Abstract
Background
High performance liquid chromatography and immunological and enzymatic methods are known as the methods for HbA1c measurement. However, the differences in the principles of the methods may cause slight discrepancies, which become problems especially in the regions where hemoglobinopathies that have high HbF concentrations are commonly seen. In this report, we discuss the effect of HbF on the HbA1c measurement by the enzymatic method (BM Test HbA1c) which can be applied to automatic analyzer of high sample throughput.
Methods
Thirty-five samples (HbF concentration: between 1.4 and 90.1%) and 68 samples (HbF concentration: <1.0%) were analyzed by high performance liquid chromatography (Tosoh G8) and BM Test HbA1c. The results by BM Test HbA1c were compared with those by the high performance liquid chromatography and International Federation of Clinical Chemistry reference method.
Results
The high and positive correlation coefficient was demonstrated between the enzymatic and International Federation of Clinical Chemistry methods for the samples with normal HbF concentration. The samples with high HbF concentration exhibited high and positive correlation coefficient, although the figure is lower than that of the normal HbF concentration group.
Conclusions
BM Test HbA1c revealed a high and positive correlation with high performance liquid chromatography and International Federation of Clinical Chemistry reference methods. However, the HbA1c values by BM Test HbA1c and high performance liquid chromatography method were lower than those by International Federation of Clinical Chemistry reference method when the HbF concentration was high. BM Test HbA1c had some deviation from the International Federation of Clinical Chemistry reference method, while no significant interference was seen when the HbF values were less than 15%. When HbF is higher than 15%, however, the interpretation of HbA1c by BM Test may require some special consideration.
Introduction
Glycohemoglobin (HbA1c), in which valine at the N terminal of β-globin is non-enzymatically and covalently bounded to glucose, is relatively stable. Its blood concentration increases with the average of blood glucose concentration and is retained until the end of life of the red cells. Thus, the HbA1c concentration reflects the average blood glucose concentration in the past 1–2 months. HbA1c is measured by high performance liquid chromatography (HPLC), immunological, or enzymatic methods. However, the difference in the principle of the methods may cause slight discrepancies.
Hemoglobin variant is known to affect results of HbA1c measurements. It may affect the elution profile of abnormal Hb and related HbA1c, and often gives falsely lower HbA1c concentration or exceptionally higher results.1,2 The antibody in the immunological measurement recognizes 4–10 N-terminal aminoacids of the glycosylated β-globin. The aminoacid replacement within this region may affect the result of the HbA1c concentration. The HbS (β6 Glu > Val) and HbC(β6 Glu > Lys) in fact gives higher result.3,4
BM Test HbA1c, one of the enzymatic methods, employs the proteinase that cuts the bond between the β2 His and β3 Leu of the β-globin, hence HbS, HbC, HbD-Los Angeles (β121 Glu > Gln), and HbE (β26 Glu > Lys) are not affected. Nevertheless, Hb Deer Lodge (β2 His > Arg) and Hb Niguarda (β3 Leu > Met) are affected. 5 International Federation of Clinical Chemistry (IFCC) reference method for the measurement of HbA1c is also one of the enzymatic methods. IFCC method uses the endpeptidase which cleaves between β6 Glu and β7 Glu and makes fructosyl hexapeptide. Then HbA1c is quantified using HPLC–ESI/MS or in two-dimensional approach using HPLC and capillary electrophoresis with UV detection.
On the other hand, HbF and HbA2 that constitute the normal Hb component by less than 1 and 4%, respectively, are expected to interfere with HbA1c measurements. Since HbF and HbA2 concentrations in the thalassemia minor are up to 3 and 6%, respectively, they have little impact on HbA1c measurements.
However, in the β-thalassemia major, hereditary persistence of fetal hemoglobin (HPFH) or δβ-thalassemia where the HbF concentration may exceed 10%, significant interference by high HbF to the HbA1c measurement is possible. Thus the discrepancy between clinical manifestation and HbA1c concentration that is lower than expected ensues. For example, HbA1c cannot be detected in the homozygote for the β-thalassemia because HbF is nearly 100% in such case even if the patient has hyperglycemia by diabetes mellitus.
Generally speaking, except for coexistence of the abnormal hemoglobins, hemoglobins other than HbF rarely increase. Thus, we target on HbF for this study. The reports on the interference by HbF with the HbA1c measurements have been few, and no reports have referred to the effectiveness of enzymatic methods in the areas where HbF is often high.6–8 In this paper, we concentrated on the effectiveness of HbA1c measurement on the high HbF samples by the enzymatic method (BM Test HbA1c) that goes on the commonly available routine automatic analyzers of high sample throughput.
Materials and methods
Materials
Thirty-five samples of abnormal hemoglobins or β0-thalassemia with HbF concentrations between 1.4 and 90.1% and 68 normal controls with no abnormality in the Hb fractionation were used. The blood was collected with EDTA as an anticoagulant and transferred to our laboratory in cooled condition within a day. The packed cells that had sufficiently been washed with saline were stored at −80℃ until the analysis was performed. They were thawed at 37℃ and used as hemolysate of approximately 100 g/L. The HbF value was measured by VARIANT™ II TURBO (Bio-Rad Laboratories).
