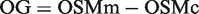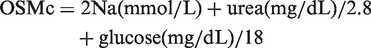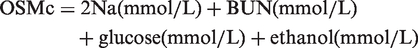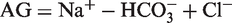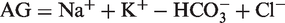Abstract
Ethylene glycol poisoning, while uncommon, is clinically significant due to the associated risk of severe morbidity or lethality and it continues to occur in many countries around the world. The clinical presentation of ethylene glycol toxicity, while classically described in three phases, varies widely and when combined with the range of differential diagnoses that must be considered makes diagnosis challenging. Early and accurate detection is important in these patients, however, as there is a need to start antidotal treatment early to prevent serious harm. In this article, we will review the literature and provide guidance regarding the diagnosis of ethylene glycol poisoning. While gas chromatography is the gold standard, the usefulness of this test is hampered by delays in access due to availability. Consequently, there are several surrogate markers that can give an indication of ethylene glycol exposure but these must be interpreted with caution and within the clinical context. An in-depth review of these tests, particularly the detection of a raised osmolar gap or an raised anion gap acidosis, will form the main focus of this article.
Introduction
Ethylene glycol, also called 1,2-ethanediol, is a viscous liquid with a sweet taste.1,2 It is nonvolatile at ambient temperatures and has a molecular weight of 62.07 g/mol. Ethylene glycol itself is a colourless fluid although the solutions it is commonly found in are often coloured in an attempt to prevent accidental ingestion. Due to the solutes depression of the freezing point of water, ethylene glycol is most commonly used as a de-icing solution or as antifreeze in coolant systems. Ethylene glycol is found in brake and hydraulic fluid, is commonly used as a solvent and is also found in household cleaners such as window cleaner.
Ethylene glycol poisoning, while uncommon, is clinically significant due to the associated risk of severe morbidity and potential lethality and it continues to occur in many countries around the world. In the United States in 2010, there were 5725 cases of ethylene glycol poisoning reported to poison control centres, seven of which were fatal. 3 In Victoria, Australia, there were 18 ethylene glycol poisonings in 2011. 4 In the United Kingdom in 2010–2011, there were 608 enquires to the National Poisons Information Service (NPIS) regarding 488 individual exposures to toxic alcohols and glycols. The most common agent involved in these exposures was ethylene glycol. 5
Many reported poisoning cases are accidental ingestions in children.4,6 A breakdown of UK data collected from the NPIS in 2010–2011 reveals that 18% of calls were for children younger than five years. 5 Intentional ingestion as a deliberate self-poisoning in adults was the most common mode of poisoning, with 67% of NPIS calls related to intentional adult overdose. Rarely, there are reported cases of homicidal intent 7 and there have been previous epidemics of ethylene glycol toxicity reported, for example due to water contamination or recreational intent.8–10
Pharmacokinetics
Gastrointestinal absorption of ethylene glycol is rapid, while dermal and pulmonary absorption is poor. 1,11 Onset of the symptoms of intoxication along with elevated serum concentrations have been documented to occur 20–30 min following ingestion12–14 whilst a peak in ethylene glycol concentration usually occurs after 1–4 h. 1,11 Ethylene glycol is not protein bound and is highly water soluble, meaning that it distributes evenly throughout body tissues with a volume of distribution of 0.5–0.8 L/kg. 11
Ethylene glycol undergoes metabolism in the liver by a series of oxidative steps starting with the enzyme alcohol dehydrogenase (ADH) (Figure 1). In a process using nicotinamide adenine dinucleotide (NAD), ethylene glycol is converted to glycolaldehyde and then rapidly on to glycolic acid.
1
Subsequent slow conversion to glyoxylic acid results in the accumulation of glycolic acid, the main metabolite responsible for the development of metabolic acidosis. A small amount of glyoxylic acid is converted to oxalate which then precipitates as calcium oxalate crystals. The elimination half life of ethylene glycol is 3–8 h.15–17 Hepatic metabolism is responsible for 80% of the elimination of ethylene glycol, while the majority of the remainder is through renal excretion of unchanged ethylene glycol.
Major pathway in the metabolism of ethylene glycol. Adapted from Brent.
2
Clinical presentation and mode of toxicity
The clinical presentation of ethylene glycol toxicity is classically described in three phases 18 with the most serious toxic effects developing from the metabolites of ethylene glycol, not the parent compound itself.
