Abstract
Background
We present a 5-year review of our UK service for plasma ethylene glycol and diethylene glycol determination in cases of acute poisoning.
Methods
Ethylene glycol and diethylene glycol have been measured on all samples received for screening for toxicity by gas chromatography-flame ionization detection over a five-year period. A detailed audit of the results has been undertaken.
Results
In this period, we received 811 requests, 56% were for first-time screening and 44% repeat analysis where a positive sample has already been received. Of the first-time screen samples, 33.5% screened positive for glycol poisoning. The mean positive ethylene glycol concentration was 1204 mg/L (range 31 to 8666 mg/L). Diethylene glycol was present in 14% of ethylene glycol positive samples but never found alone.
Conclusions
The data presented here suggest it is not essential to measure diethylene glycol since its inclusion is rarely likely to change patient management.
Introduction
Ethylene glycol (EG) can be found in a wide range of different domestic products such as antifreeze, preservative, rust remover, disinfectant and general purpose cleaner. Another glycol diethylene glycol (DEG) is sometimes used as a substitute for EG. 1 Both EG and DEG are highly toxic and poisoning is associated with severe morbidity and mortality.
The metabolites of both EG and DEG are responsible for their toxicity. EG and DEG are metabolized in the liver by alcohol and aldehyde dehydrogenase. EG yields the toxic metabolites glycoaldehyde, glycolic acid, glycoxylic acid and oxalic acid.2,3 DEG is metabolized to 2-hydroxyethoxyacetic acid (2-HEAA), diglycolic acid (DGA) and 2-oxo-1,4-dioxan.4–8 DEG is not converted to EG as the ester bond is resistant to cleavage. 9 Time to presentation is an important factor outcome and, depending on the concentration of the ingested product, accidental or intentional ingestion of as little as 30 to 100 mL of fluid containing EG and or DEG may be fatal.2–4
The clinical presentation of both EG and DEG poisoning has been generalized to three stages.10–12 Stage 1 (1–12 h) sees gastrointestinal symptoms and or central nervous system (CNS) depression, stage 2 (12–24 h) cardiopulmonary effects and stage 3 (24–72 h) renal failure. In the first few hours after ingestion, patients may remain asymptomatic with a clinical presentation that resembles ethanol intoxication. Both osmolar and anion gaps are transient features of glycol poisoning and depending on time of presentation may not be immediately apparent. 11 This is especially the case for DEG poisoning when development of metabolic acidosis and anion gap typically do not present until stage 2.12–15 Later features of both EG and DEG poisoning are renal failure but unlike EG, oxalate crystalluria is not usually seen with DEG.5,14,16 Treatment of glycol poisoning is primarily aimed at slowing or preventing the production of toxic metabolites by inhibiting dehydrogenase enzyme activity with the intravenous administration of ethanol or fomepizole (4-methylpyrazole).17–19
With patient history not always forthcoming and osmotic and anion gap non-specific clinical indications of EG and DEG poisoning, diagnosis can only be confirmed by the detection of glycols in blood. 11 Determination of EG and DEG blood concentration can also help clinicians in determining the best course of treatment, its effectiveness in reducing glycol levels and when to stop further treatment.
Methods for detection of glycols in blood include gas chromatography coupled to mass spectrometry (GC-MS)20–22 or flame ionization detection (GC-FID)23–26 and headspace gas chromatography (HS-GC). 27 Recently, improved automated enzymatic methods for EG have also been described.28,29 Of these methods, GC-FID offers a reliable testing approach which is also fast and suitable for 24 h use in laboratories with the correct equipment and skills. However, glycols can bind strongly to GC capillary columns, potentially contaminating the inlet/liner interface which can cause carryover and peak tailing. Derivatization methods are less affected by this23–24; however, incomplete derivatization may result in inaccurate results and some methods will not detect all glycols, for example phenyl boric acid does not form boric esters with DEG. 25 Typically, direct GC-FID methods involve fewer operator steps making them better suited to urgent analysis.
