Abstract
Background
Ethylene glycol is a highly toxic compound found in various household products. Cases of poisoning are rare but may be fatal unless diagnosed and treated promptly. Early recognition of poisoning is critical for the management and recovery of patients. Indirect testing is not specific for the presence of ethylene glycol. Therefore, urgent and accurate measurement should be sought if ingestion is suspected in order to determine the need for treatment with an antidote. Here, we present the validation of an automated assay for measurement of ethylene glycol on an Abbott Architect using a commercially available kit (Catachem).
Methods
Analytical parameters of imprecision, linearity, stability and bias were determined using spiked human plasma samples processed on both the Catachem assay and on an in-house gas chromatography-mass spectrometry method. Interference was assessed using samples collected into a variety of sample collection tubes and spiked with a number of alcohols.
Results
Excellent agreement was observed between the two methodologies with the enzymatic assay demonstrating linearity and precision across the relevant clinical range (50–3000 mg/L). In addition, the Catachem assay displayed no interference from a number of different sample tubes and alcohols. However, propylene glycol interference was observed at concentrations associated with excessive use (>1 g/L) and 2,3-butanediol interference observed at concentrations associated with butanone ingestion. Inspection of the enzymatic reaction profile was found to differentiate between alcohols.
Conclusions
This automated assay is suitable for the diagnosis of ethylene glycol poisoning and is now in routine use, enabling the laboratory to provide a rapid 24 h service with support by gas chromatography-mass spectrometry as necessary.
Introduction
Ethylene glycol is an odourless, colourless, sweet liquid, which is commonly used as a de-icing solution or antifreeze in coolant systems. Poisoning from ethylene glycol is uncommon, but when it occurs, there is an associated risk of severe morbidity and mortality with the oral ingestion of around 100 mL being fatal. 1 This is a global problem with many reports of accidental poisoning following ingestion by children. 2 The clinical presentation of ethylene glycol toxicity is classically described in three phases, with the most serious toxic effects developing from the formation of the metabolites of ethylene glycol (glycoaldehyde, glycolate, glyoxylate and oxalate), rather than the parent compound itself. These phases are typically described as neurological (0.5–12 h from ingestion), cardiopulmonary (12–36 h) and renal (24–72 h) and often overlap as the clinical course of poisoning progresses. Because of this, early and accurate diagnosis is vital in the management of patients presenting with suspected ethylene glycol toxicity in order to recognize the need to start treatment with fomepizole (an alcohol dehydrogenase inhibitor), ethanol or haemodialysis. 3 This is complicated by the fact that the presentation of ethylene glycol poisoning may be non-specific, especially when patients are not forthcoming about ingestion. Presentation may occur at any time following ingestion, with various symptoms, which lead to wide range of possible differential diagnoses. However, the presence of a high anion gap, associated with a metabolic acidosis and an elevated plasma osmolal gap should lead to a clinical suspicion of a methanol or glycol poisoning.
Indirect testing for ethylene glycol ingestion by measuring, e.g. lactate, osmolal gap and anion gap, although potentially helpful, is not always accurate.4,5 Accurate measurement should therefore be sought if ingestion is suspected. 3 Recent guidelines from the Association for Clinical Biochemistry and Laboratory Medicine have recommended that ethylene glycol measurement should be available within 4 h if poisoning is suspected. 3 However, this is complicated by the fact that ethylene glycol assays are typically performed using specialized equipment including liquid chromatography with UV, 6 or mass spectrometric detection, 7 or gas chromatography with flame ionization 8 or mass spectrometric detection, 9 which are only available in a limited number of specialized toxicology centres. At present, only six such centres across the UK advertise this service, located in Birmingham, Bristol, Cardiff, Glasgow, London and Sheffield. 10 These centres can be difficult to access in a cost-effective and timely manner potentially resulting in delays to diagnosis and treatment of poisoned patients, especially out of normal working hours. There is therefore a need for a rapid, automated, cost-effective assay for the measurement of ethylene glycol which is also readily available in a large number of departments in order to enable the investigation of patients with suspected poisoning. Here, we present the validation of an enzymatic assay for the measurement of ethylene glycol in human plasma on an Abbott Architect.
Materials and methods
Chemicals, materials and reagents
Stock solutions or powders of ethylene glycol, ethylene-d4-glycol, diethylene glycol, lactate, ethanol, methanol, glycolic acid, propylene glycol, isopropyl alcohol, 1-propanol, fomepizole and acetone were obtained from Sigma Aldrich (Sigma, Poole, UK). The derivitization agents N,O-Bis (trimethylsilyl) trifluoroacetamide containing trimethylchlorosilane (BSTFA-TMCS) (99:1) and dimethylformamide (DMF) were also purchased from Sigma. High-performance liquid chromatography (HPLC)-grade water and acetonitrile were obtained from Rathburn Chemicals (Rathburn Chemicals Ltd, Walkerburn, UK). The Catachem DISCRETPAK™ ethylene glycol automated assay (Oxford, Connecticut, US) was performed using an Architect c-16000 analyser (Abbott Diagnostics, Illinois, USA).
