Abstract
Background
Synthetic cannabinoids (NOIDS) are novel psychotropic drugs (NPS) currently freely sold in the United Kingdom as ‘research chemicals’. Detection of NOIDS use is not available in current routine methods. Here we describe a marker which helps determine which patients have used these substances.
Methods
In a test case, ultra-performance liquid chromatography mass spectrometry (UPLC-Tof) was used to screen the legal high Herbal Haze II, the contents of hand-rolled cigarettes and five patient samples for NOIDS and their metabolites.
Results
Analysis of legal high Herbal Haze II and cigarettes identified the third generation adamantyl-type NOIDS N-(1-adamantyl)-1-pentyl-1H-indazole-3-carboxamide (AKB-48), 5F-AKB-48 and N-adamantyl-1-fluoropentylindole-3-carboxamide (STS-135). Out of 18 potential metabolites, 1-adamantylamine (C10H17N) was detected in all five urine samples. This adamantyl-type NOID marker was incorporated into our routine LC-MS/MS urine screen. Out of 14,436 random urine samples screened over eight months, 296 (2.05%) tested positive for the adamantyl-type NOID marker.
Conclusion
We have discovered a urine marker for identifying patients smoking legal high products containing the third generation adamantyl-type NOIDS such as AKB-48 and its fluoropentyl analogue 5F-AKB-48, which are among the most popular NOIDS currently available in legal high products sold in UK. This marker can be incorporated into routine LC-MS/MS drug screening alongside classic drugs of abuse. Positive detection rates for this new legal high marker are greater than for established classic drugs that are routinely screened such as amphetamine. This work highlights the need for a flexible toxicology screening service capable of adapting to changes in drug use such as the growing popularity of legal highs/NPS.
Introduction
Synthetic cannabinoids (NOIDS) sometimes referred to as Black Mamba, spice, K2 or herbal incense are novel psychotropic drugs (NPS) which mimic the effect of cannabis. Typically legal high products are prepared by spraying the NOIDS onto herbs which users then incorporate into roll-ups and smoke usually together with tobacco. Sold as research chemicals ‘not for human consumption’, the 1968 medicines act does not apply. Consequently, these products are freely available to buy on the high street and on-line. Like Δ9-tetrahydrocannabinol (THC) found in cannabis, the psychotropic effects of NOIDS are a result of binding to cannabinoid receptors located in the brain.1–3 Unlike THC, most NOIDS are full rather than partial agonists making them far more potent. 4 Although clinical presentation varies, reported cases frequently describe catatonic states, dystonia, aggression, respiratory depression, hypotension and tachycardia.5–9 As with cannabis abuse there is evidence that regular NOID abuse is linked to an increased risk of psychosis.10–12 A number of deaths have been attributed to NOIDS.13,14
Based on their chemical structure, first and second generation NOIDS have been broadly classified into six groups: classical, napthoylindoles, napthoylnapthalenes, phenylacetylindoles, benzoylindoles and cyclohexylphenols15–17 and by 2013 all were recognized under UK Misuse of Drugs Act 1971 as class B controlled drugs. 18 Shortly afterwards third generation NOIDS not covered by the 2013 legislation, started to appear in herbal type legal highs sold in the UK, and many incorporate an adamantyl group (adamantyl-type NOIDS).
Adamantyl-type NOIDS were first identified in legal high products sold in Japan in 2012; N-(1-adamantyl)-1-pentyl-1H-indole-3-carboxamide (SDB-100) and N-(1-adamantyl)-1-pentyl-1H-indazole-3-carboxamide (AKB-48). 19 Further adamantyl-type NOIDS have since been identified, including N-adamantyl-1-fluoropentylindole-3-carboxamide (STS-135) and a flouropentyl version of AKB-48 (N-(adamantan-1-yl)-1-(5-fluoropentyl)-1H-indazole-3-carboxamide (5F-AKB48)). 20 The rationale behind the addition of an adamantyl group may be linked to reports that it can improve drug potency, by increasing the affinity for receptor binding and impeding degradation by hydrolytic enzymes.21,22 The latest 2014 Forensic Early Warning System (FEWS) report identified adamantyl-type NOIDS as the most popular third generation NOID currently on sale in UK, with 5F-AKB-48 present in 45% of 345 products seized from head shops in Aberdeen and Plymouth. 23
Detection of NOID abuse presents a particular challenge, both to the clinician and testing laboratories. NOIDS lack the distinct pungent smell of cannabis. The information provided on legal high products is limited, inaccurate, and often users are unsure of what they have actually taken. Whilst it is relatively easy to test and identify parent drugs in legal high products, detection in biological samples is more challenging. NOIDS are not detected by conventional drugs of abuse screening methods. 24 NOIDS undergo extensive metabolism, and as they are more potent than cannabis, concentrations of both parent drug and metabolites are likely to be low in biological fluids.
