Abstract
We report a case of transient human anti-mouse antibody from a 64-year-old man in a carbohydrate antigen 19-9 immunoassay using an AIA 1800 analyser that generated immune enhancement after surgical resection of recurrent cancer.
Methods
The carbohydrate antigen 19-9 concentration was measured using an AIA 1800 analyser and a UniCel Dxl 800. Size-exclusion high-performance liquid chromatography was carried out on a Superose 12 column to estimate the carbohydrate antigen 19-9 elution profile using an AIA 1800 analyser. To determine whether IgM in the patient contributed to the carbohydrate antigen 19-9 immunoassay, immunoprecipitation was performed. Furthermore, mouse immunoglobulins were added to the patient’s serum to verify that the patient’s IgM reacted with it.
Results
The carbohydrate antigen 19-9 concentration was >400 and 9.5 kU/L using an AIA 1800 analyser and using a UniCel Dxl 800, respectively. In the single carbohydrate antigen 19-9 peak, the molecular weight corresponded to IgM by size-exclusion high-performance liquid chromatography on a Superose 12 column. In the immunoprecipitation reaction and addition of mouse immunoglobulins, there was interference for anti-human IgM and mouse immunoglobulins whose recoveries were 3.2 and 14.2%, respectively. These results indicated that IgM in the patient’s serum interfered with the carbohydrate antigen 19-9 immunoassay using an AIA 1800 analyser.
Conclusion
A novel transient human anti-mouse antibody generated with immune activation in a carbohydrate antigen 19-9 immunoassay using an AIA 1800 analyser was identified in a patient with rectal cancer after surgical resection. These findings demonstrate the importance of monitoring tumour markers in patients after treatment with mouse monoclonal antibody.
Introduction
Carbohydrate antigen 19-9 (CA 19-9) is a digestive cancer-associated antigen discovered in 1979.1,2 In the serum, CA 19-9 exists as a mucin glycoprotein, which has a high molecular weight of 200–1000 kDa and has a sialylated lacto-N-fucopentaose II epitope recognized by 1116 NS 19-9 (Signet Laboratories, Dedham, MA).3,4 CA19-9 has been used as a tumour marker for patients with pancreatic cancer, 4 colorectal cancer,5,6 and gastric cancer.7,8
In some immunoassays, various forms of interference affect the results.9,10 Cole et al. 11 reported false-positive human chorionic gonadotropin test results. Covinsky et al. 12 demonstrated that an IgM lambda antibody to Escherichia coli produces false-positive results in multiple immunometric assays. Moreover, immunoglobulin binds to hormones, macro prolactin,13,14 macro thyroid-stimulating hormone, 15 and macro follicle-stimulating hormone, 16 resulting in incorrect clinical interpretation due to low or zero bioactivity. Therefore, it is important to take into account this possibility when the laboratory results disagree with the clinical course.
Here, we report a transient human anti-mouse antibody (HAMA) in an AIA 1800 CA 19-9 immunoassay detected by size-exclusion high-performance liquid chromatography (HPLC), immunoprecipitation, and the addition of mouse immunoglobulins. To our knowledge, this is the first identification of transient HAMA generated with immune activation by surgical operation in a patient previously treated with mouse monoclonal antibody.
Materials and methods
Patient
In June 2010, a 64-year-old man was diagnosed with rectal cancer with metastasis in the liver. He received palliative surgical resection for rectal cancer in June 2010 and for a metastatic tumour in the liver in November 2010. In June 2011, he received radiation to the metastatic para-aortic lymph node. The patient had followed by treatment with mouse monoclonal antibody, panitumumab for six months since September 2011 and cetuximab for one month since April 2012, and with the multikinase inhibitor regorafenib for 11 months since October 2013. During treatment with regorafenib, the para-aortic lymph node was removed in October 2014. Twenty-two days after the resection, the CA 19-9 concentration using an AIA 1800 analyser was markedly elevated at >400 kU/L, the concentration having been 16 kU/L before the operation. Furthermore, the carcinoembryonic antigen concentrations before and after the excision were 15.9 and 3.8 μg/L, respectively. Based on the disagreement with clinical data, we suspected that the high concentration of CA 19-9 was induced by a non-specific reaction. The patient did not receive blood transfusion during or after the surgery. The patient’s informed consent was obtained.
CA19-9 immunoassay
The CA19-9 concentration was measured by ST AIA-PACK CA19-9 based on a two-site immunoenzymometric assay using the AIA 1800 analyser (TOSOH, Tokyo, Japan) and the Access GI Monitor based on chemiluminescence enzyme immunoassay using a UniCel Dxl 800 (Beckman Coulter, Fullerton, CA). These analyses were performed according to the manufacturer’s instructions. The AIA-PACK CA 19-9 assay contains monoclonal mouse antibody immobilized on a magnetic solid phase and enzyme-labelled monoclonal mouse antibody. In contrast, the Access GI Monitor contains paramagnetic particles coated with polyclonal goat antibiotin antibody, mouse monoclonal-alkaline phosphatase conjugate, and mouse monoclonal-biotin conjugate.
