Abstract
Background:
The renin-angiotensin-aldosterone system (RAAS) plays a crucial role in kidney development and the progression of chronic kidney disease (CKD).
Objectives:
To identify children with low birth weight (LBW) at risk of CKD who have RAAS activation.
Design:
We conducted a prospective cohort study to evaluate whether a history of LBW contributes to the development of latent RAAS activation using urine samples from patients with short stature with no clinical kidney symptoms. Additionally, among children who had idiopathic nephrotic syndrome (INS), we examined how a history of LBW contributes to the development of latent RAAS activation using residual kidney biopsy samples.
Methods:
We prospectively evaluated angiotensinogen (AGT) using spot urine in children with and without a history of LBW, who required evaluation for short stature without kidney symptoms at registration. We also performed immunohistochemical staining of AGT using kidney biopsy specimens of subjects with and without a history of LBW who had INS. Urinary AGT was assessed as a marker of intrarenal RAAS.
Results:
In 45 children (median age 5 years), urinary AGT/creatinine (Cr) levels were significantly higher in children with a history of LBW (n = 24) than in those without (n = 21, median: 12.6 vs 6.7 µg/g・Cr, mean: 15.4 vs 9.1 µg/g・Cr, P < .01). The unadjusted mean difference between the 2 groups and the 95% confidence interval were 6.3 and (1.6, 11.1), respectively. In the immunohistochemical kidney pathological study, the positive area of AGT staining in the kidney tubules of 3 subjects with a history of LBW was more extensive than that of 3 additional subjects without a history of LBW.
Conclusion:
Our results indicated that latent intrarenal RAAS activation in children with a history of LBW persists until early school age and may contribute to the progression of CKD.
Keywords
Introduction
With advances in perinatal department care, the prognoses for preterm birth (PB) or low birth weight (LBW) infants have improved significantly. However, PB and LBW are risk factors for the development and progression of various chronic diseases, 1 including chronic kidney disease (CKD), which primarily manifests as glomerular hypertrophy and tubulointerstitial fibrosis. 2 Understanding the mechanisms of CKD progression in children with a history of PB or LBW is crucial for not only improving long-term outcomes but also reducing healthcare costs associated with end-stage kidney disease management. The Development Origin of Health and Disease (DoHaD) hypothesis, which suggests that chronic diseases develop because of a mismatch between the postnatal environment and the developmental conditions of PB or LBW infants, has already been applied to CKD progression. 1 Moreover, reports have shown that nephron deficits and reduction in nephron number are involved in the progression of CKD.2,3
The effectiveness of renin-angiotensin-aldosterone system (RAAS) inhibitors in slowing the progression of CKD has been confirmed. 4 Similar to adult CKD patients, the frequency of introducing RAAS inhibitors in pediatric patients, whose CKD was caused by chronic nephritis or congenital kidney and urinary tract abnormalities, has been increasing.5 -9 In humans, RAAS-related genes, such as renin and angiotensinogen (AGT), are heavily involved in ureteric bud and renal collecting duct formation. 10 In particular, local RAAS only acts on kidneys and differs from circulating RAAS, which circulates throughout the body and depends on hemodynamics. 11 A report revealing that AGT is the origin of the fibrosis cascade in the renal tubulointerstitium has led to numerous subsequent molecular biological studies focusing on intrarenal RAAS in CKD. 12 Urinary AGT, which reflects the activation of intrarenal RAAS, has been shown to be an indicator for the prognosis of kidney function 13 ; moreover, gestational age and urinary AGT in neonates are significantly correlated. 14
Recently, the concept of diseases characterized by compensatory glomerular hypertrophy due to hyperfiltration, resulting from reduced nephron number, has become widespread. 15 Various background factors have been reported in many studies as potential contributors to reduced nephron numbers. For example, a history of PB or LBW had been shown to be associated with inadequate formation of the ureteric bud and renal collecting duct. The relationship between the RAAS, which plays a crucial role in both kidney development and the progression of CKD, and a history of PB or LBW with unknown mechanisms of CKD progression has not yet been examined. Verification of whether intrarenal local RAAS activation, which is thought to be involved during the neonatal period, 14 persists into early school age and adolescence in those with a history of LBW before the onset of CKD would support the DoHaD hypothesis.
