Abstract
Multiple myeloma is a malignant plasma cell dyscrasia that is becoming more prevalent in an increasingly ageing population. It is a complex disease with clinical phases ranging from the premalignant monoclonal gammopathy of undetermined significance to asymptomatic (smouldering) myeloma and then symptomatic myeloma; the latter occasionally terminating in the clonal proliferation of plasma cells outside the bone marrow. We present a patient whose clonally evolved disease from monoclonal gammopathy of undetermined significance to multiple myeloma demonstrated the presence of an unusual combination of monoclonal immunoproteins. Capillary electrophoresis demonstrated the presence of three paraproteins in the gamma region (γ-region), two of which were additional to the IgGk paraprotein which migrated in the slow γ-region at initial diagnosis. Subsequent isotypic identification of the new paraproteins was not possible by immunotyping and initial immunofixation studies failed to definitively characterize the monoclonal proteins. After reduction with beta-mercaptoethanol, two paraproteins were detected by both capillary and gel electrophoresis. However, only immunofixation was able to resolve three distinct monoclonal bands, confirming the presence of free monoclonal kappa light chains in the mid-gamma region and free monoclonal heavy chains in the fast gamma region. Triple gammopathies in themselves are uncommon; this case presents a very unusual combination of paraproteins which required various electrophoretical and immunochemical techniques to identify and characterize them. The change of electrophoretic signature from the monoclonal gammopathy of undetermined significance phase to the diagnosis of multiple myeloma suggested that a number of genetically distinct subclones were present in the pretreatment clonal evolution of the disease.
Keywords
Case description
A 68-year-old female patient previously diagnosed with monoclonal gammopathy of undetermined significance (MGUS) was admitted to hospital in 2012 with progressively increasing back pain. When initially diagnosed two years earlier, she had an IgGk paraprotein (20 g/L) and a normal serum free light chain (sFLC) ratio, although the involved free light chain was slightly elevated at 25.1 mg/L (free Kappa reference interval 3.0–19.0 mg/L).
Three months prior to admission, the intact paraprotein had increased to 26 g/L, the kappa sFLC to 3554 mg/L and the Kappa/Lambda ratio was 131. She was complaining of increasing back pain, but no lytic lesions were present.
On admission, she was markedly hypercalcaemic (adjusted serum calcium 4.00 mmol/L, reference interval 2.20–2.60 mmol/L) and a grossly elevated kappa sFLC of 24680 mg/L. Further investigations revealed the presence of multiple expansile rib lesions and a marked wedge compression fracture in L4 vertebra.
A needle core biopsy of one of the rib lesions demonstrated the presence of a diffuse monotonous infiltrate of plasma cells (100%), which were exclusively CD138+ on immunophenotyping. Consequently, the patient was diagnosed with symptomatic multiple myeloma (MM) with multiple plasmacytomas.
Various laboratory parameters during the evolution of the patients’ disease.