Gas chromatography-mass spectrometry analysis
The gas chromatography-mass spectrometry (GC-MS) quantitative assay for ethylene and diethylene glycol has been in routine use within the Department for over two years and has yielded acceptable performance throughout as evidenced by ongoing satisfactory external quality assessment (EQA) performance. It was therefore seen as both selective and sensitive and deemed suitable to act as the reference method for this evaluation study.
The instrumentation consisted of a Thermo Trace 1300 Gas Chromatograph coupled to a Thermo ISQ LT Single Quadrupole Mass Spectrometer (Thermo Scientific, Hemel Hemstead, UK) using an electron-impact ion source. The column used was a Thermo TG-5MS Column (30 m length, 0.25 mm ID, 0.25 µm film). Helium was used as the carrier gas at a flow rate of 1 mL min−1. Initial oven temperature was 70℃ for 0.25 min followed by a temperature-programmed increase of 65℃ min−1 to 250℃, then another increase of 125℃ min−1 to 310℃ where temperature was held for 1 min. Split injection was used at a ratio of 1:30 with an inlet temperature of 250℃. Samples were analysed in the selected ion monitoring mode with the characteristic ions used for quantitation as follows; ethylene glycol 191 and ethylene-d4-glycol 195. Instrument control and sample analysis were performed using Chromeleon (Version 7.0, Dionex).
For the GC-MS analysis, five calibration standards (50, 250, 500, 1000 and 2000 mg/L) were prepared by appropriate dilution of a stock ethylene glycol solution in water. Internal quality controls (IQC) at two levels (300 and 1000 mg/L) were independently prepared in the same way. Internal standard for this method was 1000 mg/L ethylene-d4-glycol in 100% acetonitrile.
Sample preparation (GC-MS)
Standards or samples (50 µL) were manually pipetted into 1.5 mL microfuge tubes. To this, 50 µL of working internal standard was added. Samples were vortex-mixed for 30 s and centrifuged at 9000 g for 5 min. Supernatant (20 µL) was transferred into a clean 1.5 mL glass vial and 20 µL of DMF and 300 µL of BSTFA were added. Vials were capped, vortex-mixed for 10 s and placed onto a heating block for 10 min at 90℃. After 10 min, samples were removed, vortex-mixed for 10 s, allowed to cool and transferred to the GC-MS autosampler ready for injection.
Catachem enzymatic method
The Catachem ethylene glycol procedure is based on the affinity of a specific glycerol dehydrogenase enzyme from a bacterial source to catalyse the oxidation-reduction reaction of ethylene glycol in the presence of nicotinamide adenine dinucleotide. This two-point kinetic procedure, using 3 µL of sample, is monitored at 340 nm, and the increase in absorbance is directly proportional to the concentration of ethylene glycol in the sample. Assay parameters for the Architect System were provided by Catachem. 11 The calibrator (single point 1550 mg/L), quality control (QC) at two levels (560 and 2480 mg/L), diluent and activator reagent were supplied with the kit.
Method validation
Interference studies
Drug-free plasma samples from a healthy volunteer were spiked with a mixture of commonly encountered compounds similar to ethylene glycol at 7000 mg/L, reflecting concentrations far in excess of those likely to be encountered or achieved physiologically to ensure the specificity of the technique. Compounds used were lactate, ethanol, glycolic acid, propylene glycol, methanol, isopropyl alcohol (2-propanol), 1-propanol, beta-hydroxybutyrate, 2,3-butanediol, acetone and diethylene glycol. In a separate experiment, blood was collected from a healthy volunteer into a number of different collection tubes. Tubes types used were plain serum, EDTA, lithium heparin, fluoride oxalate, sodium citrate, serum separator and cross-match (BD Vacutainer®). Samples were processed and analysed as above to test for interference by each tube type in both methods. Lack of interference was defined as a measured ethylene glycol concentration of <50 mg/L, as per ACB-NPIS Guidelines. 3 In order to assess the effect of fomepizole on the measured ethylene glycol concentration, plasma samples containing approximately 500 mg/L of ethylene glycol were analysed before and after the addition of 120 mg/L fomepizole, a concentration which reflects the mean peak concentration following a single oral dose of 100 mg/kg. 12 Acceptable performance was defined as no significant difference in measured ethylene glycol concentration in the neat and spiked samples.
Interference of propylene glycol and 2,3 butanediol on the enzymatic method was investigated by spiking drug-free plasma samples from a healthy volunteer with propylene glycol at values of approximately 1000, 2000, 3000, 4000, 5000, 6000 and 7000 mg/L and 2,3 butanediol at values of 90, 225, 451, 676, 901, 4506 and 9012 mg/L. Samples were then processed and analysed by both methods with the maximum acceptable concentration of propylene glycol or 2,3 butanediol defined as a measured ethylene glycol concentration of <50 mg/L in the presence of the interferent.
