Abstract
Background
Measurement of aldosterone and/or renin is essential to aid the differential diagnosis of secondary hypertension, guide strategy for therapeutic management of hypertension and assess adequacy of mineralocorticoid replacement.
Aim
The objective was to establish normative data for aldosterone and renin using the Immunodiagnostic Systems specialty immunoassay system platform in a Caucasian population.
Methods
Following informed consent, 365 subjects were recruited to this study. Subjects were ambulatory and attended clinic for blood pressure measurement and phlebotomy between the hours of 7:00 and 11:00. Blood pressure was measured according to the 2013 European Society of Hypertension/Cardiology guidelines. The inclusion criteria: age ≥18 years, BMI <30 kg/m2, non-pregnant, blood pressure <140/90, normal electrolytes and kidney function and not taking prescribed/over the counter medications. Ninety-four subjects were excluded based on these criteria. A total of 271 volunteers (females n = 145), aged 18–65 years formed the reference cohort. Blood for aldosterone/renin was collected into ethylenediaminetetraacetic acid specimen tubes. Samples were kept at room temperature and transported within 30 min of blood draw to the laboratory for immediate processing (centrifugation, separation and freezing of plasma). Plasma was stored at −20℃ prior to analysis on the Immunodiagnostic Systems specialty immunoassay system instrument.
Results
The established reference intervals in an Irish Caucasian population for renin: females: 6.1–62.7 mIU/L, males: 9.0–103 mIU/L, for aldosterone: females: <138–1179 pmol/L, males: <138–670 pmol/L, respectively.
Conclusion
This study demonstrates that reference intervals for aldosterone and renin should be gender specific. These automated immunoassays offer rapid stratification of patients with refractory hypertension and will better facilitate the optimization of therapeutic management.
Introduction
Hypertension is a common condition and is a significant cause of cardiovascular morbidity and mortality. Approximately 90% of people with hypertension have ‘essential’ hypertension, where no clear identifiable cause is found. 1 The remainder have an underlying secondary cause attributable to renal or adrenal pathology. Biochemical assessment of the renin-angiotensin-aldosterone system (RAAS) has recently assumed a much greater role in clinical practice largely attributable to the emerging evidence that primary aldosteronism (PA) is a much more common cause of hypertension than previously thought, accounting for as many as 5 to 13% of cases.2–5 The detection of PA is of particular importance, not only because it provides an opportunity for a targeted treatment, but also because it has been extensively demonstrated that patients affected by PA are more prone to cardiovascular events and target organ damage than patients with essential hypertension.6,7 The current mainstay of biochemical investigation of RAAS is by antibody-based radioimmunoassay for plasma aldosterone concentration (PAC), and plasma renin activity (PRA) or direct renin concentration (DRC). The ratio of aldosterone to renin, aldosterone renin ratio (ARR), first proposed by Hiramatsu et al., 8 commonly using PAC:PRA, is recommended as the most reliable available means of screening for PA.9,10 It is argued that renin measurement has more impact on the derivation of the ARR than aldosterone measurement. 11 However, the clinical importance of aldosterone measurement cannot be overstated. Reliable measurement is critical both to definitively confirm or exclude PA, and to adrenal venous sampling, the results of which largely determine whether a patient is a candidate for surgery or medical management. 12 Schirpenbach et al. 13 highlighted many of the deficiencies with direct (without extraction) immunoassay measurement of aldosterone, in particular the lack of comparability of results produced using these different assays. More recently, a comprehensive study assessing two automated direct aldosterone immunoassays (Liaison and Immunodiagnostic Systems specialty immunoassay system [IDS-iSYS] methods), again found significant proportional bias in result comparison between assays. These authors concluded that translating the aldosterone results of these assays into clinical practice mandates the establishment of method-specific reference ranges. 14
Sex-specific differences in the RAAS have been well described. 15 Oestrogens increase angiotensinogen concentrations and decrease renin concentrations, angiotensin-converting enzyme activity, the density of angiotensin II receptor type 1 and aldosterone production. 15 Pizzolo et al. 16 reported higher aldosterone and renin concentrations in the luteal phase compared to the follicular phase in healthy normotensive females (n = 33) but no difference in the mean ARR. 16 Recently, Kerstens et al., 17 in a study to determine reference values for the ARR in normotensive individuals showed that circulating aldosterone, PRA and PRC are influenced by gender and suggested that sex-specific reference intervals were needed.
The development of superior techniques to investigate the enzymes and hormones in RAAS has proved challenging. 18 A new era of diagnostics is emerging with the advent of automated immunochemiluminometric assays (ICMA) for PAC and DRC that are guideline acceptable 9 with the aldosterone (PAC) assay referenced to liquid chromatography-tandem mass spectrometry (LC-MS/MS) and the renin (DRC) assay to the WHO International Standard (IS) code 68/356. These routine high-throughput methods have the potential to better facilitate the investigation of patients with resistant and endocrine-related hypertension. However, to ensure accuracy of result interpretation and patient safety, laboratory specialists need to be knowledgeable of the analytical characteristics and performance of both their aldosterone and renin assays. Further, it is paramount that the performance of these assays is reviewed regularly in liaison with the clinical users of the laboratory service.
