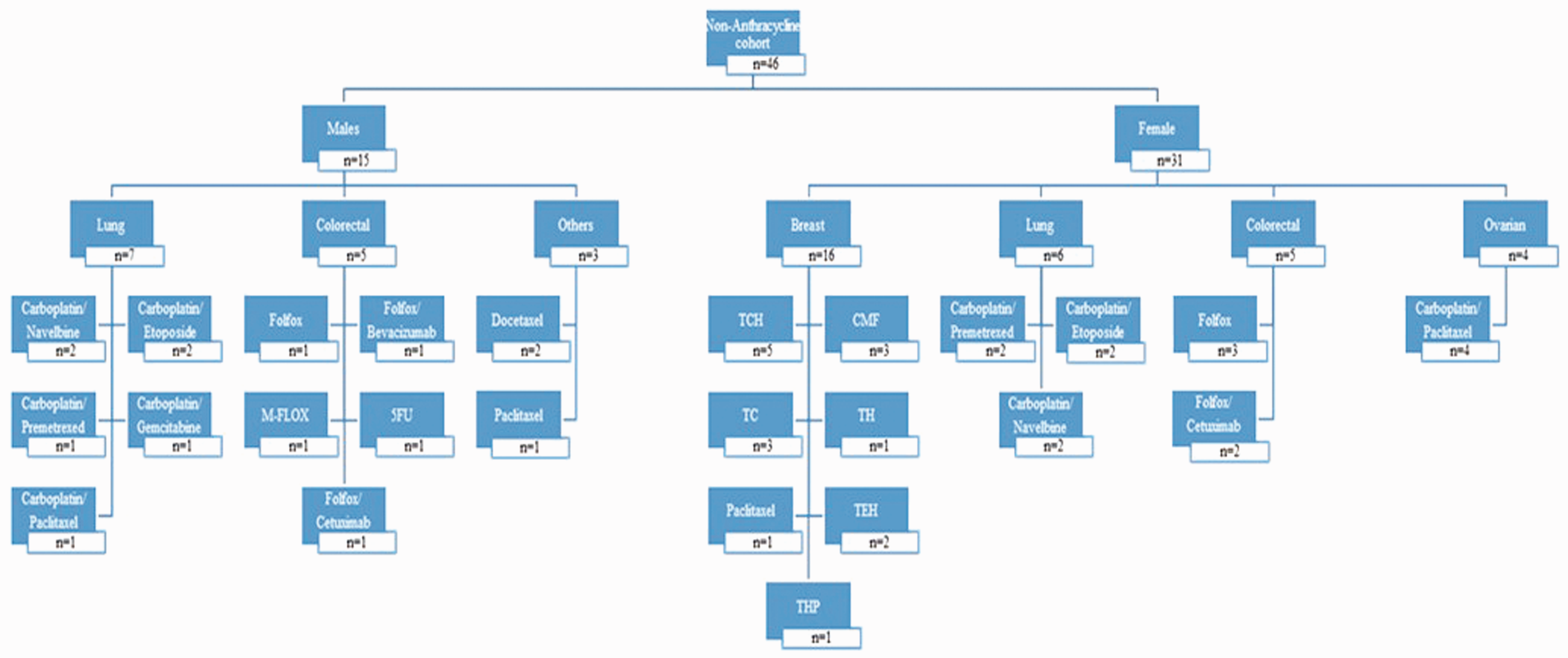
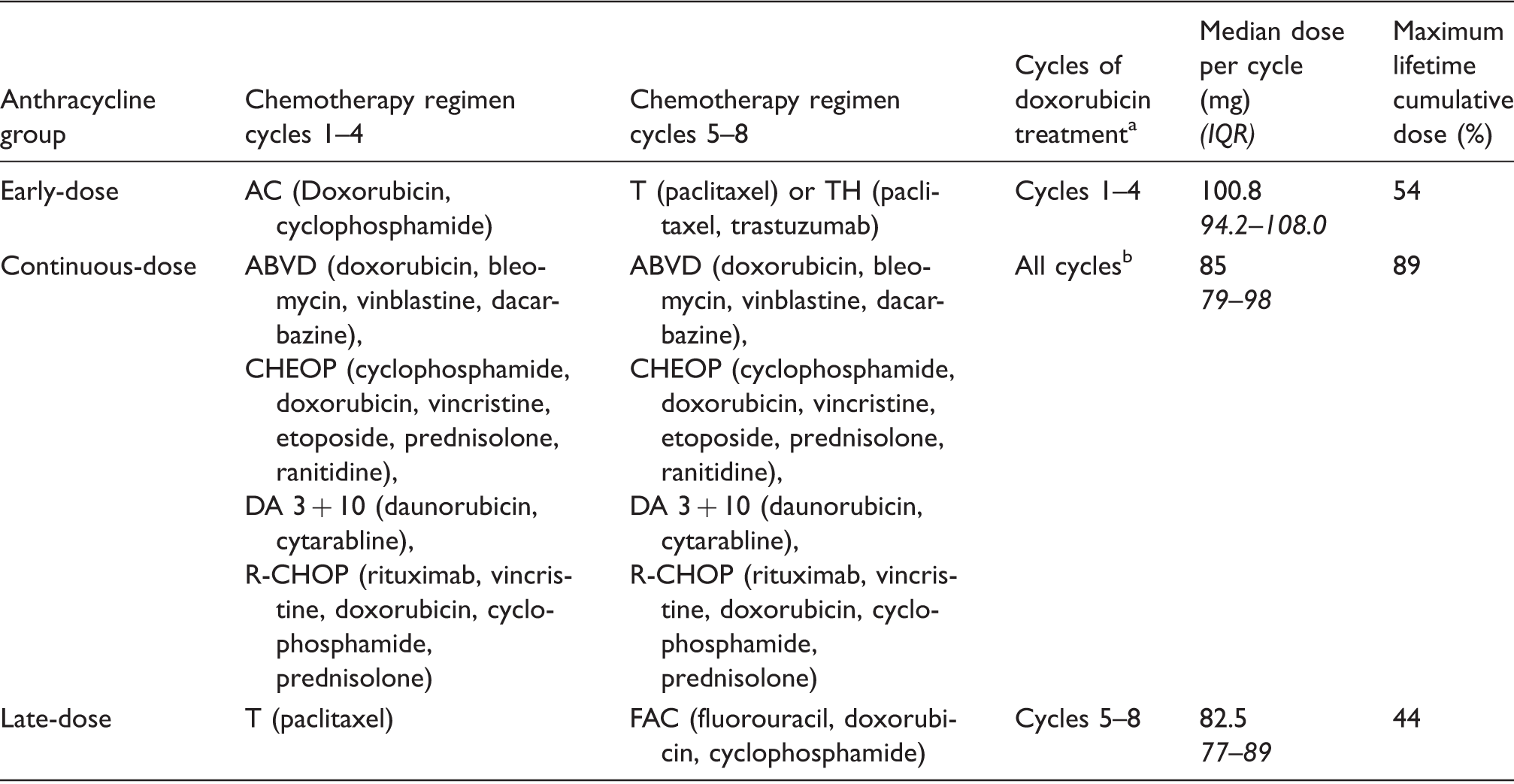
Ethical approval in accordance with the Declaration of Helsinki was attained from the Mater University Hospital Ethics Committee (Ref: 1/378/1387), and all patients provided signed consent to participation. A cohort study was designed in which we prospectively recruited 84 patients diagnosed with oncological or haematological malignancies who were naïve to prior chemotherapeutic treatment. Details of all chemotherapeutic agents were recorded, and patients were classified into two treatment cohorts, based on whether they received an anthracycline (n = 38 patients) or a non-anthracycline (n = 46 patients) regimen, Figures 1 and 2, respectively. The anthracycline and non-anthracycline cohorts were predominantly composed of female breast cancer patients. Lung and colorectal cancer diagnoses were also frequently observed among both female and male patients in the non-anthracycline cohort. Doxorubicin (Adriamycin®) was the main anthracycline used with one patient receiving daunorubicin. Patients in the anthracycline cohort were assigned based on dosing regimen (early-dose, continuous-dose and late-dose group) according to the cycles in which an anthracycline agent was administered (Table 1). A diverse array of chemotherapeutic strategies was utilized, with regimens consisting of between four and eight treatment cycles.
