Abstract
Background
Accumulating evidence shows an over-abundance of Fusobacterium nucleatum in colorectal tumour tissues. Although stool DNA testing of Fusobacterium nucleatum might be a potential marker for the detection of colorectal tumours, the difficulty in detecting Fusobacterium nucleatum in stool by conventional methods prevented further explorations. Therefore, we developed a droplet digital polymerase chain reaction (PCR) assay for detecting Fusobacterium nucleatum in stool and investigated its clinical utility in the management of colorectal tumours in a Japanese population.
Methods
Feces were collected from 60 healthy subjects (control group) and from 11 patients with colorectal non-advanced adenomas (non-advanced adenoma group), 19 patients with colorectal advanced adenoma/carcinoma in situ (advanced adenoma/carcinoma in situ (CIS) group) and 158 patients with colorectal cancer of stages I to IV (colorectal cancer group). Absolute copy numbers of Fusobacterium nucleatum were measured by droplet digital PCR.
Results
The median copy number of Fusobacterium nucleatum was 17.5 in the control group, 311 in the non-advanced adenoma group, 122 in the advanced adenoma/CIS group, and 317 in the colorectal cancer group. In comparison with that in the control group, the Fusobacterium nucleatum level was significantly higher in the non-advanced adenoma group, the advanced adenoma/CIS group and the colorectal cancer group.
Conclusions
This study illustrates the potential of stool DNA testing of Fusobacterium nucleatum by droplet digital PCR to detect individuals with colorectal tumours in a Japanese population.
Introduction
In 2012, an estimated 1.4 million new cases of colorectal cancer (CRC) and 693,900 CRC-related deaths occurred worldwide. 1 Because more than 95% of patients with CRC would benefit from curative surgery if diagnosed at an earlier or precancerous stage, 2 it is important to develop highly sensitive and specific assays to detect CRC at the early stage that are non-invasive, inexpensive and easy to perform. The main approach to CRC screening throughout the world is the fecal occult blood test (FOBT). 3 However, one research group reported that the sensitivity of the FOBT for the diagnosis of colorectal tumours was only 26% for carcinoma and 12% for advanced adenoma detection. 4 Furthermore, the FOBT also carries the risk of false-positive results in patients with haemorrhoids, ulcers and inflammatory bowel disease.5–7 To avoid the disadvantages of the FOBT, screening methods with greater sensitivity and specificity are strongly required.
Fusobacterium species (a group of non-spore-forming, anaerobic gram-negative bacteria) is an oral bacteria group of the human microbiome. The members of the Fusobacterium species are highly heterogeneous, and some have been recognized as opportunistic pathogens implicated in periodontitis,8–10 inflammatory bowel diseases,11–13 pancreatic abscess14,15 and hepatic abscess.15–17 In addition, the over-abundance of Fusobacterium nucleatum (Fn) in CRC tissues has been reported,18–22 and Rubinstein et al. 23 demonstrated that Fn is a cause rather than a consequence of CRC. Thus, accumulating evidence indicates that Fn could play an important role in CRC development. In light of these previous reports, we speculated that the detection of Fn in stool could be a potential marker of colorectal tumours. However, previous studies have shown a difficulty in detecting Fn in the stool by quantitative real-time PCR. 24 Therefore, we developed a highly sensitive method for detecting Fn in stool by use of droplet digital PCR and investigated the association of the abundance of Fn in stool with the development and progression of CRC in a Japanese population.
