Abstract
Patient safety is a leading challenge in healthcare and from the laboratory perspective it is now well established that preanalytical errors are the major contributor to the overall rate of diagnostic and therapeutic errors. To address this, the European Federation of Clinical Chemistry and Laboratory Medicine Working Group for Preanalytical Phase (EFLM WG-PRE) was established to lead in standardization and harmonization of preanalytical policies and practices at a European level. One of the key activities of the WG-PRE is the organization of the biennial EFLM-BD conference on the preanalytical phase to provide a forum for National Societies (NS) to discuss their issues. Since 2012, a year after the first Preanalytical phase conference, there has been a rapid growth in the number of NS with a working group engaged in preanalytical phase activities and there are now at least 19 countries that have one. As a result of discussions with NS at the third conference held in March 2015 five key areas were identified as requiring harmonisation. These were test ordering, sample transport and storage, patient preparation, sampling procedures and management of unsuitable specimens. The article below summarises the work that has and will be done in these areas. The goal of this initiative is to ensure the EFLM WG-PRE produces work that meets the needs of the European laboratory medicine community. Progress made in the identified areas will be updated at the next preanalytical phase conference and show that we have produced guidance that has enhanced standardisation in the preanalytical phase and improved patient safety throughout Europe.
Introduction
Patient safety is a leading challenge in health care. A systematic approach is necessary to improve patient safety and to reduce the probability of patient harm from unnecessary exposure to diagnostic or therapeutic procedures.
1
It is now well established that preanalytical errors are the major contributor, from the laboratory perspective, to the overall rate of diagnostic and therapeutic errors.2–5 Although the term ‘preanalytical phase’ first began to appear in publications already back in the 1970s, almost two decades were needed to realize that the preanalytical phase is an area that can no longer be neglected.6,7 Obviously, a wide systematic approach to limit the burden of preanalytical errors at an international level is only possible under the umbrella of formally established bodies, which should take the lead in standardization and harmonization activities. Otherwise, most individual efforts done at national level will remain isolated. Unfortunately, until recently, such body did not exist at a European level. The European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) has decided to address that need by establishing the Working Group for Preanalytical Phase EFLM (WG-PRE) in 2012.
8
This group has grown since then in size and activity, and currently has 13 members and two expert consultants from the diagnostic industry.
9
The main EFLM WG-PRE activities in Europe focus on the following key goals:
Promote the importance of the preanalytical phase. Assess the quality of the current preanalytical policies and practices by performing surveys. Define the best practices and provide recommendations and guidance for some most critical preanalytical phase activities and procedures. Encourage harmonization of preanalytical policies and practices. Educate laboratory professionals by providing educational materials, organizing conferences, courses, webinars, etc.
The aim of this article is to present the overview of the past and current activities, as well as the vision and mission of EFLM WG-PRE in leading the way at European level in standardization and harmonization of preanalytical policies and practices.
Preanalytical phase activities within EFLM national societies
One of the key activities of the WG-PRE is the organization of the biennial EFLM-BD conference on the preanalytical phase in partnership with Becton Dickinson (BD). The third conference was held in Porto (Portugal) in March 2015. 10 The conference covered a number of important topics, including unnecessary test requesting, implementation of EU needle stick injury directives, harmonization of fasting requirements,11,12 influence of physical factors and contrast media on laboratory tests,13,14 order of draw, transportation, centrifugation and preanalytical external quality assessment (EQA).15–20 The Porto conference was attended by over 600 professionals, up from 250 in Parma (Italy) in 2011 and 400 in Zagreb (Croatia) in 2013. The increase in conference attendance highlights the growing interest and recognition that the preanalytical phase is achieving.
