Abstract
Background
Early diagnosis of thyroid cancer is hampered by the inability of fine-needle aspiration biopsy (FNAB) to accurately classify ∼30% of cases while preoperative cancer staging detects lymph nodal involvement in only half of cases. Liquid biopsy may present an accurate, non-invasive alternative for preoperative thyroid nodule assessment. Thyrotropin receptor (TSHR) mRNA, a surrogate marker for circulating cancer cells (CTC), may be an option for early detection of malignancy from peripheral blood, but requires methodological improvements. We aimed to investigate if TSHR mRNA can be detected in low sample volumes by employing an ultrasensitive method – droplet digital PCR (ddPCR).
Methods
Less than 5 mL of blood was collected from 47 patients with thyroid nodules (25 benign and 22 malignant). RNA was isolated from the fraction of mononuclear cells where CTCs segregate. Samples were analysed for the presence of TSHR mRNA by ddPCR.
Results
Thyrotropin receptor mRNA was detectable in 4 mL sample volumes, with the test having good specificity (80%) but modest diagnostic accuracy (68.1%). Combining TSHR mRNA with ultrasound features and FNAB diagnosis, the test reaches high rule-out performances (sensitivity = 90% and NPV = 88.2%). Strikingly, TSHR mRNA correctly classified all samples with thyroid capsule invasion, lymph node metastasis and extrathyroidal extension. If aggressiveness is defined using these parameters, TSHR mRNA test reaches 100% sensitivity and 100% NPV for detecting high-risk cases.
Conclusions
Employing ddPCR for TSHR mRNA improves its measurement by enabling detection in sample volumes common for laboratory testing. The test displays high prognostic performance, showing potential in preoperative risk assessment.
Keywords
Introduction
Preoperative thyroid cancer diagnosis is performed by fine-needle aspiration biopsy (FNAB), a reliable non-surgical method. However, roughly a quarter of patients receive an indeterminate result that leads to overdiagnosis and unnecessary treatments.1,2 Additionally, although a minimally invasive procedure, FNAB presents discomfort for the patients. Another problem in preoperative decision-making is that up to 50% of patients with papillary thyroid cancer (PTC) have cervical lymph node (LN) involvement, and only half of them can be identified through preoperative neck ultrasound. 3 Implementation of a validated circulating molecular marker would present a step forward in preoperative diagnosis and/or staging by presenting a non-invasive adjunct marker for early cancer diagnosis and selection of candidates for lymphadenectomy.
Liquid biopsy (LB) is a diagnostic concept based on the analysis of tumour-derived products present in body fluids, predominantly blood.4,5 The field of liquid biopsy as a substitute or complement to conventional biopsy has been actively explored over the past decade. A test for thyrotropin receptor (TSHR) mRNA analysed in peripheral blood of patients with thyroid nodules has been actively investigated by a group from Cleveland Clinic. 6 TSHR mRNA serves as a surrogate marker for circulating thyroid cancer cells. Although other tissues express TSHR, the group demonstrated that by using specific primers, thyrocyte-originating TSHR mRNA can be detected from the fraction of peripheral blood mononuclear cells. 7 Their subsequent research showed the test can improve preoperative diagnosis,8–10 and assist in detecting LN metastasis in papillary thyroid microcarcinomas (PTmCs). 11 Studies from independent research groups did not fully corroborate these results, with one study reporting TSHR mRNA correlates with aggressiveness 12 and another one failing to confirm its effectiveness. 13 Interestingly, the only study on European patient cohorts did not investigate its preoperative diagnostic or prognostic applicability. 14 Also, the European Thyroid Association (ETA) guidelines on molecular FNAB diagnostics do not include any comment on this test, 15 while the American Thyroid Association (ATA) 2015 guidelines state the test should be investigated more thoroughly. 3 Taking this into account, the usefulness of TSHR mRNA warrants further investigation in the target sample cohort.
Although many studies have explored the usefulness of LB in various malignancies, there are still many analytical challenges that complicate its translation to the clinic. One of them is that circulating cancer cells (CTCs) occur at very low levels in the circulation. 5 For that reason, higher volumes of blood (usually 7 mL) are required for their analysis. Also, unless specialized equipment for their sorting is applied, they are isolated along with white blood cells (WBCs), meaning that any DNA/RNA originating from CTC will exist in a large amount of unnecessary material from WBCs. Droplet digital PCR (ddPCR) has shown superiority in situations where the amount of the desired molecule is very low comparing to the background.16,17 This method has not been previously applied to the detection of circulating TSHR mRNA in patients with thyroid cancer.