Recently, Immunodiagnostic Systems (IDS) has launched two newly developed ICMA for PAC and DRC using ethylenediaminetetraacetic acid (EDTA) plasma for use on the IDS-iSYS automated immunoassay platform. The objective of the current study was to verify the analytical performance characteristics and establish normative data for PAC, DRC and ARR using the newly developed ICMA on the IDS-iSYS automated system in an Irish Caucasian population.
Materials and methods
The study was conducted simultaneously in two major teaching hospitals with a standardized protocol for subject selection, phlebotomy, blood pressure (BP) measurement and analytical parameters. The import of the study, the pro-forma and the rationale for the requested information was explained to all subjects who volunteered to participate in this study. The study pro-forma included a section requesting the details of all medications (prescribed and/or over the counter (OTC) medications be accurately recorded together with the date of the last dose/prescription. Those recruiting and consenting participants were instructed to give examples of particular medication types (antihypertensives and oral contraceptive pill [OCP]/hormone replacement therapy [HRT]) and also to explain the term OTC medications and again give examples, e.g. non-steroidal anti-inflammatory drugs (NSAID). Subjects recruited to this study were not taking prescribed/OTC medications for a minimum of three months.
Ethical permission for this collaborative study was obtained in accordance with the Declaration of Helsinki and was granted by each Institution’s Clinical Research Ethics Committees.
Study inclusion criteria
The study inclusion criteria were: age ≥18 years, BMI <30 kg/m2, non-pregnant, BP <140/90, normal electrolytes and kidney function (estimated glomerular filtration rate [eGFR] > 60 mL/min/1.73 m2), non-smoker, Caucasian and not taking prescribed/OTC medications for a minimum of three months.
Participants
Following informed written consent, the authors recruited 365 participants from the local population to this study. Subjects had unrestricted salt intake, were fasted overnight, ambulatory and required to attend clinic for BP measurement and phlebotomy between the hours of 7:00 and 11:00. BP was measured according to the 2013 European Society of Hypertension/Cardiology guidelines. 19 All subjects were seated for 10–15 min prior to having whole blood (10 mL) drawn and collected into appropriate specimen tubes (Becton Dickinson/Sarstedt plastic vacutainer), potassium EDTA plasma for the measurement of PAC/DRC and plain (serum) for the measurement of sodium, potassium and creatinine. The eGFR was calculated using the Modification of Diet in Renal Disease (MDRD) Study equation. Specimen tubes were kept at room temperature (RT) and transported within 30 min of blood draw to the laboratory for immediate processing (centrifugation, separation and freezing of plasma). Plasma was stored in a temperature-controlled freezer at −20℃ for a maximum of six months prior to analysis on the IDS-iSYS instrument. Serum samples were analysed for electrolytes and creatinine within 2 h of blood draw. At this visit, basic clinical information including age, gender, ethnicity, body mass index (BMI), current medication usage (prescribed/OTC) smoking and pregnancy status was recorded. In reproductive female participants, no record of the stage of the menstrual cycle was taken at the time of sample collection.
Baseline demographics of the study cohort.