In the final session of the conference, all EFLM National Societies (NS) were invited to present their activities and interests. The aim of this initiative was to gather all representatives of EFLM NS and learn about their activities at national level through which they have addressed preanalytical phase quality. More specifically, our aim was to learn how many NS have already established preanalytical working groups, whether they have performed some surveys related to preanalytical phase or have they published national recommendations or guidelines related to the preanalytical phase. We were also interested to know what challenges and problems NS face and how they plan to deal with these in the future. We assumed that there were already many active groups across Europe, and it was our intention to create an open forum to discuss common issues of interest, share experiences and see how we could mutually benefit from each other. All EFLM NS were invited to participate, 20 accepted the invitation and were represented in Porto. Each NS had 10 min for their presentation.
Preanalytical challenges identified by EFLM NS
From NS presentations, we learned that the first official preanalytical group was launched in 1995 in Germany, followed in 1999 by Spain, then Italy in 2005. Since 2012, a year after the first Preanalytical phase conference, there has been a rapid growth in the number of NS with a working group engaged in preanalytical phase activities, and there are now at least 19 countries that have one (Figure 1). We believe that recent WG-PRE activities, at least to some extent, are mirrored by the increasing number of nations with a preanalytical working group.
Number of guidelines and surveys published by EFLM National societies (NS). The NS on the x axis are aligned from left to right, respective to their year of origin.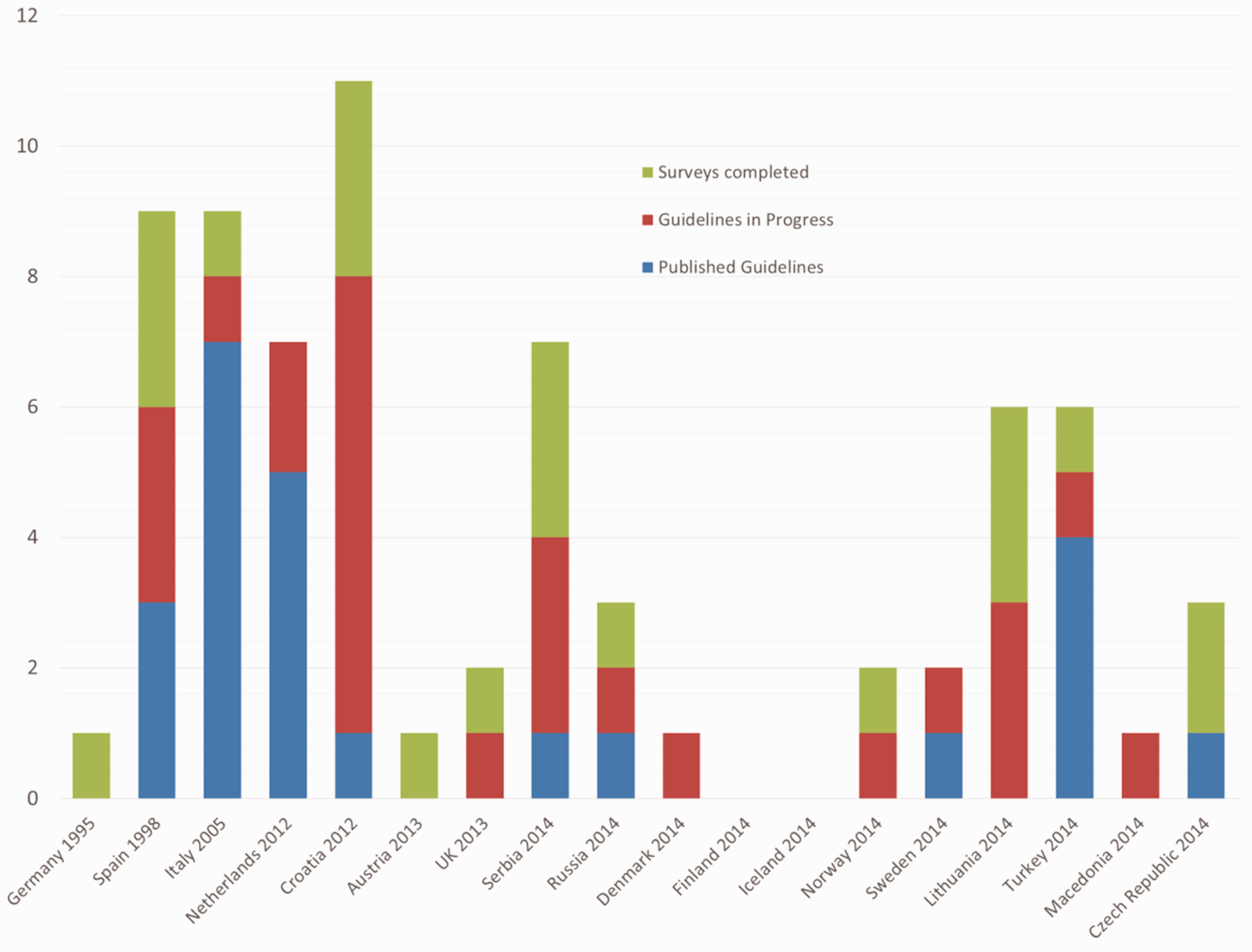