In response to concerns from clinicians from our own specialist Poison Unit that cases of DEG poisoning in the UK were being missed, we extended our EG methods in 2009 and launched a new GC-FID direct method for the simultaneous detection of the toxic glycols EG and DEG. Here, we present a five-year analysis of this service from 2009 to 2013 to determine the frequency of EG and DEG poisoning. We have compared our experience with audit data published by the National Poisons Information Service (NPIS) regarding glycol poisoning in UK hospitals.30–33
Methods
EG and DEG concentrations were determined using a direct method based on that of Williams et al.,
25
which was optimized for routine analysis. The extraction consists of protein precipitation with 500 μL of serum mixed with 750 μL of ethanol (containing 2,3-butanediol 300 mg/L as internal standard) and spun for 10 s. The mixture is then centrifuged to produce a clear supernatant of which 1 μL is injected. An Agilent Technologies 6890 N Gas Chromatograph (Berkshire, UK) with flame ionization detector (FID) was used with Agilent GC Chemstation Rev. A. 09. 01 [1206] software and a Restek Rtx-200 capillary column Crossbond trifluoropropylmethylpolysiloxane 30 m × 0.53 mm i.d. 3 μm film thickness (Buckinghamshire, UK). The total run time is 4.5 min per sample. For both EG and DEG the method is linear to 1000 mg/L. At EG and DEG concentrations <100 mg/L, the intra-precision for the method is <5% and interassay precision <7% (
Samples
As some serum separator tubes may contain glycols to prevent confusion from potentially interfering chromatography peaks, plasma samples only are accepted for analysis (EDTA, lithium heparin or fluoride oxalate).
Statistics and calculations
Data were gathered from the laboratory computer records of all requests made for glycol screening between 2009 and 2013, Telepath version 4 (iSoft Laboratory Systems Ltd).
Results
Requests