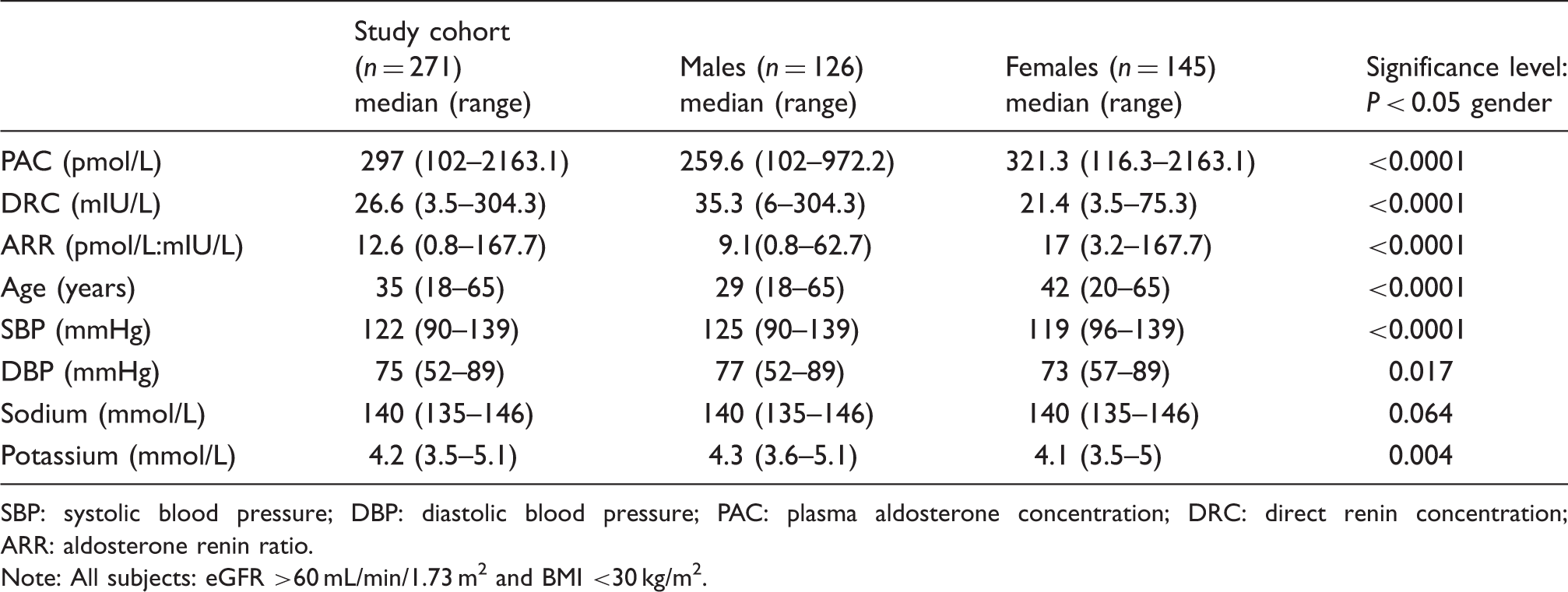
SBP: systolic blood pressure; DBP: diastolic blood pressure; PAC: plasma aldosterone concentration; DRC: direct renin concentration; ARR: aldosterone renin ratio.
Note: All subjects: eGFR >60 mL/min/1.73 m2 and BMI <30 kg/m2.
Analytical methods
The IDS-iSYS is an automated analyser based on chemiluminescent technology. The DRC assay is a sandwich ICMA employing two monoclonal antibodies, a magnetic particle solid-phase capture antibody and an acridinium-labelled tag antibody. Concentration of renin is directly proportional to light (expressed in relative light units) emitted by the acridinium label and measured by the system luminometer. The DRC assay is calibrated against the WHO (National Institute for Biological Standards and Control) IS code 68/356.
The PAC assay is a competitive one-site immunometric assay that uses a biotinylated monoclonal antibody bound to streptavidin-coated magnetic particles. Acridinium-labelled aldosterone competes with sample aldosterone for the limited amount of biotinylated antibody. Concentration of aldosterone is inversely proportional to light emitted by the acridinium label and measured by the system luminometer. The IDS-iSYS PAC assay is referenced to LC-MS/MS. Results from each assay were used to calculate the ARR, i.e. PAC in pmol/L divided by the DRC in mIU/L to give the ARR in pmol/L: mIU/L.
Each laboratory undertaking this study is accredited to ISO15189:2012 standard for medical laboratories. An IDS-iSYS analyser was available at each site. During the study period and across both sites, one reagent, calibrator and control lot were used. Patient samples were analysed in batches at each site. Aliquots of patient samples were exchanged between sites to ensure results were in accord. Internal Quality Control (IQC) performance between sites was reviewed to ensure results were within specified limits.
Assay volume is an important factor to consider when introducing a new assay (adult/paediatric patients). We recommend a minimum volume of 500 µL of plasma to assay both analytes simultaneously. We verified the manufacturer’s claims for time to first test result and throughput. From successful calibration and assay initiation, the first result is released within 43 min and test throughput is confirmed as 76 reportable tests per hour.
Precision and trueness studies were performed in accordance with the Clinical and Laboratory Standards Institute (CLSI) experimental protocol 15 guideline 20 (replicates (minimum of three) each day for five days) using manufacturer independent quality control (Randox Immunoassay Speciality II; levels 1–3 for DRC and BIO-RAD Lyphochek Immunoassay Plus Controls; levels 1–3 for PAC) or pooled patient plasma. Additionally, the aldosterone assay was compared to LC-MS/MS. Split aliquots of patient samples (n = 48) were sent to an accredited LC-MS/MS laboratory in Manchester for analysis as part of our assay verification protocol. This laboratory is a referral laboratory and was engaged commercially for the analysis of these samples. The method has been published in detail. 21
Statistical analysis
Reference values within the IDS-iSYS PAC and DRC assay reportable ranges were used to establish the respective reference intervals. In 11 of 271 control subjects (4%), aldosterone values were below the assay reportable range (102.5 pmol/L) and for statistical purposes were assigned the arbitrary figure of 102 pmol/L.