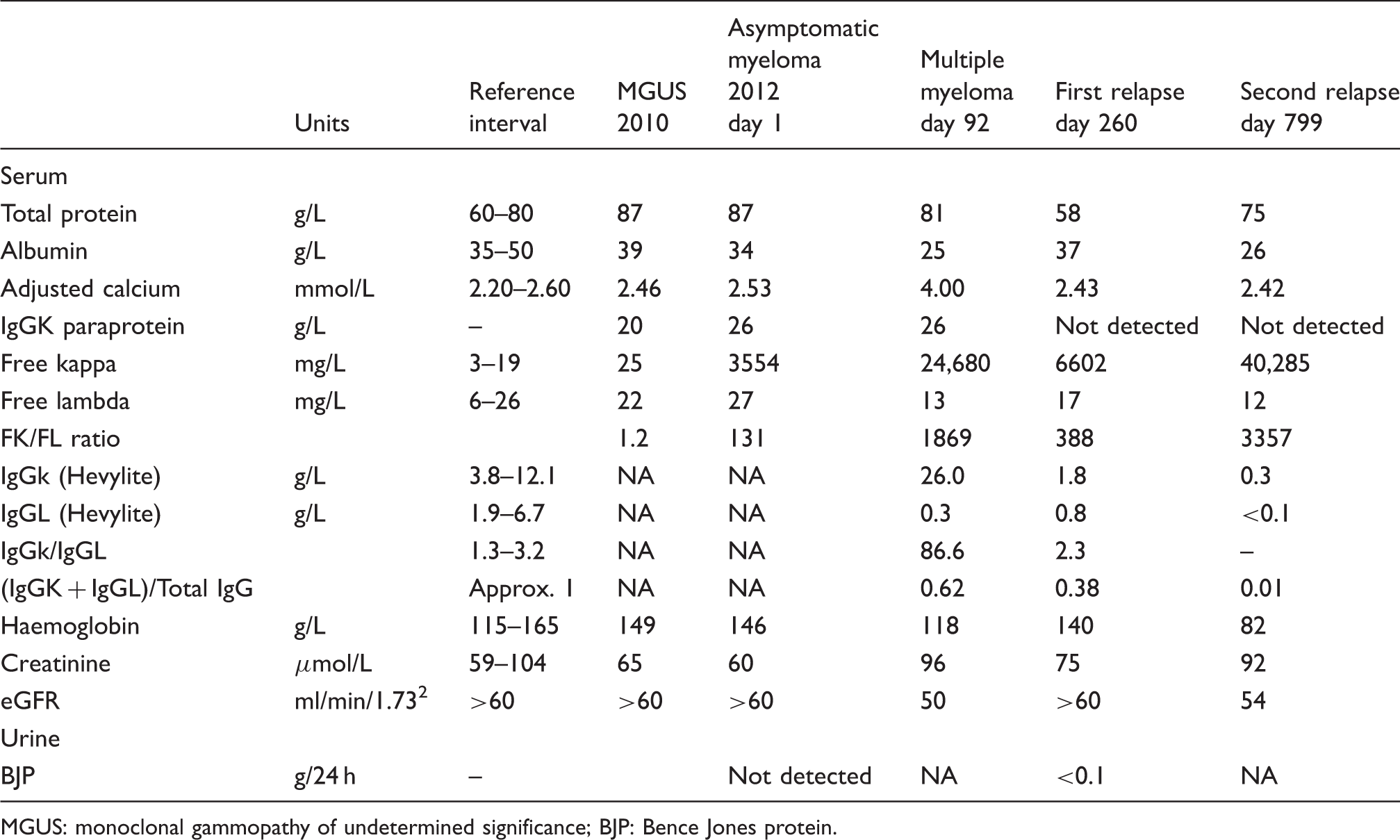
MGUS: monoclonal gammopathy of undetermined significance; BJP: Bence Jones protein.
Serum protein electrophoresis (SPEP), immunotyping (IT) and immunofixation (IFE) were performed using the Sebia Capillarys 2 and Sebia Hydrasys (Sebia, Evry, France). The IgG heavy chain matched pair and sFLCs (Hevylite® and Freelite®; The Binding Site, UK) were quantified using specific antibodies on the SPAplus (The Binding Site, UK) and the Roche Modular c502 (Roche Diagnostics, Mannheim, Germany), respectively.
SPEP demonstrated the presence of three paraproteins in the γ-region, two of which were additional to the IgGk paraprotein, which migrated in the slow γ-region at initial diagnosis (Figure 1). The IT profile (samples pretreated with specific antisera, which result in the altered mobility of the resultant antigen–antibody complex) failed to definitively identify the isotype of the two unknown monoclonal components.