The total number of requests received for EG and DEG analysis over the five-year audit period (2009 to 2013) was 811 from 93 different UK locations. During the period under study other 24-h services for urgent EG analysis closed. The area from which samples were received widened, from predominantly central England to take in the whole of the England from the South Coast to the Scottish borders. Of these requests, 56% (451) were for first-time screening and 44% (360) repeat analysis (monitoring) following an initial positive sample. Ages ranged from <1 year to 89 years (mean 41 years). Using the Mann-Whitney test, the median age of males (43 years) was significantly higher ( Summary of total requests for glycol analysis received from 2009 to 2013.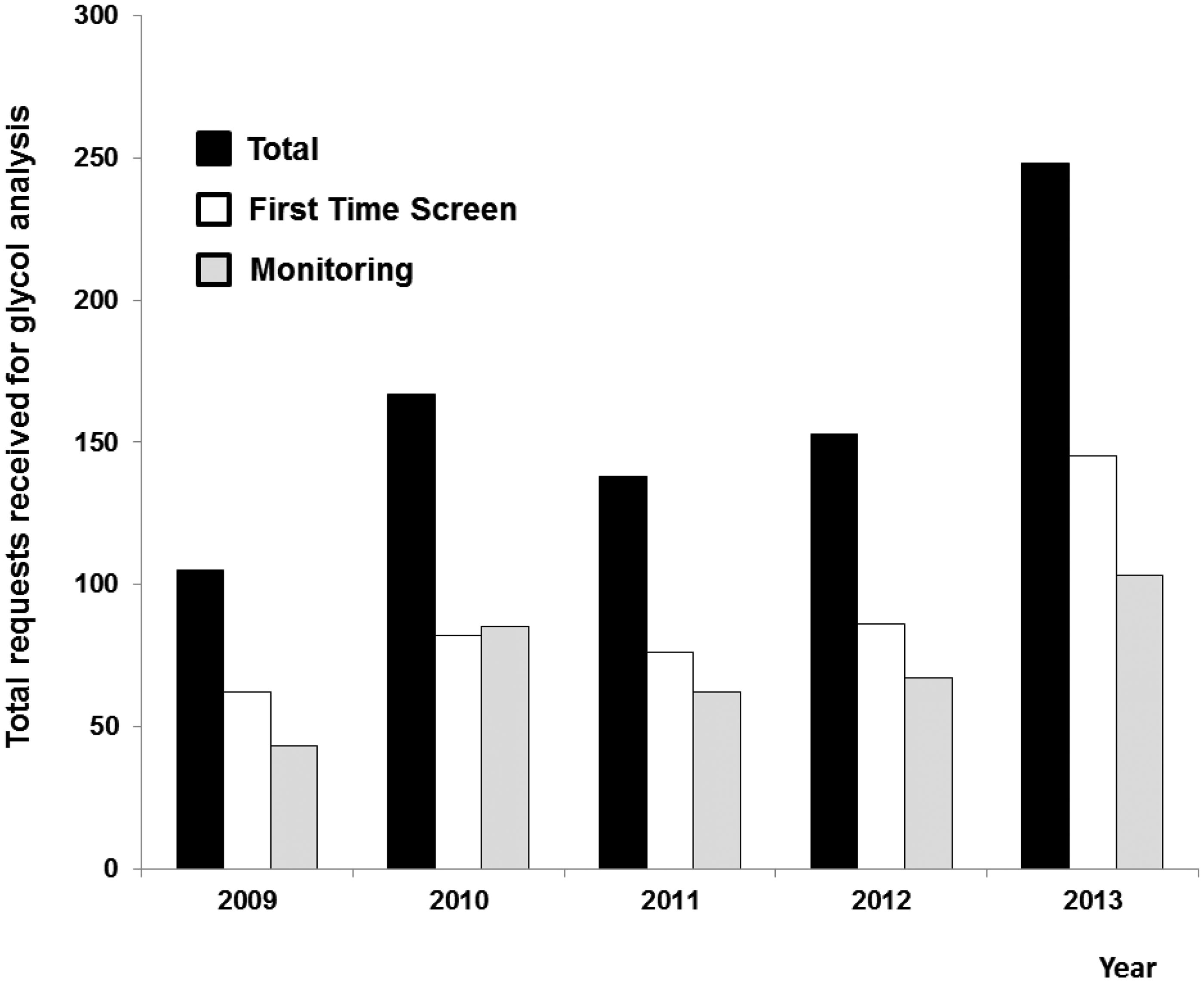
Screening
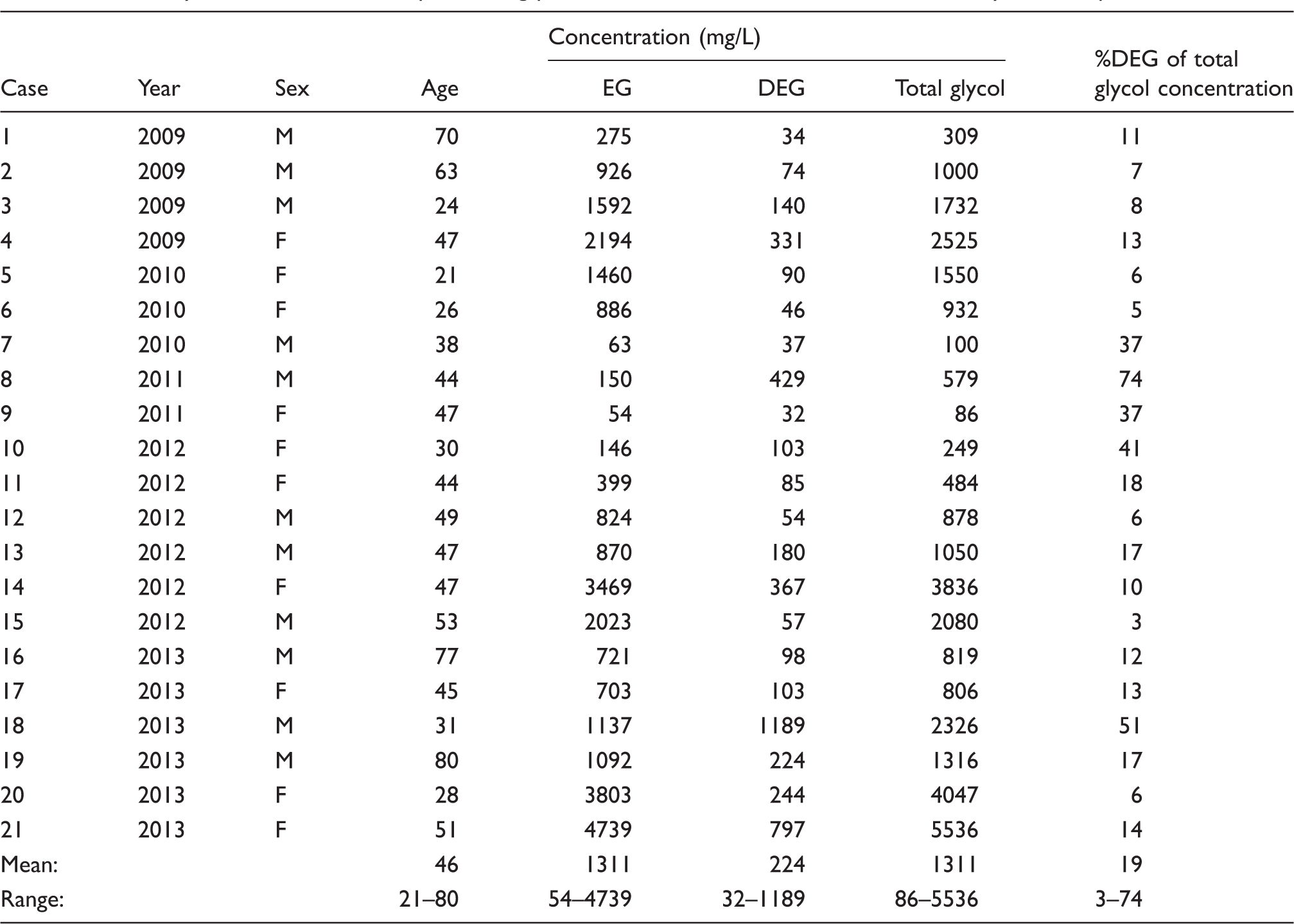
Summary of 21 first screen samples testing positive for both EG and DEG within the five-year audit period.
The number of first-time screening requests testing positive for EG by year and month (total of five years requests) are presented in Figure 2.
Annual and monthly trends in first-time screening requests testing positive for EG.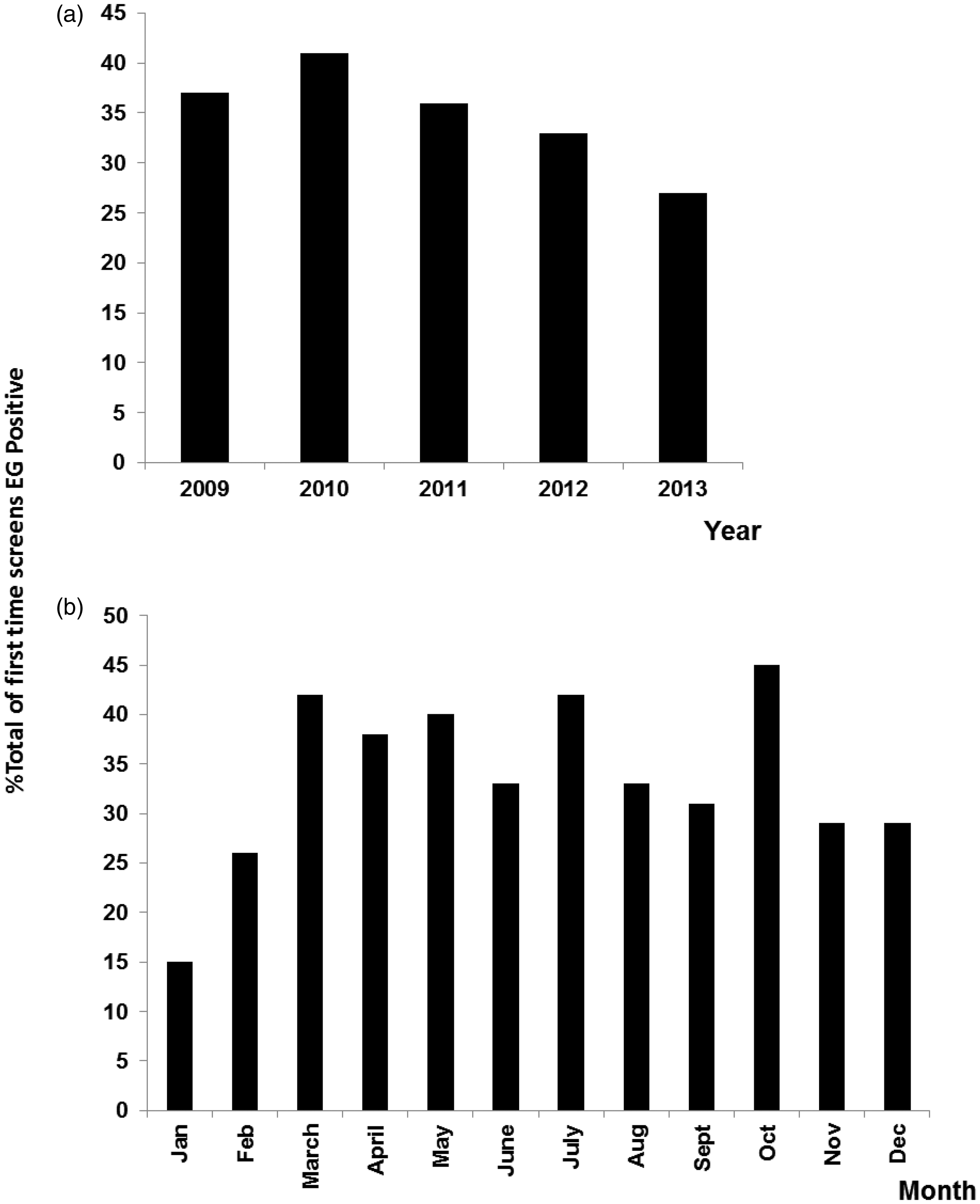
According to NPIS data for 2010–2011 and 2011–2012, a total of 1315 enquiries were made regarding glycol poisoning, 548 of which originated from hospital sources.31,32 Some 60% (329) of these cases were classified as systemic exposure and 66% (216) also reported antidote treatment was given.
No other glycols such as propylene glycol were detected in the total 811 samples tested. In many cases where the glycol screen was negative, a further request for methanol screening by GC-headspace was added. Over the five-year audit period, none of these requests were found to be methanol positive.
Monitoring
Further requests for monitoring were received for 76% (114) of the first time screens that tested positive (mean EG concentration 1204 mg/L, range 31 to 8666 mg/L). Typically, for each case of glycol poisoning three (range 2 to 11) further monitoring requests were received over a period of 1 to 11 days. When DIEG was detected the mean first screen value was 164 mg/L, range 32 to 1189 mg/L.