Here, we report a prospective study to identify children with LBW at risk of CKD who exhibit RAAS activation.
Materials and Methods
We conducted a main prospective study and additional analysis at Kobe University Hospital, Kakogawa Central City Hospital, and Takatsuki General Hospital. In the main prospective study, we evaluated the difference in the level of urinary RAAS marker in children with short stature with no clinical kidney symptoms between those with and without a history of LBW from August 2019 to December 2023. As an additional analysis, among children with idiopathic nephrotic syndrome (INS), we compared immunohistochemical staining of RAAS markers in subjects with and without a history of LBW from September 2016 to December 2023.
Study Design
Our overall objective of the study was to identify which children with LBW at risk of CKD have RAAS activation. The objective of the main study was to evaluate how a history of LBW contributes to the development of latent RAAS activation using urine samples from patients with short stature with no clinical kidney symptoms. The aim of the additional analysis was to evaluate how a history of LBW contributes to the development of latent RAAS activation using residual kidney biopsy samples from patients with INS.
Setting
The overall setting of the main study included a sample size of 50 cases, with the observation period defined as the duration of continuous regular follow-up at our department. Evaluation points were defined as regular medical follow-up visits, and background factors were extracted from electronic medical records at the time informed consent was obtained. We prospectively collected the following data during the evaluation for short stature: blood pressure, urinalysis (including urinary AGT, urinary neutrophil gelatinase-associated lipocalin [NGAL], urinary β2 microglobulin [β2MG]), blood examination (including serum creatinine [Cr], serum AGT, serum cystatin C [CysC]), CysC-estimated glomerular filtration rate (CysC-eGFR), and kidney ultrasonography findings. We also collected data on sex, gestational age, birth height, and birth weight. For the additional analysis, neither sample size nor observation period were defined for the retrospective study. Evaluation points for the additional analysis were set as regular medical follow-up visits, and background factors were extracted from electronic medical records at the time informed consent was obtained. Immunohistochemical staining of AGT was used as a marker of intrarenal RAAS, and angiotensin II (AT2) was used as a marker of systemic RAAS.
Participants
The subjects in the main study comprised children aged under 10 years who had no clinical kidney symptoms and had undergone evaluation for short stature on admission. The children were divided into 2 groups: those with and without a history of LBW. We excluded those with a genetic abnormality, bone metabolic disease, cardiopulmonary disease, congenital anomalies of the kidney and urinary tract, and CKD. Participants in the main study were categorized into 3 subgroups (ie, children with a history of PB and LBW, those with a history of LBW but no PB, and those without a history of PB or LBW) for the subgroup analysis. The subjects in the additional analysis comprised children who underwent kidney biopsy during remission of INS. The children with INS were divided into 2 groups: those with and without a history of LBW.
Variables
The primary outcome of the main prospective study was the difference in the level of urinary AGT/Cr in children with no clinical kidney symptoms between those with and without a history of LBW. The overall exposures in the main prospective study were background, kidney symptoms, various blood and urine findings, and kidney ultrasound findings. The outcome of the additional analysis using kidney biopsy specimens was differences in immunohistochemical staining of AGT and AT2 in INS subjects with and without a history of LBW. The overall exposures in the additional analysis were background, kidney symptoms, various blood and urine findings, and kidney ultrasound findings.
Data Sources
Urinary and serum total AGT was evaluated using a commercially available enzyme-linked immunosorbent assay Kit (#27412 Human Total Angiotensinogen Assay Kit – IBL, Immuno-Biological Laboratories Co. Ltd., Fujioka, Japan) using spot urine and serum samples. A commercially available mouse monoclonal antibody (#10417 anti-human angiotensinogen [104AT 601.2.80] mouse immunoglobulin G [IgG] monoclonal antibody, Immuno-Biological Laboratories Co. Ltd., Fujioka, Japan) was used for immunohistochemical staining of AGT as a marker of intrarenal RAAS. A commercially available human, rabbit polyclonal antibody (H002-12 Anti-Human Angiotensin II, Phoenix Pharmaceuticals Co. Ltd., Burlingame, CA) was used for immunohistochemical staining of AT2 as a marker of systemic RAAS. Kidney biopsies were performed by pediatric nephrologists. We measured the glomerular diameter by picking up only the glomeruli with a vascular pole. After measuring all glomeruli, we averaged the diameter of the 3 longest glomeruli. A mean glomerular diameter greater than 220 µm was considered glomerular hypertrophy. 13
Bias
For the main study, we did not include healthy children without short stature who have a history of LBW. Therefore, there may have been selection bias because we only included children with a history of LBW who had short stature on admission. Furthermore, because the additional analysis was a retrospective study, various biases are likely.