These working groups have produced a vast amount of work in their relatively short life and have performed 21 surveys and produced 19 sets of guidelines with a further 22 planned (Figure 1). Of the guidelines and surveys performed, a significant proportion was around phlebotomy procedures. We also learned that 8 out of 22 countries had already established some form of national preanalytical EQA scheme, and many societies have been engaged in organization of scientific educational events on some preanalytical topics.
During their presentations at the Porto meeting, all NS were asked to address the issues of harmonization of preanalytical phase policies and procedures at a national and European level, and the joint statement of all participating NS was that:
Harmonization of preanalytical phase policies and practices is possible and necessary at national level in each individual country in Europe. Harmonization of preanalytical phase policies and practices is possible and necessary at international level. EFLM NS are willing to work with the EFLM to achieve harmonization of the preanalytical phase in Europe and help developing internationally acceptable guidelines and recommendations and implementation of those at national level.
When asked to define which preanalytical steps they think are the most critical and need immediate harmonization, eight areas were suggested for consideration (Figure 2). The five most popular areas identified by NS were: test ordering, sample transport and storage, patient preparation, sampling procedures and management of unsuitable specimens.
Key preanalytical steps identified by EFLM NS as the most critical and in need of immediate harmonization.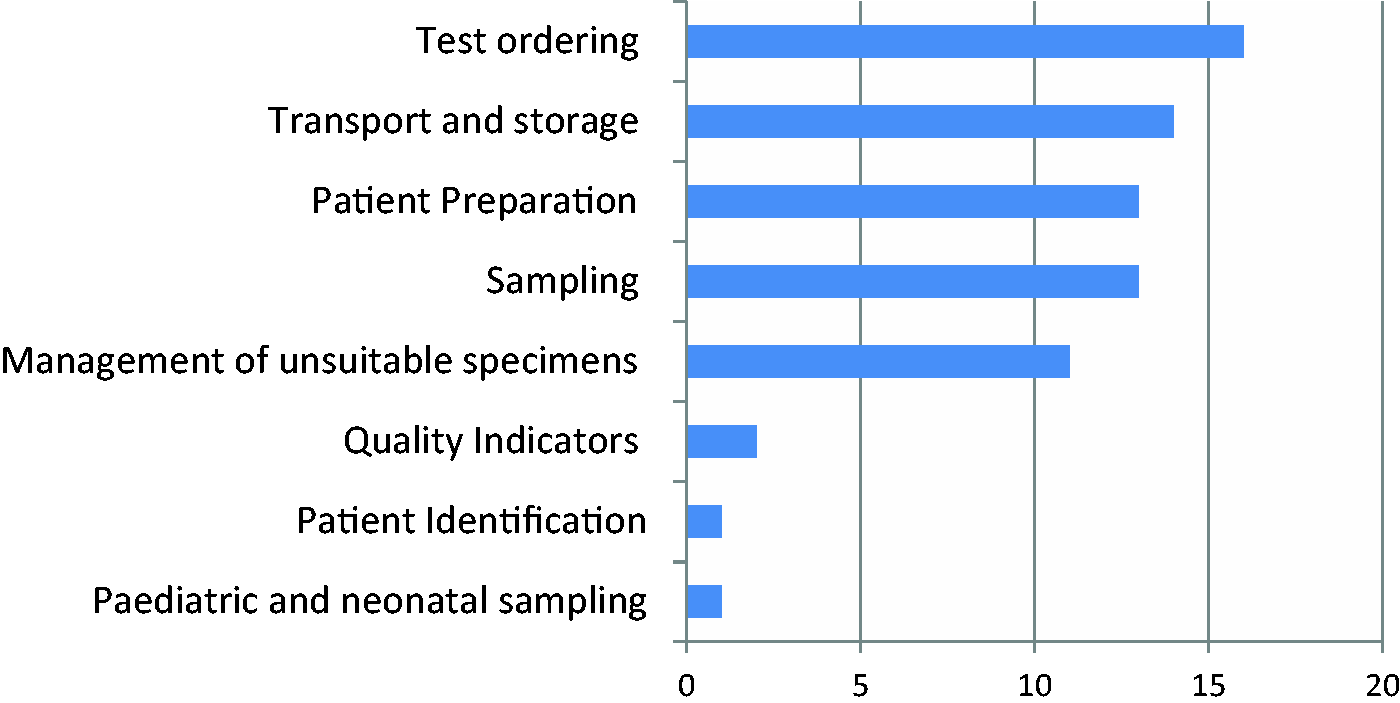
These key issues will certainly help WG-PRE to better target its future work. Moreover, given the momentum that has been achieved and the growing number of countries, experts and professionals involved in the field across Europe, there is a need to join these efforts and take advantage of it. A pragmatic approach would be a bi-directional collaboration between individual societies and EFLM WG-PRE. Work that has already been done at the national level should be identified and translated to the wider audience at the European level. Furthermore, some tasks and responsibilities