Repeat patients
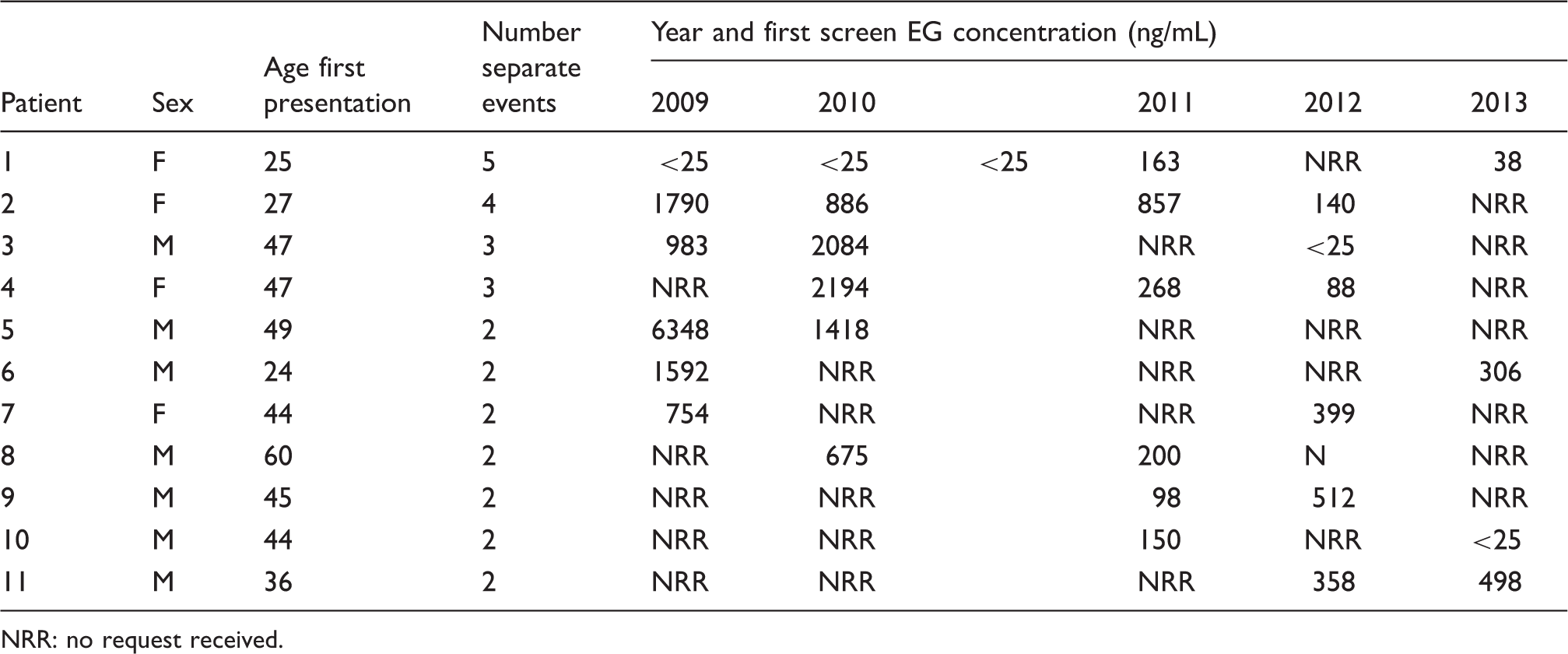
Summary of 11 patients where multiple requests for screening for glycol poisoning were received over the five-year audit period.
NRR: no request received.
Discussion
Over the five years covered by this audit we received just under 500 requests for first-time assessment of patients for suspected glycol poisoning of which 33.5% tested positive. Of these 151 positive cases, 64% of the patients were male and 36% female.
The general recommendation to clinicians is that where there is a high suspicion of glycol poisoning, treatment should be started immediately to prevent morbidity and mortality. 11 High suspicion of ethylene glycol overdose often includes no clinical history, with an unexpected raised osmolar gap or just metabolic acidosis, both non-specific markers for glycol poisoning. This helps to explain why only one in three of the samples we received for first-time screening were actually positive for glycol poisoning.
