Abstract
Objective
Primary aldosteronism is a curable cause of hypertension which can be treated surgically or medically depending on the findings of adrenal vein sampling studies. Adrenal vein sampling studies are technically demanding with a high failure rate in many centres. The use of intraprocedural cortisol measurement could improve the success rates of adrenal vein sampling but may be impracticable due to cost and effects on procedural duration.
Design
Retrospective review of the results of adrenal vein sampling procedures since commencement of point-of-care cortisol measurement using a novel single-use semi-quantitative measuring device for cortisol, the adrenal vein sampling Accuracy Kit.
Results
Technical success rate of adrenal vein sampling increased from 63% of 99 procedures to 90% of 48 procedures (P = 0.0007) after implementation of the adrenal vein sampling Accuracy Kit. Failure of right adrenal vein cannulation was the main reason for an unsuccessful study. Radiation dose decreased from 34.2 Gy.cm2 (interquartile range, 15.8–85.9) to 15.7 Gy.cm2 (6.9–47.3) (P = 0.009). No complications were noted, and implementation costs were minimal.
Conclusions
Point-of-care cortisol measurement during adrenal vein sampling improved cannulation success rates and reduced radiation exposure. The use of the adrenal vein sampling Accuracy Kit is now standard practice at our centre.
Introduction
Primary aldosteronism (PA) is an important secondary cause of hypertension caused by renin-independent aldosterone secretion from one or both adrenal glands. PA is curable by adrenalectomy in cases of unilateral hypersecretion.1,2 The finding of a unilateral adrenal lesion on computed tomography (CT) imaging may be misleading in determining lateralization of aldosterone hypersecretion, and some patients without CT-evident adrenal lesions have surgically correctible lateralized hypersecretion emphasizing the importance of adrenal venous sampling (AVS) studies. Discordance rates between imaging and functional studies using AVS are typically 30–40%.2–5 Therefore, AVS is considered as an essential investigation in patients with PA being considered for adrenalectomy.2,6,7
AVS is technically challenging and is only performed in major centres by interventional radiologists with experience in the procedure. 2 Despite this, successful bilateral cannulation is reported in about 60–70% of cases in many centres.2,3 Venography is inaccurate for determining successful cannulation of the adrenal veins. Successful cannulation is determined by a significantly higher cortisol concentration in the adrenal vein sample than a paired peripheral plasma sample. 8 This finding is usually confirmed retrospectively after the procedure has concluded due to the slow turnaround time of routine automated cortisol immunoassay methods.
Intraprocedural cortisol measurement has been found to increase the success rates of AVS by providing feedback to the interventionist before study completion, but such approaches have involved standard or modified laboratory equipment and methods for the quantitative measurement of cortisol by immunoassay.9–12 These modified methods are still relatively slow, significantly prolonging the procedure and, in order to provide a collocated service to the interventional suite, may require significant infrastructure expenditure.
A novel approach to intraprocedural cortisol measurement for AVS was reported in a multicentre, controlled study in Japan utilizing a device called the AVS Accuracy Kit (AAK, Trust Medical Co., Kyoto, Japan). The use of the AAK device improved success rates from 60% (n = 145) to 94% (n = 148), compared with no intraprocedural cortisol measurement. 13 It has since been demonstrated to improve success with routine use in a centre utilizing a C-arm CT-assisted AVS protocol. 14 We present our experience with incorporating the use of this device into a non-CT guided AVS protocol.
Materials and methods
Study design
We collated retrospective biochemical, imaging and clinical data on all patients who underwent AVS at Royal Perth Hospital (RPH), Perth, Western Australia between January 2012 and December 2017. AVS was performed by interventional radiologists (DR and GvS) using a sequential cannulation protocol (Appendix 1). Ninety-nine AVS studies were conducted before implementation of the AAK device in May 2016 and 48 after implementation.
Intraprocedural and laboratory cortisol measurements
Each heparinized sample for intraprocedural cortisol measurement was centrifuged for 2 min at 2000 × g. Plasma (100 μL) was then pipetted into the sampling port of the AAK. Two minutes after pipetting, an assessment was made of the approximate cortisol concentration (Figure 1). The 2-min reading time was based on data from the manufacturer and extensive local validation before clinical implementation.