could be delegated to individual NS. Once completed, the outcomes of such projects might then be used by other NS. The EFLM WG-PRE feels the responsibility to initiate and carry on a dialogue on that. NS presentations in Porto were the very first step, and we believe that a lot more can be achieved by collaboration between NS and EFLM WG-PRE in the future.
The WG-PRE has discussed the key areas that would benefit most from standardization and harmonization during its recent meeting and below is the summary of the work that has already been done in these areas. When considering our future activities, we will certainly bear in mind the on-going and planned activities across Europe and see how to complement the work already done and avoid unnecessary overlaps.
The contribution of EFLM WG-PRE to some ongoing and future preanalytical phase projects in Europe
Test ordering
Inappropriate test requesting has long been known as a waste of resources and results in inappropriate patient pathways. It is also known that demand for laboratory testing is increasing disproportionately to medical activity. This is often due to defensive medicine where clinicians over-request to ensure they do not miss anything. 21 However, it is challenging to measure the problem, with rates of inappropriate requests being somewhere between 5% and 95%.22,23 Inappropriate tests are those that could be avoided with no detriment to patient care, but when considering appropriate utilization of the laboratory, it is important to include tests which were not requested but which would have been clinically relevant at the time of the initial request. 24 The area should therefore ensure that all appropriate tests are performed and inappropriate tests suppressed. There are various mechanisms to ensure appropriate test ordering, both via reducing inappropriate tests and by ensuring the correct tests for a condition are performed. 25 The ultimate aim of looking at appropriate test ordering is, as well as improving the patient journey and reducing costs, to standardize the care received regardless of location.