We aimed to investigate if measurement of TSHR mRNA is applicable to a European patient cohort and if the ultrasensitive ddPCR will enable its detection in lower sample volumes. We performed a prospective study to validate the potential of TSHR mRNA in early detection of thyroid cancer and preoperative risk stratification, that is, the diagnostic and prognostic performance of TSHR mRNA in a Serbian cohort of patients.
Material and methods
Patients
A total of 47 whole blood samples were collected preoperatively from patients undergoing surgery for thyroid nodules at the Centre for Endocrine Surgery, Clinical Centre of Serbia, Belgrade. The study included 25 benign thyroid lesions, that is, 22 goiters and 3 follicular thyroid adenomas (FTAs) and 22 malignant tumours, that is, 10 papillary thyroid microcarcinomas (PTmCs), 7 papillary thyroid carcinomas (PTCs), 4 Hürthle-cell thyroid carcinomas (HTCs) and 1 poorly differentiated thyroid carcinoma (PDTC).
Ultrasound (US) examination and FNAB were performed several weeks before surgery. Ultrasound reports were examined in search for the presence of ultrasound features suspicious for malignancy according to ATA guidelines. 3 Samples positive for the presence of hypoechogenicity, microcalcifications, intranodular hypervascularity or irregular margins were classified as high risk for malignancy, whereby samples without these features were considered as low risk of malignancy. FNAB smears were evaluated according to Bethesda System for Reporting Thyroid Cytopathology. 1 Indeterminate cases (Bethesda III–V) that appeared at a rate of 35% in our cohort were classified as test positives when calculating diagnostic performance of FNAB. Non-diagnostic cases (Bethesda I) were excluded from this analysis. Upon surgery, ultrasound and pathology reports generated by two independent pathologists were collected and personal, preoperative and postoperative clinical data were recovered. That included age, gender, ultrasound features, FNAB diagnosis, final histopathological diagnosis, tumour size, the presence of extrathyroidal extension (Ei) and lymph node metastasis (LNM) and the degree of neoplastic infiltration (DI). DI was evaluated according to Basolo et al. 18 (A – totally encapsulated, B – not encapsulated without tumour capsule invasion, C – thyroid capsule invasion and D – extrathyroidal extension) and used as a binomial variable in calculating prognostic test performance, whereby the cut-off for aggressiveness was set between B and C. All patients had their blood drawn several weeks after FNAB and before surgery.
All patients were informed and gave their consent to use their biological material for research purposes. The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee at the Centre for Endocrine Surgery, Clinical Centre of Serbia, Belgrade no. 562/2.
RNA extraction and reverse transcription
We collected 4 mL of whole blood in EDTA-coated tubes and peripheral blood mononuclear cells (PBMC, buffy coat) were separated using Lymphocyte Separation Medium (Capricorn Scientific, Ebsdorfergrund, Germany) according to the manufacturer’s instruction. Total RNA was extracted using 400 mL of TRIzol reagent (Ambion, Carlsbad, CA, USA). Reverse transcription was performed with 1 µg of RNA using Revert Aid reverse transcriptase (Thermo Fisher Scientific, Waltham, MA, USA) and random hexamer primer in a two-step running method: 5 min at 70°C and 5 min at 4°C for primer annealing followed by 15 min at 25°C, 60 min at 42°C, 15 min at 70°C and 5 min at 4°C. Complementary DNA (cDNA) was stored at −20°C until further use.