Diagnostic and therapeutic characteristics of the anthracycline cohort. Diagnostic and therapeutic characteristics of the non-anthracycline cohort. Doxorubicin dosing characteristics for early, continuous and late-dose anthracycline groups. Treatment cycle duration between 14 and 28 days for all regimen. Regimen consisting of up to eight treatment cycles.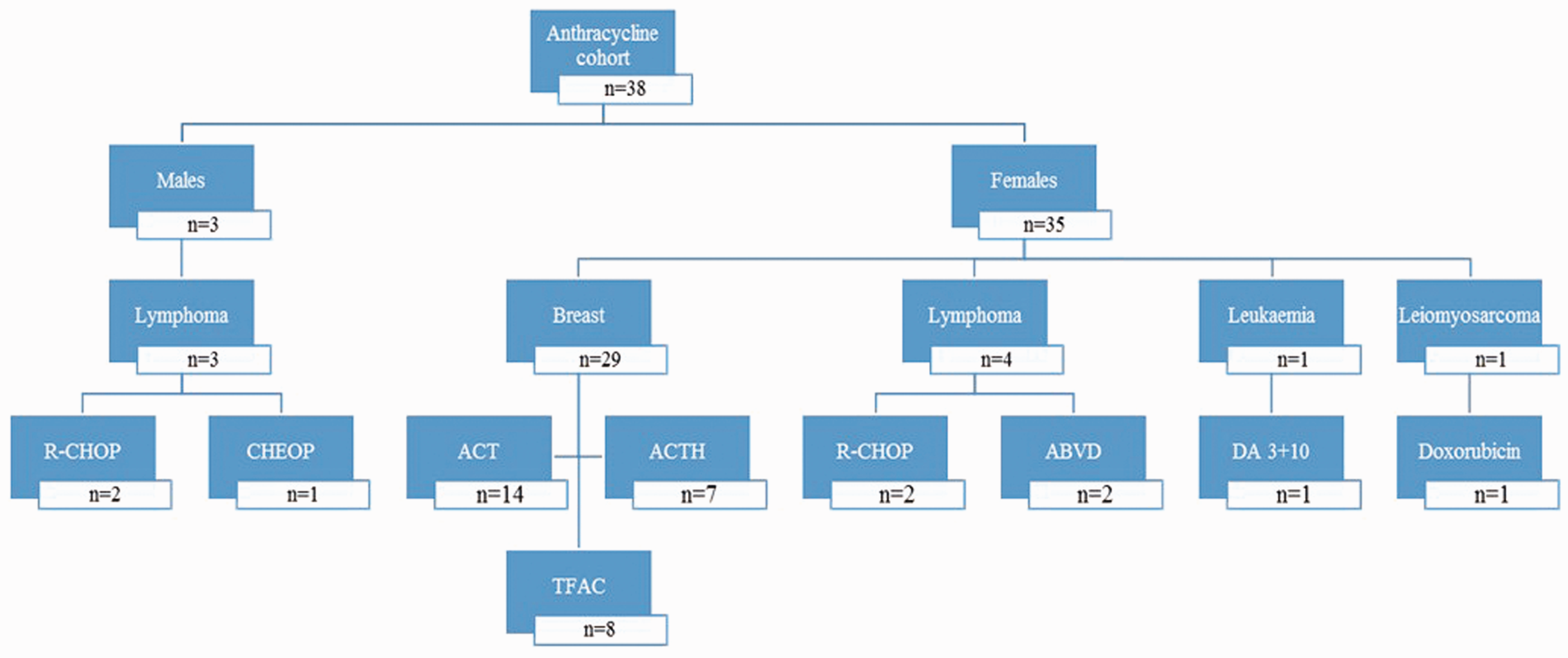
Laboratory analysis
A 10 mL serum sample collected in standard serum gel tubes was obtained from each patient prior to commencing chemotherapy, and subsequently on the first day of each chemotherapy cycle (14–28 day duration). This resulted in a total of 606 individual samples for hs-cTnI analyses. Samples were centrifuged at 3500 g for 10 min and an aliquot was frozen at −70℃ until analysis. Samples were analysed using an Abbott Diagnostics Architect STAT hs-cTnI assay (Abbott Diagnostics, Illinois, USA) as reported previously 18 and performed on the Abbott Architect i2000sr platform. The reported limit of detection of this assay is 1.2 ng/L, range 0–50,000 ng/L, 10% coefficient of variation (CV) at 3 ng/L. 19 All patient ECHO results and routine laboratory data were recorded.
Statistical assessment
An unpaired t-test was used to assess patient ages between cohorts (normal distribution as evaluated using D’Agostino-Pearson omnibus normality test). hs-cTnI data were assessed using a unisex 99th percentile (25 ng/L) 20 together with gender-specific 99th percentiles of 16 ng/L for females and 34 ng/L for males 21 as determined in the HUNT study 18 and by Apple and colleagues. 22 hs-cTnI data were evaluated for normality using the D’Agostino-Pearson omnibus normality test for normal distribution (GraphPad Prism, version 6.05). This resulted in the application of non-parametric methods to all hs-cTnI assessments. Mann-Whitney tests were used to compare measurements between patients treated with specific chemotherapeutic agents. Friedman tests were applied to assess hs-cTnI during cycles of chemotherapy among each of the anthracycline groups and the non-anthracycline cohort, with Wilcoxon tests comparing 5th and final cycle measurements where appropriate. Two-tailed Spearman correlations were used to examine the relationship between chemotherapeutic drug dose and hs-cTnI measurements.