High resolution mass analysis, for example time of flight mass spectrometry (Tof), has been used to identify NPS in legal high products.25,26 Tof has also been used to investigate NOID metabolism using in-vitro studies to identify potential markers for screening, with hydroxlated and carboxylated metabolites a common finding.27,28 This has led to development of specific screening methods using immunoassay29,30 and mass spectrometry.31–35 These specialist tests exclusively screen for NOIDS and metabolites and do not include other drugs.
We are a UK NHS laboratory that provides a specialist clinical toxicology service, including screening for classic drugs of abuse and NPS. In 2014 we were referred samples from a clinical incident of multiple patients suspected of using NOIDS, for which the legal high product, hand-rolled cigarettes and patient urine samples were sent to us for analysis. We have used ultra-performance liquid chromatography mass spectrometry (UPLC-Tof) to identify a novel marker for the third generation adamantyl-type NOIDS in urine. We have then incorporated this marker into our routine drugs of abuse ultra-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) screen, which includes other classic drugs of abuse and legal highs. Here we present data for the positive detection rate of this marker for adamantyl-type NOID use over eight months of routine screening (July 2014 to February 2015).
Experimental
Chemicals and solutions
All solvents and water were purchased from Fisher Scientific® (Leicestershire, UK) and were LC-MS/MS grade. Leucine encephalin, 1-adamantylamine and internal standard 1-(1-adamantyl)ethylamine hydrochloride (rimantadine) were brought from Sigma-Aldrich (TX, USA). Reference drugs used for UPLC-Tof were purchased from Cerillant® Sigma-Aldrich, LGC® (Luckenwalde, Germany) and Purechemicals.net (London, UK).
Validation samples
Herbal legal high Herbal Haze II (1 g, London WC1N 3XX), two hand-rolled cigarettes and five random urine samples (test patient samples) were received from a secure mental health facility for NPS screening by UPLC-Tof (January 2014). Drug free urine used as blanks for method development, validation, calibrators and controls were provided by laboratory volunteers.
Routine urine samples for drugs of abuse screening
Random urine samples (14,477) collected into plain white universals were received for routine urine drug screening by LC-MS/MS for both clinical and medico-legal purposes from locations across the UK.
Preparation of non-biological samples
For legal high and tobacco products 1 mg/mL stocks were prepared in methanol by sonicating for 10 min and centrifuged for 5 min. For analysis, supernatants were then further diluted in methanol to give ∼1000 μg/L extracts.
Preparation of urine samples
For both LC-MS/MS and UPLC-Tof analyses urine drugs were extracted and concentrated using a method developed in-house. Into a 2 mL vial was added 400 µL of urine, 10 µL of IS mix and 600 µL of a reagent containing acetonitrile, ethyl acetate and 500 mmol/L ammonium carbonate buffer (pH 8). After mixing, the bottom layer was removed by centrifugation and the upper drug-containing layer transferred to a fresh vial and dried under a stream of nitrogen. The dried extracts were then reconstituted in 100 µL of loading buffer.
UPLC-Tof analysis
The UPLC-Tof system used was a Waters ACQUITY UPLC® (binary solvent manager, sample organizer, sample manager and column manager) interfaced with a hybrid Waters Xevo G2 QTof detector, with electrospray ionization in positive ion mode (Waters, Co., Milford, MA). Two methods were used for NPS qualitative screening, a NOID specific method and a general drug screen that includes over 1300 drugs and metabolites.
The NOID specific method includes a collision-induced dissociation (CID) mass spectral library containing over 100 first, second and third generation NOIDS and their metabolites and was developed in-house. It uses a Waters ACQUITY UPLC® BEH C18 1.7 µm 2.1 × 100 mm column protected with a BEH C18 1.7 µm 2.1 × 5 mm VanGuard pre column as a stationary phase, maintained at 50℃. Drug separation was performed over a total of 12 min using a gradient of 5 mmol/L ammonuim formate pH 3 (solvent A), and 0.1% (v/v) formic acid in 100% acetonitrile (solvent B) at a flow rate of 500 µL/min. Data acquisition was performed using Waters MassLynx software at a cone voltage of 25 V, capillary voltage 3.0 kV, desolvation gas flow of 900 L/h at 500℃, with a scan rate of 0.2 per sc in centroid mode from m/z 30 to 600 (MSE function).