The hemolysate samples were measured by HPLC (Tosoh G8) and enzymatic method (BM Test HbA1c) in duplicate. JCCRM411 (Lot4) was used as the primary standard for accuracy evaluation. The results by HPLC and BM Test HbA1c were compared with the IFCC reference method.
The study was approved by the Institution Review Board of Yamaguchi University Hospital and informed consent was obtained prior to sample collection.
Results
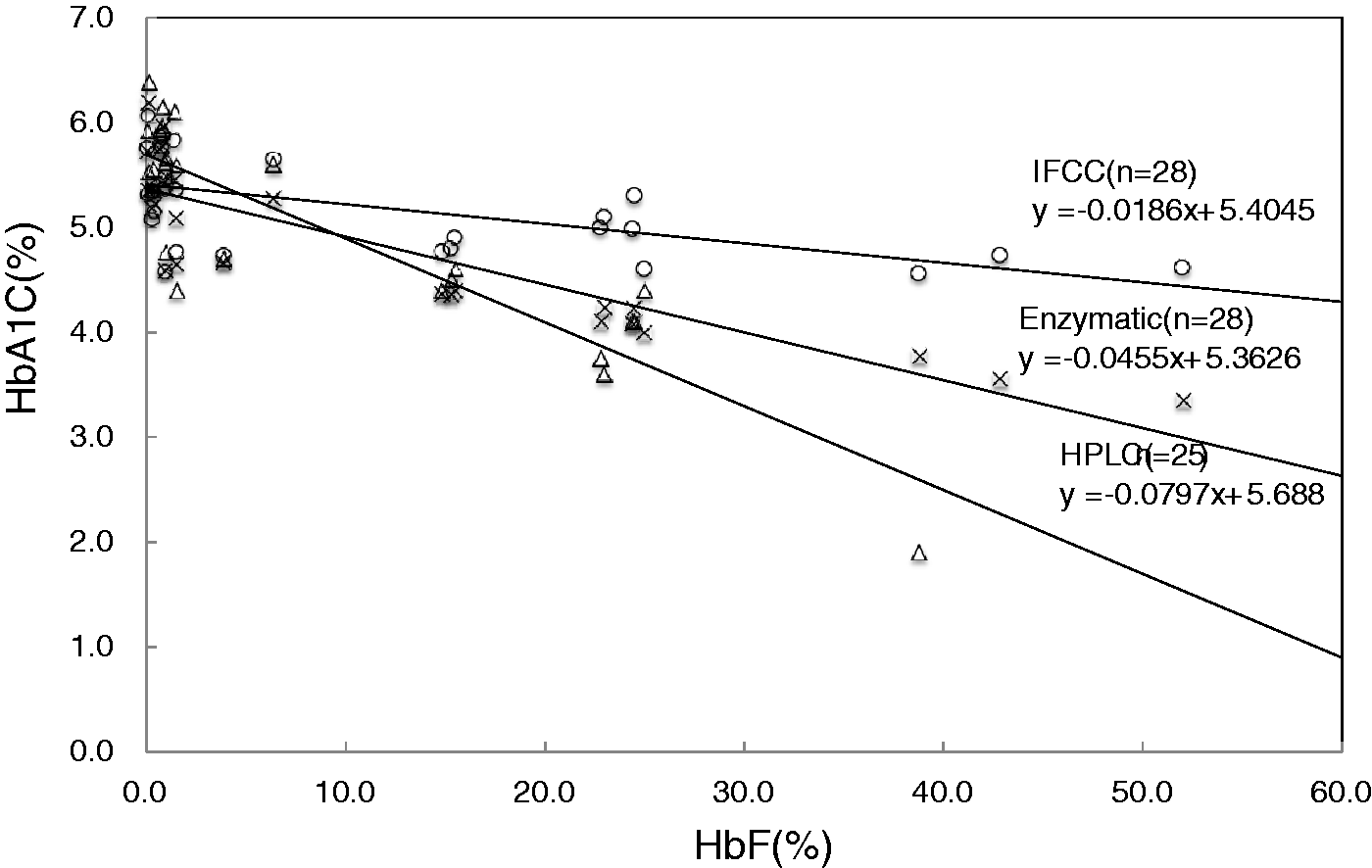
The results of HbA1c measurements by enzymatic method for 34 samples with HbF concentrations between 1.4 and 90.1% are presented in Table 1(a) using HPLC and IFCC as reference. The HPLC method was not measurable in seven samples (about 80%) with the HbF concentration 35% or higher. The high and positive correlation coefficient was demonstrated between enzymatic method and IFCC or HPLC method (Figure 1). The 28 samples whose HbA1c concentrations were around the reference concentration (between 4.6 and 6.2%) showed lower results in both methods when the HbF value increased (Figure 2). This is also true for the IFCC method because the correlation (HbA1c versus HbF) was −0.0186. However, it is −0.0455 in the enzymatic method and −0.0797 in the HPLC, which are 2.5- and four-fold higher than the IFCC, respectively.