Linearity and lower limit of quantitation
For the GC-MS method, assay linearity was calculated by a least-squares linear regression analysis of the peak area ratios of analyte to internal standard versus nominal analyte concentration. The regression parameters of the slope, intercept and correlation coefficient were calculated by the weighting factor, 1/x2. An acceptable r2 value was deemed to be >0.95. For both methods, the lower limit of quantitation (LLOQ) and upper limit of quantitation (ULOQ) were defined as the lowest or highest analyte concentration for which the values of precision (relative standard deviation, RSD) and accuracy (relative error, RE) were ≤ 20%, and the signal-to-noise ratio was ≥ 30 (LLOQ on GC-MS only). RE was calculated using RE (%) = ([measured concentration−nominal concentration]/nominal concentration) × 100.
Precision and accuracy
To evaluate precision, IQC samples, either prepared as outlined above (GC-MS) or supplied with the Catachem kit, were analysed on the same day, and over a period of 11 consecutive days. The bias of the GC-MS method was assessed by comparison with mean ethylene glycol values obtained from the LGC toxic alcohol external quality assurance (LGC TAK) scheme. To evaluate the accuracy of the enzymatic method, 11 spiked plasma samples covering the reportable ethylene glycol concentration range were analysed enzymatically and by GC-MS. Acceptable performance was defined as no significant difference between the results obtained by the two methods.
Matrix effects
For the GC-MS assay, the matrix effect was assessed in five separate experiments at a relevant IQC concentration (700 mg/L) by comparing the concentration of drug measured when the material was made either in HPLC-grade water or drug-free donor plasma. The mean percentage difference between the two should ideally be <10% and statistically insignificant when the replicates are compared using a paired t-test. Statistical analysis was performed using Analyse-it.
Stability
The stability of ethylene glycol in plasma samples was studied for both methods using a single IQC concentration (nominal value of 1100 mg/L), measured in triplicate after 0 h at room temperature, 24 h at room temperature (25℃) and 2–8℃, after seven days at 2–8℃ and −20℃ and after long-term storage at −20℃ (28 days). A sample was considered to be stable in the biological matrix, when the calculated concentrations were 90–110% of those of the freshly prepared samples.