The Minitab Release (version 16) statistical software was used to illustrate the data. The frequency distributions for PAC/DRC/ARR in healthy Caucasian normotensive Irish men and women were established. The requirement to partition for gender was evaluated using the Harris and Boyd’s standard deviate test. 22 Outliers were assessed in accordance with the criteria proposed by Dixon 23 and Reed et al. 24 This approach to statistically significant outliers is supported by the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) working group. 25 Boxplots were used to illustrate the data for PAC/DRC/ARR in healthy male and female subjects. The box portion of the box and whisker plot includes 50% of the data, the lower, median (represented by a line) and upper quartile. The whiskers extend to the minimum and maximum values. Disconnected points are potential outliers.
Partitioning for gender
The data were divided according to gender and then subjected to the Harris and Boyd’s standard deviate test
22
to investigate whether partitioning for gender was required.
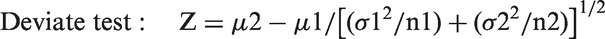
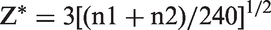
Briefly, the statistical Z value is compared with a critical Z* value. If the calculated Z value is greater than the critical Z* value (Z* = 3), partitioning is recommended.
Outlier removal
Outliers are atypical, infrequent observations that lie outside the true distribution of a healthy reference population. 26 Histograms representing the frequency distributions of the data were visually examined for the presence of outliers. Outliers were not automatically excluded. Outliers were first checked to out rule transcriptional, preanalytical and analytical (haemolysis, lipaemia) errors or the inclusion of inappropriate reference values. An outlier was removed based on the difference between an extreme observation and the next observation (D), divided by the range of all observations including the extremes (R). This is referred to as the D/R ratio.23,24 An outlier was excluded when D was one-third or greater than the range R as proposed by the IFCC working group. 25 When multiple outliers are on the same side of the distribution, the one-third rule may fail to identify the extreme outlier, and therefore mask slightly less extreme outliers. In this case, the one-third rule should be applied to the least extreme outlier. If the rule rejects this outlier then the more extreme outliers will automatically be rejected. If the rule does not reject this outlier then all the outliers should be included or an alternative technique that considers all the outliers together should be applied. 25
Establishing the reference intervals
To determine the reference interval, the IFCC non-parametric method, which does not assume Gaussian type distribution, was employed. 25 For the analyte assessed, the lower and upper reference limits were estimated at the 2.5th and 97.5th percentiles, respectively. The percentiles are determined by excluding a required percentage of the values in each tail of the reference distribution. In this method, the data are ranked. First, n is equal to the total number of values in the distribution. The values are sorted in ascending order with rank 1 given to the minimum value; the next value has rank 2 and so on, until the maximum reference value has the rank n. The reference limits are determined according to rank number equal to 0.025(n + 1) and 0.975(n + 1), respectively. The percentiles represent the original reference value that corresponds to the computed rank value.
Results
Performance of the IDS-iSYS PAC and DRC assays
The between-run analytical coefficient of variation (CVA%) at a mean PAC of 238 pmol/L, 442 pmol/L and 1648 pmol/L was 9.71%, 9.37% and 3.83%, respectively. The mean bias of the assay as determined by comparison to LC-MS/MS 21 (n = 48, target range: 100–2037 pmol/L) was 38 pmol/L (C.I. 6.7–69.3), Passing–Bablok regression: IDS-iSYS = 1.117 LC-MS/MS (CI: 1.029–1.213)–12.67 (Confidence Interval (CI) −39.15–12.72), P = 0.65 and Spearman correlation, r = 0.986. Using pooled patient EDTA plasma, we determined the limit of quantitation (LoQ) for PAC to be 138 pmol/L. The latter was the lowest PAC reproducibly measured (n = 10) with an intra-assay CVA% of ≤10%. Notably, at a mean PAC of 126 pmol/L within run assay precision (n = 10) was 15.9%, less than half the reported intraindividual biological variation of 37% 27 and hence, clinically acceptable.
The between-run analytical CVA% at a mean DRC of 14 mIU/L, 100.3 mIU/L and 390.2 mIU/L was 7.7%, 8.4% and 4.9%, respectively. The mean bias of the assay as determined by proficiency testing through the Randox International Quality Assessment Scheme (RIQAS) (n = 15, target range: 17.7–116.5 mIU/L) was 0.5 mIU/L (C.I. [−0.7–1.8]).
Analytical performance characteristics for the PAC and DRC assays.