Study Size
For the prospective urine biomarker study in children with a history of LBW who had no clinical kidney symptoms, we calculated a sample size of 50 children. At our institution, the number of patients who undergo evaluation for short stature with a history of LBW is approximately 20 per year. We assumed that approximately 30% to 40% of children would be ineligible because of non-consenting issues (ie, 12-14 children × 4 years = 48-56). Therefore, to satisfy these assumptions, we calculated a sample size of 50 children over 4 years.
Quantitative Variables
Urine protein concentration was measured to calculate an early morning urine protein Cr (P/C) ratio. The severity of hematuria was assessed by counting the number of red blood cells (RBCs) per high-power microscopic field in a centrifuged early morning urine sample; fewer than 5 RBCs per high-power field indicated the absence of hematuria. We calculated the CysC-eGFR of each patient using their serum CysC and height. 16 Hypertension was defined according to the International Pediatric Hypertension Association criteria. 17 We defined birth at less than 37 weeks of gestation as PB and a birth weight of less than 2500 g as LBW. PB was used exclusively as a grouping variable for the subgroup analyses of the main prospective study.
Statistical Analysis
Comparisons of categorical and continuous variables were conducted using t-tests; if unequal variances were detected, we used the Wilcoxon rank-sum test. Fisher’s exact test was used to examine associations between categorical variables.
For the subgroup analysis of the main study, we first divided the subjects into 3 groups: children with a history of PB and LBW, those with a history of LBW but no PB, and those without a history of PB or LBW. Those that showed significant differences in the Kruskal-Wallis test were further analyzed using pairwise comparisons between groups using Dunn’s test to correct for multiple testing. We performed all analyses using JMP version 11.0 (SAS Institute Japan Ltd., Tokyo, Japan). All data are expressed as medians (interquartile ranges [IQRs]) and means (standard deviations [SDs]), and the threshold for significance was P < .05.
Results
Clinical Characteristics and Results of Biomarkers in the Main Prospective Study
A total of 65 children who underwent examination for short stature were evaluated on admission to our hospital (Figure 1). Of these, 45 children (27 boys and 18 girls) were eligible for the main prospective study, which comprised 21 children with a history of LBW and 24 children without a history of LBW. Among children with a history of LBW, 14 children had a history of PB.

Patient flow chart. Of the 65 children who underwent evaluation for short stature on admission, 45 children were included in the study. Abbreviation: LBW, low birth weight.
Table 1 shows the comparison of clinical characteristics of children with and without a history of LBW. The median age of children with and without a history of LBW was 5.0 and 4.6 years, respectively. There were no significant differences in the sex ratio or median age between children with and without a history of LBW. There was no significant difference in urinary Cr levels between the groups. None of the children had significant hematuria, proteinuria, or abnormal levels of urinary NGAL/Cr. In 3 cases, CysC values were recorded as 0 because of sampling errors (eg, poor storage conditions or technical errors during measurement), which made it impossible to calculate the eGFR. Thus, CysC was analyzed in all cases except these 3. Four children (10.8%) had hypertension. Systolic blood pressure and the proportion of children with hypertension were not significantly higher in those with a history of LBW than in those without a history of LBW. Serum AGT/Cr levels differed significantly between children with and without a history of LBW. For the primary outcome for the main prospective study, urinary AGT/Cr levels were significantly higher in children with a history of LBW than in those without a history of LBW (median: 12.6 vs 6.7 µg/g・Cr, mean: 15.4 vs 9.1 µg/g・Cr, P < .01; Figure 2a). The unadjusted mean difference between the 2 groups and the 95% confidence interval were 6.3 and (1.6, 11.1), respectively. The comparisons of clinical characteristics of children for the subgroup analysis of the main study are shown in Table 2. Urinary AGT/Cr levels differed significantly among the 3 subgroups. The pairwise comparisons showed no significant difference in urinary AGT/Cr levels between children with a history of PB and LBW and those with a history of LBW but no PB, as well as between children with a history of PB and LBW and those without a history of LBW nor PB (Tables 3-5 and Figure 2b). Owing to the high density of overlapping data points, not all individual values could be visualized in the graph. Thus, the complete set of raw individual data is included in the Supplemental Material.