SPEP (protein 6 buffer) and immunotyping (Sebia Capillarys 2). (a) Electrophoretogram of serum indicating the three monoclonal peaks in the γ-region, (b) immunotyping demonstrating the immunosubtraction for each of the five monospecific antibodies superimposed upon the reference overlay.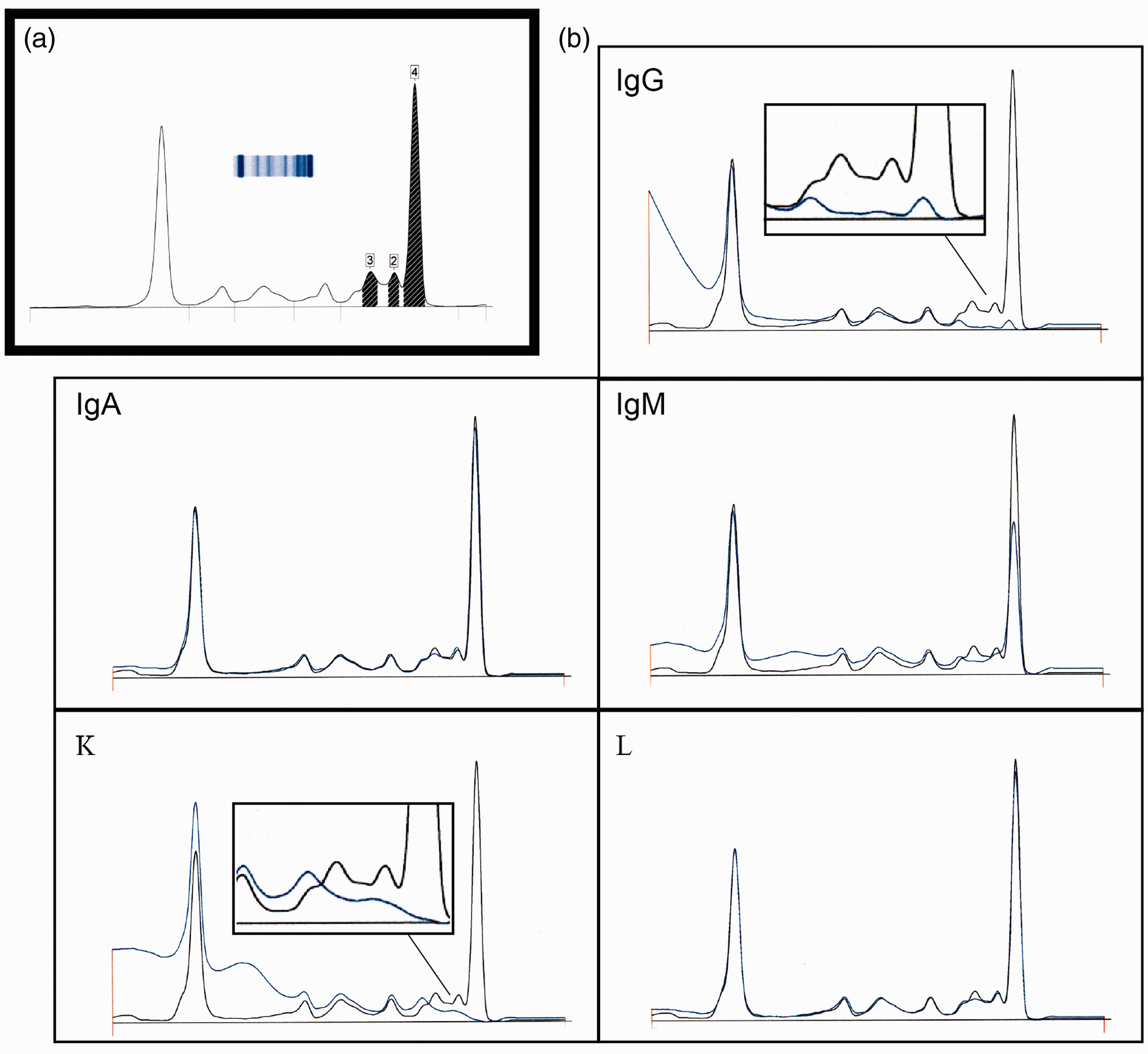
Discussion
MM is a malignant immunosecretory disorder characterized by an uncontrolled monoclonal plasma cell proliferation in the bone marrow. In approximately 85% of patients the aberrant clone secretes an intact paraprotein which is usually detected in the serum as a homogeneous band. Here, we present an unusual case of MM, exhibiting an excess of both free monoclonal, light and heavy chains, in the presence of an intact paraprotein.