Studies such as The NHS Atlas of Variation in Diagnostic Services in the UK are a useful tool in targeting variation to try and increase under-requesting areas and decrease over-requesting areas. 26
Although most NS have identified this as one of the top priority preanalytical issues, there have been only few initiatives related to test demand management at the national level in Europe. One such example is the Minimum Retesting Interval project in UK. Minimum retesting intervals can be used to put blocks into place to prevent a test being repeated in a time window that is not clinically relevant. 27 Interestingly, a recent study evaluating the effectiveness of locally developed computerized alert system based on re-testing intervals showed that unnecessary testing can be reduced up to 70% using this approach. 28
There are also some well-documented processes to achieve the reduction in inappropriate testing, such as reflective testing, where a frontline test is performed with subsequent tests added only when clinically useful in light of the original result. 29 The Netherlands has done a lot of work in this area, and a webpage containing many resources has been developed to assist laboratory professionals in implementing reflective testing in everyday routine. 30 Both these mechanisms should help to prevent defensive medicine to some extent. As previously mentioned, the final mechanism entails continuous education of requestors possibly by using specific software which flag tests that have been recently requested or else using pop-up information boxes or showing the price of tests on the request screen. It is also essential that evidence-based medicine is taught during education to highlight the need for appropriate testing. There is also an on-going project in Spain under the Pilot Group of the Appropriate Utilization of Laboratory Tests working group aimed to study and address inappropriate test requests at the national level.31–34
Reflex testing, as well as preventing some tests from being performed, also ensures that required tests are performed in a timely manner and often without recollecting specimens from the patients. An alternative approach is condition-specific requesting, a growing area where clinicians use information technology systems to click on a condition; pre-set algorithms then determine what tests are required as a frontline. Further tests are then cascaded within the laboratory, as required. 29
Transport and storage
The transport of samples, their storage within the laboratory and the conditions surrounding both situations have long been known as important preanalytical factors with varying effects on the potential test result. In 2002, the World Health Organisation (WHO) published recommendations on sample types and stabilities in various different conditions, which has been further updated and published in various languages by the Extra Analytical Quality Assurance group of the German United Society for Clinical Chemistry and Laboratory Medicine.35,36 Although this work from the German United Society for Clinical Chemistry and Laboratory Medicine is the most comprehensive, there has been considerable other work from various sources looking into this area.37–40 In fact, the Netherlands and Macedonia already have national guidelines defining the requirements for transport, with Macedonia also providing guidance on storage conditions for human samples. As a result of the wide range of work done or being done in this area, it would be a good area to collect the data and standardize. Such initiative has already been undertaken by Norway, which are in the process of creating a web resource as a single point of reference with all the known sample stabilities for different analytes, across different analytical platforms, tube types and analytical methods.
Patient preparation
Patient preparation for laboratory tests is an area of high variation not just across Europe but also within nations. The data clearly show that this is an issue many nations feel needs addressing, and an area that should be standardized across Europe to ensure patients are provided with the right information for the tests requested. This is something that does not currently occur.41,42 There are a large number of preanalytical variables that fall under the umbrella of patient preparation and can affect laboratory tests. These include diet, drugs (medication and recreational), herbal remedies, vitamins, physical activity, smoking, alcohol and many more. It is essential to standardize the recommendation and information using an evidence-based approach but also to establish robust mechanisms in order to ensure that this information reaches the person who needs it most (i.e. the patient). It is only by doing this that the patient can be assured of receiving truly accurate lab results and avoiding unnecessary follow-up investigations. The WG-PRE has already performed some work in this area and has produced a comprehensive overview and recommendations on fasting requirements for laboratory testing. 11 The conference in Porto has highlighted that there is good work already being carried out in this area by Croatia, Lithuania and Serbia, who have conducted large surveys and also plan to produce some recommendations in the near future. 43 It is the desire of WG-PRE to see how this document can be adapted and used to serve wider audience as EFLM recommendation. This is just one example demonstrating how efforts can be shared to the mutual benefit, according to the principle: all for one and one for all.