Results
The anthracycline cohort (n = 38), (mean age: 49 years, SD: 13, 95% CI 45 to 54) was significantly younger than their non-anthracycline counterparts (n = 46), (mean age: 59 years, SD: 10.23, 95% CI 56 to 62) (Unpaired t-test, P < 0.001). No patients had documented pre-existing heart disease or other co-morbidities. Prior to treatment, hs-cTnI for patients in both anthracycline and non-anthracycline cohorts was below both the gender-specific (M: 34 ng/L, F: 16 ng/L) and unisex (25 ng/L) 99th percentiles.
Median hs-cTnI measurements increased significantly among the early-dose group from 1.3 ng/L (95% CI 1.04 ng/L to 2.11 ng/L) to 31.2 ng/L (95% CI −8.24 ng/L to 153.1 ng/L) following the first five cycles of chemotherapy (Friedman, P < 0.0001), with a median relative δ increase of 2592% (95% CI 1606 to 5358%) and a median absolute δ increase of 30.7 ng/L (95% CI 19.05 ng/L to 57.77 ng/L) (Figure 3(a)). The greatest relative δ increase in hs-cTnI between cycles of chemotherapy occurred following the first cycle of treatment (median increase, 164%, 95% CI 97 to 354%), while the greatest median absolute δ increase occurred between the 4th and 5th cycles of treatment (11.9 ng/L, 95% CI 1.07 ng/L to 71.71 ng/L), correlating to the final cycle of anthracycline treatment (with cyclophosphamide) and 1st cycle of paclitaxel treatment (with trastuzumab in some cases). In the continuous-dose group, there was a statistically significant increase in median hs-cTnI measurements from 1.75 ng/L (95% CI 0.13 ng/L to 5.8 ng/L) to 21.20 ng/L (95% CI −27.62 ng/L to 125.3 ng/L) following the 5th cycle of chemotherapy (Friedman, P < 0.0001), with a median relative δ increase of 744% (95% CI −9618% to 20,199%) and an absolute δ increase of 27.85 ng/L (95% CI −54.94 