Thyrotropin receptor mRNA quantification using droplet digital PCR
Levels of TSHR mRNA were measured using droplet digital PCR, with GAPDH as endogenous control, that is, reference gene. We found that a number of studies similar to ours which explored cancer biomarkers by measuring gene expression on ddPCR were using one or several reference genes in their analyses.19–23 Their use in the expression measurements on ddPCR is not obligatory; however, due to the reaction efficiency variations that can occur in reverse transcription and cause unequal cDNA loading into the reaction mixture, the recommendation in the field is to use reference genes as an additional normalisation step to control for these variations.24,25 Primer sequences of the analysed genes were as follows:
TSHR 7
Forward 5′-GCTTTTCAGGGACTATGCAATGAA-3′
Reverse 5′-AAGGGCAGTGACACTGGTTTGAGA-′3
GAPDH
Forward 5′-ACAGTCCATGCCATCACTGCC-3′
Reverse 5′-GCCTGCTTCACCACCTTCTTG-3′
Each 20 μL ddPCR reaction consisted of 10 μL of ddPCR Supermix (Bio-Rad, Hercules, CA, USA), 2 μL of cDNA and 0.40 μL of each primer. Each reaction mixture was transferred to a cartridge along with 70 μL of Droplet Generation Oil (Bio-Rad, Hercules, CA, USA) and partitioned into droplets using the QX200 Droplet Generator (Bio-Rad, Hercules, CA, USA). Droplets were transferred to 96-well plates (Eppendorf, Hamburg, Germany) and sealed using the PX-1 plate sealer (Bio-Rad, Hercules, CA, USA). The PCR was performed in a T100 Thermal Cycler (Bio-Rad, Hercules, CA, USA). The cycling conditions were as follows: 95°C 10 min, 1 cycle; 94°C 30 s, 58.5°C 1 min, 40 cycles; 98°C 10 min, 4°C ∞. Following end-point amplification, the individual droplets were measured with the QX200 Droplet Reader (Bio-Rad, Hercules, CA, USA) and analysed using QuantaSoft software (Bio-Rad, Hercules, CA, USA) to determine the target cDNA amount. All samples with less than 10,000 droplet counts were omitted from the following analysis. The mRNA amount was calculated using the concentration value of number of copies/µL normalized to GAPDH. The results of TSHR expression are reported in relative expression units similar to other studies performed with ddPCR for gene expression analysis of cancer biomarkers.19,21 More specifically, studies dealing with thyroid cancer biomarkers analysed via ddPCR22,23 used relative expression values for subsequent statistical analysis, and as Kurohama et al., 23 we also used expression values of the gene of interest normalized to reference gene for all analyses, and established a cut-off value based on ROC analysis (described in the following subsection) expressed in relative expression units.
Statistical analysis
Statistical analysis was carried out with the SPSS 12.0.1 software package (SPSS, Chicago, IL, USA) for Windows. Mann–Whitney U test was used for between-group comparison of non-Gaussian distributed variables. Receiver operating curve (ROC) analysis was used to assess the appropriate cut-off value for TSHR mRNA expression as a diagnostic or prognostic test, and used to calculate sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and diagnostic accuracy. Additionally, Younden’s J index was calculated to confirm the selection of the cut-off point. Fisher’s exact test or Pearson correlation test and Spearman’s rho were used for correlating binomial or continuous variables, respectively. Two-tailed statistical testing was used in all cases, and probability (p value) for less that 0.05 was considered statistically significant. Power analysis was performed using G*Power 3.1 free software tool.