Capillary electrophoresis (CE) in combination with IT has proven to be an efficient approach to the successful immunochemical characterization of most paraproteins. However, the limitations of this technique are well recognized in some patients with IgM paraproteins, minute bands (especially monoclonal free light chains) 3 and its inability to identify the IgD and IgE isotypes.
Although the three paraproteins in this patient were clearly defined by CE, their subsequent characterization by IT demonstrated an unusual and uninterpretable immunosubtraction pattern. The explanation for this unpredicted subtraction is unclear; it may be due to non-specific antibody interactions, which affect the specific antiserum detection. The complete subtraction with anti-γ by all three paraproteins suggested that the heavy chain component of the three paraproteins consisted of IgG only and the almost complete absence of subtraction with anti-λ indicated that only kappa light chains would be expressed in the conformation of any or all of the paraproteins.
The successful characterization of the three paraproteins necessitated the application of IFE. This was performed before and after reduction with beta-mercaptoethanol on a Hydragel 4IF gel. The band in the mid γ-region (‘band 2’) (Figure 2), in the non-reduced sample, demonstrated reactivity with gamma heavy chain and both kappa (free and bound) and kappa free light chain antisera. The band in the fast γ-region (‘band3’) reacted with γ-heavy chains only, which corroborated the finding of heavy chain excess determined by the indirect use of the Hevylite® assay. Since three different excretory plasma cell monoclonal components were identified, potential co-migration of these proteins made the interpretation of the IFE also challenging. If no kappa free antiserum had been used, it may have been incorrectly concluded that ‘band 2’ had the same isotype as ‘band 1’. However, the differential in stain intensity between gamma and kappa (equal dilution of sample in both lanes) and the positive immunofixation with Kappa free antisera confirmed the presence of free monoclonal light chains, migrating alongside another paraprotein. Free light chain antisera should therefore be included as part of the selective IFE antibody panel, its use being especially important when there is suspected co-migration with an intact paraprotein.