Clinical biochemical examination
IgG, IgA, IgM, and rheumatoid factor (RF), which are immune response markers, were measured on a JCA-BM8030 automated analyser (JEOL, Tokyo, JAPAN).
HAMA titre
The titre of HAMA was calculated by dividing CA 19-9 using an AIA 1800 analyser by the IgM concentration.
Dilution tests
Dilution tests were performed by diluting the patient’s serum with 150 mM NaCl to assess the linearity, and the CA 19-9 concentrations were measured using an AIA 1800 analyser.
HPLC analysis
Size-exclusion HPLC was carried out on a Superose 12 column (10 × 300 mm, GE Healthcare, Buckinghamshire, England) as previously described. 14 Briefly, 200 μL of serum were injected. The elution buffer was 150 mM NaCl and 16 mM phosphate buffer (pH 7.4). The flow rate was 0.5 mL/min and the fraction volume was 0.8 mL. The Superose 12 column was calibrated using a Gel Filtration Calibration Kit with low-molecular-weight and high-molecular-weight proteins (GE Healthcare). Protein and CA19-9 concentrations were monitored at 280 nm using the Akta Explorer System (GE Healthcare) and an AIA 1800 analyser, respectively.
Immunoprecipitation and the addition of mouse immunoglobulins
Immunoprecipitation and the addition of mouse immunoglobulins were performed using polyclonal anti-human IgM14,17 and mouse immunoglobulins, using ST AIA-PACK Myoglobin (TOSHO), respectively. One hundred microliters of antiserum or mouse antibody was mixed with 100 μL of the patient’s serum and incubated at 4℃ overnight in the immunoprecipitation reaction. After centrifugation at 12,000 g for 5 min, the CA19-9 concentration of the supernatant was measured using an AIA 1800 analyser.
Results
CA19-9, IgG, IgA, IgM, and RF concentrations, and HAMA titre