Comparison of Clinical Characteristics in Children With or Without a History of Low Birth Weight.

Abbreviations: AGT, angiotensinogen; Cr, creatinine; CysC, cystatin C; eGFR, estimated glomerular filtration rate; LBW, low birth weight; NGAL, neutrophil gelatinase-associated lipocalin; β2 MG, β2 microglobulin.
All data except for * were presented using median values (interquartile range) and mean value ± standard deviation. All data except for * were analyzed using the Wilcoxon rank-sum test.
Forty-two children with data for CysC and CysC-eGFR.
Evaluated 43 children with data for serum angiotensinogen.
Evaluated 44 children with data for NGAL/Cr.
Evaluated 37 children with data for systolic blood pressure.
Analyzed using the Fisher’s exact test.
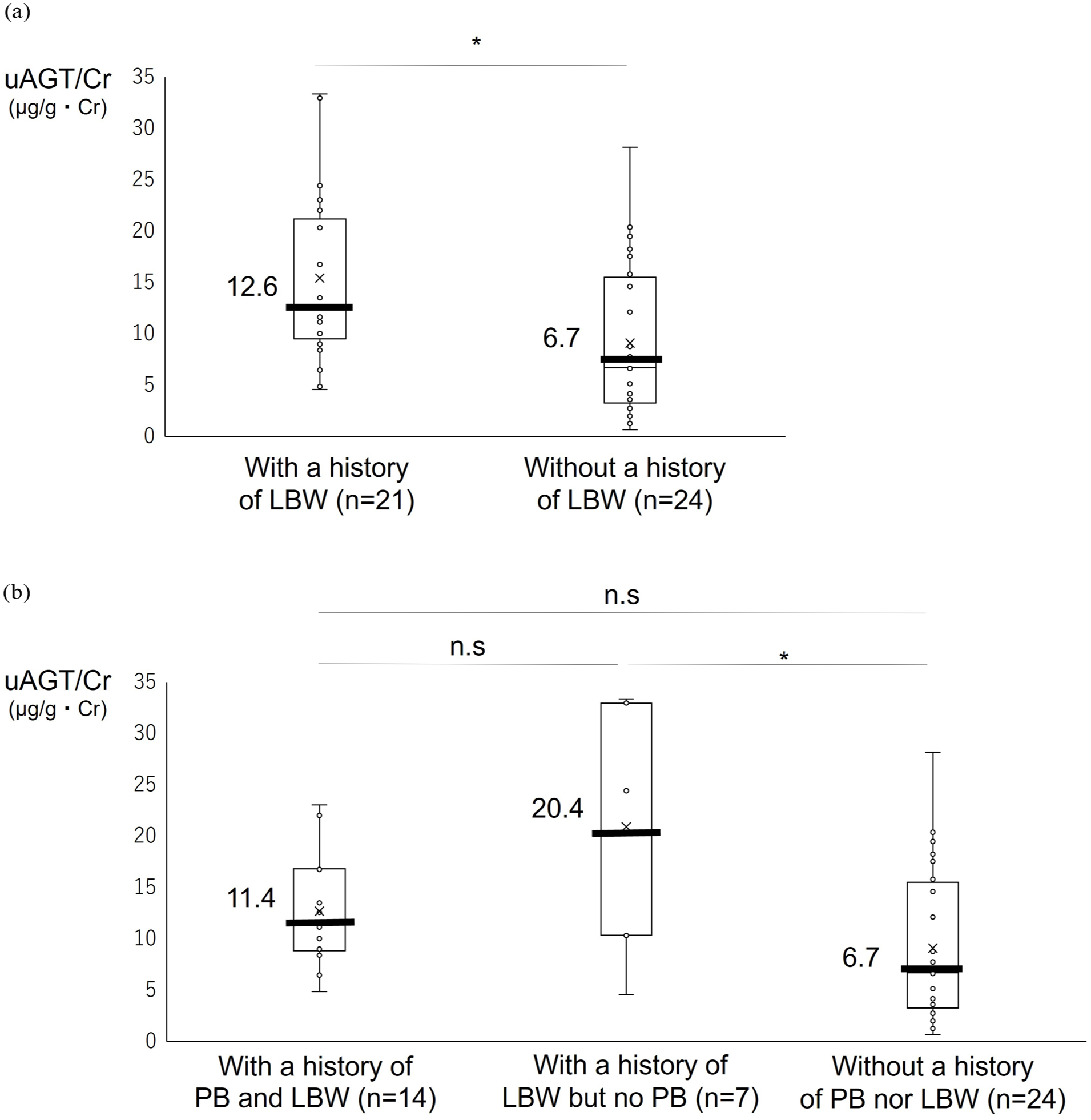
Comparison of the urinary angiotensinogen/creatinine level. (a) Children with and without a history of low birth weight (LBW). (b) Children with a history of preterm birth (PB) and LBW, those with a history of LBW but no PB, and those without a history of PB nor LBW. Abbreviations: AGT, angiotensinogen; Cr, creatinine; LBW, low birth weight; n.s., not significant; PB, preterm birth. *P < .05.