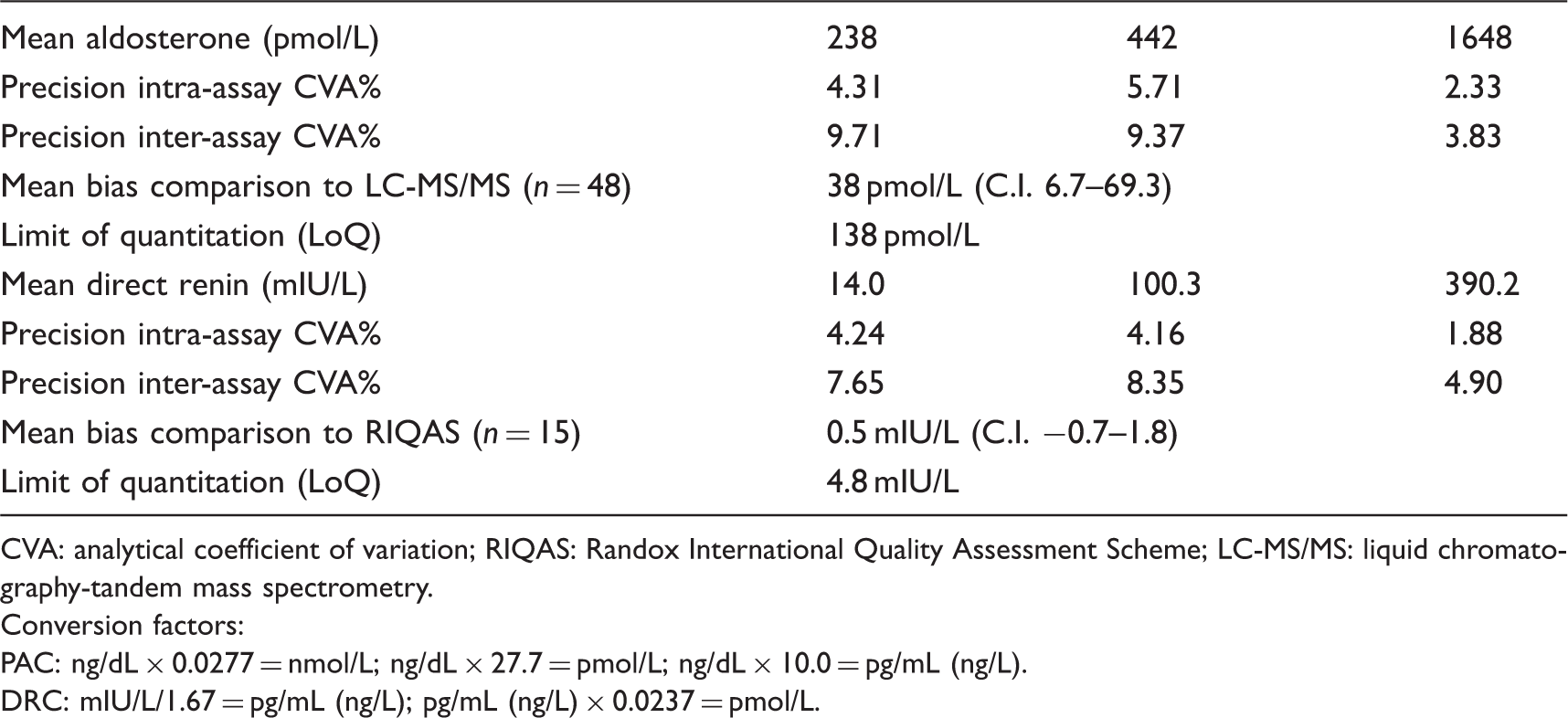
CVA: analytical coefficient of variation; RIQAS: Randox International Quality Assessment Scheme; LC-MS/MS: liquid chromatography-tandem mass spectrometry.
Conversion factors:
PAC: ng/dL × 0.0277 = nmol/L; ng/dL × 27.7 = pmol/L; ng/dL × 10.0 = pg/mL (ng/L).
DRC: mIU/L/1.67 = pg/mL (ng/L); pg/mL (ng/L) × 0.0237 = pmol/L.
Reference intervals
Partitioning for gender
Application of the Harris and Boyd deviate test to the data determined that the calculated Z for PAC, DRC and ARR in healthy male and female subjects were 3.93, 6.62 and 6.18, respectively. Each calculated Z value is >critical Z*(3.18); therefore, partitioning for gender is recommended.
Outlier handling
Once an outlier was found, that subject was removed entirely from the reference population. Initially and prior to partitioning, one male subject was removed while establishing the renin reference interval for the total cohort. Following the finding that partitioning was recommended, a further four subjects (male: n = 3, female: n = 1) were removed from the gender-specific cohorts while establishing the ARR reference interval.
Outlier removal – Example and calculation
Visual inspection of the frequency distribution of the ARR data for healthy male subjects indicated the presence of three potential outliers (data not shown). The minimum ARR value was 0.92 pmol/L:mIU/L and the maximum, 62.7 pmol/L:mIU/L giving the range (R) of 61.8 pmol/L:mIU/L. The least extreme probable outlier having an ARR value of 50.78 pmol/L:mIU/L and the next ARR observation, a value of 24.7 pmol/L:mIU/L. By applying the D/R ratio and the one-third rule to these data: (50.78–24.7)/61.8 = 0.42. The D/R is >0.33; hence, all three outliers are removed.