Materials and methods
Materials
Feces were collected from 60 healthy subjects (control group) and from 11 patients with colorectal non-advanced adenomas (non-advanced adenoma group), 19 patients with colorectal advanced adenoma or carcinoma in situ (advanced adenoma/CIS group), and 158 patients with CRC of stages I to IV (CRC group). All subjects were Japanese. Criteria of advanced adenoma are defined as adenomas of 1 cm or greater in size, or with villous components (tubulovillous or villous), or with high-grade or severe dysplasia. 25 Staging was classified according to the Union for International Cancer Control. 26 The mean age of the subjects was 32 years for the control group, 65 years for the non-advanced adenoma group, 69 years for the advanced adenoma/CIS group and 69 years for the CRC group. The subjects comprised 149 men and 97 women (sex information was unavailable for 1 subject). CRC stages included stage I in 49 patients, stage II in 29 patients, stage III in 54 patients and stage IV in 21 patients (stage information was unavailable for 5 subjects). There were no significant differences in tumour location or daily intake of lactobacillus preparation between the three groups. The study protocol was approved by the institutional review board of Yamaguchi University Graduate School of Medicine, and informed consent was obtained from each patient and the healthy volunteers.
Fecal specimens were collected in Yamaguchi University Hospital and were stored at −20℃ until DNA extraction. Fecal specimens from patients were collected prior to bowel preparation for colonoscopy or surgical treatment. Fecal samples were thawed from −20℃, and about 200 mg of each sample was used for DNA extraction. The DNA was extracted using QIAamp DNA Stool Mini Kit (QIAGEN, Tokyo, Japan) according to the manufacturer’s instructions. DNA quantification was performed using NanoDrop 2000 spectrophotometers (Thermo Scientific, Yokohama, Japan).