Sampling
Many nations feel that standardization of blood sampling is an important step forward, and the data we collected at the conference in Porto highlighted that many NS have produced or are in the process of producing guidelines for the blood sampling process. The vast majority of these nations have used the Clinical and Laboratory Standards Institute (CLSI) documents as a basis for their own national guidelines, with some amendments to fit local regulations and/or practices. The fact that many nations feel the need to produce their own guidelines despite the existence of the CLSI and WHO guidelines indicates that perhaps these documents can be further improved or adapted to fit national (legal and technical) requirements in terms of blood collection.44,45 This therefore illustrates that there is a compelling need to monitor each step of the phlebotomy process and produce evidence-based guidelines that target the key steps. There is further difficulty in this area, however, in that even once good quality standardized guidelines are produced, the staff has to be trained in the new guidelines. The training received by staff performing blood collection varies greatly across Europe, 46 and data have shown that compliance with the existing CLSI guidelines is poor. 47 The WG-PRE plans to produce an auditing guideline complete with risk occurrence chart and make this tool available to all interested laboratories, to help them identify risks with their own phlebotomy process. Taken together, this highlights the need to engage the appropriate staff to roll out new guidance, as well as the need to ensure that compliance is continually monitored to maintain standardization.
To address this aspect, WG-PRE has launched a project to produce a consensus European document defining the best phlebotomy practices. As a first step, WG-PRE has hosted in June 2015 in Paris the 1st European meeting involving stakeholders in phlebotomy from many European NS. During that meeting, an initial discussion took place about the scope and content of the document. By engaging national stakeholders from an early stage, it is our hope that as many NS as possible will commit to promoting the new guidance and facilitating its uptake via the best channels in each nation. The document will seek input from NS and other stakeholders such as nurses and laboratory technicians with blood collection responsibility. The consensus document is planned to be finalized in 2016.
Management of unsuitable specimens
The last of the big areas where preanalytical groups from around Europe would like to see standardization is the management of unsuitable specimens. By this, we mean standardizing the recommended action taken when a sample is received that is haemolysed or lipaemic, where tubes are incompletely filled, or if samples are clotted to name but a few. As well as problems with the sample or type of sample, this also includes problems with patient ID and samples that do not match request forms. It is currently unknown how much variation there is in this area. Therefore, before standardization can be started, the problem must be accurately quantified. We suspect that the vast majority of laboratories follow the advice of their instrument supplier regarding haemolysis, icterus and lipaemia, but it has been shown that these recommended levels are not always scientifically accurate. 48 However, data have been produced on the number of samples rejected from different laboratories emphasizing the vast heterogeneity existing in rejection rates. 49 The best way to collect this information would be to use data from EQA schemes, analysing rates and causes of rejections, and also assessing the variation in responses to case based scenarios. These data could then be used to provide standardized, evidence-based guidelines on the recommended action to be taken in a variety of situations. WG-PRE is planning to assist NS in this area by launching a pilot EQA preanalytical project in collaboration with the European Organisation for External Quality Assurance Providers in Laboratory Medicine. The Norwegian Quality Improvement of Primary Health Care Laboratories (Noklus) and the Norwegian Clinical Chemistry EQA Program performed a pilot preanalytical EQA scheme in 2014 where medical laboratories reported four defined errors in samples received from primary health care. The same registrations will be carried out in 2015 and 2016 to look for a potential improvement and to evaluate if this can be established as an on-going Norwegian EQA-program. Another pilot study focused on harmonizing expression of serum indices on different analytical platforms has been planned by WG-PRE, and it is projected to be completed in 2016.
Quality indicators
The International Federation of Clinical Chemistry and Laboratory Medicine WG on Laboratory Errors and Patient Safety (WG LEPS) has established a range of quality indicators, and collated data from across Europe to assess the rates of errors and their variation,50,51 possibly leading to a lower degree of interest in this area. After running for several years, the number of indicators was harmonized at a European meeting in Padova in 2014 to reduce the overall number and focus more on those that were most important and achievable. 52 There are also many laboratories now publishing data on the use of quality indicators, and how their use has led to improvement in the quality of service by the laboratory. However, the number and type of quality indicators, as well as how they are counted, still varies between laboratories and should be seen as a real hurdle for efficient comparisons. 49
Observational studies are nowadays increasingly used to look at the error rates and compliance with guidelines. At the Porto conference, 10 abstracts dealing with this subject were presented. The advantage of direct observation of specimen collection errors, when also performed on a larger scale, yields an error frequency for each practice step. When a severity grading is also added to the observed error frequency, an overall risk assessment and indication on the most important steps for corrective actions is obtained. 47
The rise in the number of laboratories collecting quality indicators has been driven not only by interest within the profession, but also by the new ISO 15189 standardization. This has also led some EQA providers to initiate schemes and allow data comparison across different laboratories.