ng/L to 162.7 ng/L) (Figure 3(b)). The greatest median relative δ and absolute δ increase in hs-cTnI between cycles occurred between the 3rd and 4th cycles of chemotherapy: 128% (95% CI 85% to 161%) and 13.7 ng/L (95% CI −3.03 ng/L to 40.27 ng/L), respectively. Finally, in the late-dose group, concentrations of hs-cTnI were comparable with the non-anthracycline group prior to the introduction of the anthracycline agent, and an increase was observed thereafter (Figure 3(c)).
hs-cTnI assessed during chemotherapeutic regimen in early-dose (a), continuous-dose (b) and late-dose (c) anthracycline groups as well as in the non-anthracycline cohort (d). Number of patients (a) n = 21, (b) n = 9, (c) n = 8, (d) n = 46. hs-cTnI assessment naïve to treatment to cycle 5, Friedman; cycle 5 to 5 months post-treatment initiation, Wilcoxon; naïve to treatment to 5 months post-treatment initiation, Wilcoxon. (- - - - -) 16 ng/L, female 99th percentile; (_ . _ . _ . ) 25 ng/L, unisex 99th percentile; (………) 34 ng/L, male 99th percentile. * P < 0.05, **P < 0.01, ****P < 0.0001.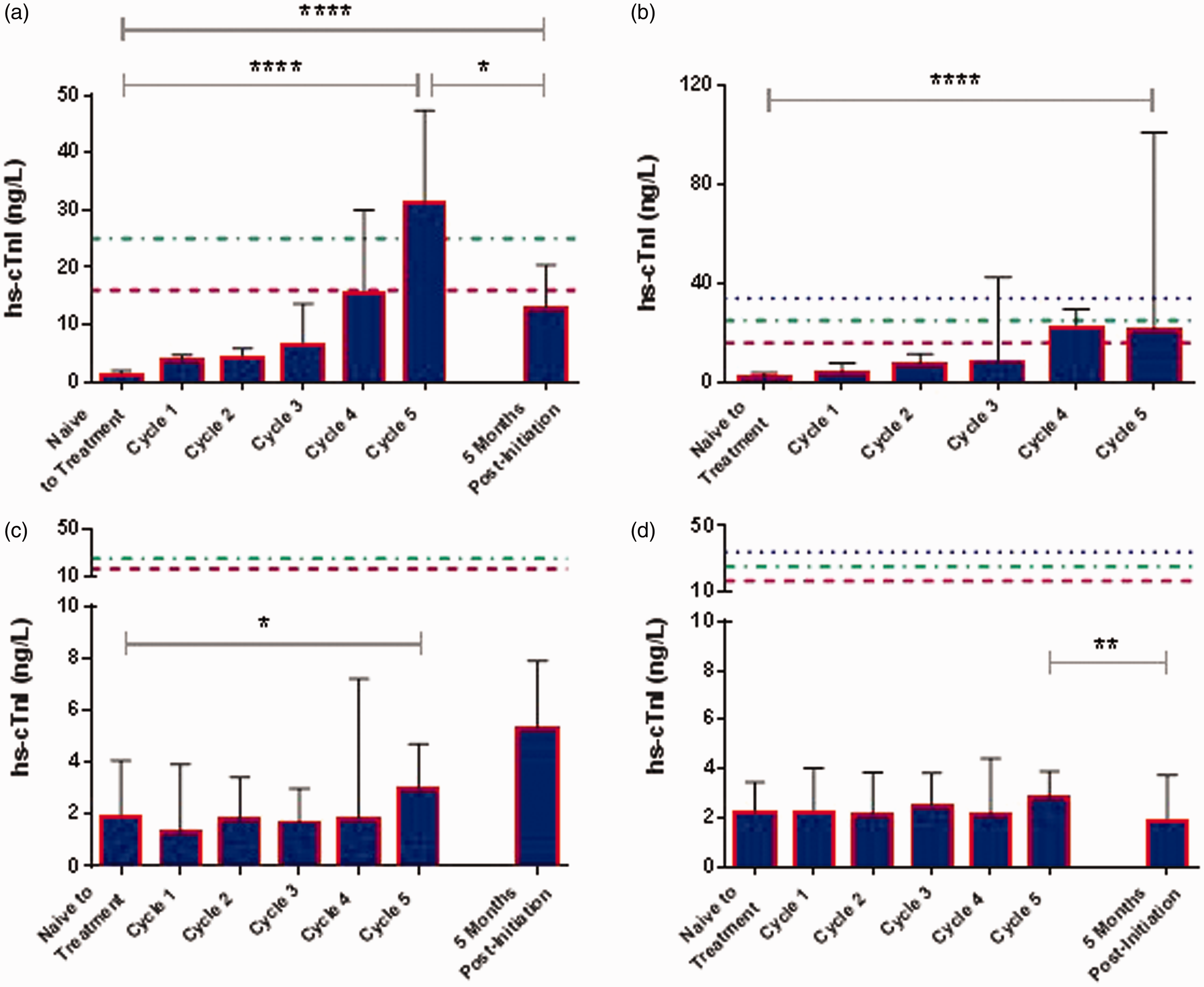
In comparison, hs-cTnI measurements among the non-anthracycline cohort were significantly below the gender-specific and unisex 99th percentiles during all cycles of treatment, with many measurements below the LOD (Figure 3(d)). While hs-cTnI values were statistically significantly lower at five months postinitiation of chemotherapy when compared with measures at cycle 5, the values were all below the 99th percentile and likely of minimum clinical significance.
With one exception, patients from all three anthracycline groups showed significant increases in hs-cTnI, whereas in the non-anthracycline cohort no significant changes were observed (Figure 4).