Photographs of the AVS Accuracy Kit taken 2 min after application of plasma to the sampling port. A dark fail line appears if the cortisol concentration is < 276 nmol/L (a); an equivocal fail line where the cortisol concentration is between 276 and 828 nmol/L (b); and no fail line if the cortisol concentration is > 828 nmol/L (c).
At the commencement of each procedure, the first peripheral sample was tested with the AAK to confirm a relatively low cortisol concentration, for meaningful comparison with adrenal samples. At least one right and one left adrenal vein sample was then assessed using the AAK kit, and further samples were assessed as required.
After each AVS procedure was completed, cortisol was measured in the laboratory by automated chemiluminescent immunoassay on an Architect analyser (Abbott Laboratories, Abbott Park, IL, USA). Inter-assay coefficients of variation (CV) were 6.6% at a concentration of 100 nmol/L, 2.7% at 556 nmol/L and 2.2% at 856 nmol/L. Aldosterone and renin were measured by automated chemiluminescent immunoassays on a Liaison or Liaison XL analyser (DiaSorin, Saluggia, Italy). Inter-assay CVs for aldosterone were 9.0% at 302 pmol/L, 5.6% at 686 pmol/L and 7.5% at 1153 pmol/L; and for renin, 9.6% at 3.9 mU/L and 6.5% at 47.4 mU/L.
Data analysis and costings
AVS was considered successful if the final quantitatively measured cortisol concentration in at least one right- and one left-sided adrenal vein sample exceeded the paired peripheral concentration by two-fold. Statistical significance of the change in success rates with and without the AAK was determined by Pearson Chi-squared test, and of the change in radiation dose by the Mann-Whitney U test, using Analyse-it for Microsoft Excel, version 3.70.1.
The additional cost of providing intraprocedural cortisol testing was determined and included reagent and disposable costs, labour and equipment, including the face-value price of the AAK. The calculated cost of an AVS procedure included staffing costs, consumables and an infrastructure loading.
Ethics, approvals and funding
Since the AAK was an unapproved device in Australia at the time of use, it was accessed by successful application, endorsed by the RPH Human Research Ethics Committee, to the Therapeutic Goods Administration under its Authorized Prescriber scheme. The AAK device was provided free of charge by Trust Medical Co. for evaluation purposes. Trust Medical Co. played no role in designing our local protocol for use of the AAK, analysis of the data or drafting of this manuscript and provided no additional funding. This retrospective study was approved by the RPH Service 1 Safety and Quality Committee (Quality Activity 21806).