Results
Thyrotropin receptor mRNA levels show a trend of rising in the circulation of patients with malignant thyroid tumours
Patient characteristics.
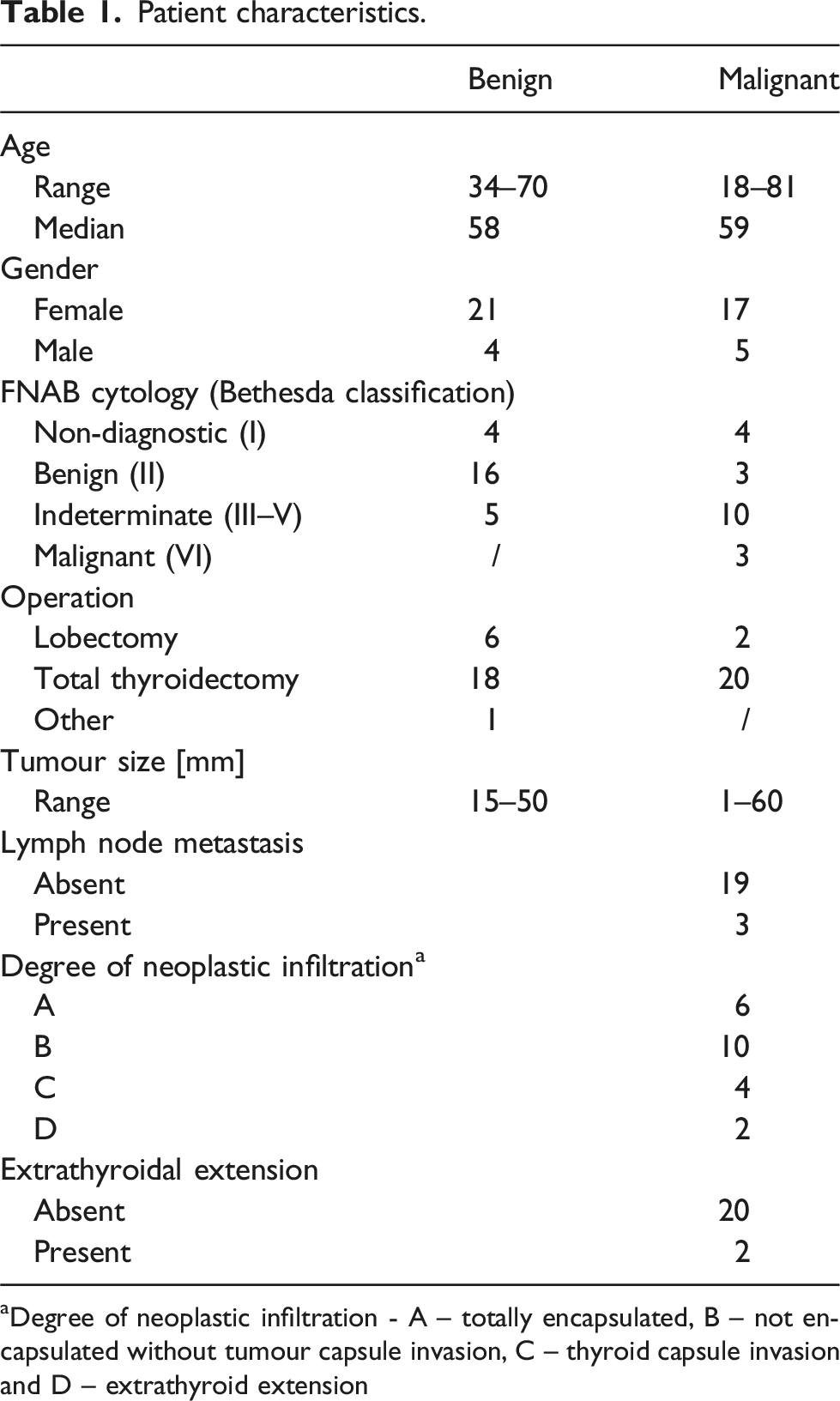
aDegree of neoplastic infiltration - A – totally encapsulated, B – not encapsulated without tumour capsule invasion, C – thyroid capsule invasion and D – extrathyroid extension
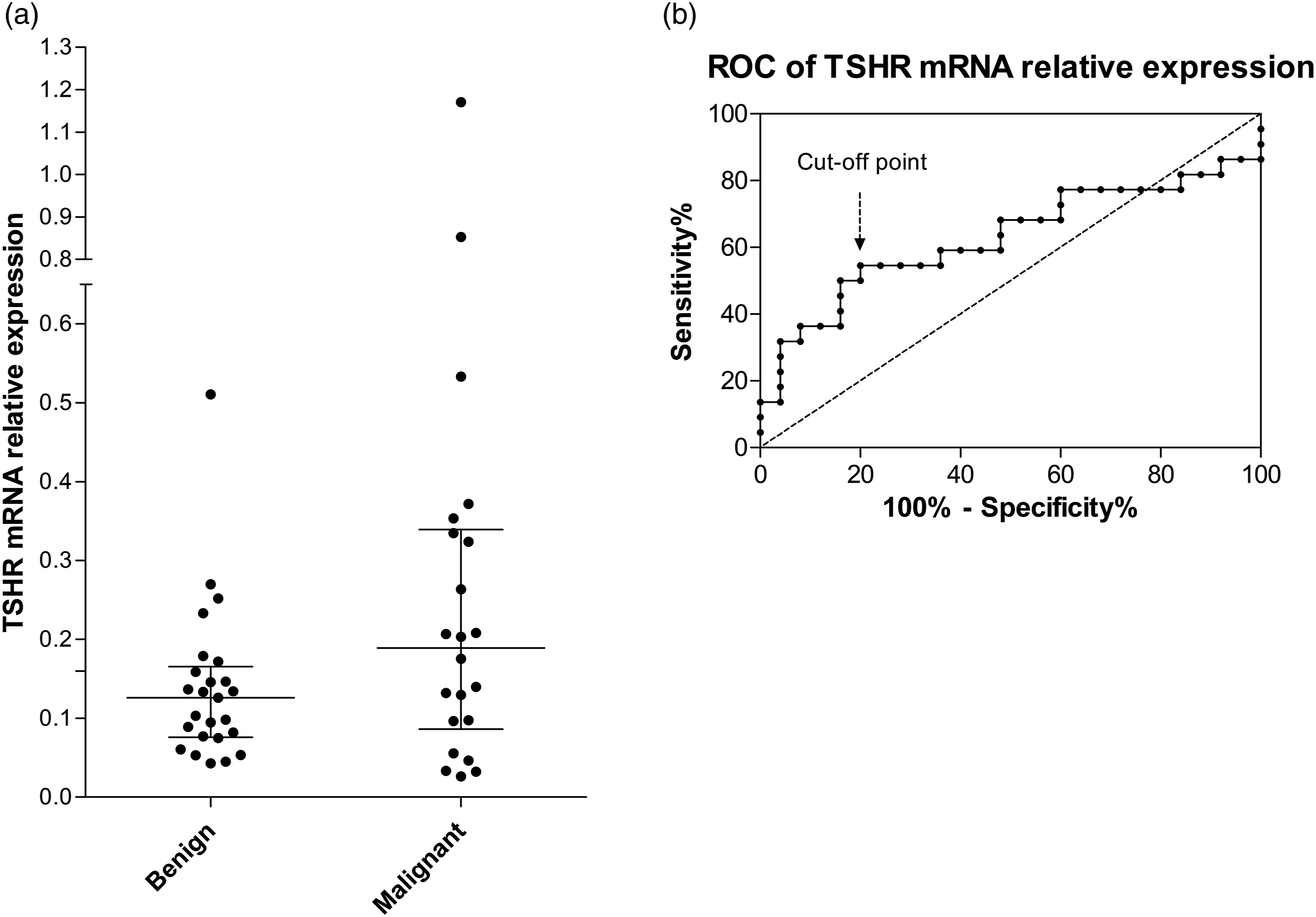
(a) TSHR mRNA relative expression levels in the circulation of patients with benign and malignant thyroid lesions. PBMC layer that contains CTC was separated with lymphocyte separation medium from whole blood samples. Specific primers were used to quantify TSHR mRNA from thyrocytes using droplet digital PCR. Lines represent median with interquartile range. (b) ROC curve of TSHR mRNA expression levels used to determine cut-off levels for test positivity. AUC = 0.627, cut-off point (0.173 relative expression units) was set at 54.5% sensitivity and 80% specificity.
Thyrotropin receptor mRNA test performance is nearing fine-needle aspiration biopsy performance metric
The diagnostic performance of TSHR mRNA, FNAB diagnosis and TSHR mRNa combined with suspicious ultrasound features and FNAB.

PPV, positive predictive value; NPV, negative predictive value.
*p < .05, Fisher’s exact test.
**p < .01, Fisher’s exact test.
TSHR mRNA classification of thyroid lesions according to histology.
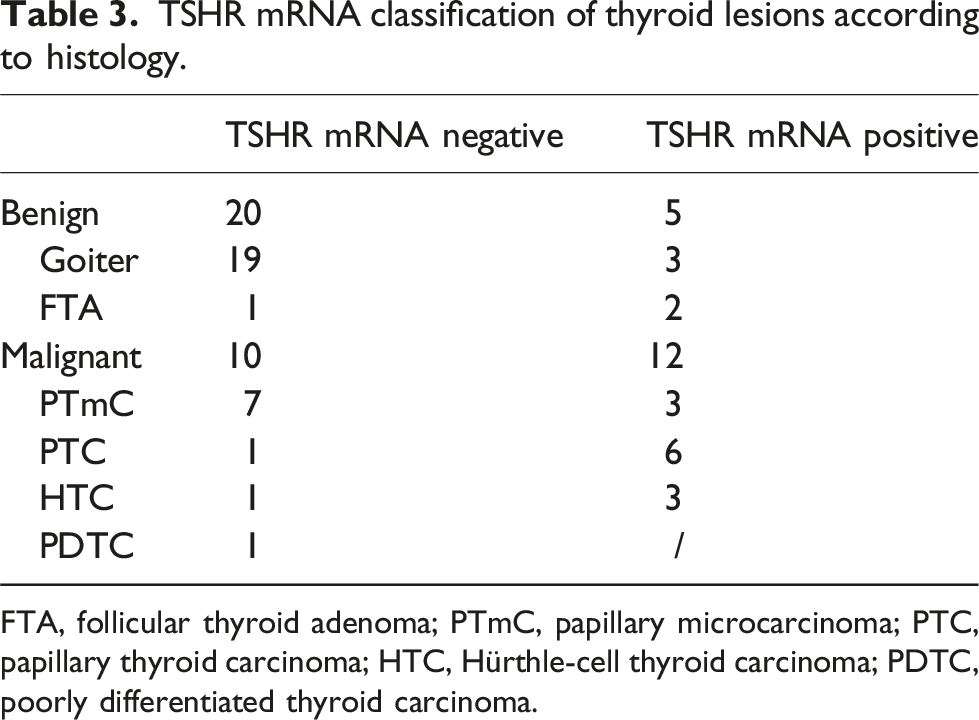
FTA, follicular thyroid adenoma; PTmC, papillary microcarcinoma; PTC, papillary thyroid carcinoma; HTC, Hürthle-cell thyroid carcinoma; PDTC, poorly differentiated thyroid carcinoma.