Phase 1 (Neurological): this is similar to ethanol intoxication, lasting from 0.5 to 12 h and involving slurred speech, ataxia, nausea and vomiting followed by increasing somnolence and coma. Ethylene glycol is responsible for the early central nervous system (CNS) effects of inebriation, while the drowsiness and encephalopathy is related to the accumulation of organic acids and associated cerebral oedema.1,2,19,20 Seizures and myoclonic jerks have been documented and maybe due to a direct toxic effect of glycolic and glyoxylic acid in addition to the hypocalcaemia associated with calcium oxalate crystal precipitation.1,2,21–23
An anion gap acidosis is created primarily as a result of glycolic acid accumulation and also as a result of lactic acid production. Lactic acid is produced in increased quantities due to impairment of the citric acid cycle secondary to NAD depletion. In addition to this, high concentrations of aldehydes and acid metabolites further inhibit the process of oxidative phosphorylation.24–26
Phase 2 (Cardiopulmonary): this occurs between 12 and 36 h after ingestion. Patients exhibit tachycardia, hypertension followed by hypotension, tachypnoea, Kussmaul’s respirations, congestive heart failure and pulmonary oedema. Acidaemia impacts negatively on cardiac contractility and contributes to hypotension and myocardial compromise. Pulmonary oedema may be secondary to cardiac failure, or in the absence of cardiac failure, it is thought to be due to direct toxicity and development of an acute respiratory distress syndrome-type picture. 27 Calcium oxalate crystal deposition in myocardium has been demonstrated on autopsy, but the relevance of this is unclear.28,29
Phase 3 (Renal): this occurs 24–72 h following ingestion and involves progressive renal injury from tubular necrosis with haematuria, flank pain and oligo-anuria. Calcium oxalate crystals precipitate in the renal tubular lumen which leads to blockage and loss of glomerular filtration. 30 In addition to this, monohydrate crystals accumulate in the proximal tubular cells and have been demonstrated to exert direct cellular toxicity. 31
In practice, the presentation of ethylene glycol toxicity can be nonspecific, especially when patients are not forthcoming with a history of deliberate or accidental ingestion. Patients may present at any time following ingestion and it is not uncommon for features from all three phases to overlap. 2 In the early phase, patients with altered mental status maybe mistaken for simple ethanol intoxication, and indeed, this may be a concomitant issue. Without the characteristic smell of ethanol, slurred speech and drowsiness may be confused for an acute neurological event.
Later with the development of tachypnoea and tachycardia, sepsis or shock should be considered and ruled out. In some cases, metabolic acidosis in a patient with little background history conjures up a wide number of causes which need to be methodically eliminated, ranging from toxicological causes to renal failure, diabetic ketoacidosis and a range of possible causes of a lactic acidosis. Consequently, the differential diagnosis is wide. In this article, we will review the literature and provide guidance regarding the diagnosis of ethylene glycol poisoning.
Diagnosis
Early and accurate diagnosis is important in the management of patients presenting with potential ethylene glycol toxicity to determine the need to start timely treatment with antidotes such as fomepizole or ethanol. In this section, we will discuss the evidence regarding the diagnostic process for ethylene glycol toxicity. Specifically, we will review the role of each of the following in making a diagnosis of ethylene glycol toxicity: the history, direct quantitative testing for ethylene glycol itself and indirect testing including measurement of the osmolar gap, pH and anion gap acidosis, lactic acidosis, glycolic acid, hypocalcaemia, urinary fluorescence and oxalate crystal detection and colorimetry.
History
Some cases of ethylene glycol toxicity present with a definite history of ingestion, for example when caregivers witness children drink from a container, when adults present with unintentional ingestion or in cases where adults freely volunteer the information. In other cases, the history may not be forthcoming, either because the patient is unwilling to volunteer information or because their consciousness is altered and they are unable to. In cases with a positive history of ingestion, the estimated volume of ingestion and the concentration of the fluid may be helpful in estimating the ingested dose. However, the accuracy of such estimations and how it relates to serum concentrations is unclear. Work has been done to establish the average volume of a ‘swallow’ or ‘mouthful’ and also to establish whether people can estimate the volume of fluid in containers. The estimated volume of a mouthful varies with age and gender and appears to have wide confidence intervals. For example, children aged 18–66 months have an estimated swallow volume of 5–10 mL with a range of 1–29 mL. 32 In a study of 152 doctors and nurses, there was a considerable variation in the accuracy of estimation of visually estimated fluid volumes with some subjects over-reporting by up to 700%. 33 While calculations for estimating the peak ethylene glycol serum concentration from dose have been published, their accuracy has not been validated. 14
Also of relevance from the patient history, is whether there has been any co-ingestion. Concurrent ingestion of ethanol may delay symptoms and signs and the interpretation of tests such as the osmolar gap is also influenced by the presence of ethanol.
Direct quantitative testing
Gold standard: gas chromatography
Gas chromatography with flame ionization detection is the method of choice for the quantitative measurement of ethylene glycol in blood.11,34 Gas chromatography is a specialized technique which is often only offered by centralized laboratories due to the costs involved in maintenance and validation of an infrequently performed assay. The NPIS of the United Kingdom and the Association of Clinical Biochemists recommend that while this is a specialized and infrequently needed test, urgent measurement of ethylene glycol concentrations are needed for the optimal management and advises a turn around time of less than 4 h. 35 The limited availability of this test means that blood samples often need to be transported for off-site analysis. Delay in diagnosis and delay in out-of-hours screening for toxic alcohols can lead to delays in initiating treatment. This can prevent treatment being initiated within the recommended 6 h as described by current American Academy of Clinical Toxicology practice guidelines.1,36 Therefore, clinicians need to consider other indirect tests, as discussed below, to help them make rapid decisions about antidote treatment.