The second general drug screening method was used as described by Rosano et al. 36 with additional mass spectra library additions performed in-house. It has a CID mass spectral library containing over 1300 drugs and metabolites, 10% of which are NPS. For routine screening the adamantyl-type NOID marker was incorporated into this second general drug screening method (C10H17N RT 2.88 min f:135.1174), and was used for the routine confirmation of all urine UPLC-MS/MS screen positive results.
For both methods leucine encephalin (2 ng/µL in 50/50 (v/v) acetonitrile/water containing 0.1%(v/v) formic acid) was used as a lock-mass calibrant and analysed for two ions at 556.2271 Da and 120.0813 Da every 30 s at a flow rate of 5 µL/min throughout the run. Sample injection volumes were 10 µL. Criteria used for drug identification was a mass accuracy of 5 ppm for parent compound (function one) and qualifier fragment (function two), with an average isotope fit within 20% of the calculated ratio and a target RT ± 0.3 min. 36
UPLC-MS/MS urine drug screen
The UPLC-MS/MS system comprised a Waters ACQUITY® UPLC system (binary solvent manager, sample organizer, sample manager and column manager) and Xevo TQD detector with positive electrospray ionization. Chromatographic separation was performed using a Waters ACQUITY® UPLC HSS C18 1.8 µm 2.1 × 150 mm column, and HSS C18 1.8 µm 2.1 × 5 mm VanGaurd pre column maintained at 30℃ with a total run time of 6 min using a gradient of 5 mmol/L ammonuim formate with 0.025% (v/v) formic acid (solvent A), and 0.025% (v/v) formic acid in 100% methanol (solvent B) and injection volume of 15 µL. The drug testing panel includes 26 drugs and metabolites, and for detection it uses multiple reaction monitoring (MRM). Creatinine is included as an integrity checker to identify adulterated samples. 37 For 1-adamantylamine (adamantyl-type NOID marker) quantifier and qualifier ions were 152.3/135.1 m/z and 152.3/107.2 m/z, respectively, with a RT of 3.19 min. Rimatadine was used as IS with monitoring of a single transition 180.3 > 163.2 m/z (RT 3.67 min). Criteria used for positive identification were a transition ion ratio (>10) of ±50% and target RT ± 0.2 min.38–42 Included in each analysis was a five-point adamantyl-type NOID marker calibration curve (0–80 μg/L) and two controls ±25% of the positive cut-off threshold of 5 μg/L, all prepared in blank urine collected from a drug free volunteer.
Results
Validation samples
Legal high product Herbal Haze II came in a green metal foil sachet. The contents (1 g) were a light green plant like material with a pungent sweet smell. UPLC-Tof screening using the NOID specific method detected third generation NOIDS AKB-48 (C23H31N3O RT 10.66 min f:135.1174), 5F-AKB-48 (C23H30FN3O RT 9.56 min f:135.1175), 5F-PB-22 (C23H21N2O2F RT 6.19 min f:232.1135) as well as traces of STS-135 (C24H31FN2O RT 8.7 min f:135.1164). AKB-48, 5F-AKB-48 and STS-135 are all adamantyl-type NOIDS. UPLC-Tof screening using the general drug screen was negative.
Examination of the contents of the two hand-rolled cigarettes revealed in addition to tobacco a small amount of light green material identical in appearance to the contents of legal high product Herbal Haze II (see Figure 1). UPLC-Tof screening using the NOID specific method detected AKB-48, 5F-AKB-48 and 5F-PB-22. UPLC-Tof screening using the general drug screen detected nicotine only.
Contents of hand-rolled cigarettes from validation test case containing synthetic cannabinoid herb material. 1. Hand-rolled cigarette. 2. Tobacco contents containing light green herb material. 3. Contents legal high herbal haze II.
Summary of possible AKB-48 metabolites used for screening five test patient urine samples by UPLC-Tof using NOID specific method.

Validation UPLC-Tof method for adamantyl-type NOID marker
For the UPLC-Tof general drug screening method, the limit of detection of the adamantyl-type NOID marker determined using drug-spiked urines collected from 10 different volunteers was <2 μg/L for detection of the parent compound only, and 5 μg/L for parent compound and qualifier ion.