Immunofixation of serum (Sebia Hydrasys) with monospecific antisera. (a) Standard IFE, (b) IFE postreduction with beta-mercapoethanol and (C) extended high-resolution IFE demonstrating diffuse monoclonal staining between \'band 2\' and \'band 3\' for anti-γ and diffuse monoclonal staining between \'band1\' and \'band2\' for anti-κ. Additional kappa band observed in beta region.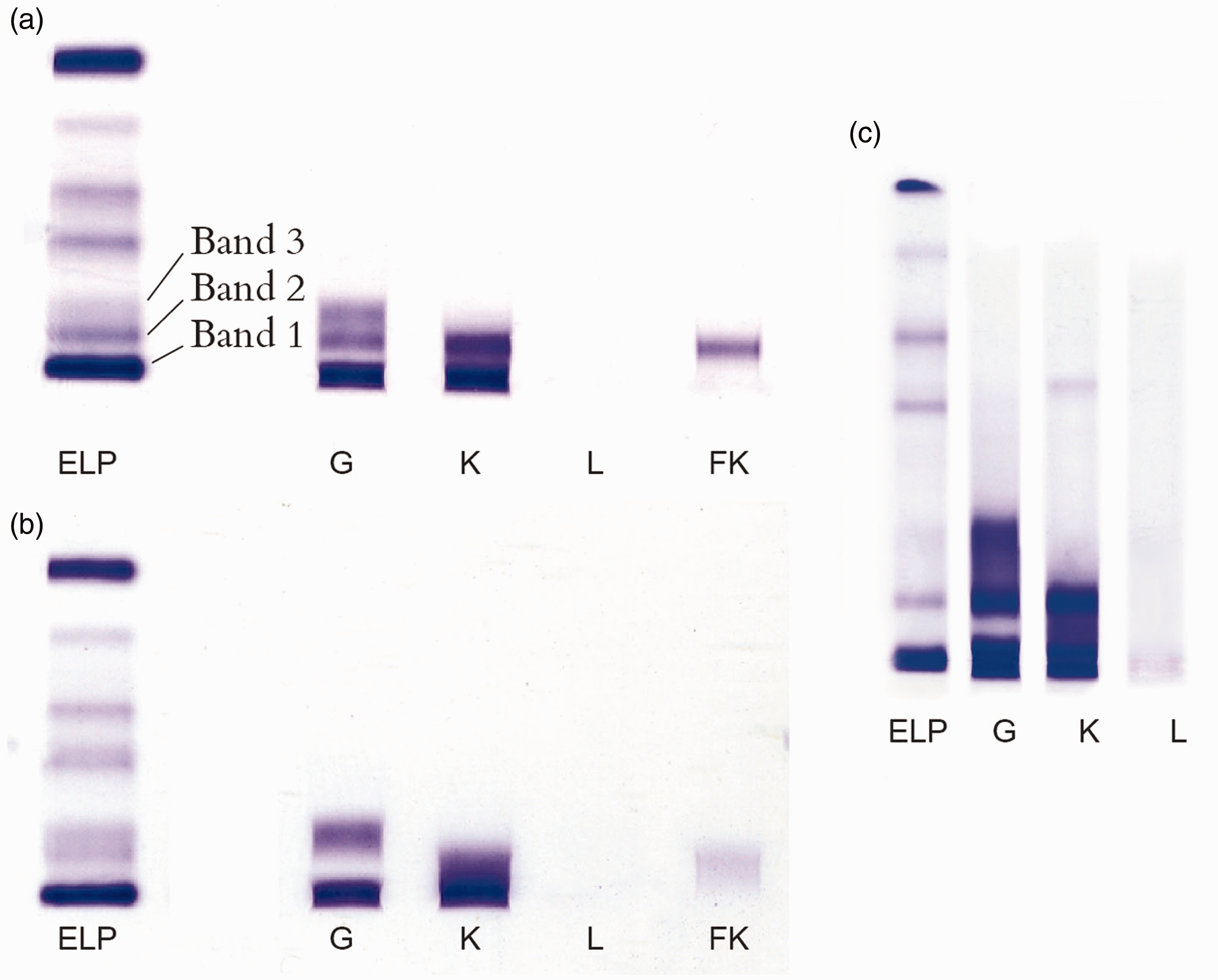
Pretreatment with beta-mercaptoethanol, an approach which has proven to be very useful in the isotypic identification of paraproteins with polymerized forms of IgM, IgA and free monoclonal light chains, assisted in the characterization of ‘band 2’ and ‘band 3’. The absence of immunofixation with anti-γ in ‘band 2’ (reduced) suggested two possibilities. Either a small dimerised IgGk has been reduced to the molecular weight of the monomer in ‘band 1’ or polymerized free γ-heavy chains have been reduced to one homogeneous band in ‘band 3’, while ‘band 2’ now contains free monoclonal light chains only.
SDS agarose gel electrophoresis of ‘band 3’, after extraction from a high resolution agarose gel and MALDI-TOF proteomics of the same, confirmed the molecular weight of the free γ-heavy chains to be 82 KDa (data not included). This most likely represents a dimer of truncated γ-heavy chains.
The migration of the free monoclonal light chains appears to be different in the two media. The minute bump, which is observed in ‘peak 1’ after subtraction with anti-γ, in conjunction with its complete subtraction with anti-κ, suggests the presence of co-migrating free monoclonal kappa light chains. However, IFE demonstrates that the same paraprotein is migrating in ‘band 2’ on gel electrophoresis. The shift in electrophoretic mobility may be due to the difference in the pH employed in the two media, agarose gel 8.6 and Capillarys 10.0.