It should be noted that despite the accuracy of gas chromatography, there is poor correlation between serum ethylene glycol concentrations and clinical symptoms. Even absence of ethylene glycol concentrations on gas chromatography should be interpreted with caution, particularly in a patient with a metabolic acidosis, as there have been documented cases of late presentations of ethylene glycol poisoning with no ethylene glycol detected. 36 The explanation for toxicity with low or undetectable serum ethylene glycol is that the parent compound may be completely metabolized by the time the patient presents, leaving the clinician with the problem of an undifferentiated metabolic acidosis. These situations are uncommon however and since the opportunity for antidote therapy has already passed, treatment will likely be unchanged with the patient needing haemodialysis. 36
Enzymatic assay
Enzymatic assays exist for the detection of ethylene glycol and are currently in use with in veterinary medicine. 37 Such assays have been available for 30 years and have been the subject of continued experiments to try to improve analytical accuracy. 38 The main difficulty until recently was poor specificity and consequent false-positive results in the presence of other alcohols such as propylene glycol, 1,3-butanediol, 2,3-butanediol, 1-octanol and 1,3-propanediol. 37 Recent work by Juenke et al. 39 has experimented with a modified version of the existing veterinary assay in human plasma samples. The authors report reduced false positives despite the presence of interfering substances. Commentators have noted that this assay would provide advantages over gas chromatography in terms of reduced running costs making it potentially more widely available and a reduction in turn around times as results can be made available as quickly as 30 min. 40 As yet this technique does not appear to have been widely embraced or validated in human clinical trials, so cannot be fully endorsed; however, it holds promise for improving the efficiency and simplicity of the diagnostic process.
Indirect testing
When patients present with possible ethylene glycol toxicity and the gold standard gas chromatography ethylene glycol assay is not available or there is a delay in results, other tests are necessary in order for clinicians to make early decisions about treatment and disposition. In this situation, there are a number of indirect tests which can help to make a presumptive diagnosis or at least give an indication of the likelihood of toxicity. These tests will now be further elaborated on.
Osmolar gap
Ethylene glycol is an osmotically active particle and the osmolar gap has been widely used as a surrogate measure to help detect the presence of unmetabolized ethylene glycol.
41
The osmolar gap is the numerical difference between the measured osmolality (mOsmol/kg) of a blood sample and an estimated osmolarity (mOsmol/L) which is calculated using the results from routinely measured osmotically active particles such as sodium, urea and glucose. If the osmolar gap is larger than expected, this is an indication that there may be additional osmotically active particles in a patient’s blood, of which ethylene glycol is a possible cause. With a molecular weight of 62.07, the relative contribution of ethylene glycol to the osmolar gap is less than other alcohols such as ethanol or methanol.42–45 A serum ethylene glycol of 3 mmol/L (21 mg/dL), a reportedly lethal concentration, should theoretically produce an increase in overall osmolality of only 4 mOsm/kg H2O while an even more significant concentration of 8 mmol/L (50 mg/dL) could produce a rise of 8–10 mOsm/kg H2O in the osmolar gap.
46
An elevated osmolar gap is nonspecific and has many different causes as discussed later. The osmolar gap is calculated as in equation (1)
The measurement of osmolality for detection of toxic alcohols is traditionally done by freezing point depression due to inaccuracies associated with the vapour pressure method when measuring volatile substances. This is not strictly a problem with ethylene glycol given it is nonvolatile. 47 However, use of the osmolar gap as a screening tool for ethylene glycol is not a commonly used test which means clinicians are poorly trained at interpreting the results. Similarly, some clinical biochemistry laboratories may be less familiar with the test as it is a section of the laboratory that generally has a low workload. This means caution must applied when interpreting individual numeric results. In addition to these issues, the use of the osmolar gap is beset by a number of theoretical problems and potential pitfalls which we will now elaborate on.
The formula
The first problem with using the osmolar gap to detect ethylene glycol is which formula to use to calculate the OSMc. There have been more than 15 different formulae proposed and the validity of each of these has long been debated.46–55 Most of the formulae in widespread use include contributions from sodium, urea and glucose with a few including ethanol.
45
In general, the variability of each of these formulae is explained by differing coefficients for the relative contribution of each of the substrates.
54
Two of the most frequently used calculations in the United States and Europe are as follows in equations (2) and (3)
The second formula includes ethanol, which if not taken into account may lead to error. In 2001, Purssell et al. 56 suggested the addition of a coefficient: ethanol (mmol/L)/3.7. In 2012, a very close approximation of ethanol (mmol/L)/4.0 was revalidated using 603 retrospective cases and 32 prospective spiked samples. 57 A great deal of research effort has gone into the search for the most accurate formula, the results of which are difficult to compare and ultimately inconclusive.45–49,52–57
The reference range
Attempting to find the most accurate calculation brings us to the second problem which is intrinsically linked to the validity of any formula. That is, what is the normal range for an osmolar gap? An accepted reference interval is not truly known. It is anticipated that normal, healthy subjects will have a difference between the measured and calculated serum osmolality. This difference is made up by unmeasured solutes, such as proteins, which are not included in the OSMc.
A normal range for the osmolar gap has been shown to vary from −5 to 9 mOsm in one group of healthy volunteers, 55 while in cohorts of patients presenting to the Emergency Department, the range has been reported to be as wide as −5 to 15 mOsm58 and −10 to 20 mOsm. 59 In addition to this, the reference interval for the osmolar gap was found to vary over time within a single laboratory, further decreasing the reliability of a standardized range. 60 All of this variability makes a definitive reference interval difficult to define.