Droplet digital PCR
We performed droplet digital PCR to count the absolute copy number of Fn. The PCR reaction was performed with 40 ng of DNA, 1× ddPCR Supermix for Probes (BioRad, Hercules, CA, USA), 0.25 µM of each primer and 0.125 µM of probe in a total volume of 22 µL followed by droplet generation using an automated droplet generator (BioRad). The sequences of the Fn primer and probe set were as follows: forward primer, 5′-AAGCGCGTCTAGGTGGTTATGT-3′; reverse primer, 5′-TGTAGTTCCGCTTACCTCTCCAG-3′ and probe, 5′-FAM-CAACGCAATACAGAGTTGAGCCCTGCATT-TMARA-3′.
27
The PCR amplicon length is 108 bp from 730429 to 730536 of NCBI reference sequence NC_003454.1 (Fn subsp. nucleatum ATCC 25586 chromosome, complete genome). Cycling conditions included preheating at 95℃ for 10 min followed by 40 cycles of denaturation at 94℃ for 30 s, annealing at 60℃ for 60 s and final heating at 98℃ for 10 min. After amplification, the PCR plate was transferred to a QX100 droplet reader (BioRad), and fluorescence amplitude data were obtained by QuantaSoft software (BioRad) as shown in Figure 1.
One dimensional plots of Fusobacterium nucleatum (Fn) copy numbers by droplet digital PCR.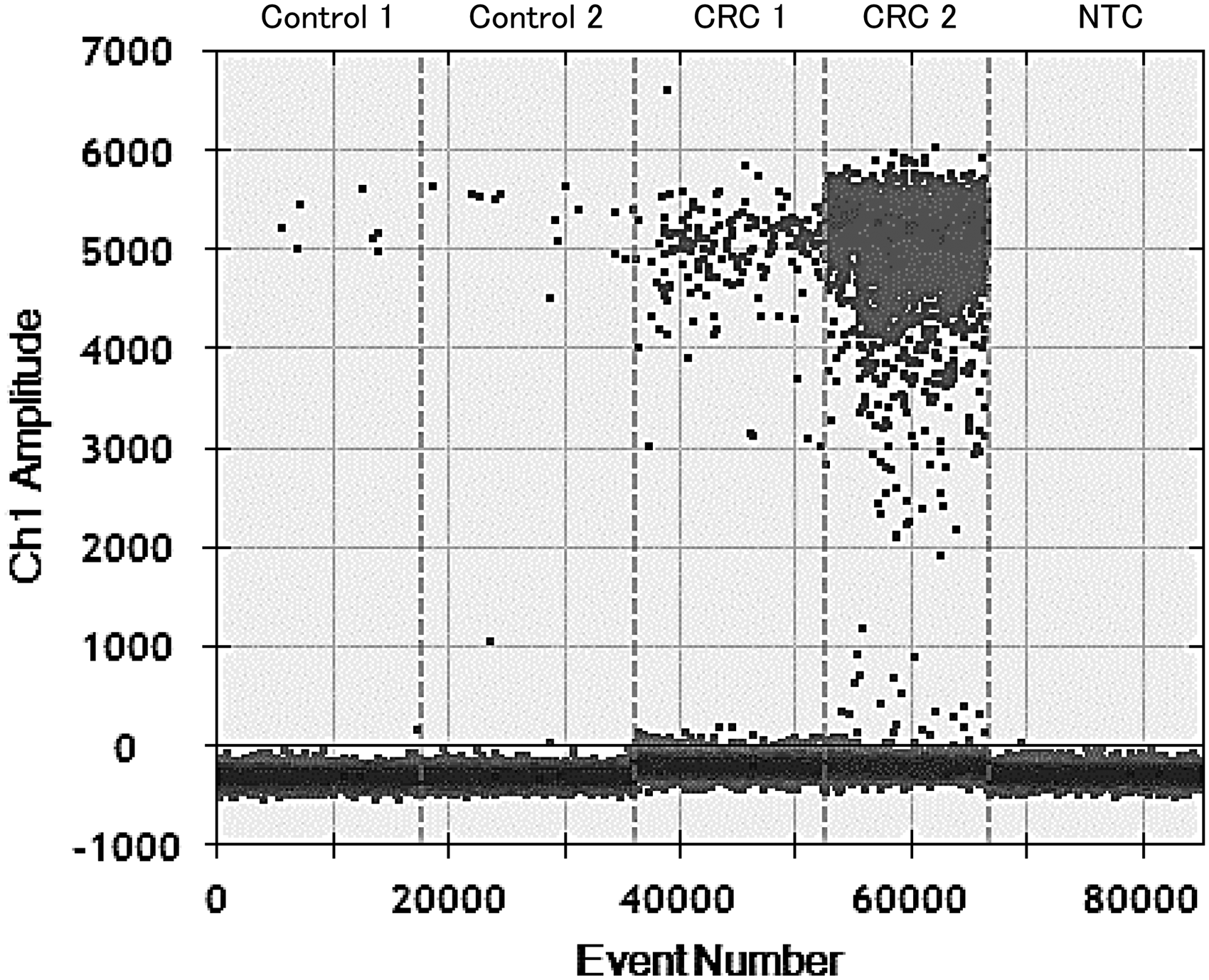
Statistical analyses
To compare variables, the Mann-Whitney U test, chi-square test, and Fisher’s test were used, and a P value of less than 0.05 was considered statistically significant. Statistical analyses were performed with StatFlex Ver. 6 (Artec, Osaka, Japan), GraphPad InStat Ver. 3, and GraphPad Prism Ver. 6 statistical software (GraphPad Software, La Jolla, CA, USA).