The evident fusion in the CE between ‘peak 2’ and ‘peak 3’ and the positive staining with γ-heavy chains only in the same region by IFE would suggest that the free γ-heavy chains represented by ‘peak 3’, extend anodally to include ‘peak 2’. An additional free kappa band in the beta region was detected in the extended high-resolution IFE; this probably represents either a higher molecular weight polymer of free monoclonal light chains or a complex formed by their binding to another serum protein. 4
The presence of free gamma heavy chains in the serum by SPEP was first described by Franklin 5 and has been defined in a spectrum of lymphoproliferative disorders, but rarely in the setting of MM. 6 Gamma heavy chain disease (γ-HCD), a rare disorder with approximately 130 cases reported to date, is characterized by the monoclonal production of truncated γ-heavy chains which are mostly dimers, 7 without any associated light chains. The molecular weight of the monomeric γ-heavy chain ranges from 27 to 49 KDa and consist principally of the Fc region. 8
A search of the literature has only identified two cases of a B-cell lymphoproliferative neoplasm, where free light chains have been identified in the presence of free γ-heavy chains. However, the monoclonal free light chains in these patients were not evident by SPEP and were detected by the Freelite® assay only.9,10
Prior to the availability of the Freelite® assay, all of the 13 reported cases of biclonal γ-HCD co-existed with various IgG or IgM intact paraproteins. 9 A recent paper identified 3 of 15 patients with γ-HCD who had monoclonal light chains. 10 However, none of these patients had free monoclonal light chains identified in the serum by electrophoresis.
The appearance of γ-heavy chains in this patient, rather than being associated with γ-HCD, is more likely indicative of clonal evolution of MM. The simultaneous appearance of both γ-heavy chains and the exponential rise in free kappa light chains is possibly due to a subclone that has lost its ability to pair the heavy and light chains. Alternatively, the unusual spectrum of monoclonal proteins present in this patient may indicate the presence of a number of immunologically diverse subclones, which include those which have truncated ability for the production of intact immunoglobulins and secrete free monoclonal light chains and free monoclonal heavy chains, respectively.
It is thought that there are two driving forces in the dynamics of clonal evolution which contribute to the diversification seen in MM. Firstly, there is the intrinsic genetic instability of aggressive myeloma subclones and secondly the selective pressures introduced by therapies during the course of the disease. 11
The very unusual picture seen in the evolution from MGUS to symptomatic MM, without the intervention of therapy suggested the former to be the case. Abnormal cytogenetics were confirmed by fluorescence
The revised 2014 International Myeloma Working Group guidelines, 12 not published at the time of the patient’s progression to MM, now enable therapeutic intervention to take place before the presence of overt clinical symptoms. The guidelines would have located this patient (FLC ratio > 100) in an ultrahigh risk subgroup with smouldering myeloma and at imminent risk of progression and therefore requiring therapy, even in the absence of CRAB features (hypercalcaemia, renal failure, anaemia and bone lesions).
This case emphasizes the benefit of clearly identifying monoclonal proteins (including monoclonal free heavy chains) at diagnosis and throughout the course of the disease. The identification of the monoclonal free heavy chains and monoclonal free light chains at diagnosis enabled the subsequent monitoring of this patient using both the Freelite® and Hevylite® assays. Light chain escape and heavy chain escape were therefore detected at an earlier time point than would have been possible by traditional SPEP and IFE. The inclusion of the Hevylite® assay enabled a more complete picture of the biochemical relapse to be obtained in this patient and may also provide a useful diagnostic tool in the work-up of suspected cases of γ-HCD.
In summary, this present case demonstrates a very unusual combination of monoclonal immunoproteins, all of which are conspicuous by CE. An apparently simple triple gammopathy was shown to consist of an intact IgGk paraprotein, monoclonal free γ-heavy chains and monoclonal free kappa light chains with co-migrating bands, the identity of which depended on the media selected for electrophoresis and prior reduction of the serum with beta-mercaptoethanol.
The unusual electrophoretic signature in conjunction with the Freelite® and Hevylite® results observed at diagnosis, suggested that significant intraclonal heterogeneity was present at that point. Light chain escape and heavy chain escape were subsequently demonstrated at first and second relapses. We believe this is the first reported case of heavy chain escape in MM. 13 The potential for heavy chain escape should be borne in mind when monitoring MM patients.
Footnotes
Acknowledgements
We would like to thank Dr Stephen McClean (University of Ulster) for performing the MALDI-TOF proteomics on the protein extracts.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written informed consent was obtained from the patient.
Guarantor
WID.
Contributorship
WID wrote the paper. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