Despite such controversy, the most commonly applied concentration to determine the presence of unmeasured osmotic solutes is an osmolar gap greater than 10 mOsm.46–55 Knowing, however, that the normal range for osmolar gap can be as low as −5 or −10 mOsm, it is possible for a patient to present with a toxic ingestion and an osmolar gap that is within the reference range but actually represents a significant change from their normal baseline. For example, a patient with a pre-existing osmolar gap of −5 mOsm who then ingests ethylene glycol and has a blood ethylene glycol concentration of 50 mg/L may only increase their osmolar gap to 5 mOsm which remains below the reference interval of 10 mOsm. This situation could lead a clinician to underestimate a potentially toxic ingestion. The potential for such false negatives is one argument as to why the measurement of the osmolar gap should not be regarded as a failsafe screening tool. Reader’s attention is also drawn to the fact that variable units are used to report ethanol and ethylene glycol in the literature which can be a source of confusion making it important for clinicians to double check the units before interpreting the result.
In 1996, Glaser 46 published a review of the evidence for the use of the osmolar gap as a screening tool for detecting ethylene glycol poisoning. The authors reviewed various published formulae for calculating osmolality and the osmolar gap. They concluded that the wide range of variability in the normal population as well as the variability from the differing calculations made the osmolar gap unsuitable as a screening tool. Later, in 2004, Purssell et al. 53 conducted a further review which came to a similar conclusion, stating that the available evidence was inconsistent and did not provide the expected level of evidence for the widespread use of the osmolar gap as a screening tool. In summary, these reviews looked at the evidence provided by five major studies done between the 1970s and the 1990s which aimed to establish the most accurate formula for osmolality estimation. In 1975, Dorwart and Chalmers 54 evaluated 13 different forumulae in 715 patients; in 1984, Bhagat et al. 61 compared three formulae in 100 serum samples; in 1987, Worthley et al. 62 tested five formulae in 300 patients; in 1993, Hoffman et al. 58 conducted a linear regression analysis from the results of 321 patients; and in 1997, Osypiw et al. 63 examined six formulae in 212 patients. Each of these studies purported superior results by using slightly different combinations of coefficients for the solutes in their formulae, leaving a clear conclusion difficult to establish.
Since 2004, there have been three further studies looking to address the utility of the osmolar gap as a screening test.48,49,60 The first, by Khajuria and Krahn, tested three formulae on a sample of both 49 prospective and 136 retrospective patients poisoned with either methanol or ethylene glycol. One of their formulae reported sensitivity of 1.00 and specificity of 0.86 for the detection of methanol or ethylene glycol (they do not differentiate between them) when using a cut off concentration for osmolar gap of greater than 10 mOsm. 60 However, that same study reported a drift in the osmolar gap reference range of 15 mOsm over a six-year period, calling into question the reliability and applicability of their reported sensitivity.
In the second study by Lynd et al., 49 the authors examined the ability of the osmolar gap to predict ethylene glycol and methanol in ‘concentrations high enough to need antidote therapy’. The study included 131 patients for analysis with only 20 patients with detectable ethylene glycol or methanol concentrations. The need for antidote therapy was defined as an ethylene glycol concentration greater than 3 mmol/L. The authors also used an osmolar gap cut off of 10 mOsm and reported a sensitivity of 85–90% but a specificity of only 22–50%.
The third study, by Krasowski et al., was a retrospective analysis of laboratory records and identified 470 samples where gas chromatography had been performed. The cohort included patients with ethylene glycol, methanol, propylene glycol and/or isopropanol poisoning and they considered serum concentrations of greater than 1.62 mmol/L (10 mg/dL) for any of these alcohols as a positive result. The authors reported a sensitivity of 0.82 and specificity of 0.85 when using an osmolar gap cut off of 20 mOsm. 48 When using an even higher cut off at 30 mOsm, Krazowski et al. report that the sensitivity dropped to 0.49 but the specificity increased to 0.95. The use of different reference intervals and different concentrations for positive results complicates comparison between these cohorts. The major limitation with all of these studies, however, is their small sample sizes and their retrospective nature.
Unfortunately, none of these studies are large enough or robust enough to conclusively solve the debate over which calculation or which reference range is definitively the most accurate. However, we can say that using a higher cut off for the upper limit of the osmolar gap reference interval has been shown to improve specificity and therefore reduce the likelihood of false-positive results.48,49,51 The finding of an osmolar gap greater than 20 or 30 mOsm, when other causes of a raised osmolar gap have been excluded, is increasingly suggestive of underlying ethylene glycol toxicity. Improved specificity comes at the cost decreased sensitivity and the absence of a raised osmolar gap cannot be taken as evidence to discount ethylene glycol toxicity.
The clinical context
Finally, adding further to the controversy, there are a number of other clinical situations where the osmolar gap would be expected to be raised. Herein lies another problem with the use of the osmolar gap, that is, how to interpret it in relation to the clinical situation. The osmolar gap is only an indirect marker of poisoning and may also be raised by conditions such as renal failure,64,65 sepsis or shock,66–69 mannitol therapy,70,71 alcoholic and diabetic ketoacidosis72–79 and hyperglycaemia. 72 In the undifferentiated patient with a decreased level of consciousness, it is possible for more than one of these processes to be occurring at one time which may yield false-positive results when interpreting the osmolar gap. It is therefore important that the clinician takes steps to rule out or consider all the alternative causes for an elevated osmolar gap.