Patient identification
The CLSI guidelines state that patients must state their name, address, ID number and/or date of birth if conscious. Any discrepancy must be recorded and reported as determined by local policy. If the patient is unconscious and emergency testing is required, this practice may not be applicable to safeguard patient’s life. The fact that the Porto conference showed that there was some interest in standardizing this aspect indicates that some NS feel that the topic is still a matter of debate. In its recent publication, WG-PRE showed that tube labelling and patient identification were the most critical steps during phlebotomy. 47 This underlines the need for the role of WG-PRE in providing recommendation and encouraging harmonization of patient and sample identification throughout Europe. To address this need, WG-PRE has recently published an article aimed to raise the awareness about the need for immediate improvement of patient identification and tube labelling processes and calling for harmonization of these important preanalytical steps. 53
Paediatric and neonatal sampling
As with general venous sampling, there is significant interest in providing standardized guidelines for capillary blood sampling. Croatia and the Netherlands have already performed a lot of work in this area. 54 There are some significant problems with capillary blood collection over venous blood collection in addition to the benefits of being able to test multiple analytes on a small sample, most significantly the fact that haemolysis and lipaemia cannot be detected until specific sensors for measuring cell-free haemoglobin in whole blood (for example) are developed. As with venous testing, there are guidelines available from both the CLSI and WHO.44,55 The work published by Croatia shows that there are deviations from the guidelines.56,57 A detailed look at practices across Europe is needed, with the aim to produce good evidence-based recommendations that take into account the real world situation. Again, valuable documents produced at the national level might serve here as a good starting point.
Discussion
The eight topics discussed above have been identified by the growing number of preanalytical phase national working groups as key areas for standardization. The EFLM WG-PRE used the Porto conference on the preanalytical phase to showcase what is happening in the growing preanalytical community and viewed the feedback from the conference and national working groups as an opportunity to be told what our colleagues felt we should be doing to move the area. As discussed, eight areas were identified and the EFLM WG-PRE will aim to address these over the next 2 years and feedback our progress at the 4th EFLM-BD European Conference on Preanalytical phase in 2017 in Amsterdam (The Netherlands). A summary of the planned work for this period is:
Publish and share the opinion paper on patient identification. Develop the WG-PRE website as a central source of information. Follow-up the Norwegian project on transport and storage and publicize this. Publicize the Croatian and Serbian work on patient preparation. Publish a consensus document on venous blood sampling involving all relevant stakeholders. Establish a scenario-based EQA scheme focussing on the preanalytical phase. Assess the practices related to detection and management of unsuitable specimens, across Europe. Publish and share the opinion paper on patient identification.
53
The end result of all this work is to ensure the EFLM WG-PRE produces work that is desired by the European laboratory medicine community. It is the aim of the group to be able to report back on all these issues at the next preanalytical phase conference and show that we have produced guidance that has enhanced standardization in the preanalytical phase and improved patient safety throughout Europe. It is also our aim to place all the guidelines and evidence in a central place on the Web, which will become the first place to look when laboratories need evidence or guidance on whatever issue of the preanalytical phase.
The fact that European NS have agreed that harmonization is necessary and possible at a national level as well as across Europe is extremely encouraging and will be our driving force in the future. We also recognize the willingness of European NS to work with EFLM WG-PRE to achieve harmonization of preanalytical phase in Europe. Harmonization will take time and will need considerable effort. We believe this is feasible, but only through a joint effort, collaboration and by sharing our resources. EFLM WG-PRE will do its best to assist and contribute to this ambitious plan.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
AMS.
Contributorship
AMS, GL and MC put the original draft together which was edited by all authors. All authors were involved in the collection of data and creation of figures