Validation of LC-MS/MS method for adamantyl-type NOID marker
The limit of quantitation (LOQ) defined as the lowest drug-spiked urine calibrator concentration with a signal to noise >10, minimum of 10 data points across each peak, drug concentration ± 20% of nominal value (%accuracy) and %CV < 20% (n = 5) for our adamantyl-type NOID marker was <2 μg/L. To enable UPLC-Tof confirmation the positive cut-off threshold for the routine LC-MS/MS screening method for adamantyl-type NOID marker was set above the LOQ at 5 μg/L. The coefficient of determination was R2 > 0.99 (0–100 μg/L). Carryover at 10,000 μg/L was <1 μg/L and below the LOQ. At 10,000 μg/L no cross-talk was observed between quantifier and qualifier MRM for the adamantyl-type NOID marker and the IS rimantadine. Inter-assay precision at ±25% of the 5 μg/L positive cut-off threshold (n = 10 analysed over 2 weeks) was 9.9% and 11.1%, respectively. Matrix effects (ME) were assessed according to the method of Gandhi et al. 28 Absolute matrix effects (AME), true recovery and process efficiency were all within our acceptable target for a screening method of 100 ± 30%.
Urine screening results
From July 2014 to February 2015 we received 14,465 urine samples from 7232 individuals for urine drugs of abuse screening by LC-MS/MS. Of these samples 29 (0.2%) were rejected after the integrity check (creatinine concentration <1.0 mmol/L) indicated adulteration. The majority of our workload (86%) are clinical requests with 6% originating from our own Accident & Emergency (A&E) and inpatients, 16.5% from other hospitals, 63% from mental health trusts and 0.7% other. The remaining 13.8% of requests were for private testing for medico-legal purposes such as workplace testing. More samples were received from males 10,711 (74%) compared to females 3601 (24.9%) with 153 (1.1%) sex not stated. The mean age of patients tested was 34.6 years.
Summary of LC-MS/MS urine screen %positive monthly detection rates for adamantyl-type NOID marker.
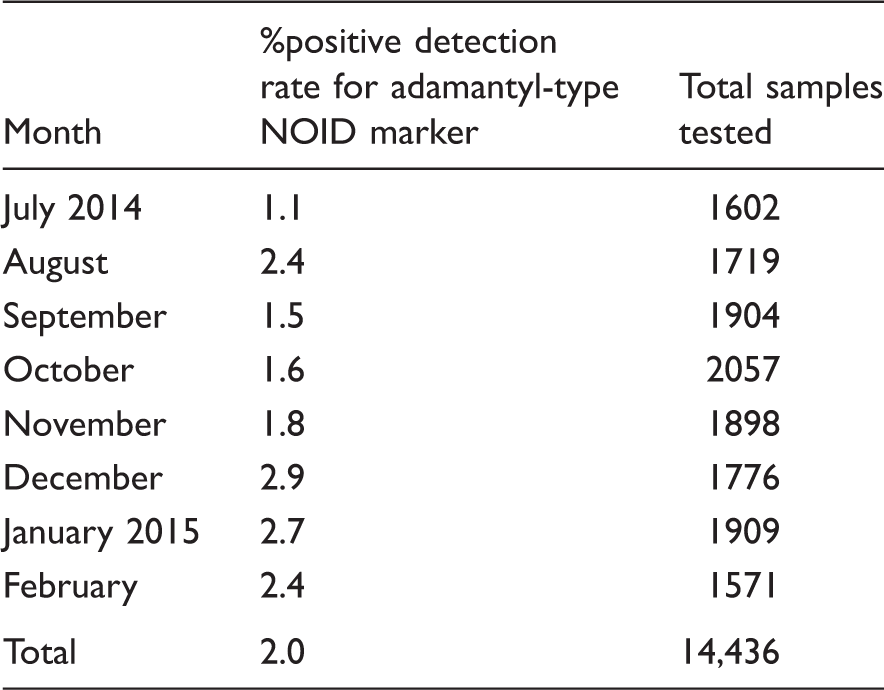
Overall, 180 individuals tested positive for the adamantyl-type NOID marker which is 2.5% of the total 7232 individuals tested over the eight months. The majority of these 180 individuals (86.5%) were male. The mean age was 37 years (range 3 days to 56 years). The youngest patient to test positive was a three-day-old male infant with this request querying intrauterine drug exposure which we confirmed. Repeat testing was responsible for the remaining 116 positive adamantyl-type NOID marker results we reported. One 53-year-old male tested positive for the adamantyl-type NOID marker on 19 different occasions. It was confirmed by the case worker that this individual is a regular user of ‘Black Mamba’ a common generic street name used for any herb type legal high which is typically added to smoking products.