Establishing the reference interval
Established reference Intervals for PAC, DRC and ARR in healthy male and female subjects.
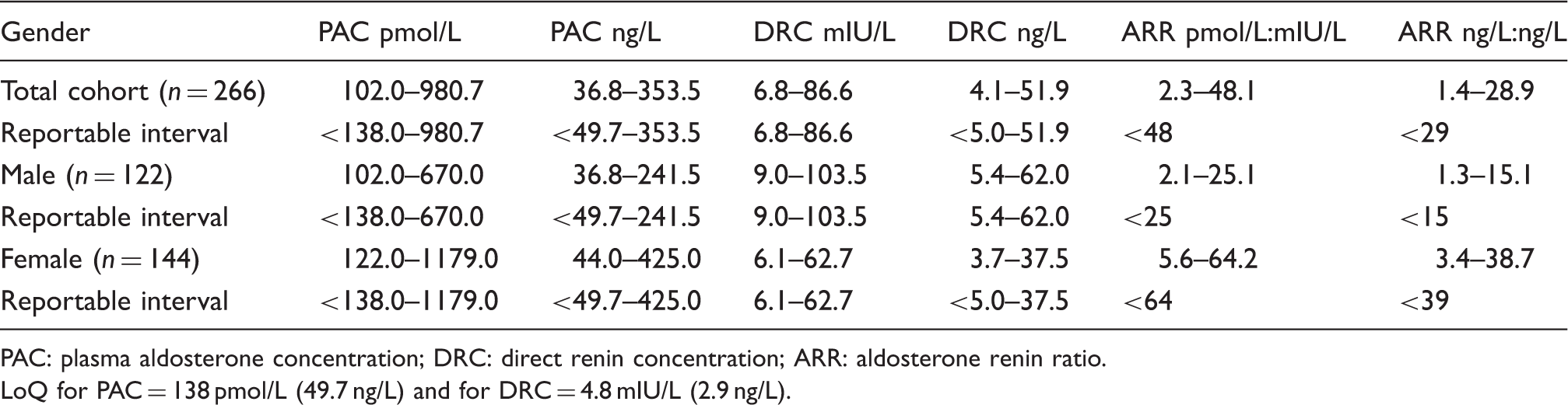
PAC: plasma aldosterone concentration; DRC: direct renin concentration; ARR: aldosterone renin ratio.
LoQ for PAC = 138 pmol/L (49.7 ng/L) and for DRC = 4.8 mIU/L (2.9 ng/L).
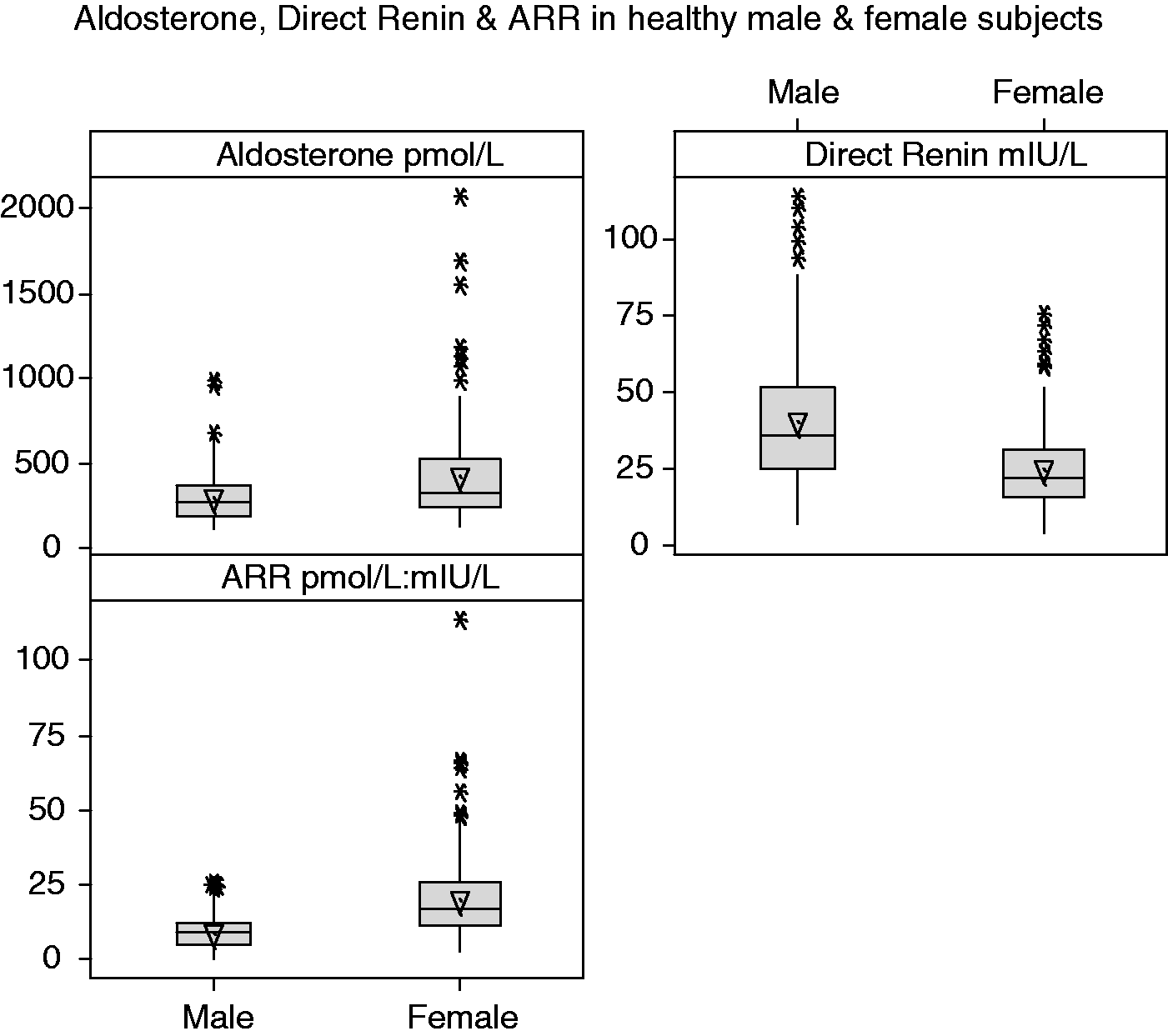
Boxplot of PAC, DRC and ARR in healthy male (n = 122) and female (n = 144) subjects.
Discussion
Laboratory diagnostics underpin the majority of investigations in clinical medicine. Biochemical assessment of the RAAS is not unique in this regard. It is apparent that existing methods for measuring aldosterone and renin vary considerably and there are significant differences in results and the units used to report results between different laboratories.9,10,18,28–30 It is universally agreed that better diagnostics in this area would have significant patient benefit, not only in better diagnosis of cases of adenoma-related hypertension, but in the future tailoring of anti-RAAS therapeutics.31–33 LC-MS/MS offers analytical superiority; however, aldosterone is a challenging assay requiring mass spectrometers with higher orders of sensitivity, which are currently costly and not as accessible to routine clinical laboratories as an automated immunoassay.
We performed a verification of the analytical performance specifications for the IDS-iSYS PAC and DRC ICMAs in two major teaching hospital laboratories. The assays were found to be precise, CVA ≤10% at low, medium and high analyte concentrations and demonstrated minimal bias (Table 2). The functional sensitivity of the DRC assay was verified at 4.8 mIU/L and established for the PAC assay at 138 pmol/L.
Deficiencies in automated assays for aldosterone measurement without extraction have been highlighted previously with particular focus on analytical sensitivity and calibration.12,13