Results
The rate of successful bilateral cannulation during AVS was 63% without and 90% with the AAK (P = 0.0007). To exclude improved technical prowess as a cause of this difference, we compared year-by-year success rates between 2012 and 2017, which were consistent with historical results and did not show significant variation prior to implementation of the AAK (Figure 2).
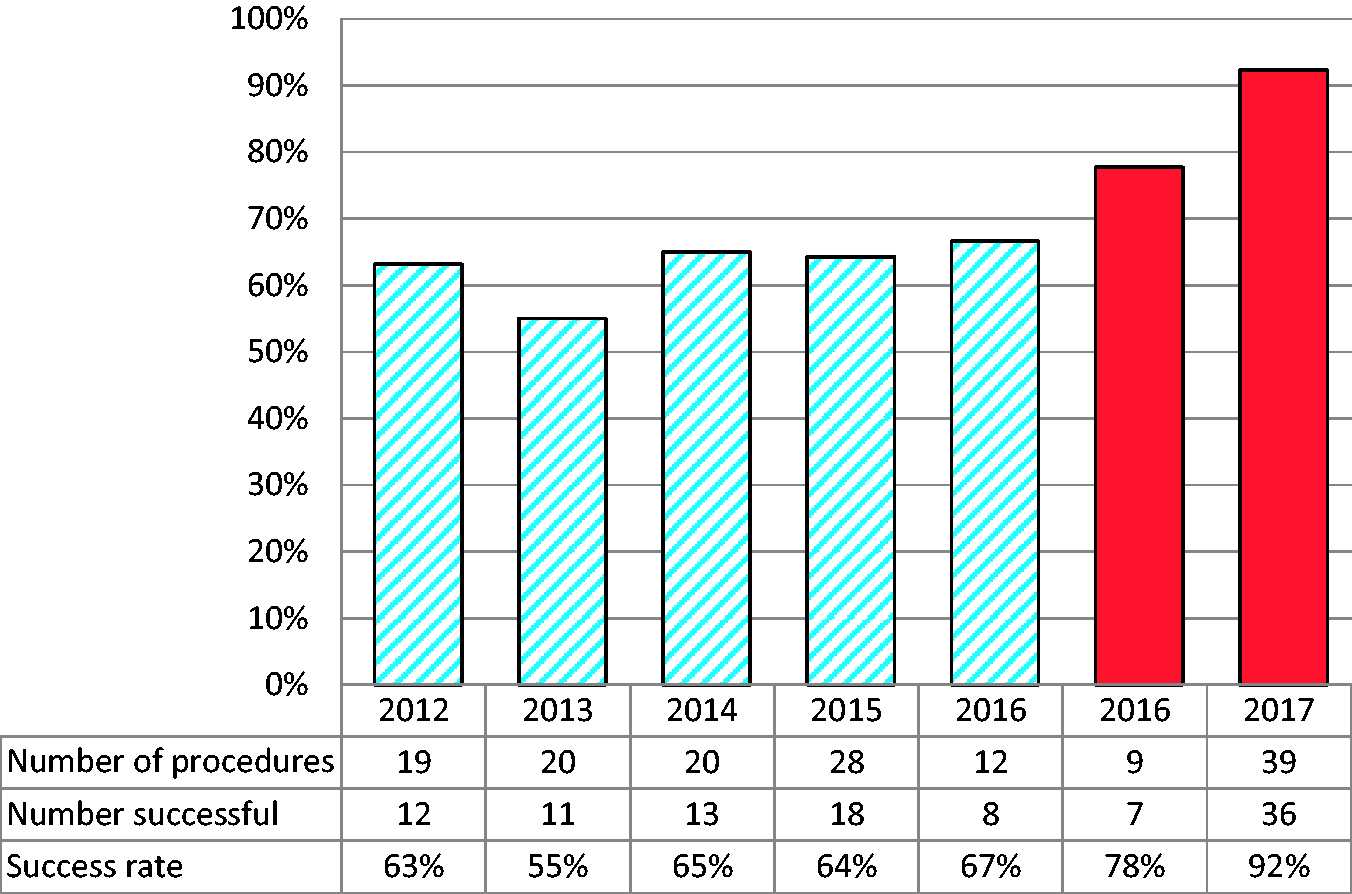
Technical success rates of AVS at Royal Perth Hospital, before and after implementation of the AVS Accuracy Kit (AAK). Striped bars: AVS procedures conducted prior to implementation of the AAK. Solid bars: AVS procedures conducted after implementation of the AAK. Difference before and after implementation of AAK, P = 0.0007; difference between 2016 and 2017 calendar years after implementation of AAK, P = 0.17.
Five of 48 cases using the AAK were unsuccessful. Of these, four had been due to failure of cannulation of the right adrenal vein. All failures, which were confirmed by formal laboratory measurement of cortisol, were predicted at the time of the AVS by the AAK. One of these cases demonstrated suppression of aldosterone secretion on the successfully cannulated left side, in a patient with a right adrenal lesion seen on CT. Prior to use of the AAK, there were 37 failed procedures, 30 of which were due to failure of right-sided cannulation.
No failed cases using the AAK had been considered for a repeat AVS procedure at the time of writing. Of 37 failed procedures prior to implementation of AAK, three AVS procedures had been repeated, all of which had been successful on repeat testing.
Seven of 62 successful cases performed prior to implementation of the AAK showed non-lateralized aldosterone secretion, compared with 7 of 30 cases performed with the AAK. This was a non-significant difference (P = 0.12).
The incremental cost per procedure of providing intraprocedural cortisol measurement was estimated at AUD$160, including reagent costs, disposables and labour cost of a medical scientist to attend the procedure. The mean number of adrenal blood samples taken per procedure was 5.9 prior to implementation of AAK and 5.4 after implementation (P = 0.05). After the first 10 cases, a mean of 4 AAK devices were used per procedure. The total cost of each AVS procedure was estimated to be AUD$2790, including the AAK protocol.