Wilcoxon comparison of hs-cTnI concentrations at initiation of treatment and following the 5th cycle of chemotherapy among both anthracycline (a) and non-anthracycline cohorts (b). Dashed, dotted lines and significance values are as per Figure 3.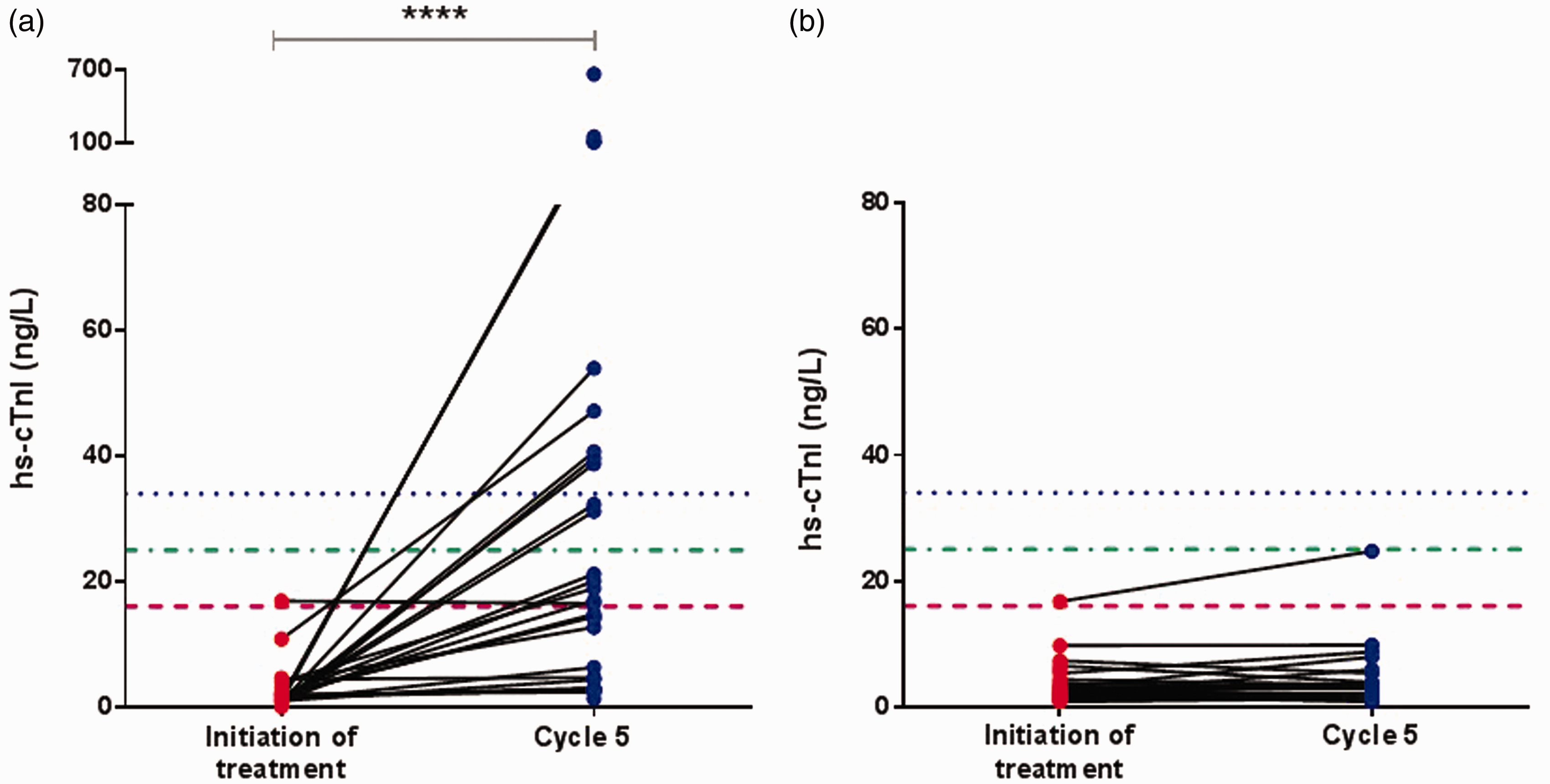
There was no direct correlation between anthracycline dose and hs-cTnI concentrations (average Spearman r < −0.22).
Fifteen patients (18%) in the study cohort died. In total of 41 patients (49%), 18 in the anthracycline cohort (10 who had completed chemotherapy) and 23 in the non-anthracycline cohort (5 who had completed chemotherapy) had follow-up hs-cTnI evaluation at six months with only 2 patients (anthracycline cohort) with persistently raised hs-cTnI.
No patients in either cohort had significant sepsis, and where C-reactive protein (CRP) was measured, elevated values were not associated with an elevated hs-cTnI. Moreover, there was no definitive correlation between hs-cTnI and white cell count (Spearman r = −0.2766, P < 0.05, 95% CI −0.52 to 0.004).
In our evaluation of patients receiving cyclophosphamide (maximum dose was <750 mg/m2) among the non-anthracycline cohort, no significant increase in hs-cTnI was detected following the first five cycles of chemotherapy (Friedman, P > 0.05). Similarly, in patients treated with paclitaxel, no significant changes were identified (Friedman, P > 0.05). No significant difference in hs-cTnI was observed between patients in the early-dose group who received trastuzumab (n = 7) when compared with those who did not (n = 14), (Mann-Whitney, P > 0.05). Similarly, no significant elevations of hs-cTnI were identified among patients of the non-anthracycline cohort treated with trastuzumab (n = 9), (Friedman, P > 0.05).