Results
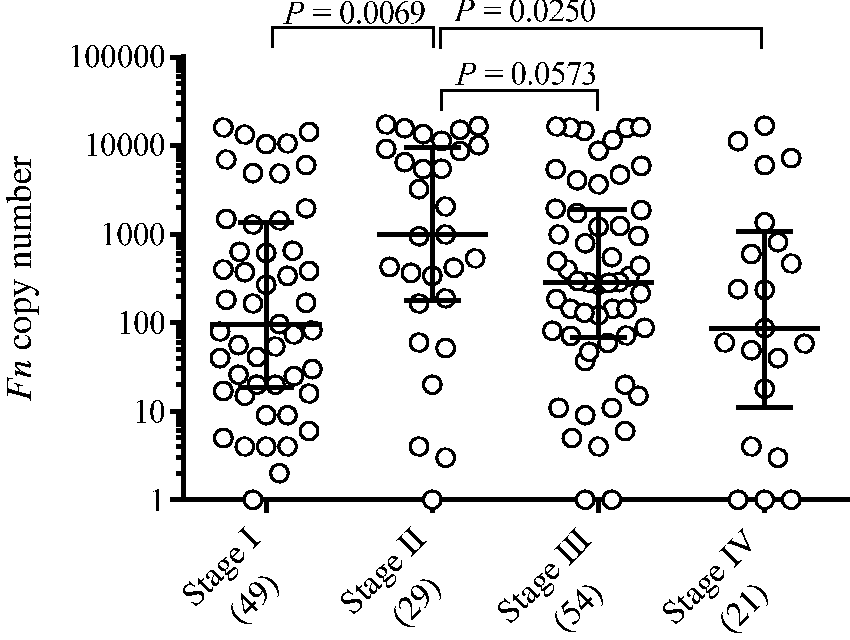
The median copy numbers of Fn were 17.5 (range, 0 to 5793) in the control group, 311 (range, 1 to 5305) in the non-advanced adenoma group, 122 (range, 1 to 9481) in the advanced adenoma/CIS group and 317 (range, 0 to 17343) in the CRC group. In comparison with that in the control group, the Fn level was significantly higher in the non-advanced adenoma group (P = 0.0147), the advanced adenoma/CIS group (P = 0.0060) and the CRC group (P < 0.0001) (Figure 2). There were no significant differences in Fn levels between the three groups. The Fn level was significantly higher in stage II CRC than in stage I CRC (Figure 3): the median copy number was 97 in stage I CRC, whereas it was 999 in stage II CRC (P = 0.0069, Figure 3). This trend or association was also observed between stage II and stageIII (median copy number of 288, P = 0.0573) and between stage II and stage IV (median copy number of 87, P = 0.0250).
Copy numbers of Fusobacterium nucleatum (Fn) in the control, non-advanced adenoma, advanced adenoma/CIS, and CRC groups. Each sample is indicated by a closed circle. The box plots show