Comparison of Parameters That Showed Significant Differences Among the 3 Subgroups (ie, Children with a History of Preterm Birth (PB) and Low Birth Weight (LBW), Those Who Had a History of LBW But No PB, and Those Without a History of PB or LBW).
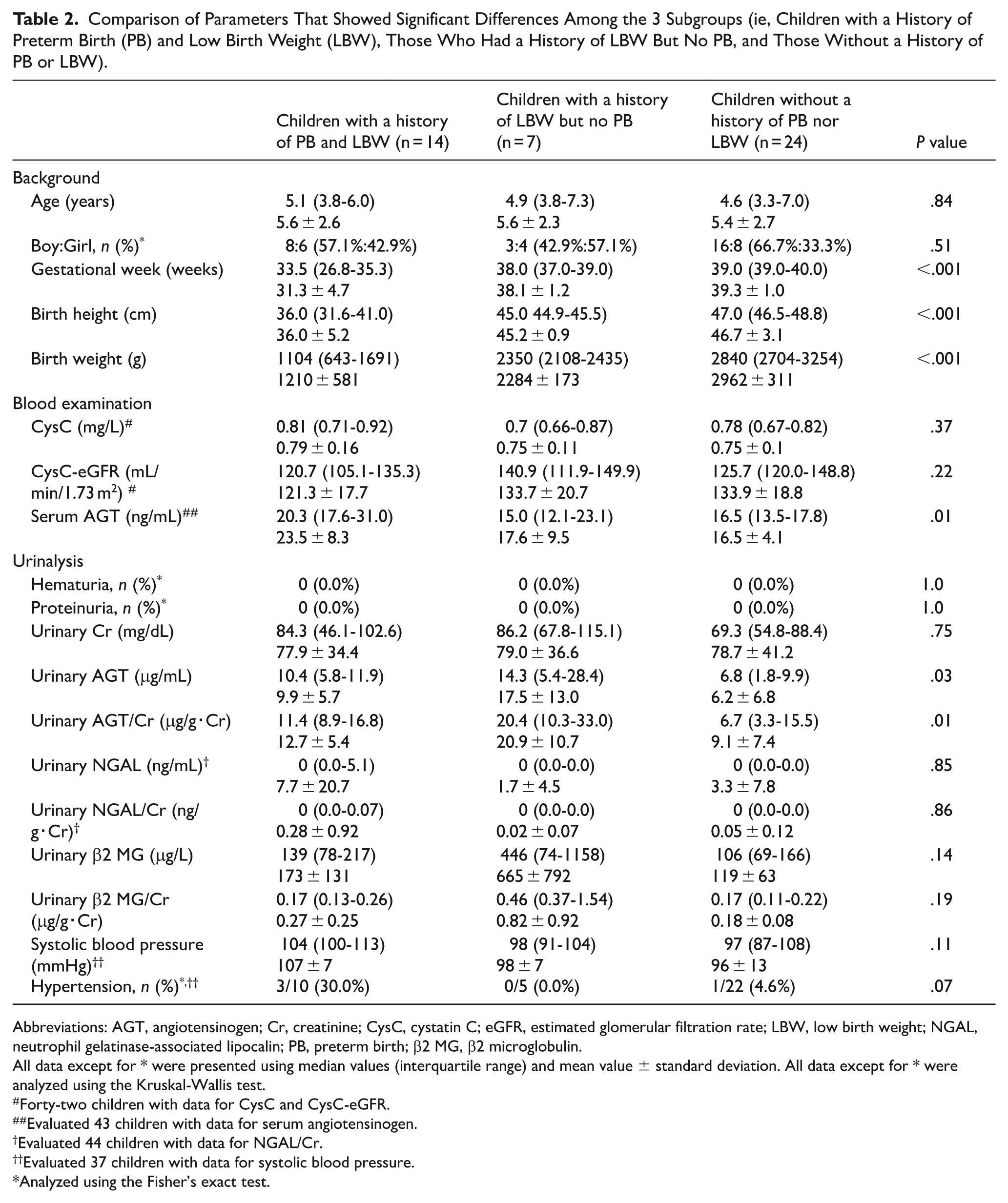
Abbreviations: AGT, angiotensinogen; Cr, creatinine; CysC, cystatin C; eGFR, estimated glomerular filtration rate; LBW, low birth weight; NGAL, neutrophil gelatinase-associated lipocalin; PB, preterm birth; β2 MG, β2 microglobulin.
All data except for * were presented using median values (interquartile range) and mean value ± standard deviation. All data except for * were analyzed using the Kruskal-Wallis test.
Forty-two children with data for CysC and CysC-eGFR.
Evaluated 43 children with data for serum angiotensinogen.
Evaluated 44 children with data for NGAL/Cr.
Evaluated 37 children with data for systolic blood pressure.
Analyzed using the Fisher’s exact test.
Comparison of Parameters that Showed Significant Differences Among the 3 Subgroups Between Children With a History of Preterm Birth (PB) and Low Birth Weight (LBW) and Those Who Had a History of LBW But no PB.

Abbreviations: AGT, angiotensinogen; Cr, creatinine; LBW, low birth weight; PB, preterm birth.
All data were presented using median values (interquartile range) and mean value ± standard deviation. All data were analyzed using the Dunn’s test.
Evaluated 20 children with data for serum angiotensinogen.
Comparison of Parameters That Showed Significant Differences Among the 3 Subgroups Between Children With a History of Preterm Birth (PB) and Low Birth Weight (LBW) and Those Without a History of PB Nor LBW.

Abbreviations: AGT, angiotensinogen; Cr, creatinine; LBW, low birth weight; PB, preterm birth.
All data were presented using median values (interquartile range) and mean value ± standard deviation. All data were analyzed using the Dunn’s test.
Evaluated 36 children with data for serum angiotensinogen.