Even after eliminating alternative causes for an elevated osmolar gap, the result may still be raised, normal or low depending on the timing of an ingestion. 80 The osmolar gap is usually most significantly elevated in the first few hours following ingestion and the degree of osmolar gap elevation may not correlate with physical symptoms. 81 The timing of the ingestion is therefore extremely important and leads on to another surrogate marker which is an anion gap metabolic acidosis which will be discussed next.
Anion gap
As previously mentioned, the major metabolites of ethylene glycol are organic acids which are not only responsible for cellular injury but also cause the raised anion gap that is seen in those with significant, established, ethylene glycol poisoning.2,3,44,51 Glycolic acid is the major cause of raised anion gap metabolic acidosis in ethylene glycol poisoning.1,86,87 Importantly, a raised anion gap will only be present following metabolism of the parent ethylene glycol compound to an organic acid. On the basis of the half life of ethylene glycol, the development of a raised anion gap has been estimated to take more than 3 h and can be prolonged if the patient co-ingests ethanol, due to preferential metabolism of ethanol over ethylene glycol by ADH.2,86,87 Therefore, it is possible that a patient may not have a raised anion gap if they present early for assessment and are assessed prior to significant conversion of the ethylene glycol to the toxic and negatively charged, acidic metabolites. Initially, a patient is more likely to exhibit a raised osmolar gap than an anion gap.
44
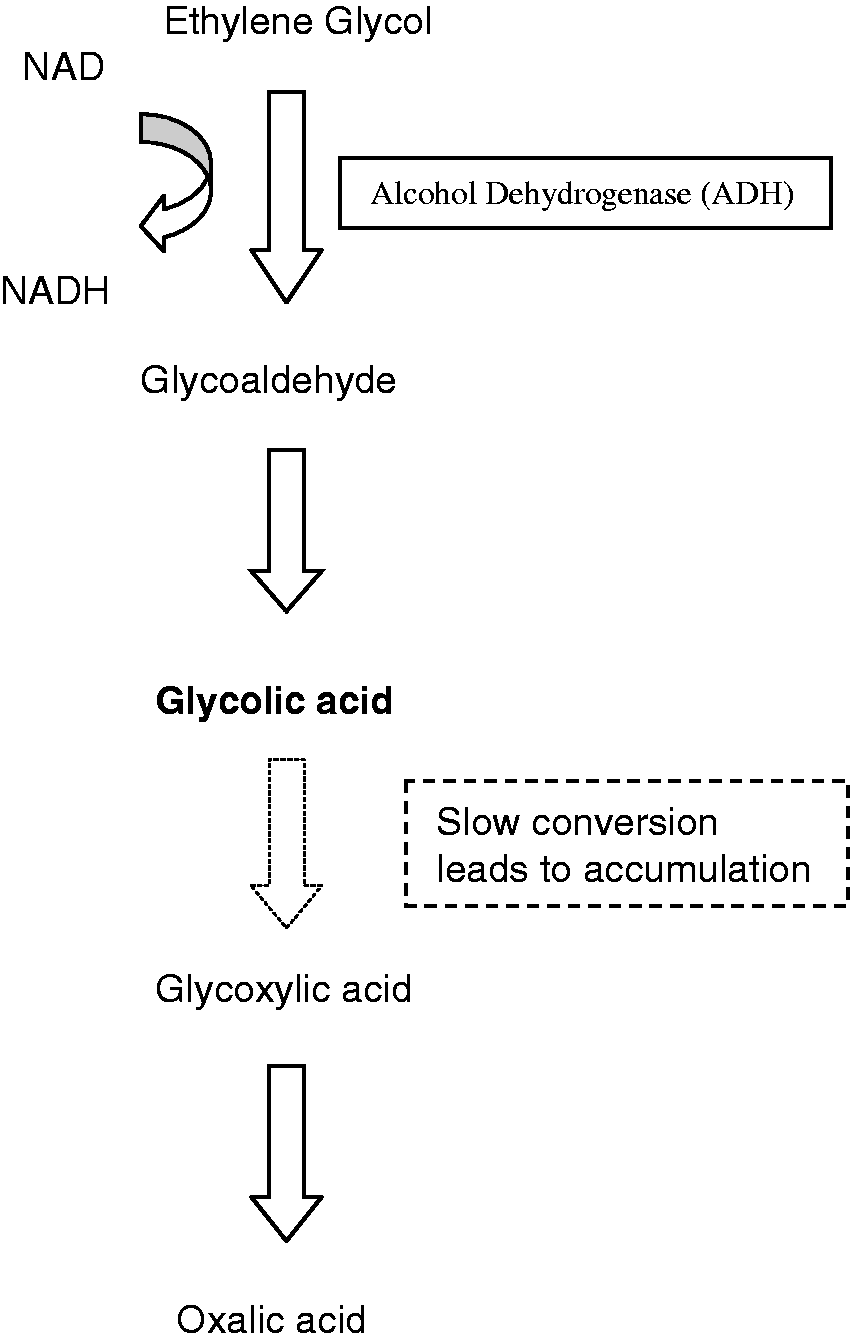
A number of hours later, the exact time being dependent on the amount of ethylene glycol ingested and the co-ingestion of ethanol (and potentially treatment in hospital), the osmolar gap may be returning to normal while an anion gap acidosis is becoming evident (Figure 2). Rarely, there have also been reported cases where both the osmolar gap and the anion gap have been normal.