No procedural complications were reported in cases performed using the AAK. Prior to implementation of AAK use, complication rates and severity were in line with international experience and included self-limiting femoral vein dissections, and haematomas in approximately 5% of cases. No patients required inpatient admission for management of complications.
The median radiation dose attributable to the AVS procedure was 34.2 Gy.cm2 (interquartile range [IQR], 15.8–85.9) before implementation of the AAK and 15.7 Gy.cm2 with the AAK (P = 0.009; n = 36 [data unavailable for most recent 12 procedures]).
Discussion
Our retrospective study demonstrated a significant improvement in the success rate of AVS by the use of a rapid semi-quantitative cortisol measurement device, albeit less dramatic than those findings from the initial multicentre randomized trial using the same device. 13 Such an improvement is likely to enhance patient access and acceptability of this specialized procedure.
Cannulation of the right adrenal vein is more difficult than the left and is responsible for the majority of failed studies in most published series. Failure of cannulation of the right adrenal vein remained responsible for most cases of failed AVS after implementation of the AAK in our study. Of note, one such failed study did show contralateral suppression in a patient with a known right-sided adrenal lesion; such a result is considered clinically meaningful, despite technical failure. 15
The relatively low cost of implementing the AAK at our service was in part due to the absence of any new infrastructure requirements. Our laboratory, located in a large tertiary referral, teaching hospital, was able to provide a portable centrifuge, trolley and pipette for the duration of each AVS procedure. Although all of these items have associated costs and depreciation, most are standard equipment and would be contained within the inventories of major laboratories. Accounting for the likely reduction in the need for repeat procedures, we estimate the cost increment of providing point-of-care cortisol testing with the AAK at AUD$80 per procedure. Depending on local costs of AVS, which vary widely, 16 and practices in relation to repeating failed procedures, it may be even more cost-effective in other centres.
Although our AVS procedures were not timed, we did not discern any change in the length of time taken to complete each procedure, and radiation exposure was reduced by more than 50%.
Our study is limited by its non-randomized, retrospective design, using a historical control cohort. We cannot exclude some effect of increased technical expertise with greater experience with the procedure, but the year-by-year data presented in Figure 2 and the absence of any other changes in technology, technique or personnel during this period suggest that most of the improvement is due to the AAK. Our data may not be directly comparable with other specialist centres. However, we present real-world data, supporting the conclusions of the multicentre randomized study by Yoneda et al., 13 utilizing the local expertise of two interventional radiologists with over 30 years of collective experience with AVS.
PA is an often curable cause of resistant, premature or familial hypertension in around 10% of cases. 17 Case detection has increased with awareness of the condition. 18 Contrary to earlier understanding, most patients are not hypokalaemic at presentation, and this realization has led to an increase in screening for the condition by measurement of the plasma concentrations of aldosterone and renin and calculation of a ratio.17,18 Increased rates of diagnosis will inevitably lead to greater demand for AVS. This procedure requires significant and concentrated expertise, and as such is offered in relatively few centres. Increasing success rates is crucial as a strategy for improving access to AVS, as capacity could theoretically be improved by up to 20–30% in many centres by avoiding the need for repeat procedures, depending on routine local practices. The rapidity of the result, ease of use and low start-up cost of the AAK make it an attractive option for the point-of-care measurement of cortisol during AVS. Its availability during AVS is now considered standard care at our centre.
Footnotes
Acknowledgement
Nicola Sawyer for operating the POCT testing device.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The AAK device was provided free of charge by Trust Medical Co. for evaluation purposes. Trust Medical Co. played no role in designing our local protocol for use of the AAK, analysis of the data or drafting of this manuscript and provided no additional funding.
Guarantor
Samuel Vasikaran.
Contributorship
MP contributed to protocol development, data extraction and analysis, approvals documentation and wrote the first draft of the manuscript. DR and GvS performed all AVS procedures, extracted and analysed radiation data and reviewed the manuscript. MT, PG, MG and SV contributed to protocol development and reviewed the manuscript.