As FNAB is a gold standard in preoperative assessment of thyroid nodules, we calculated the diagnostic performance of FNAB in our cohort and compared it to TSHR mRNA (Table 2). FNAB gives an indeterminate result in up to 30% of cases (35% in our cohort); however, since those cases are appointed to surgery, we counted them as FNAB test positive. Non-diagnostic cases were excluded from this analysis. While diagnostic accuracy of FNAB was indeed higher than that of TSHR mRNA (78.9%), specificity and PPV values of TSHR mRNA were on par or higher than FNAB performance.
With the aim to increase the test performance, we added ultrasound (US) features in the test. US features that were considered suspicious for malignancy included the presence of hypoechogenicity, microcalcifications, intranodular hypervascularity or irregular margins, according to the ATA guidelines. 3 The test using TSHR mRNA and US resulted in a higher sensitivity (68.2%) and slightly higher diagnostic accuracy (72.3%) but at the expense of specificity (76%) (Fisher’s exact test, p = .003, Table 2).
Finally, when both preoperative tests used in the clinic for nodule assessment (US features and FNAB) are combined with TSHR mRNA, the test sensitivity reaches a high value correctly detecting 90% of cases with a high level of accuracy for the negative test result (NPV = 88.2%) (Fisher’s exact test, p = .003, Table 2).
Circulating Thyrotropin receptor mRNA detects aggressive forms of thyroid cancer
Finally, we looked at the levels of this marker in the aggressive forms of thyroid cancer to assess its prognostic performance. Due to the low sample size, we were unable to perform statistical tests with single clinicopathological parameters. Therefore, we calculated the aggressiveness based on the presence of the following clinicopathological markers of tumour severity, that is, thyroid capsule invasion, extrathyroidal extension and metastasis to the lymph nodes. By using power analysis, we confirmed that our sample size used in the analysis of TSHR mRNA prognostic performance was sufficient. We calculated the sample size needed to observe the selected effect size for a test with power = 0.8. We estimated the effect size from Liu et al. 12 by calculating Cohen’s d from TSHR mRNA measurement in patients with LNM and capsular invasion. The effect size was considered large according to Cohen’s d criteria 27 (3.01 and 2.37 for LNM and capsular invasion, respectively). With alpha = 0.05 and power = 0.8, we calculated that the sample size needed to observe a large effect size (0.5– 0.6 in Fisher’s exact test) is N = 18– 22 samples. Thus, given that a priori effect size is large, we concluded that our sample size used in the analysis of prognostic performance of TSHR mRNA (N = 22) is appropriate for detecting the effect, according to the estimates from power analysis.
TSHR mRNA classification of cases according to clinicopathological parameters of aggressiveness.

The prognostic performance of TSHR mRNA test to classify patients according to aggressiveness.
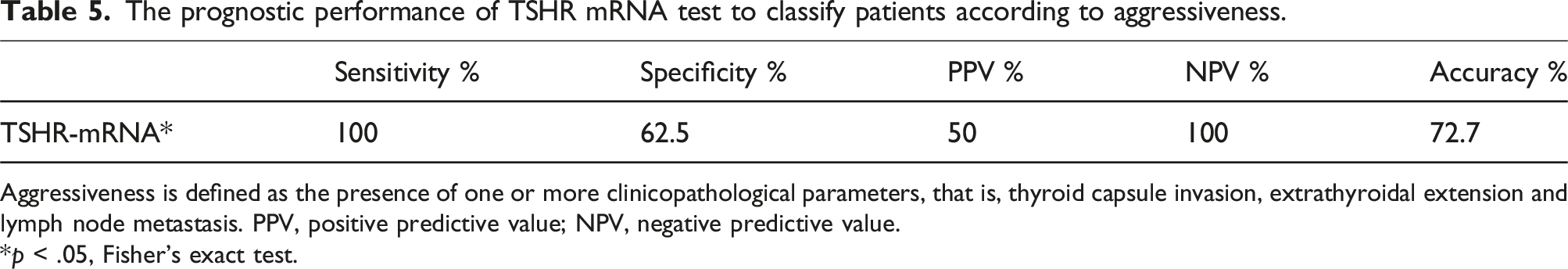
Aggressiveness is defined as the presence of one or more clinicopathological parameters, that is, thyroid capsule invasion, extrathyroidal extension and lymph node metastasis. PPV, positive predictive value; NPV, negative predictive value.