The time courses of CA 19-9 and IgM concentrations and HAMA titre are shown in Figure 1. At the first collection of serum after lymph node excision on 24 October 2014, the CA19-9 concentration was >400 kU/L (measurement range 1–400 kU/L without dilution) using an AIA 1800 analyser. In contrast, the 19-9 concentration using a UniCel Dxl 800 was 9.5 kU/L (measurement range 0.8–2000 kU/L without dilution). The CA 19-9 concentration using an AIA 1800 analyser gradually decreased to less than the normal reference range (<37 kU/L) on 2 December 2014. The IgG, IgA, IgM, and RF concentrations remained unchanged (Supplemental Table 1). Moreover, the titre was gradually decreased in accordance with the reduction of the CA 19-9 concentration (Figure 1). On the other hand, the CA 19-9 concentration using a UniCel Dxl 800 remained within the reference range (<35 kU/L) through 24 October 2014 to 2 December 2014.
Time course of CA 19-9, IgM concentrations, and HAMA titre. The solid lines indicate CA 19-9 concentration (circle) and IgM concentration (triangle), respectively. The dot line indicates HAMA titre (square). The titre of HAMA was calculated by dividing CA 19-9 by IgM concentration. The arrow indicated the period of prescription for regorafenib.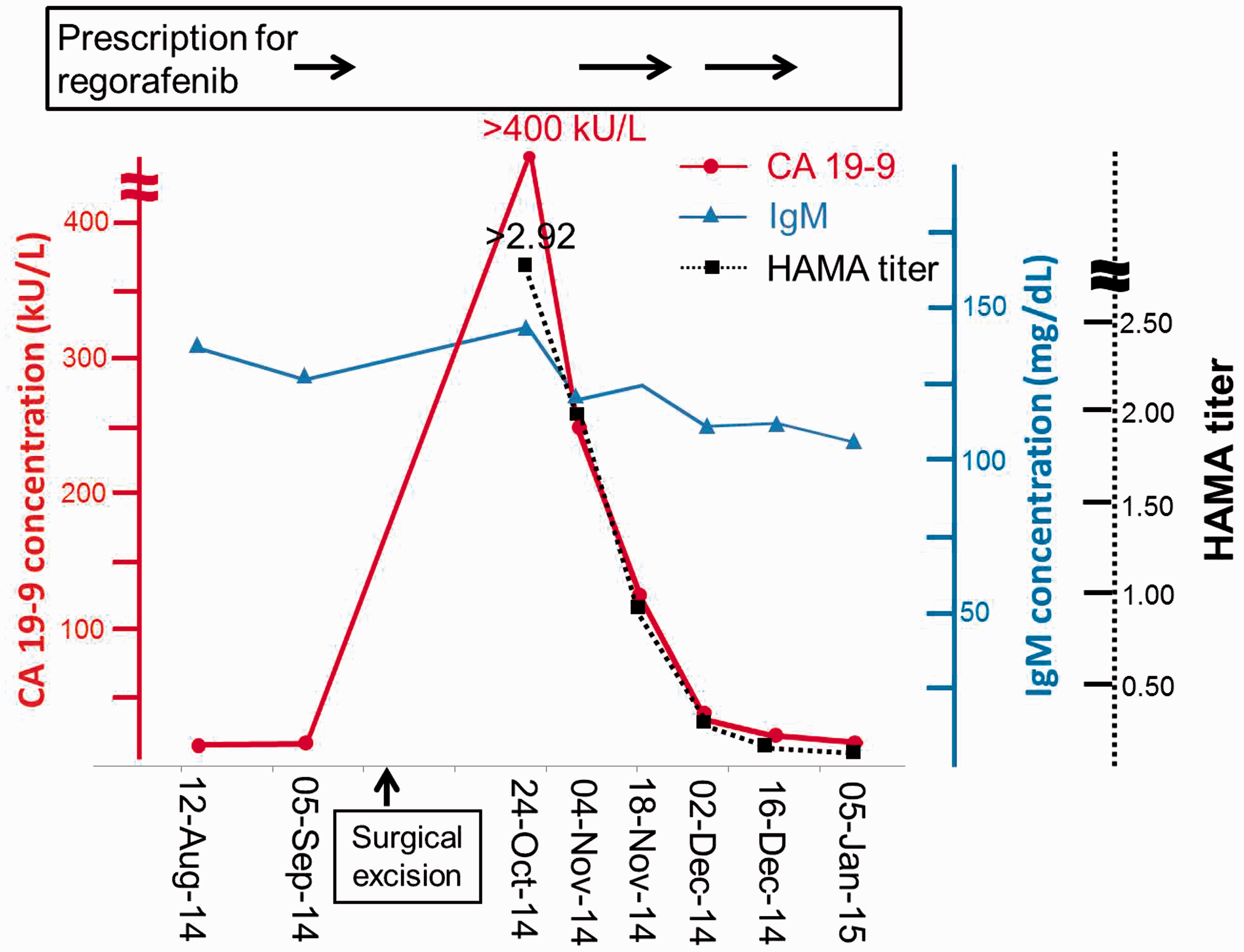
Dilution tests
Dilution tests were performed using 150 mM NaCl. There was no linearity of CA 19-9 (Supplemental Figure 1).
Characteristics of HPLC analysis
The patient’s serum collected on 24 October 2014 was subjected to HPLC using a Superose 12 column. A single CA 19-9 peak was noted, of which the molecular weight corresponded to IgM (900 kDa) (Supplemental Figure 2).
Immunoprecipitation and addition of mouse immunoglobulins
Immunoprecipitation and addition of mouse immunoglobulins.
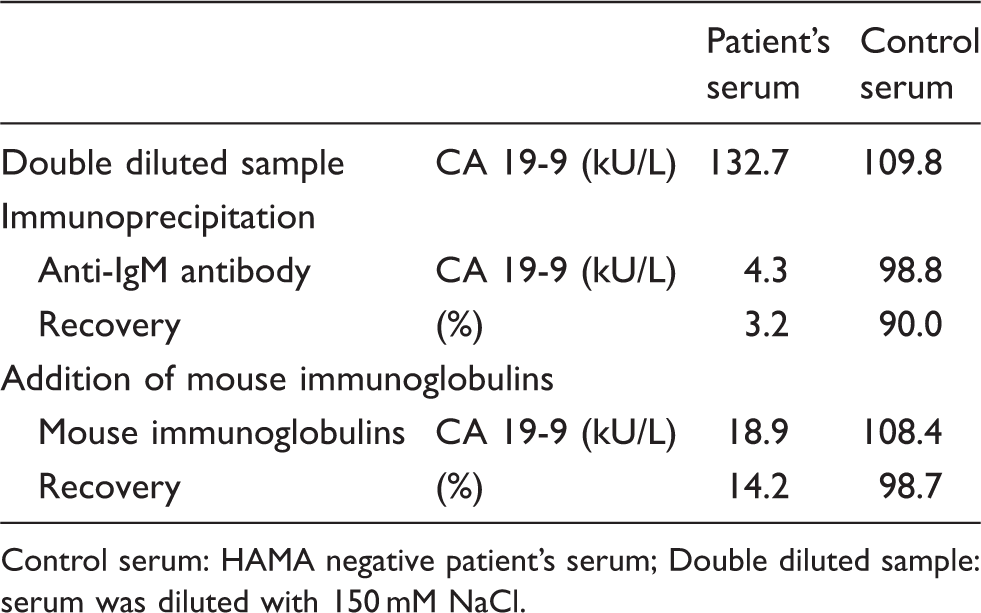
Control serum: HAMA negative patient’s serum; Double diluted sample: serum was diluted with 150 mM NaCl.