Comparison of Parameters That Showed Significant Differences Among the 3 Subgroups Between Children With a History of Preterm Birth (PB) But No Low Birth Weight (LBW) and Those Without a History of PB nor LBW.

Abbreviations: AGT, angiotensinogen; Cr, creatinine; LBW, low birth weight; PB, preterm birth.
All data were presented using median values (interquartile range) and mean value ± standard deviation. All data were analyzed using the Dunn’s test.
Evaluated 30 children with data for serum angiotensinogen.
Additional Analysis of Histological Examination
Group 1 consisted of 3 children (2 boys and 1 girl) with a history of LBW who underwent kidney biopsy during remission of INS. They had neither hypertension nor sonographic anomalies of the kidney or urinary tract. At the time of the kidney biopsy, none of the patients exhibited hematuria or proteinuria on urinalysis, and their examination revealed normal kidney function and no signs of autoimmune disease. Their median values for gestational age, birth weight, and age at biopsy were 36 weeks, 2482 g, and 5.5 years, respectively. Group 2 consisted of 3 children (2 boys and 1 girl) without a history of LBW who underwent kidney biopsy during remission of INS. They had neither hypertension nor sonographic anomalies of the kidney or urinary tract. At the time of the kidney biopsy, none of the patients exhibited hematuria or proteinuria on urinalysis, and their examination revealed normal kidney function and no signs of autoimmune disease. Their median values for gestational age, birth weight, and age at biopsy were 39 weeks, 2810 g, and 7.2 years, respectively. The kidney biopsy findings of all subjects were similar. Light microscopic findings of the kidney biopsy revealed minor glomerular abnormality without glomerular hypertrophy, crescents, glomerulosclerosis, or interstitial fibrosis. There were no significant depositions of IgG, Immunoglobulin A, Immunoglobulin M, C3, C4, C1q, or fibrinogen. Electron microscopy showed no findings of thin basement membranes or significant electron-dense deposits.