Statistical analysis
Measured concentrations of ethylene glycol in plasma before and after addition of fomepizole were compared by a paired t-test. Concentrations of ethylene glycol determined enzymatically and by GC-MS were compared by Bland–Altman and Passing and Bablock analyses. Concentrations of ethylene glycol measured in IQC made up with HPLC-grade water or drug-free plasma were compared by a paired t-test. All statistical analyses were performed using Analyse-it for Microsoft Excel, Leeds UK. See http://www.analyse-it.com.
Results
Interference
For the GC-MS analysis, typical chromatograms are depicted in Figure 1. No interfering signals were observed from any of the potential interferences investigated; or when a variety of blood collection tubes were used. Retention times were within 0.1 min in each case when compared with an analytical standard and the relevant internal standard. For the enzymatic method, interference was observed, i.e. false-positive ethylene glycol values seen, with propylene glycol (propane-1,2-diol) at concentrations >1000 mg/L, and with 2,3-butanediol at concentrations >90 mg/L. Concentrations of ethylene glycol in plasma samples spiked with fomepizole were 5% higher than in samples not spiked, but this difference was not significant (P = 0.17).
Representative chromatographs for ethylene glycol (b) and ethylene-d4-glycol (a) when measured by GC-MS. Ethylene glycol was at a concentration of 250 mg/L. Retention time was 2.23 min. Ions used were 191 and 195 for ethylene glycol and ethylene-d4-glycol, respectively.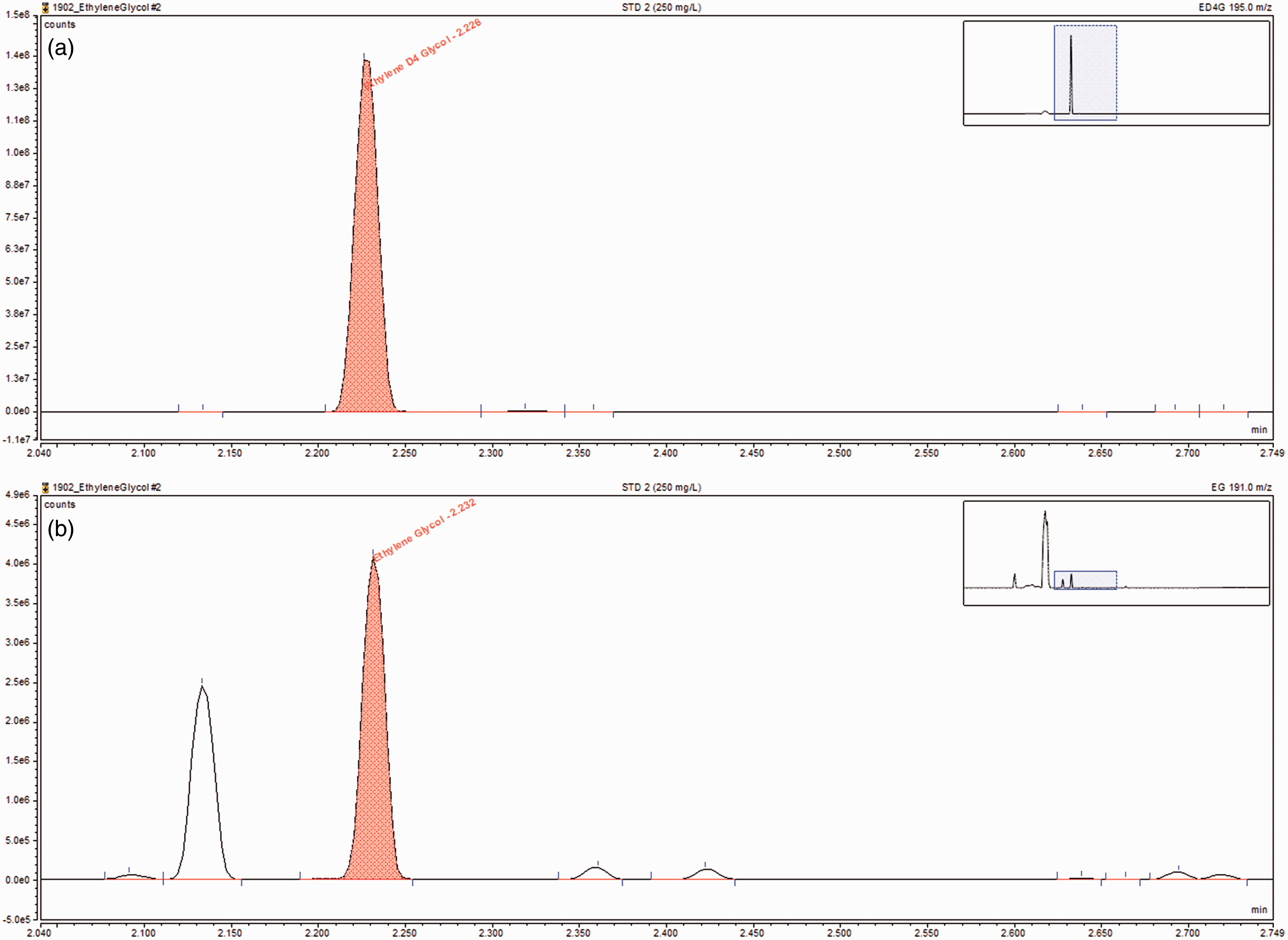
Linearity, ULOQ and LLOQ
Linearity of ethylene glycol assays.
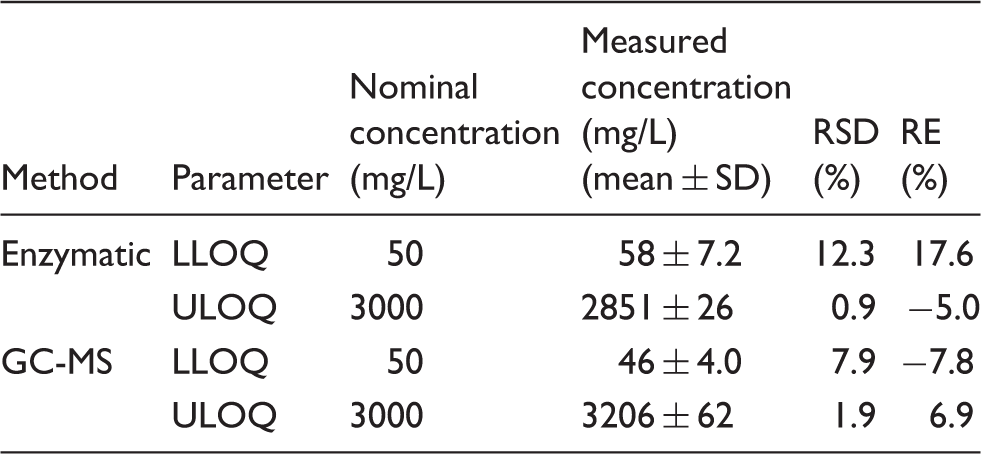
Note: Both were linear from 50–3000 mg/L. R2 for the GC-MS assay was 0.99.
RSD: relative standard deviation; RE: relative error; LLOQ: lower limit of quantitation; ULOQ: upper limit of quantitation.
Precision, accuracy and matrix effect
Imprecision of ethylene glycol assays.
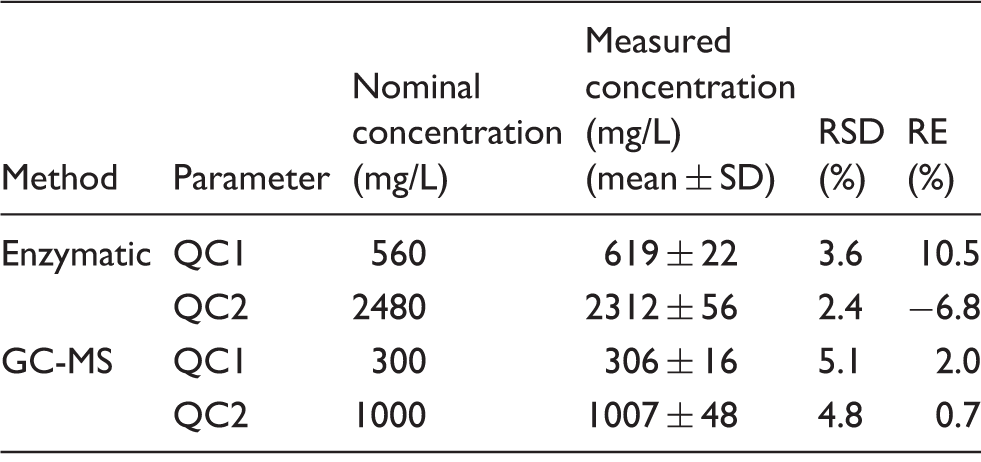
Note: RSD at all levels were <6% with RE <11%.