Prospective comparison of hs-cTnI assay with cTnI assay performance
In a prospective evaluation of the hs-cTnI values which would not have been quantifiable by conventional cTnI assays, a total of 573 measures out of the 606 assayed were below the 10% CV limit of 30 ng/L observed in many conventional cTnI assays. 19
ECHO imaging
ECHO imaging was performed in all patients in the anthracycline cohort (n = 38) and in 21 of the 46 patients in the non-anthracycline cohort. No patient presented with diastolic or systolic dysfunction prior to commencement of chemotherapy. Two patients in the anthracycline cohort (early-dose group, both female) showed evidence of Grade I diastolic dysfunction (LVEF of >55%), while two patients in this cohort demonstrated 5% decreases in LVEF. One patient in the non-anthracycline cohort had a Grade I diastolic dysfunction (EF of 55–60%).
Discussion
It is recognized that there are strong links between cancer and heart disease and a growing clinical need for optimized cardio-oncology patient management. We have shown that the specificity and improved sensitivity of the hs-cTnI assay (with an LOD of 1.2 ng/L) offer improved diagnostic/monitoring capability over what was available previously with conventional cTnI assays (with 10% CVs of 30 ng/L). 19 Because of the insensitivity of ECHO for subtle cellular changes, 23 hs-cTnI assay offers a complementary diagnostic tool in patients receiving treatment known to predispose to cardiotoxicity.
In patients treated with a doxorubicin-containing regimen, hs-cTnI measurements increased significantly following serial assessment of the first five cycles of chemotherapy among our early-dose and continuous-dose groups. These results correlate with findings in a previous study which showed increases in hs-cTnI, three months into chemotherapy in patients treated with an anthracycline-based regimen. 24 There were no changes in hs-cTnI in patients in the late-dose group prior to the introduction of an anthracycline agent. However, a small but significant increase in hs-cTnI was observed thereafter, confirming that anthracycline-mediated cardiomyocyte injury can be monitored using hs-cTnI. There is no agreed consensus in the serial measurements as to whether relative or absolute δ changes in troponin correlates best with extent of damage, and we report both in this study.25,26
The significant increase in median relative and absolute δ in hs-cTnI during the first five cycles of treatment among the early-dose group (2592% and 30.7 ng/L, respectively) is indicative of significant cardiomyocyte injury, given the awareness that smaller absolute δ increases in hs-cTnI in a non-ACS clinical setting are considered to be significant. 27 The greater rises in hs-cTnI were seen in the early-dose group whose maximum cumulative dose of doxorubicin was 53% and median dose per cycle was 101 mg, while the continuous group was administered 89% of maximum dose, characterized by a median dose per cycle of 85 mg. Where we did not observe a dose-dependent hs-cTnI rise, there was a greater absolute δ increase in hs-cTnI in the early-dose group suggesting that the continuous regimen which utilized smaller doxorubicin doses per cycle resulted in less acute cardiomyocyte injury. This is a key finding which suggests that cardiomyocyte injury, as indicated by hs-cTnI release, is somewhat dependent on the mode of administration, which warrants validation in larger patient cohorts.
The decrease in hs-cTnI values below peak measures for almost all patients of the anthracycline cohort evaluated following the initial eight-cycle assessment (either post-completion of chemotherapy and during subsequent chemotherapy) was suggestive of a decrease in subclinical cardiomyocyte damage following completion of anthracycline treatment. This suggestion warrants further investigation but highlights the utility of hs-cTnI to identify dynamic changes within the cardiomyocyte during chemotherapy which may be linked with cardiac injury.
Generally, anthracycline-related cardiomyopathy is a long-term consequence of this treatment and follow-up of incidence of heart failure and cardiomyopathy in this patient cohort are required to define clinical decision thresholds and elucidate the true association between early hs-cTnI elevations, anthracycline dose, cardiotoxicity and heart failure.
hs-cTnI was evaluated prior to commencing each cycle of chemotherapy, which provided an assessment of cardiac status the furthest time from the prior cycle (typically between 14 and 21 days for our anthracycline cohort). Recent literature has suggested that significant cardiac damage leads to troponin release from the troponin complex which persists for a number of days, a theory established in the pathobiology of ACS. 28 This is in comparison to the early releasable pool or cytosolic source of troponin which appears to induce a transient increase in troponin which occurs on the first day of injury. 29 As elevations of hs-cTnI were detected at least 14 days post-administration of the prior cycle of chemotherapeutic agents, these concentrations would indicate that increases most likely reflect chronic cardiomyocyte injury. These findings provide important data regarding the treatment-related background elevations, which is essential knowledge when assessing and differentiating oncology patients with potential ACS and non-ST elevation MI.