*p < .05, Fisher’s exact test.
Discussion
In this study, we evaluated the test performance of detecting thyroid-specific TSHR mRNA as a surrogate marker for circulating thyroid tumour cells in understudied, European cohort of patients. We were able to detect the transcript in a sample volume lower than other studies by using a powerful detection method that is droplet digital PCR. The test performance of TSHR mRNA was high for preoperative risk assessment, but modest for early cancer detection.
Our results showed that measurement of TSHR mRNA is successful for detecting aggressive cases and thus cancer staging. Several studies investigated the association of TSHR mRNA measurement with clinicopathological parameters. Chia et al. detected higher TSHR mRNA levels in lymph node-positive than node-negative patients, although the difference was not significant. 8 Two subsequent studies on PTmC showed that cervical nodal metastases occurred more frequently with tumours over 5 mm and positive TSHR mRNA2 28 and that in PTmC patients over 45 years TSHR mRNA can predict LN involvement. 11 Recent study from China correlated preoperative TSHR mRNA expression levels with patient age, capsular invasion status, lymph node metastasis and BRAFV600E mutation status. 12 Our study, the first European prognostic study, corroborates these findings as all patients with extrathyroidal invasion, capsular invasion and LN metastasis were accurately classified as positive using TSHR mRNA measurement.
In the prognosis of thyroid cancer, predicting recurrence rather than survival is a more relevant oncologic outcome due to low mortality of differentiated thyroid cancer. 29 LN metastasis is the most important independent factor for disease recurrence.30–32 Preoperative detection of cervical lymph node involvement is performed via physical examination and ultrasound that is not precise for detection of central LN compartments,33,34 resulting in only 20–30% of patients being accurately examined with US. If recurrence does occur, a second operation in the central neck compartment carries higher complication rate. 35 Performing a prophylactic central lymph node dissection (pCND) is still a matter of debate as clinical studies have reported no clear benefit for pCND36–38; however, they suffer from the lack of power. 39 Until a consensus is reached, surgeons are advised to take an individualised approach in pCND decision-making, 40 and our test shows a potential in assisting in this process. In our cohort, no patients with LN metastases were missed by TSHR mRNA measurement leading to 100% sensitivity and 100% negative predictive value of the test. This test can therefore be used as an adjunct rule-out method for refining the group of patients that would benefit from initial lymphadenectomy.
Our results showed the test has high specificity for detecting malignancy, with a solid PPV, while the overall accuracy was strengthened with the addition of suspicious US features in the test. This is very similar to the test performance reported by the Cleveland Clinic group.6,8,9,41 In our sample cohort, these parameters were nearing, but stayed below FNAB test performance, possibly due to small sample size. However, as opposed to FNAB, TSHR mRNA combined with US presents a non-invasive option for preoperative evaluation of thyroid nodules, and may therefore be an intriguing possibility that deserves further investigation. It is noteworthy that when all three preoperative markers are combined, such a test reaches high percentages for sensitivity correctly detecting 90% of cases, and being highly reliable when excluding malignancy with an NPV of 88.2%. In this way, TSHR mRNA raises the rule-out performances of common preoperative tests for nodule assessment.
Another matter that was addressed was the sample volume necessary for the analysis of TSHR mRNA originating from CTC. Although CTC shedding is an early event in the development of cancer42,43,44 and despite the common view that primary tumours release numerous malignant cells into blood vessels, their levels are still quite low in the circulation. 5 Therefore, CTC analysis requires that higher volumes of blood (>5 mL, commonly 7 mL) are taken from the patients. By employing sensitive, droplet digital PCR, we were able to detect malignancy, especially aggressive types of thyroid cancer, in 4 mL of starting material.