RSD: relative standard deviation; RE: relative error; QC: quality control.
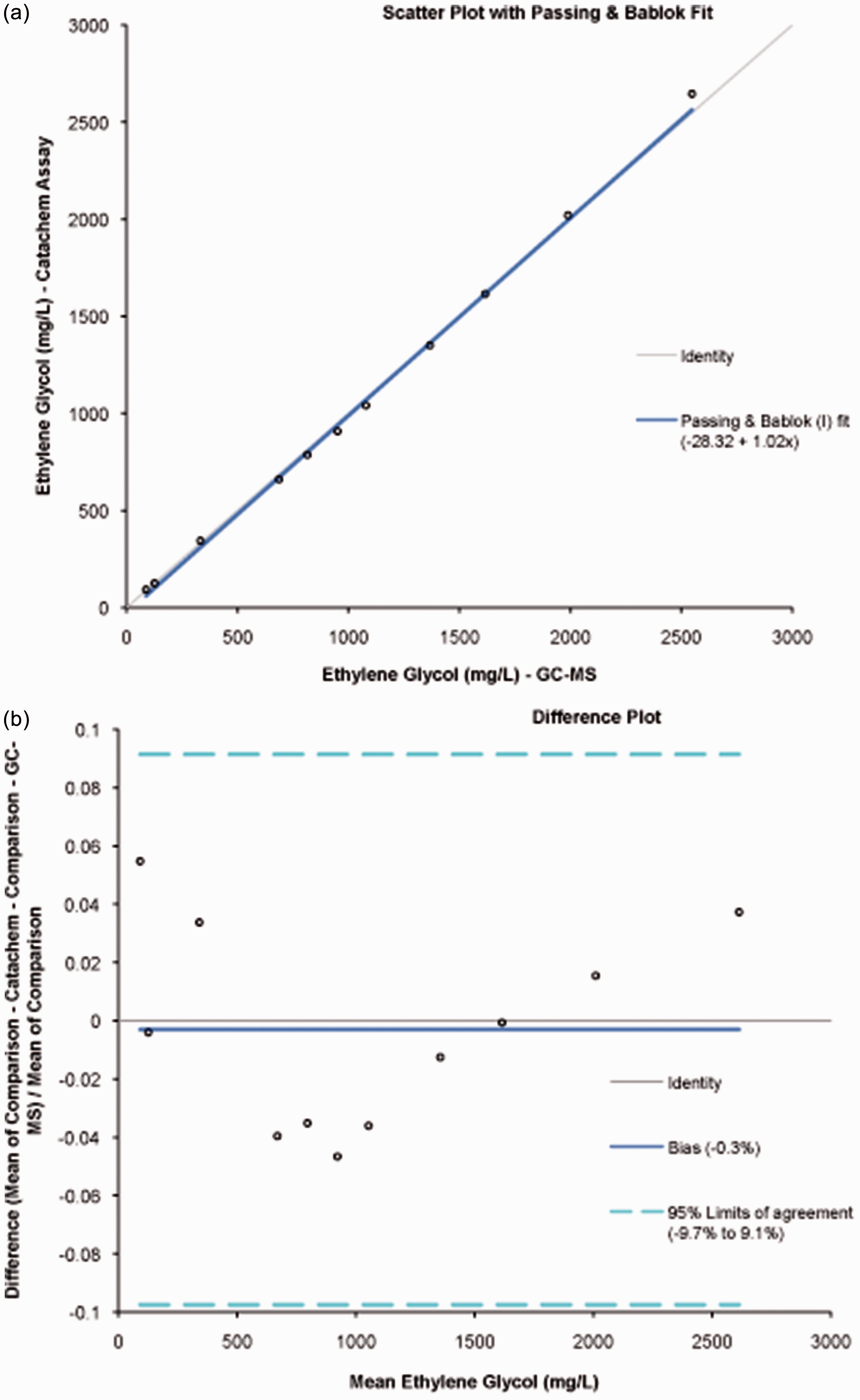
Comparison of enzymatic and GC-MS measurement of ethylene glycol shown by Passing Bablok regression (a) and Bland–Altman difference plots (b). Eleven spiked plasma samples covering the reportable range were analysed using both the enzymatic method and GC-MS.
Stability
Stability of ethylene glycol when measured either by GC-MS or enzymatically.

Note: Values in brackets are %CV of replicate analysis. Changes in ethylene glycol concentration were not significant (P > 0.05), with the exception of storage at 24 h at room temperature when ethylene glycol was measured by the enzymatic method (P = 0.03). *%CV cannot be calculated for this replicate due to a missample of one replicate.