Discussion
In January 2014, we received patient samples as well as suspect materials for screening for NPS from a secure mental health facility, where there was a high suspicion that patients were abusing a synthetic cannabinoid (NOID) type legal high. By testing the suspected legal high product (Herbal Haze II), and contents of associated hand-rolled cigarettes, we were able to confirm the suspicion of NOID involvement with the detection of third generation NOIDS, including the adamantyl-type NOIDS AKB-48, 5F-AKB-48 and STS-135. None of these adamantyl-type NOIDS were detected in the patient urine samples. This is not unexpected as NOIDS are known to undergo extensive metabolism.27,28 We then used UPLC-MS/Tof to also screen for 18 potential adamantyl-type NOID metabolites, seven of which were proposed by us, and 11 by Gandhi et al. 28 The goal of this study was to identify any adamantyl-type NOID metabolite(s) with potential as a screening marker. Only one metabolite, 1-adamantylamine (C10H17N), was detected in all five urine samples from the test case. Using our method we failed to detect any of the 11 major metabolites proposed by Gandhi et al. 28 Likewise Gandhi et al. 28 failed to identify our proposed metabolite 1-adamantylamine.
There are several major differences between our metabolite study and that of Gandhi et al. 28 In addition to the extraction method, Gandhi et al. 28 used human hepatocytes incubated in a certified Cerilliant standard of 10 µmol/L AKB-48 at 37℃ for up to 3 h. For our metabolite study we tested random urine from actual patients smoking a legal high herbal blend of different adamantyl-type NOIDS (AKB-48, 5F-AKB-48 and STS-135), any of which could be responsible for yielding the 1-adamantylamine we detected, either through metabolism or as a breakdown of the legal high product during smoking. The 1-adamantylamine may also be present as an artefact of manufacturing, and indeed was detected in Herbal Haze II when re-analysed at high concentration (1 mg/mL). This highlights the advantage of using real patient samples when investigating potential markers for screening in biological samples.
NOIDS are far more potent than cannabis. 4 As demonstrated in Figure 1 in the test case, only about 5% of the cigarette contents are legal high material. Legal high products are sold as not for human consumption and as such do not include instructions for use and this presents a particular risk to new users unfamiliar with the potency of NOIDS, as they are more likely to unintentionally overdose. Often the only information available to clinicians on what a patient may have taken is the packaging, which can be misleading. This uncertainty in identifying what an individual has taken further highlights the need for relevant NPS screening, such as our admanatyl-type NOID marker, to support both patient diagnosis and management.
Our admanatyl-type NOID marker has now been in routine use for over eight months. Overall the positive detection rate was 2.0%. To put this into the context over the same period the positive detection rate for amphetamine, metamphetamine plus ecstasy (MDMA and MDA) was 1.7%. According to the most recent FEWS report the most popular NOIDs currently on sale in UK in 2014 were the adamantyl-type NOIDS. 23
Our adamantyl-type marker can easily be incorporated into a routine urine LC-MS/MS screen with minimal financial implications. This enables us to report results in a clinically relevant time frame of hours rather than days. Whilst our marker is well suited for clinical use the inability to identify the actual parent adamantyl-NOID may make it unsuitable for forensic use.
In developing our adamantyl-type NOID marker we have taken a pragmatic approach and we do not have any data on the positive or false negative rate for example. Clinical details from the patient group under study are notoriously unreliable and herbal legal high products contain a blend of NOIDS. An important part of ongoing validation of all our methods includes feedback from users. For example there is a possibility that patients maybe on legitimate medication that contains an adamanatyl group, though this has not been observed so far. What is significant from our routine screening of 14,436 urine samples is we have yet to receive a single challenge to any of the positive adamantyl-type NOID marker positive results we have reported. Many questions still remain unanswered about our admanatyl-type NOID marker including the detection window in urine, which adamantyl-type NOIDS our marker is specific for, and if it is a metabolite or artefact of manufacturing, and we are continuing to work with our clinicians to help determine these.
In conclusion we have developed and demonstrated the use of a marker for the most common third generation NOIDS currently in use in the UK and used it to look for NOID use in over 13,000 patient urines. This marker can easily be incorporated into routine LC-MS/MS drug screening panel alongside classic drugs of abuse making it ideal for clinical use where accurate and rapid results are needed.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Both authors run a clinical service in the United Kingdom for Toxicology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sandwell & West Birmingham Hospital NHS Trust, Department of Clinical Biochemistry.
Ethical approval
Not applicable.
Guarantor
LF and JB.
Contributorship
LF undertook the research and wrote the first draft. JB edited the manuscript.