These authors while appreciating the appeal of automated systems (speed, efficiency) advocate for accuracy of aldosterone measurement and suggest that the best way to achieve this is by calibration against mass spectrometry. The IDS-iSYS aldosterone assay is referenced to LC-MS/MS. In addition, we found aldosterone was detectable in 260 of 271 (96%) normal healthy subjects.
Using a standardized pretesting blood-sampling protocol and the Harris and Boyd deviate test, we determined that partitioning of reference intervals for gender was recommended for PAC, DRC and ARR (Table 3). We established reference intervals (total cohort) for PAC (<138–980.7 pmol/L) and DRC (6.8–86.6 mIU/L) that are broadly in accord with those provided by the manufacturer (Upright PAC: 102.5–1196.6 pmol/L and DRC: 5.3–99.1 mIU/L defined in Caucasian adults [n = 243, aged 18–65 years, with normal BP, BMI and using whole blood collected into EDTA between 7 and 10 am]). Further, the reference interval we established for DRC is almost identical to that quoted by De Bruin et al. 34 of 6–85.5 mIU/L in their study of 80 normotensive individuals (males [n = 25], aged 16–75 years) using an ICMA calibrated to the WHO IS code 68/356.
We found that female gender had a positive effect on aldosterone and ARR and a negative effect on renin. These findings support the contention for the necessity of gender-specific intervals made by Kerstens et al. 17 However, unlike the latter study, our study was appropriately powered to yield gender-specific reference intervals. Moreover, using different assays for both aldosterone and renin than those employed in Kersten’s study, 17 our findings are further corroborated by those of Newton-Cheh et al. in the Framingham study, 35 and the first large population based study reported thus far by Hannemann et al. 36
This gender disparity will likely impact medical decision threshold values for the ARR in clinical practice and further study of these implications is warranted.