the median with interquartile range (25th percentile and 75th percentile). Copy numbers of Fusobacterium nucleatum (Fn) by CRC stage. Each sample is indicated by a closed circle. The bars show the median with interquartile range.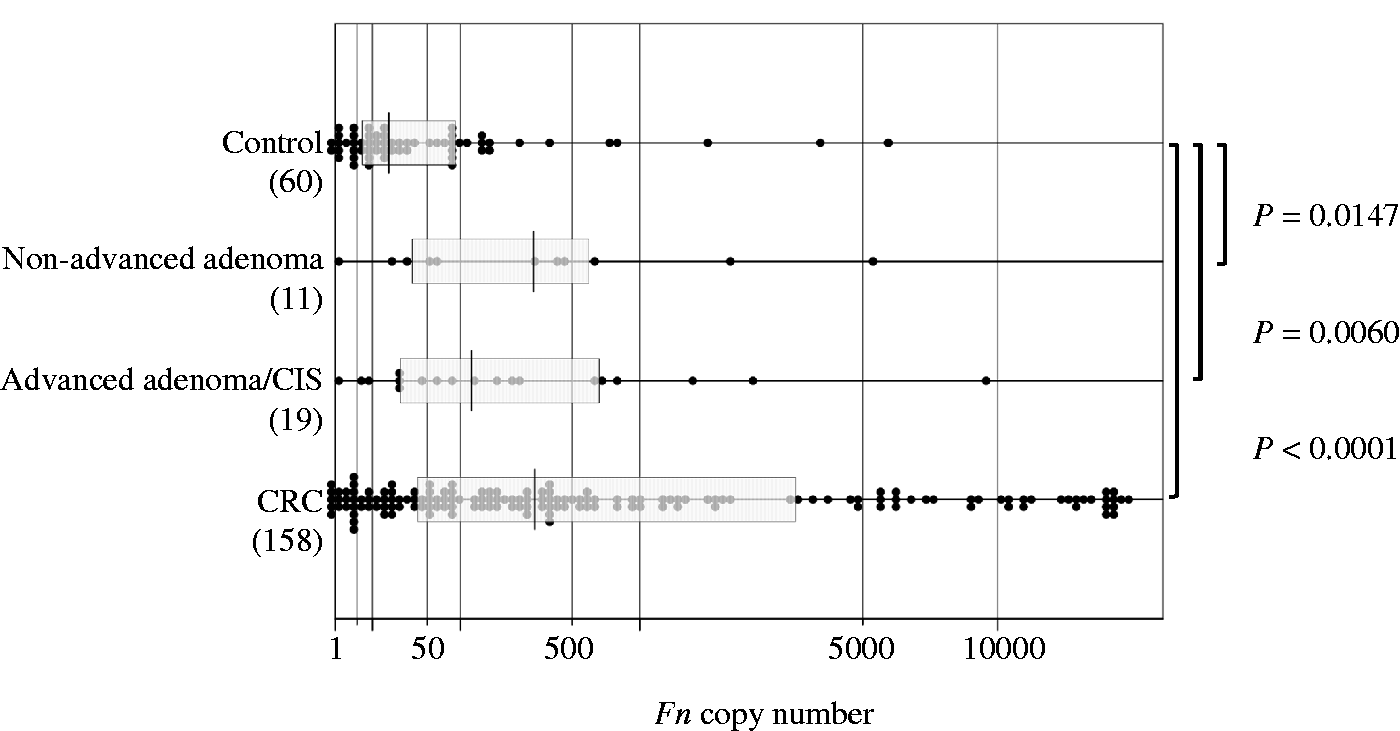
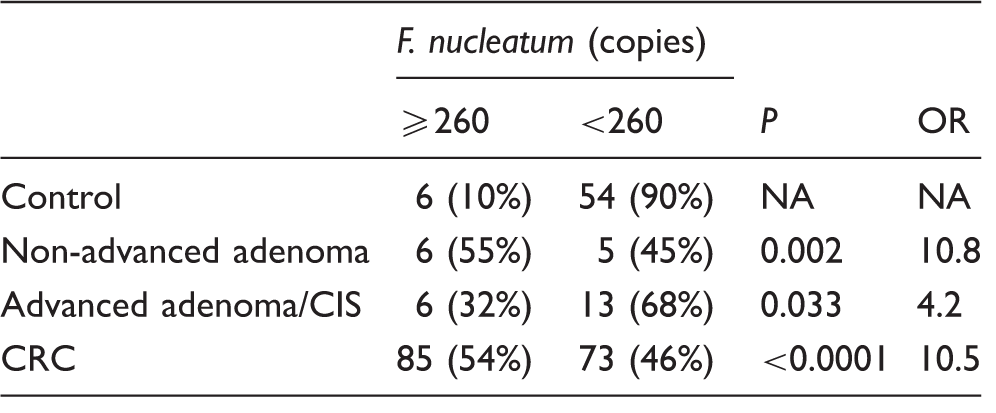
The result of the receiver-operating characteristic (ROC) curve analysis is shown in Figure 4. The area under the ROC curve was 0.75, and the best cut-off point to discriminate between control and CRC was 260 copies of Fn, resulting in a sensitivity of 54% and specificity of 90%. More than 260 copies of Fn were found in only 10% (6/60) of the healthy individuals in the control group, whereas an over-abundance (>260 copies) of Fn was found in 55% (6/11) of the non-advanced adenoma group (odds ratio [OR] = 10.8, P = 0.002), in 32% (6/19) of the advanced adenoma/CIS group (OR = 4.2, P = 0.033) and in 54% (85/158) of the CRC group (OR = 10.5, P < 0.0001, Table 1). Positive and negative predictive values were 50% and 92% for non-advanced adenoma, 50% and 81% for advanced adenoma/CIS and 93% and 42% for CRC detection, respectively.