We observed no significant cardiac troponin release in patients receiving trastuzumab. Given the extensive literature detailing the cardiotoxic potential of this agent, 30 our results albeit in a small cohort, suggest that cardiomyocyte injury is not a major factor with this treatment at doses less than the maximum administered dose of 8 mg/kg. While most patients of the anthracycline cohort also received cyclophosphamide and paclitaxel, other agents known to cause cardiotoxicity, we do not suspect either agent is solely mediating increases in hs-cTnI, although a synergistic potentiation of cardiotoxicity may be possible. Given the existing knowledge in the literature specifically regarding anthracycline toxicity and the fact that our patients treated with cyclophosphamide, paclitaxel and trastuzumab as part of non-anthracycline regimens (typically of higher doses) did not demonstrate hs-cTnI elevations, this evidence points to anthracyclines as the main cause of cardiomyocyte necrosis and hs-cTnI release in this study.
We observed normal hs-cTnI measurements among patients treated with non-anthracycline regimens following the first five cycles of chemotherapy and at six-month follow-up suggesting that the value of hs-cTnI monitoring appears to have optimal clinical utility in patients receiving anthracycline-based therapies, unless cardiac injury is clinically suspected. While a comparison of hs-cTnI measures between patients receiving anthracycline and non-anthracycline-based chemotherapeutic regimen may introduce a control bias in the evaluation of anthracycline-mediated increases in hs-cTnI, it was not possible to create a placebo cohort in this study.
Monitoring potential anthracycline-mediated cardiotoxicity using hs-cTnI
It is recognized that hs-cTnI measurement is of value as a prognostic indicator which is particularly relevant for patients who receive an anthracycline agent. In addition, NACB guidelines have previously promoted routine cTnI measurement among patients receiving Adriamycin® (doxorubicin). 31 We have shown optimal assessment of patients receiving anthracycline therapy is met by high-sensitive assays which have clear benefit over assays with a 10% CV at 30 ng/L. Routine monitoring of end-organ damage in patients receiving chemotherapy is commonplace, rises in creatinine and alanine transaminase indicative of kidney and liver injury, respectively. Where we cannot identify the cause of the injury from biochemical data alone, elevated concentrations are an indicator to monitor patients more closely. In a similar way, hs-cTnI or hs-cTnT offers more sensitive tools for risk stratifying patients for closer clinical and cardiology monitoring.
Future roles of hs-cTnI as a means of monitoring the efficacy of cardioprotective strategies
Cardio-oncology collaborative efforts have gained considerable momentum in recent years encompassing both primary and secondary cardioprotective strategies. Agents such as ß blockers, ACE inhibitors, angiotensin receptor blockers and dexrazoxane are used as prophylactic therapies, with no definitive evidence to date as to which is the optimal strategy.10,32–35 Additionally, studies to date have been limited by the lack of a real-time sensitive biomarker of change within the myocyte environment which may indicate injury. Extrapolating results from this study would suggest an important role for hs-cTnI in future clinical trials aimed at determining which agent minimizes cardiac injury. The additional sensitivity of the assay and the ability to facilitate timely detection of subclinical cardiomyocyte injury provides the platform for a well-informed and convenient means of evaluating the efficacy of such cardioprotective strategies during treatment with potentially cardiotoxic chemotherapeutic agents.
Limitations
Patients in the anthracycline group were significantly younger than those in the non-anthracycline cohort, but we have not controlled for differences in age, gender and cancer type. However, it is unlikely that any of these differences would significantly influence the changes in hs-cTnI observed in this study. Our study was not powered to assess the concentrations of hs-cTnI which were linked to the development of long-term or irreversible cardiotoxicity but rather emphasizes that increases were indicative of subclinical cardiac injury which warrants further investigation and treatment.
Conclusions
The enhanced sensitivity of hs-cTnI offers a biomarker with potential clinical utility in determining the optimal mode of administration of anthracycline chemotherapy to ensure oncological efficacy whilst minimizing cardiac injury. On the basis of evidence from this study, hs-cTnI has potential as a biomarker in future clinical trials, in determining which adjunct cardioprotective regimens optimally minimize cardiomyocyte injury.
Footnotes
Acknowledgements
We thank the staff of the Department of Clinical Biochemistry and Diagnostic Endocrinology and the Oncology/Haematology ward for their support in the facilitation of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MCF is in receipt of a small grant from Abbott Diagnostics. All other authors have no relationships relevant to this paper to disclose.
Ethical approval
Ethical approval was attained from the Mater University Hospital Ethics Committee (Ref: 1/378/1387).
Guarantor
MCF.
Contributorship
MCF designed research; MJ collected and analysed data and wrote manuscript; MCF, POG, CK and NM reviewed and edited the manuscript and approved the final version of the manuscript.