Upon immunohistochemical evaluation, the positive area of AGT staining in the kidney tubules of group 1 was more extensive than that of group 2. None of the glomeruli of any child showed AGT staining. AT2 staining was weakly positive in the kidney tubules in both groups to a similar degree (Figure 3).

Immunohistochemical pathological evaluation. Group 1 consisted of 3 children (1-a, 1-b, 1-c) with a history of low birth weight who underwent kidney biopsy during remission of idiopathic nephrotic syndrome. Angiotensinogen (AGT) staining in the kidney tubules was positive and extensive. None of the glomeruli of any child showed AGT staining. Angiotensin II (AT2) staining was negative or weakly positive in the kidney tubules. Group 2 consisted of 3 children (2-a, 2-b, 2-c) without a history of low birth weight who underwent kidney biopsy during remission of idiopathic nephrotic syndrome. AGT staining in the kidney tubules was patchy and positive. None of the glomeruli of any child showed AGT staining. AT2 staining was negative or weakly positive in the kidney tubules. Abbreviations: AGT, angiotensinogen; AT2, angiotensin II.
Discussion
We showed that the levels of urinary AGT, as a marker of intrarenal RAAS, in early school-aged children without clinical kidney symptoms who had a history of LBW were significantly higher than in those without a history of LBW. In children who had pathological glomerular hypertrophy with and without a history of LBW, kidney immunohistochemical staining of AGT was negative in the glomeruli. However, in only the child with a history of LBW, the renal tubules were AGT-positive. To the best of our knowledge, this is the first prospective study to evaluate the relationship between a biomarker of intrarenal RAAS and LBW.
It has been reported that more than 60% of nephrons are formed between 20 and 36 weeks of gestation; thus, PB is a risk factor for reduced nephron number. 18 In addition, body weight at birth and the number and size of nephron units are strongly correlated. 19 No studies have specifically examined a direct relationship between smaller kidney size and low renin or AGT production in children with short stature. A reduction in the number of nephrons leads to glomerular hypertension and hypertrophy in the remaining nephrons, which triggers a vicious cycle of progressive loss of functioning units. 20 Suzue et al found a significant correlation between gestational age and the level of urinary AGT in neonates 14 and concluded that immature kidney development in utero reflects a high level of the intrarenal RAAS marker. AGT, which is 1 of the components that make up RAAS, is secreted from the proximal tubule and induces the effect of AT2. Although renin, aldosterone, and serum AGT are established biomarkers of circulating RAAS, intrarenal RAAS has recently gained attention. Activated intrarenal RAAS plays an important role in the pathogenesis of hypertension and CKD. 10 In addition to the impact of gestational age on the level of urinary AGT in neonates, our results demonstrated that children with a history of LBW before the onset of CKD have a significantly higher level of urinary AGT than those without a history of LBW. Our findings suggest that in children with a history of LBW, there is latent RAAS activation even before the onset of CKD. Our prospective cohort study on LBW subjects is currently ongoing, and we will be obtaining longitudinal urinary AGT values to examine changes in urinary AGT values beyond school age in the future. Furthermore, if any of the subjects develop CKD, we will be able to comprehensively examine how RAAS markers contribute to the development of CKD by comparing children who do and do not develop CKD.