Receiver-operating characteristic curve of Fusobacterium nucleatum to discriminate between control and colorectal cancer. Fusobacterium nucleatum levels in stool. CIS: carcinoma in situ; CRC: colorectal carcinoma; NA: not available; OR: odds ratio.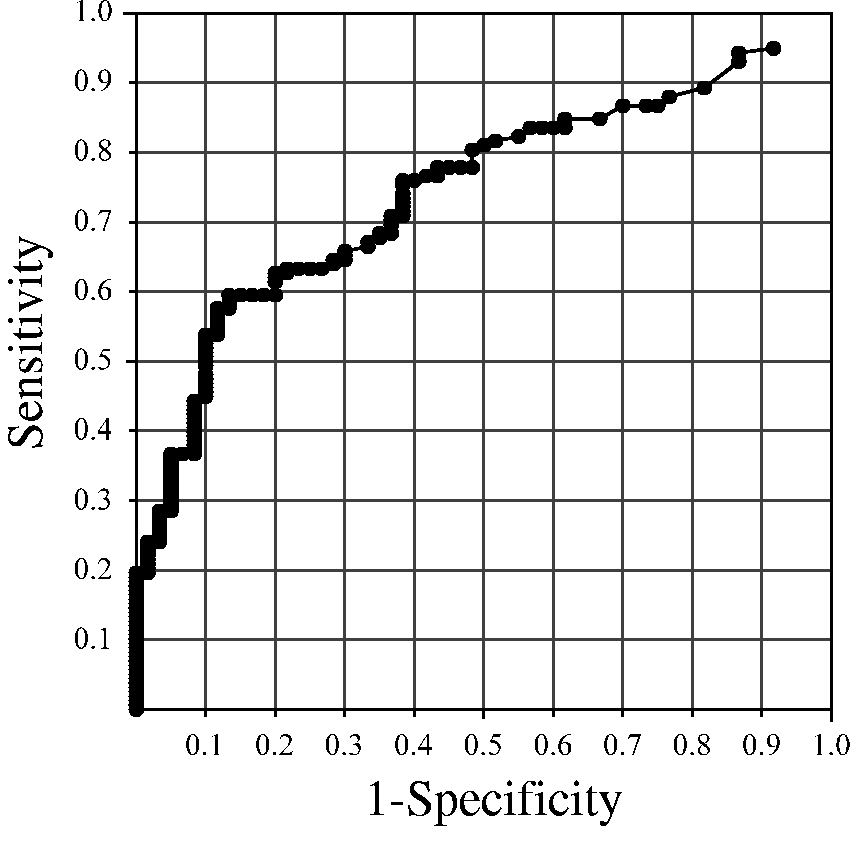
Discussion
Previous studies have shown that Fn is detected in the tissues by quantitative real-time PCR but not in the stool, 24 possibly because Fn adheres to and invades into colorectal mucosa. 23 In the present study, however, droplet digital PCR enabled the highly sensitive detection of Fn even in the stool because droplet digital PCR is 1000 times more sensitive than quantitative real-time PCR. 28 The copy number of Fn in stool was higher in the CRC group than in the control group, corresponding to the findings of a report with small sample size. 29 This study is the first, to our knowledge, to report the association of Fn level in stool with CRC in Japanese subjects. We found that the Fn level in stool was higher in both the non-advanced adenoma group and the advanced adenoma/CIS group than in the control group, suggesting that Fn in stool may affect the early stage of colorectal tumourigenesis. In addition, the Fn level in stool was higher in patients with stage II than in those with stage I CRC, implying that Fn in stool may have an influence on the infiltration of CRC because stage II reflects deeper invasion than stage I. This is the first study to date to associate the Fn level in stool with the early stage of tumourigenesis and cancer progression.