We acknowledge the age difference between female and male subjects (median age [range]: 42 [20–65] versus 29 [18–65] years, respectively) and that the study was not powered to investigate the influence of age. The import is that others have found renin decreases and the ARR increases with progressing age. 35
Dietary salt influences aldosterone, and a potential limitation of this study may be the lack of an objective assessment of the sodium intake of participants. Of note, the salt intake of the reference cohort, in accord with the Endocrine Society Clinical Practice Guideline was unrestricted prior to testing, 9 suggesting that subjects would have been on their normal diet or the average diet of the Irish population. A recently published paper assessed the salt consumption levels in Irish men and women using urinary data and found them to be high. 37 Estimated intakes in men and women aged 18–64 years, were 11.1 g/day and 8.5 g/day, respectively, and in those over 65 years of age, 11.0 g/day and 6.8 g/day, respectively. 37
We endeavoured to ensure that subjects recruited to this study were not taking any medications (prescribed/OTC, e.g. antihypertensives, OCP/HRT/NSAID) known to affect the ARR.38–40 Notwithstanding, we acknowledge that we are reliant on the accurate completion of the pro-forma by study participants. Further, in reproductive female participants, no account of the stage of the menstrual cycle was taken. The significance is that false-positive ARRs have been found in normal women tested during the luteal phase of their menstrual cycle when renin concentration is used.16,41
The ARR, used as the primary screening tool for PA, is a highly variable test with sensitivity ranging from 70 to 80% and specificity ranging from 70 to 90%. 5 A limitation of the present study is that we examined only normotensive individuals. However, ARR values in patients with PA have been shown to be considerably higher than those in healthy controls and essential hypertensives (EH) patients.42–46 Preliminary clinical studies, using this control cohort (n = 266) and 15 patients in whom PA was confirmed, were undertaken to determine the appropriate ARR cut-off for case detection of PA in our population. Receiver Operating Characteristics analysis determined that an ARR cut-off at 35.4 pmol:mIU/L provides diagnostic sensitivity and specificity of 100% and 94%, respectively. Sustarsic et al., using these same assays in healthy controls (n = 261), EH (n = 203) and PA patients (n = 140), determined that an ARR at 30.5 pmol:mIU/L discriminated between EH and PA subjects with a diagnostic sensitivity and specificity of 98.6% and 81.8%, respectively. 42 Of note, for discrimination between PA and healthy controls an ARR cut-off of at 24.8 pmol:mIU/L resulted in a diagnostic sensitivity and specificity of 100% and 94%, respectively.
We are currently reporting gender-specific reference intervals for the ARR together with the following interpretative comment, There is overlap in the ARR between individuals with and without PA. Assuming adherence to pretesting criteria (e.g. off potentially interfering medications) an ARR <25 pmol:mIU/L makes PA highly unlikely. However, the likelihood of PA increases significantly as the ARR rises above >35 pmol:mIU/L. All patients with positive screens require further investigation.
The advent of precise, accurate PAC and DRC ICMA that use a single sample type (EDTA plasma) and referenced to LC-MS/MS and WHO IS 68/356, respectively, is significant, as it will allow the use of international reference intervals and harmonization of medical decision threshold values. The test throughput and rapid turnaround times move these assays into the realm of the routine clinical biochemistry laboratory and hence will permit more timely and effective patient management.
Conclusion
We established the reference intervals for PAC, DRC and ARR in healthy Caucasian normotensive men and women using the IDS-iSYS immunoassay system. The finding of significant differences between the genders is an important consideration as to how these reference intervals are applied in the stratification of patients with refractory hypertension and optimization of therapeutic management of patients with hypertension, Addison’s disease and congenital adrenal hyperplasia.
Footnotes
Acknowledgements
We wish to express our gratitude to all the volunteers who made this study possible. Special thanks to the scientific and nursing staff of the Departments of Clinical Biochemistry at the Mater Misericordiae University Hospital, Dublin, Galway University Hospital and Bon Secours Hospital, Galway.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Mater & Mater Private Hospital Research Ethics Committee: Date of Approval: 22 January 14, Ref. No: 1\378\1614; Galway University Hospitals & Bon Secours Hospital System Clinical Research Ethics Committee: Date of Approval: 9 September 2013 and 14 September 2013, Ref. No: C.A. 969
Guarantor
MF.
Contributorship
POS and MF – conception, design, recruitment, consenting of participants, sample collection, assembly and analysis of data. JJB – recruitment, consenting of participants, sample collection analysis, quality and accuracy of laboratory methods. NG recruitment, consenting of participants, collection of samples, and renin method verification studies. MCD – recruitment consenting, phlebotomy, and measurement of BP of study participants. POS – first draft of manuscript. All authors reviewed, edited and approved the final version of the manuscript.