81
Visual schematic of relationship between osmolar gap and anion gap in ethylene glycol poisoning. Adapted from Mycyk et al.
80
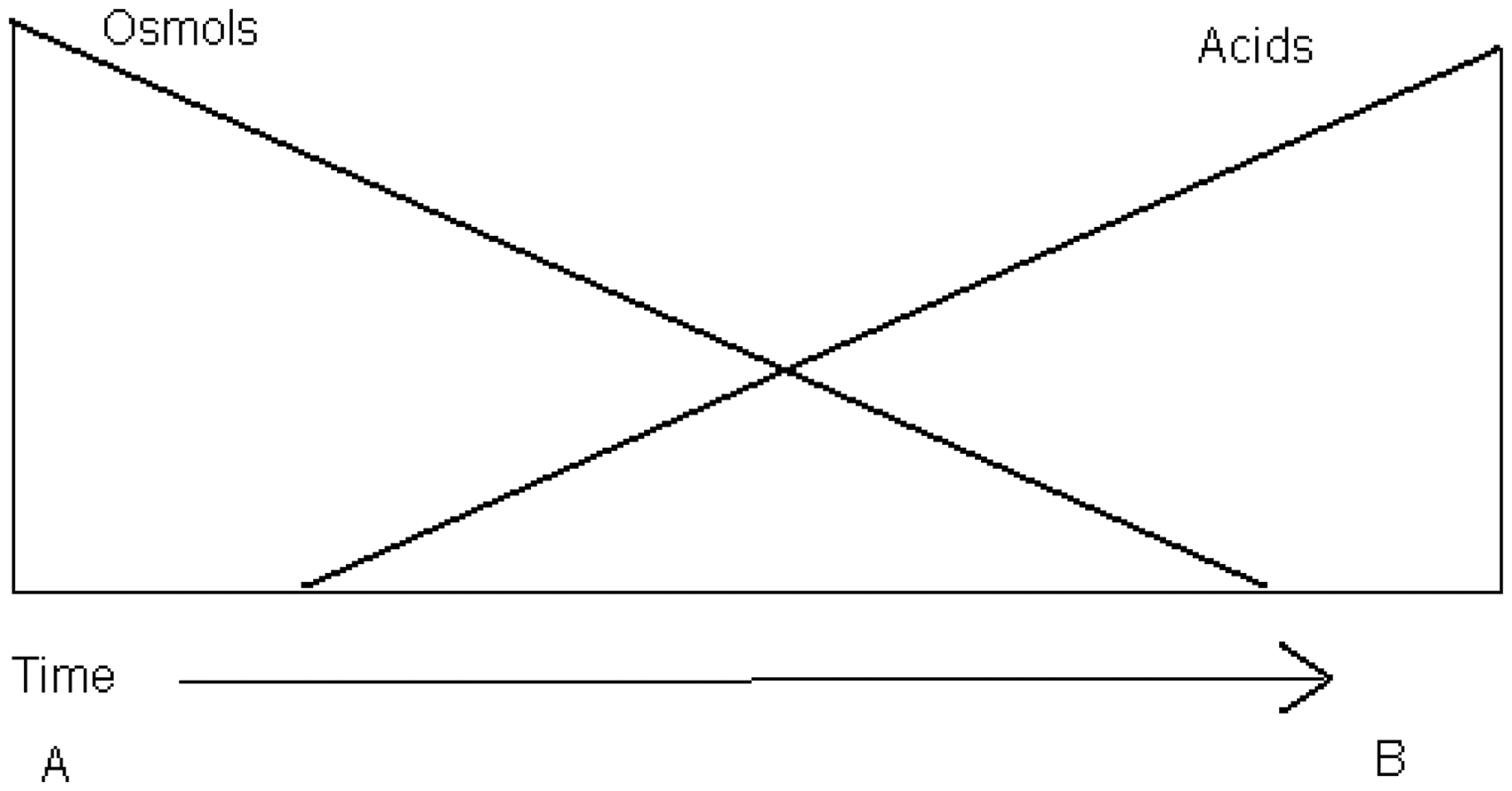
The anion gap (AG) is generally calculated using the following formulae
There is no published data to be able to establish which of these formulae is most reliable, but our clinical practice is to use the first of these formulae. The reference range most often quoted is 8–12 mEq/L or 8–16 mEq/L.82,80 If the calculated gap is greater than expected, this is taken as evidence of presence of unmeasured negatively charged molecules. Again, there is a wide differential for the possible causes of a raised anion gap acidosis and the clinician must consider them all.