Discussion
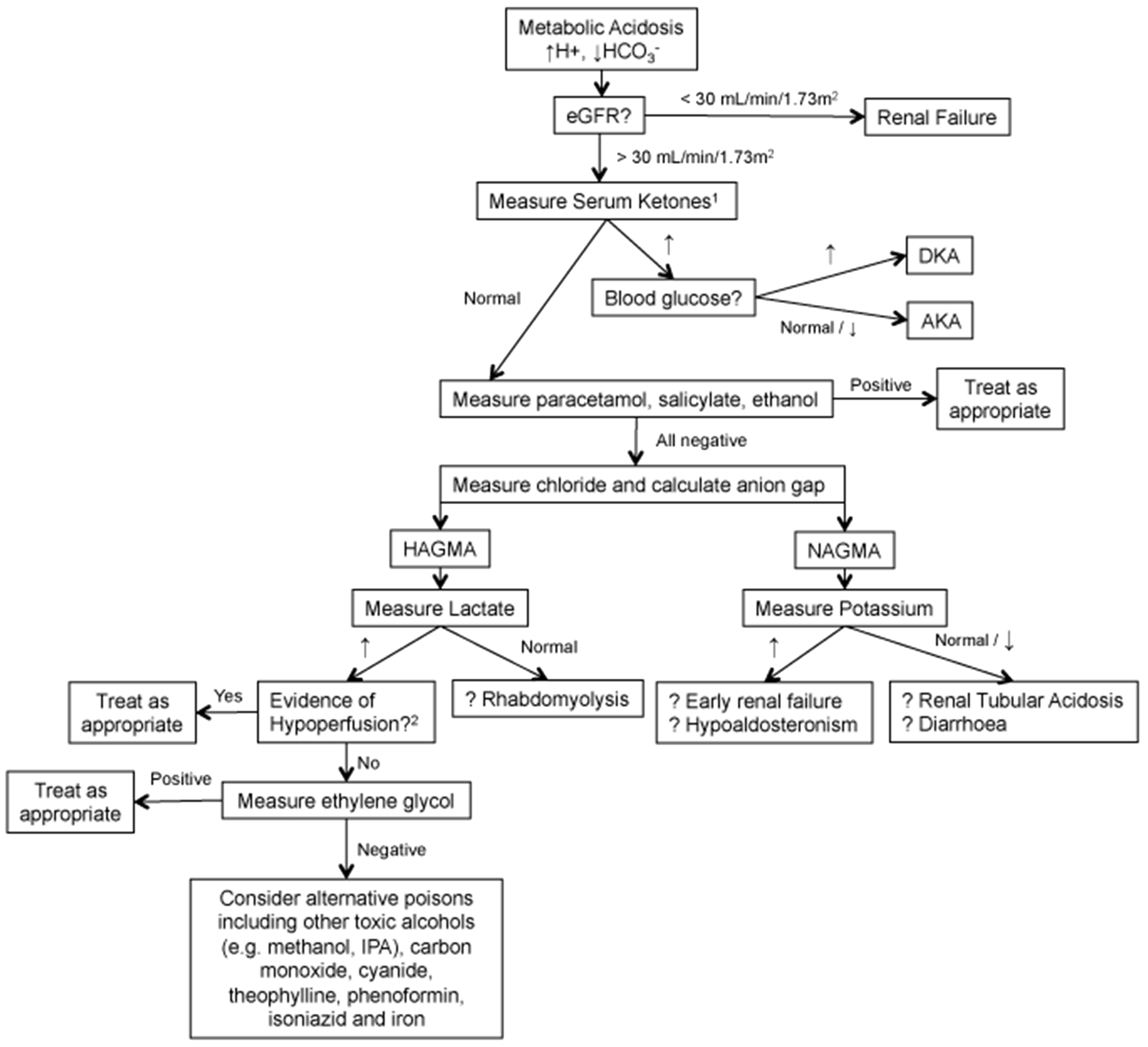
Ethylene glycol is a toxic compound whose ingestion may be fatal. Urgent treatment (fomepizole or ethanol) should therefore be given to any patient who has taken it. Although considered rare, the latest NPIS national report places ethylene glycol as fourth highest in a ranking of agents commonly involved in NPIS consultant referrals in 2014/15 (92 instances) behind only paracetamol (433), drugs of misuse (201) and unknown drug intoxication (145, which, it could be argued, may contain toxic alcohols). 13 The large number of referrals indicates the difficulty in diagnosis of ethylene glycol poisoning, as presentation may be non-specific with patients presenting at any time following ingestion with various symptoms (neurological, cardiopulmonary and renal) leading to a wide differential diagnosis. A suggested investigative pathway for patients with metabolic acidosis is shown in Figure 4. Ethylene glycol ingestion can only be confirmed by testing for the presence of the compound in blood. Therefore, clinical laboratories supporting emergency departments have a requirement to provide urgent measurement of ethylene glycol in order to aid in the investigation of patients who are suspected of taking the substance, or have unexplained anion or osmolal gaps. Measurement has typically been provided by specialist referral toxicology centres, using manual chromatographic techniques, which may not be readily be available 24/7, or if available, may be so at a prohibitive cost.