Kostic et al. 22 reported that through recruitment of tumour-infiltrating immune cells, Fn generates a proinflammatory microenvironment that is conducive for colorectal neoplasia progression. Rubinstein et al. 23 reported that Fn is a cause of CRC rather than a consequence. 23 Fn adheres to and invades colorectal cells and induces oncogenic and inflammatory responses to stimulate their growth through its unique FadA adhesin, 23 which binds to E-cadherin, activates β-catenin signalling, and differentially regulates inflammatory and oncogenic responses. The increased expression of FadA in CRC correlates with the increased expression of oncogenic and inflammatory genes. 23 Interestingly, the FadA level in tumours and even in normal mucosa from patients with colorectal adenomas or adenocarcinomas is more than 10–100 times higher than that in normal colorectal mucosa from normal individuals. 23 Because the FadA gene is unique to Fn, copy numbers of Fn in stool may reflect the FadA level and hence may be an ideal potential diagnostic marker to identify individuals at risk for developing adenomas and/or CRCs. However, because we have not investigated the level of FadA in stool, further examination will be required to prove our speculation.
Our study implies a potential for Fn to be a biomarker of CRC predisposition. We found an over-abundance of Fn in stool in 10% of the non-cancerous individuals and thus cannot exclude for them the possibility of developing CRC in the future. As a limitation of our study, however, the mean age of the control group was much younger than that of the colorectal tumour group; therefore, a prospective study is required to investigate whether Fn levels in stool actually increase the risk of CRC.
Another speculation is that the high level of Fn in stool may be related to colorectal tumours with a molecularly distinct subtype. Tahara et al. 20 reported that CRC tissues with a high level of Fusobacterium had a high degree of CpG island methylator phenotype (CIMP) and a high rate of gene mutations overall except for the TP53 gene. CIMP is a noted feature of chronic inflammation in normal tissues of patients with ulcerative colitis30,31 and in Epstein-Barr virus-associated gastric carcinoma.32–35 Interestingly, Tahara et al. 20 reported that all Fusobacterium-high CRC tissues were CIMP-1 or CIMP-2 positive, and none were CIMP negative. CIMP-1 CRC is associated with BRAF mutation and microsatellite instability caused by the hypermethylation of MLH1. 20 CIMP-2 CRC is characterized by methylation of a limited group of genes and mutation in KRAS. In contrast, CIMP-negative cases show less frequent methylation changes and very frequent TP53 mutation and chromosomal instability.36,37 Although our study suggests that a high level of Fn in stool may reflect CIMP status in colorectal tumour tissues, because we did not investigate the CIMP status of the colorectal tumour tissues, further studies are needed to elucidate this association.
In conclusion, we developed a highly sensitive stool DNA test of Fn and found that the Fn level in stool was significantly higher in patients with colorectal tumours than in the control group. Because this study suggests that stool DNA testing of Fn may be useful to detect individuals with colorectal tumours and to predict the predisposition to colorectal tumours, confirmatory studies using independent data sets are needed to support our findings. In the future, the regulation of Fn may provide a new preventive strategy against colorectal tumours.
Footnotes
Acknowledgments
We are very grateful to Ms. Naoko Okayama and Mr. Hidekazu Mizuno of Yamaguchi University Hospital, Ms. Tomomi Hoshida, Dr. Toshihiko Matsumoto, and Dr. Kiyoshi Ichihara of Yamaguchi University Graduate School of Medicine for their invaluable help in analysing the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by JSPS KAKENHI grant no. 25460687.
Ethical approval
This study was approved by the Ethics Committee of Yamaguchi University (reference number, H26-110).
Guarantor
YS.
Contributorship
YS and KS contributed equally to this work. YS researched data, wrote the manuscript, contributed to the discussion, and reviewed/edited the manuscript. KS, TH, MN, and NO researched data, contributed to the discussion, and reviewed/edited the manuscript. HM, TM, SH, TT, SH, YS, SH, MO, HN, IS and TY contributed to the discussion and reviewed/edited the manuscript. All authors reviewed approved the final version of the manuscript.