Discussion
Clinical investigators have reported various forms of interference in CA 19-9 immunoassays.18–20 Berth et al. 20 demonstrated RF interference in the immunoassay. Another report showed that a heterophilic antibody results in falsely elevated CA 19-9 concentrations. 19 Furthermore, Monaghan et al. 18 reported low-molecular-weight interference in an apparently healthy male.
In order to identify the interference, we first performed size-exclusion HPLC using a Superose 12 column. A single CA 19-9 peak corresponding to IgM was noted. From this result we suspected interference with IgM in the CA 19-9 immunoassay using an AIA 1800 analyser.
We then performed immunoprecipitation with anti-IgM antibody and the addition of mouse immunoglobulins to the patient’s serum. The CA 19-9 concentration using an AIA 1800 analyser was dramatically decreased (Table 1). These results indicated that the IgM in the patient’s serum reacted with the mouse immunoglobulins. In other words, the patient has an IgM type HAMA.
Heterothallic antibodies, most common with HAMA, are attributable to blood transfusion, vaccination, animal antibody treatment, and other unknown mechanisms. 10 In this study the patient did not receive a blood transfusion during these periods. In contrast, the CA 19-9 concentration reached over 400 kU/L using an AIA 1800 analyser at 22 days after the resection. Since that point, the CA 19-9 concentration gradually decreased (Figure 1).
The patient had been treated with mouse antibodies, panitumumab and cetuximab. Panitumumab is a fully human monoclonal antibody directed against the epidermal growth factor receptor (EGFR). 21 In contrast, cetuximab is a half-human, half-murine antibody and is also known for its EGFR blocking effects. 21 Line et al. 22 reported that 16.7% of healthy adults injected murine monoclonal antibody produced HAMA. Moreover, Oei et al. 23 demonstrated that HAMA in CA-125 is produced during monitoring of ovarian cancer patients treated with murine monoclonal antibody. Further, Azinovic et al. 24 reported that 35% of patients with B-cell malignancies developed HAMA following radioimmunotherapy. Finally, Ott et al. 25 investigated whether there was a correlation between HAMA development and clinical outcome in patients treated with the trifunctional antibody catumaxomab, composed of mouse and rat immunoglobulin. The report indicated that HAMA production after catumaxomab administration may be associated with beneficial humoral effects and prolonged survival. We did not have a chance to investigate whether HAMA in the patient developed against mouse antibodies, panitumumab and cetuximab, because the patient had received the antibody treatments at another hospital, and serum samples had not been collected during these treatment periods. Although HAMA development in this case against panitumumab and cetuximab was not clear, it may have been a beneficial effect of the metastatic tumour excision.
In the view of the therapeutic course, the surgery could have enhanced the generation of a pre-existing specific antibody, although treatment with mouse monoclonal antibody had been finished for more than 12 months before HAMA was detected. Abe et al. 26 reported that the presence of an IgM-like substance after one month of ovarian cyst surgery was associated with a significant increase of plasma CA 125 concentration. In that report, the patient’s serum did not react with Scantibodies Heterophilic Blocking Reagent, indicating that the IgM-like substance was not HAMA. In this study, there were interferences with anti-human IgM and mouse immunoglobulins, indicating the production of IgM-type HAMA. Moreover, the patient was previously treated with mouse monoclonal antibody.
To the best of our knowledge, this is the first reported case of a patient previously treated with mouse monoclonal antibody with identified transient HAMA generated with immune activation by surgical operation.
In conclusion, we report for the first time a transient HAMA generated with immune enhancement in a CA 19-9 immunoassay using an AIA 1800 analyser detected by size-exclusion HPLC, immunoprecipitation, and the addition of mouse immunoglobulin. This report demonstrates the importance of monitoring tumour markers in patients after treatment with mouse monoclonal antibody. We urge clinical investigators to consider the possibility of HAMA when the laboratory data are not consistent with the clinical course.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This study was supported by the Running Funds from the Division of Laboratory and Transfusion Medicine of Hokkaido University Hospital.
Ethical approval
Written consent has been obtained prior to use of data from patient’s medical record.
Guarantor
CS.
Contributorship
KN, KY, and HS designed the study. TM advised HPLC analysis. KK and CS performed the medical consultations. KN researched literature and wrote the manuscript. CS corrected the manuscript. All authors reviewed and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