CKD progression, induced by RAAS activation, characterized primarily by renal interstitial fibrosis, has been demonstrated in animal models. Elevated serum RAAS levels in mice have been shown to cause renal fibrotic damage. 21 Aldosterone plays a crucial role in organ fibrosis; it not only induces the secretion of fibrosis-promoting factors, such as plasminogen activator inhibitor-1 and transforming growth factor-β, but also causes mitochondrial damage in cultured proximal tubular cells. 22 Although fibrosis of the renal tubulointerstitium contributes considerably to the progression of CKD, it is often absent in pathological kidney tissue during the early stages of CKD. We have previously demonstrated that pathological kidney findings in children with decreased kidney function or proteinuria show compensatory glomerular hypertrophy and hyperfiltration due to a reduced number of nephrons but no tubulointerstitial fibrosis or vascular lesions. 15 Our case with a history of PB and LBW exhibited similar glomerular hypertrophy and no tubulointerstitial findings, alongside strongly positive AGT staining in the tubules. Pathologically, latent RAAS activation may affect the renal tubules before the manifestation of interstitial fibrosis. Future studies examining pathological findings of patients who exhibit fibrosis of the renal tubulointerstitium using fibrotic markers will be valuable.
Latent RAAS activation has been explored in various kidney diseases. In autosomal dominant polycystic kidney disease, a cystic disorder, hypertension occurs before the onset of CKD. In these patients, hypertension indicates the involvement of intrarenal RAAS activation before the development of CKD. 23 Additionally, Shono et al demonstrated positive histopathological immunostaining of AGT in the kidneys of PB newborns who died of pulmonary hypoplasia and 2 to 10-year-old microhematuria patients with histological minor glomerular abnormalities. 24 Staining of AGT in the proximal tubule tissue of the kidney is stronger in newborns at 29 or 36 weeks gestation than in children aged 2 or 10 years. Furthermore, immature kidney development could induce latent RAAS activation during the neonatal period. Our biomarker and pathological investigations revealed that latent intrarenal RAAS activation present during the neonatal period in children with a history of LBW persists until early school age.
Limitations
First, our sample of children with a history of LBW in the main study was limited. In the additional analysis, we evaluated immunohistochemical pathological staining on 3 cases per group. The sample size of the additional analysis was small because there was a limited number of cases who underwent kidney biopsy among those with both kidney disease and a history of PB and LBW; moreover, cases with the same underlying kidney disease and a history of PB and LBW were even more limited. Second, a direct relationship between latent intrarenal RAAS activation and CKD was not verified. Nevertheless, our findings did suggest latent intrarenal RAAS activation in children with a history of LBW. Future studies should investigate intrarenal RAAS biomarkers in children with a history of PB or LBW who go on to develop CKD. A prospective cohort study in the same subjects is currently ongoing, which will enable such an investigation.
Conclusion
Our biomarker and pathological study indicated that latent intrarenal RAAS activation in children with a history of LBW persists until early school age and may contribute to the progression of CKD.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719251379198 – Supplemental material for Latent Intrarenal Renin-Angiotensin-Aldosterone System Activation Could Persist Until Early School-Aged in Children with a History of Low Birth Weight
Supplemental material, sj-docx-1-bmi-10.1177_11772719251379198 for Latent Intrarenal Renin-Angiotensin-Aldosterone System Activation Could Persist Until Early School-Aged in Children with a History of Low Birth Weight by Shingo Ishimori, Shinya Ishiko, Junya Fujimura, Shuhei Aoyama, Yuka Kimura, Hideaki Kitakado, Chika Ueda, Yuta Inoki, Yu Tanaka, Tomoko Horinouchi, Tomohiko Yamamura, Nana Sakakibara, China Nagano and Kandai Nozu in Biomarker Insights
Footnotes
Acknowledgements
ORCID iDs
Ethical Considerations
All procedures in this study involving human participants were performed in accordance with the ethical standards of the Kobe University Graduate School of Medicine (B190011, B240170) and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent for Publication
All patients’ parents/guardians consented to the publication of any relevant data on the basis of anonymization of all personal data.
Consent to Participate
Written informed consent was obtained from all patients’ parents/guardians for this study according to institutional ethics.
Author Contributions
Shingo I. prepared the manuscript. Shingo I., Shinya I., and J.F. collected the clinical data. S.A., Y.K., H.K., C.U., Y.I., Y.T., T.H., T.Y., N.S., C.N., and K.N. revised the article. All the authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a Grant-in-Aid for Scientific Research (KAKENHI) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (grant numbers 23K07242 [to SI]) and Kawano Masanori Memorial Public Interest Incorporated Foundation for Promotion of Pediatrics.
Declaration of Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