1,2,82
A common tool clinicians use to remember the list of causes of a raised anion is the pneumonic MUDPILES which stands for
Lactic acidosis
Acid base interpretation in ethylene glycol poisoning may be complicated by a raised serum lactate. 83 There are three potential mechanisms to explain a raised lactate in ethylene glycol poisoning. The first is that the metabolites of ethylene glycol can cause a raised lactate. 86 Although lactate production in ethylene glycol poisoning is less well understood, it is likely that its production would follow the same temporal association as other acid production (Figure 1). The second mechanism is that there may be an alternative and concurrent cause for lactataemia such as tissue hypoxia. Third, depending on which type of laboratory equipment is used, the lactate assay may be falsely elevated secondary to interference with the assay from glycolic acid.83–89 It appears that glycolic acid may be responsible for this artefact due to structural similarity with lactate. 90 Analytical machines used for point-of-care testing have been associated with falsely elevated lactate measurements. In particular, machines such as the GEM Premier 4000 and the Radiometer ABL series have demonstrated this effect.83,87,88 The error is only noticed once the test is repeated with an alternative machine using a different method of measurement. The observed difference in the lactate concentration, between the falsely elevated value and the repeated value is referred to as a ‘lactate gap’. The Vitros Fusion 5.1, the Architect c8000 and the Radiometer Analyzer which are more specialized, nonpoint-of-care analysers, do not seem to produce false positives and may be capable of detecting a lactate gap. 83 The iSTAT, Roche BG analysers and the Bayer series (now operated by Siemens) which are some of the most widespread point-of-care machines have mixed reports from both case reports and trials investigating the issue with spiked samples.83,85,91
There are two main considerations with an elevated lactate. The first is that in the case of an undifferentiated patient with a raised anion gap acidosis, a clinician’s diagnostic process may be falsely influenced by a raised lactate. The lactate could be interpreted as adequate explanation for the observed anion gap acidosis and therefore the diagnosis of ethylene glycol poisoning may not be pursued leading to delay in diagnosis and more importantly, delay in treatment.
The second consideration is that a falsely elevated lactate or a lactate gap may serve as an indirect clue toward diagnosing ethylene glycol poisoning. There have been several reported cases where the diagnosis of ethylene glycol poisoning was made because clinicians or laboratory staff noticed a lactate gap when a sample was retested on a different machine in order to confirm an unexpectedly high lactate.83,85,88 An unexplained lactic acidosis, especially one that is worsening or where a lactate gap is observed, should be considered suggestive of ethylene glycol toxicity until results of quantitative assessment are available.
pH
Metabolic acidosis as signified by an arterial pH less than 7.35 reflects the accumulation of acid species from metabolism of ethylene glycol. Development of metabolic acidosis during ethylene glycol poisoning usually occurs in parallel with the development of an anion gap but there has been a small series of 14 patients reported who presented with nonanion gap metabolic acidosis. 92 In the 1999 Practice Guidelines for treatment of ethylene glycol, the American Academy of Clinical Toxicology included ‘arterial pH less than 7.3’ as part of a set of criteria for the presumptive diagnosis of ethylene glycol toxicity. 1 This recommendation is based on limited clinical data and represents general consensus, Level III evidence.
pH does appear to correlate very strongly with development of acute renal failure and can be used prognostically. In one retrospective review of 41 presentations of ethylene glycol poisoning, an initial pH of less than 7.3 on presentation predicted acute renal failure with a sensitivity of 1.00 and a specificity of 0.82. 93 An unexplained metabolic acidosis, especially one that is progressive should be considered as extremely suggestive of ethylene glycol toxicity until results of a specific ethylene glycol assay are available.
Glycolic acid measurement
Some authors advocate measuring glycolic acid concentrations as another indirect indicator of ethylene glycol poisoning.84,94 A technique for simultaneous detection of glycolic acid and ethylene glycol using gas chromatography has been described. 95 However, a laboratory which is unable to analyse ethylene glycol concentrations using gas chromatography would also not be in a position to process a glycolic acid concentration. The detection of glycolic acid is only of use in the setting of a delayed presentation where the entire parent ethylene glycol has been metabolized. In other settings, the analysis would also detect parent ethylene glycol, which would qualify for a gold standard diagnosis of ethylene glycol poisoning.
Colorimetry
In 2008, Shin et al. 96 described a rapid, semi-quantitative method for the detection of ethylene glycol using inexpensive reagents and colorimetry of saliva samples. While the initial results appeared promising, the results were in vitro only and the technique is yet to be prospectively tested on saliva from animals or humans exposed to ethylene glycol. It is possible that in the future, this method may be used to give a qualitative result which will aid initial treatment while formal quantitative results are being processed.
Urine fluorescence
Using urinary fluorescence to detect ethylene glycol ingestion was popularized by Winter et al. 97 who reported the technique in 1990. The method has a number of pitfalls in a similar way to measurement of the osmolar gap. Further studies which have looked to validate this technique have shown that it is unsuitable as a screening test.98–100
Detection of fluorescence relies on the principle that certain preparations of ethylene glycol have sodium fluorescein added to the solution by manufacturers. This fluorescence is intended to act as a dye to make detection of leaks in coolant circulation of cars easier for mechanics. The fluorescein can then be detected in urine when it is visualized under ultraviolet light such as a Woods lamp, as was originally described by Winter et al.