We have therefore validated a commercially available enzymatic method to measure ethylene glycol on an Abbott Architect c-16000 analyser. This assay has previously been validated on Hitachi 917 and Olympus AU400 platforms.
14
Assay reagents are stable on-board the analyser for 60 days and unopened kits can be stored for approximately two years offline according to lot number. Analytical parameters were provided by the assay manufacturer. The method showed acceptable linearity across the relevant range (50–3000 mg/L, Table 1), acceptable imprecision (RSD <10%, Table 2) and excellent agreement with the GC-MS assay used as the reference method (Figure 2). The enzymatic method showed no interference from a variety of blood collection tubes and a number of compounds related to ethylene glycol, with the only interference observed being from propylene glycol at concentrations >1000 mg/L and 2,3-butanediol at concentrations > 90 mg/L. These compounds did not interfere in the GC-MS assay. Inspection of the reaction curves on the c-16000 analyser showed clear differences between the reactions of propylene glycol, 2,3-butanediol and ethylene glycol (Figure 3), with propylene glycol showing a rapid, non-linear reaction on addition of substrate when compared with the linear reaction of ethylene glycol. Attempts to avoid the interference by blanking after the propylene glycol reaction had completed, followed by the reading of the continued ethylene glycol reaction were unsuccessful due to an insufficient change in absorbance values. Unfortunately, the time interval between read-points could not be altered on the Abbott c-16000. The reaction of 2,3-butanediol was curvilinear when compared with that of ethylene glycol (Figure 3). Fomepizole, an inhibitor of alcohol dehydrogenase and antidote for ethylene glycol and methanol poisoning, was found not to interfere when present at peak concentrations associated with supratherapeutic use of this drug.
12
It should be noted that the concentration of fomepizole added (120 mg/L) far exceeds the typical target range of 8–25 mg/L suggested by the manufacturer.
15
Comparison of ethylene glycol, propylene glycol and 2,3 butanediol reaction curves on the enzymatic method using an Abbott C-16000 analyser. Each read-point represents a time window of 18 s. The blank reading is made between 14 and 16 s. Reagent is added at the 16th read-point and absorbance read from 24 to 33. Concentrations of ethylene glycol, propylene glycol and 2,3 butanediol used are chosen to represent concentrations expected in cases of excessive ingestion. A suggested investigative pathway for patients with metabolic acidosis. (a) Measure beta-hydroxybutyrate. (b)Investigate for hypoxia, severe anaemia, shock, haemorrhage, hypotension, congestive cardiac failure. DKA: diabetic ketoacidosis; AKA: alcoholic ketoacidosis, HAGMA: high anion gap metabolic acidosis, NAGMA: normal anion gap metabolic acidosis.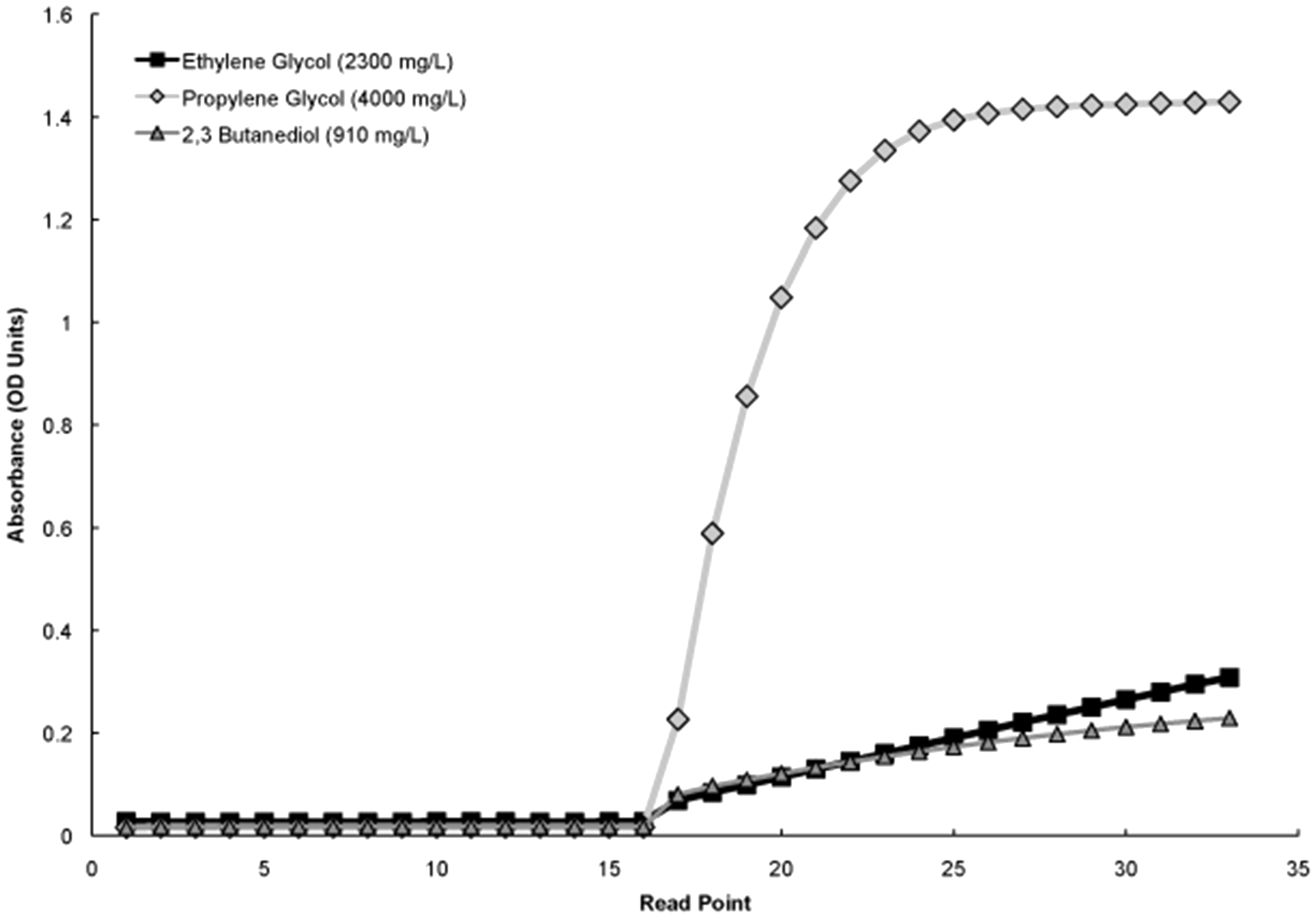
Propylene glycol (1,2-propanediol) is an odourless, viscous liquid which may be used commercially as a substitute for ethylene glycol in automotive antifreeze and is used as a preservative, emollient and vehicle for both oral and intravenous medications, typically benzodiazepines. Although considered non-toxic, there have been reports of propylene glycol causing decreased levels of consciousness, seizures and acidosis when used in pharmaceuticals16–19 and also death following its consumption (as antifreeze). 