However, our test missed a proportion of malignant cases. Looking more thoroughly into the histology of false negatives, 9/10 were less aggressive malignancies (non-aggressive cases of PTmC and PTC and minimally invasive HTC), and a PDTC with low expression of TSHR due to dedifferentiation. 26 It is possible that in cases of lower tumour burden (PTmC tumours less than 1 cm) and non-aggressive cases, lower numbers of CTCs are present in the circulation. Contrary to that, higher tumour burden might explain the two false positive FTAs with tumour size over 30 mm. Milas et al. have observed that false positive cases occur in large thyroid goiters, 10 while Aliyev et al. found a correlation with tumour focus size and TSHR but a reverse correlation with specimen weight and TSHR positivity in the cohort of PTmCs. 11 Additionally, we observed a positive correlation of TSHR mRNA with the size of the tumour in malignant cases while it was milder than the association of the marker with tumour aggressiveness. In our opinion, this speaks in favour of the hypothesis of TSHR mRNA being detectable in cases burdened with tumour cells in number, but more dominantly, in prometastatic behaviour, reflected in the presence of extrathyroidal extension and metastases to the lymph nodes, regardless of tumour size. The false positive goiters that we detected all had suspicious ultrasound features and two had a Bethesda IV FNAB diagnosis. Studies in mice showed that CTCs with metastatic potential are released even before a histological confirmation can be made 44 ; however, we can only speculate that this was the possible scenario for those cases. Milas et al. also suggested that cases with Hashimoto thyroiditis could present a risk for false positives 6 ; however, in our cohort, we did not observe a correlation with chronic lymphocytic or Hashimoto thyroiditis (data not shown), similarly to Liu et al. 12 In summary, the diagnostic performance of our test was not high primarily due to its lower diagnostic sensitivity, that is, missing out on a number of malignant cases; however, their non-aggressive nature points out the test is well suited for detecting progressive thyroid cancer due to its high prognostic sensitivity for detecting aggressive cases.
The limitations of our study include the low sample size used for the prognostic performance assessment, and the lack of aggressive variants among the PTmC. The strength of our study lies in the diversity of histological diagnosis included in the investigation that reflects the real-world situation of a busy clinic for endocrinology. Additionally, the final histopathological diagnosis was confirmed by two independent pathologists increasing the reliability of the clinicopathological data collection.
Conclusions
In summary, TSHR mRNA is detectable via ddPCR in sample volumes common to laboratory tests and able to detect malignancy with high specificity but modest overall diagnostic performance in a European sample cohort. TSHR mRNA test has 100% sensitivity and 100% NPV for detecting aggressive thyroid cancer variants and may be used as a rule-out test in planning the extent of surgery and lymphadenectomy. Large clinical studies are warranted to confirm this. Finally, our results contribute to the pool of evidence that TSHR mRNA is a useful marker in various study cohorts, and stimulates further research in the field of liquid biopsy for DTC.
Supplemental Material
Supplemental Material - Detecting thyrotropin receptor mRNA from peripheral blood of patients with differentiated thyroid cancer rules out non-aggressive cases
Supplemental Material for Detecting thyrotropin receptor mRNA from peripheral blood of patients with differentiated thyroid cancer rules out non-aggressive cases by Jelena R Janković Miljuš, Uršula Prosenc Zmrzljak, Rok Košir, Milan Jovanović, Ilona Đ Đorić, Jelena V Rončević, Tijana M Išić Denčić and Sonja A Šelemetjev in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to acknowledge Dr Dubravka Cvejić for the critical review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the ddPCR Grant Challenge initiative from Labena Ltd., Verovskova 64, 1000 Ljubljana, Slovenia, and by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Agreement No 451-03-9/2022-14/200019).
Ethical approval
The study was approved by the Ethics Committee at the Centre for Endocrine Surgery, Clinical Centre of Serbia, Belgrade no. 562/2.
Guarantor
J.J.M.
Contributorship
Conceptualization: J.J.M.; patient selection: J.J.M. and V.Ž.; clinical data acquisition: V.Ž.; sample acquisition and processing: J.R.; methodology, U.P.Z. and R.K.; investigation: J.J.M., J.R. and U.P.Z.; formal analysis: R.K., I.Đ. and T.I.D.; data curation: J.J.M, S.Š., T.I.D and I.Đ.; writing – original draft preparation: J.J.M.; writing – review and editing: S.Š., I.Đ., T.I.D., U.P.Z. and R.K.; funding acquisition: J.J.M. and S.Š. All authors have read and agreed to the published version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