The first problem then is that not all sources of ethylene glycol contain sodium fluorescein meaning that sensitivity can never be 100%. Further to this, fluorescence has been found to be pH dependent and will be absent in urine that has a pH of less than 4.5.97,98,100 Authors have described a technique which involves measuring pH prior to detecting fluorescence and then adjusting the pH with sodium hydroxide accordingly. 99 However, this method adds a level of complication to the process and to date has not been formally validated.
Urinary fluorescence is time dependent, the sensitivity drops markedly from 4 to 6 h postingestion. 97 This means that for late presentations or time-unknown cases, the technique becomes unreliable.
Although Winter et al. reported up to 100% sensitivity when samples were collected prior to 4 h postingestion, later studies have reported sensitivities of 35–42% 99 with poor inter-rater reliability (simple agreement 72.5%, j = 0.25, 95% CI 0.13–0.37). 98
We therefore feel that, at this time, there we cannot recommend the routine use of urine fluorescence in the algorithms for ethylene glycol diagnosis; further work is needed to determine the validity of this technique and any potential role that it may have.
Calcium oxalate crystals on urinalysis
Oxalic acid precipitates as monohydrate and dihydrate calcium crystals in the renal tubules and in the urine within 4–8 h of ingestion of osmolar gap.2,93 These crystals can be identified by their typical ‘top of a picket fence’ appearance when viewed under polarized light, although the monohydrate crystals may be mistaken for hippuric acid crystals. 2
Detection of oxalate crystals supports the diagnosis of ethylene glycol toxicity but is not specific. Oxalate crystals can be found in naturally occurring plant tissues giving rise to potential false positive results.2,101 Detection of oxalate crystals is also not a sensitive finding. In one series of 12 patients with confirmed ethylene glycol toxicity, only four had calcium oxalate detected in their urine. 102 While detection of crystals is potentially important, it is not pathognomonic. This is because crystalluria is a common finding in normal individuals with high dietary intake of foods containing either oxalates or vitamin C.
From a practical standpoint, it is unknown how many hospitals would have facilities available for polarized light microscopy out of hours. We feel that the low specificity along with a lack of out-of-hours facilities limits the usefulness of this technique.
Hypocalcaemia
A decreased ionized calcium concentration may suggest ethylene glycol poisoning as a result of complex formation between oxalic acid and calcium. This typically occurs more than 6–8 h postingestion, once a significant amount of ethylene glycol has been metabolized. 81 This finding may help to differentiate ethylene glycol toxicity from the toxicity caused by other alcohols like methanol where calcium sequestration does not occur. The degree of hypocalcaemia may be related to degree of toxicity and has been reported in association with QT prolongation on electrocardiogram. 103 The finding of urinary calcium oxalate at the same time as serum hypocalcaemia may be more specific for ethylene glycol poisoning than either finding in isolation 2 ; however, hypocalcaemia is best considered a supplementary finding rather than diagnostic.
Summary
Ethylene glycol poisoning remains an important toxicological problem, with potential for significant morbidity and potentially mortality. The clinical presentation varies depending on the timing of ingestion and there are a wide number of differential diagnoses which must be considered alongside or as an alternative to ethylene glycol poisoning. Serious toxicity is caused by the major metabolites of ethylene glycol and not by the parent compound.
Despite recent advances in enzymatic assay detection of ethylene glycol, the gold standard diagnostic test remains gas chromatography which gives a quantitative assessment of exposure. The usefulness of this test is hampered by delays in access due to availability. Consequently, clinicians are often forced to rely on surrogate markers of toxicity in order to make rapid treatment decisions.
Assessment of the osmolar gap is an indirect indication of ethylene glycol exposure and is most useful when performed soon after ingestion. Despite many attempts to improve the accuracy of this test, the osmolar gap still has a low sensitivity and specificity which means the results must be interpreted with extreme caution. An osmolar gap greater than 10 mOsm is the most widely used cut off to indicate potential poisoning and pre-existing negative osmolar gaps can lead to false-negative results. Ultimately, a very elevated osmolar gap increases the likelihood of the presence of ethylene glycol while a moderately raised, borderline, or even ‘normal’ osmolar gap must be interpreted with caution.
A metabolic acidosis with a raised anion gap is another useful marker of potential ethylene glycol poisoning. The results must be interpreted within the clinical context, particularly in relation to the timing of ingestion. Again, the wide range of differential diagnoses for a raised anion gap acidosis, along with a low sensitivity render this test unable to conclusively rule out or rule in ethylene glycol poisoning.
Several other tests such as looking for a lactic acidosis, positive urinary fluorescence, detection of calcium oxalate crystals in urine and the presence of serum hypocalcaemia can all be suggestive of ethylene glycol toxicity but do not give a definitive diagnosis. In the future, we may be able to rely on colorimetry to give us a qualitative indication of the presence of ethylene glycol but this method, while promising, requires prospective validation before it will have any impact on the day-to-day management of this poisoning.
In our opinion, from a practical standpoint, where there is a diagnosis of suspected ethylene glycol poisoning from the history available and/or the results of the different indirect tests discussed above, we feel that the clinician is best advised to err on the side of caution and treat with appropriate antidotal therapy until the results of a specific ethylene glycol assay (quantitative gas chromatography) are available.
Footnotes
Acknowledgement
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical Approval
Not applicable.
Guarantor
DJMc.
Contributorship
All authors contributed to the design, analysis and drafting of this article.