20 Toxic effects were observed at concentrations of 1000–5000 mg/L with fatalities reported at concentrations of > 4000 mg/L. Although we observed interference from propylene glycol in the enzymatic assay, provided adequate controls are put in place, we do not believe that this interference should prevent its routine. Firstly, the reaction curves of samples giving a positive result will be inspected by laboratory staff to check for the presence of the characteristic reaction curve of propylene glycol (Figure 3) prior to results being released. Any such samples exhibiting potential interference will then be re-analysed using GC-MS. Secondly, in those instances when drugs contain propylene glycol as an excipient in their formulation, the expected concentrations of propylene glycol observed in the circulation following ingestion of such drugs would be too low to cause interference in the ethylene glycol assay. This is supported by a number of studies in which patients had been given drugs containing propylene glycol, and where average peak concentrations of propylene glycol of 119 mg/L, 21 147 mg/L (range, 48–425) 22 and 278 mg/L (range, 54–711), 19 were all below the level at which propylene glycol is shown to interfere in the enzymatic ethylene glycol method. 2,3-Butanediol is a colourless, viscous liquid used as a precursor to various plastics and rubbers. It has been suggested that it may be formed as a novel product of intermediary metabolism in individuals with chronic alcoholism, with a maximum concentration of 34 mg/L (375 µmol/L) observed in chronic alcoholics. 23 This concentration is lower than that which caused the interference that we observed for the enzymatic assay in the present study. The enzymatic conversion of 2-butanone (methyl ethyl ketone), a compound found in denatured alcohol preparations, to 2,3-butanediol has been observed in Swedish alcoholics who have consumed such preparations. 24 2,3-Butanediol, at concentrations of up to 315 mg/L (3.5 mmol/L), have been observed in individuals who have consumed solutions containing 2-butanone. Values this high would interfere with the enzymatic method outlined here and have also been reported to interfere with other GC and enzymatic methodologies.14,24,25 Such interference is potentially problematic, but can be mitigated by inspection of the reaction curves of positive results as described above for propylene glycol. Moreover, because of the unusual nature of denatured alcohol consumption, we believe that it should not preclude the implementation of this method. It is interesting to note that the modified Catachem method used by Juenke et al. 14 also showed interference from propylene glycol at concentrations > 1000 mg/L and from 2,3-butanediol at a similar concentration to that described in the present study (250 mg/L). 14
In conclusion, we have successfully validated an automated assay for the accurate and reliable measurement of ethylene glycol in human plasma. The assay is readily available 24/7 in order to support the investigation of poisoned patients across the country.
Footnotes
Acknowledgements
We would like to thank the laboratory staff at Heartlands Hospital for their kind assistance and expertise during this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by Department of Clinical Chemistry, Immunology & Toxicology, Heart of England NHS Foundation Trust, UK.
Ethical approval
None applicable.
Guarantor
SG.
Contributorship
AJL and SG conceived and designed the study. AFR, AJL, LL and SG carried out all experiments and data analysis. AJL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