Indications for starting antidote treatment for glycol poisoning include a blood glycol concentration ≥200 mg/L with the addition of dialysis at concentrations ≥500 mg/L.7,17,19 Of the 151 positive cases of glycol poisoning detected, 66% had a total glycol concentration ≥200 mg/L and 50% ≥ 500 mg/L. The highest glycol concentration we measured was for EG at 8700 mg/L in a 49-year-old male. Follow-up has revealed this patient was successfully treated and survived. For the majority (76%) of the 151 cases of glycol poisoning, we also received repeat samples for monitoring treatment. The number of repeat samples sent varied between cases from 2 to 11, and were received over a period of 1 to 11 days. Generally, repeat requests were sent for each day of treatment. From our mean data, we can therefore approximate that for each episode of glycol poisoning antidote treatment was typically given over three days.
From our audit data, we have also identified a small group of individuals that were repeatedly tested for suspected glycol poisoning. Typically, these individuals present once a year and in the majority of cases glycol poisoning was detected (83%). With multiple requests for first-time screening and monitoring these repeat self-harming individuals were responsible for a significant amount of our workload, almost 14% (111 separate requests).
Over the five years, our annual workload increased more than two-fold, from 105 total requests in 2009 to 248 in 2013. This increase appears to be related to other services for ethylene glycol closing or no longer operating out of hours and there is no evidence in our results that actual cases of poisoning have increased over this time. For example, out of hours services in Liverpool and Cardiff ceased during the time under study. Glycol poisoning is often perceived as a more likely occurrence in autumn and winter months; however, examination of our audit data revealed no correlation between the detection of glycol poisoning and month of the year (see Figure 2).
Our study is the first in the UK to include data on the frequency of DEG poisoning. As part of the validation of our method in 2010, we analysed five antifreeze and nine screen wash products on sale in the UK for glycols. While all 14 products tested positive for EG, two were also found to contain DEG proving its availability in the UK. In the current audit, DEG was detected in approximately 14% of EG positive samples, but was never detected on its own which dispels the suggestion that by only measuring EG that some case of glycol poisoning would be missed. Typically, when present DEG increased the total toxic glycol load by an average of 19%, but in two patients DEG increased the total glycol concentration by over 50% (see Table 1).
Few UK services other than our own are capable of screening for any other glycols besides EG. The data presented here suggest it is not essential to measure DEG since its inclusion is rarely likely to change patient management. We have only ever detected DEG when EG is present. While there is a risk that by not screening for DEG the total glycol load may be underestimated, we saw no evidence that clearance of DEG in patients receiving treatment was any different to how EG responded.
Although cases of EG and DEG poisoning are relatively uncommon, due to the difficulty in making an accurate diagnosis and complexity in patient management, it remains one of the most popular referrals to the National Poison’s Information Service (NPIS) clinical toxicology consultants.30–33 While it is not stated how many of these cases underwent screening, during the same period we received 168 first-time requests for EG and DEG analysis.
Accurate diagnosis of glycol poisoning, with results available in a timely manner can equate to considerable cost savings for patient treatment. With blood screening the only way to make a definitive diagnosis of glycol poisoning, there have been repeated calls to increase availability of testing.7,10,11,30–33 However, the cost of providing such highly specialized, low throughput, services at a time when laboratories are under considerable pressure to make savings is putting these services at risk with those in the North West, London and South Wales either closing or not operating out of normal working hours. It would appear that a number of cases of overdose are not having any levels measured and this is a concern as it suggests patients may not be correctly diagnosed and treated. Local laboratories often make decisions on whether to refer patient samples to us and our experience is that there is no standardization in this decision process. If vital specialist services such as this are to be preserved, clearly there needs to be a review how such resources are funded.
Footnotes
Acknowledgements
The authors thank the staff of the Clinical Biochemistry Department at SWBH NHS Trust.
Declaration of conflicting interests
Both authors run a service in the United Kingdom for ethylene and diethylene glycol screening in blood.
Funding
Sandwell & West Birmingham Hospital NHS Trust, Department of Clinical Biochemistry funded this study.
Ethical approval
Not applicable.
Guarantor
LF.
Contributorship
LF designed the audit, analysed the data and wrote the first draft manuscript. JB edited and approved the final version of the manuscript.