Scatter plots showing relationships of BM Test HbA1c with HPLC method (a) and IFCC method (b). BM Test HbA1c revealed positive correlation with HPLC method (a) and IFCC method (b) in the reference range of HbF. However, in the high HbF concentration group, correlation was not so high as the normal HbF group. Scatter plots showing relationships of HbF concentration with HbA1c value of IFCC method, enzymatic method, HPLC method. The 28 samples of HbA1c reference label by IFCC method were measured by enzymatic and HPLC. As the HbF concentration increases, enzymatic and HPLC HbA1c values decrease. HbA1C measurements by enzymatic method, HPLC, IFCC for samples with HbF concentrations between 1.4 and 90.1%. TV: theoretical value; HPLC: high performance liquid chromatography; IFCC: International Federation of Clinical Chemistry.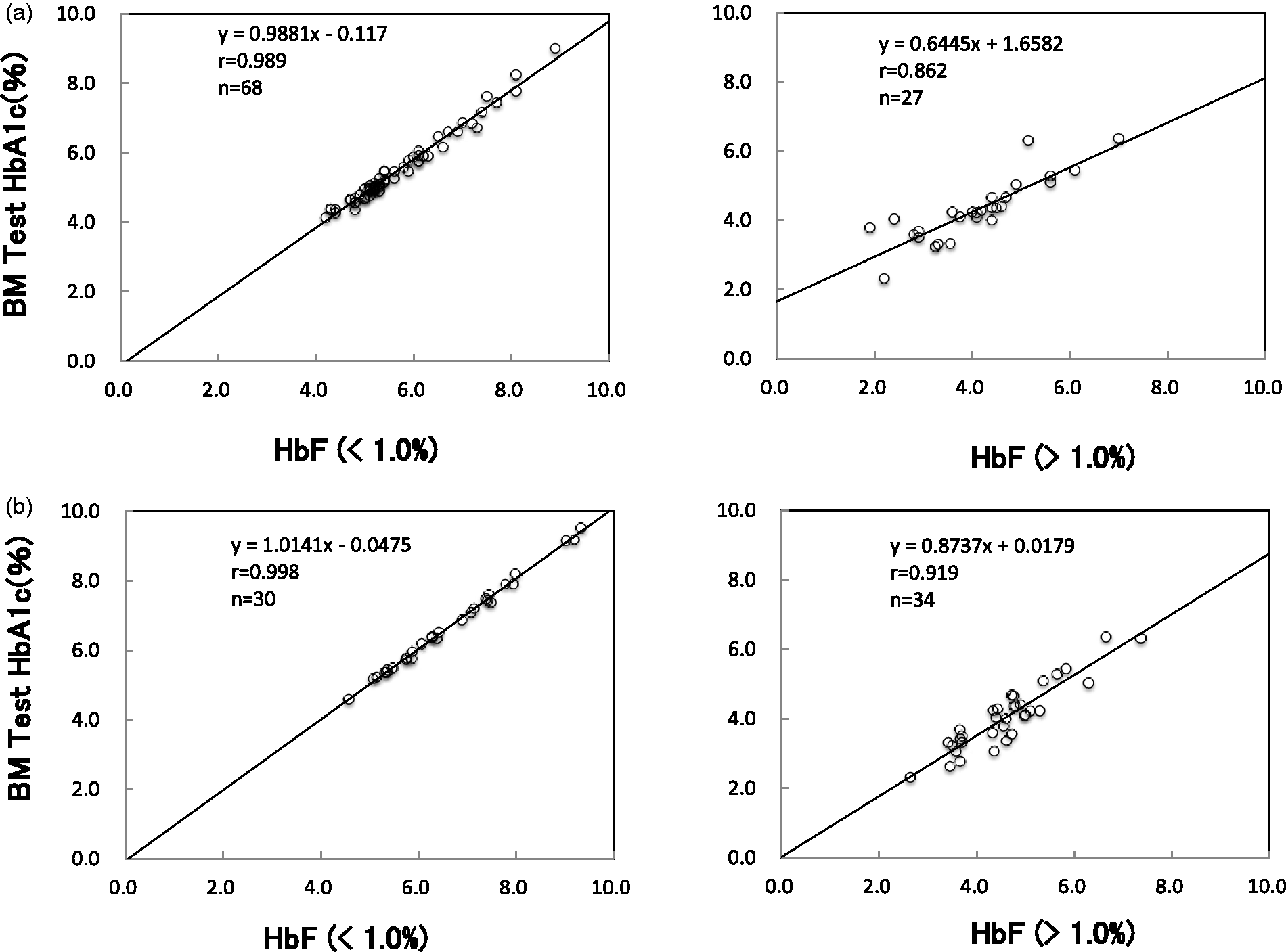
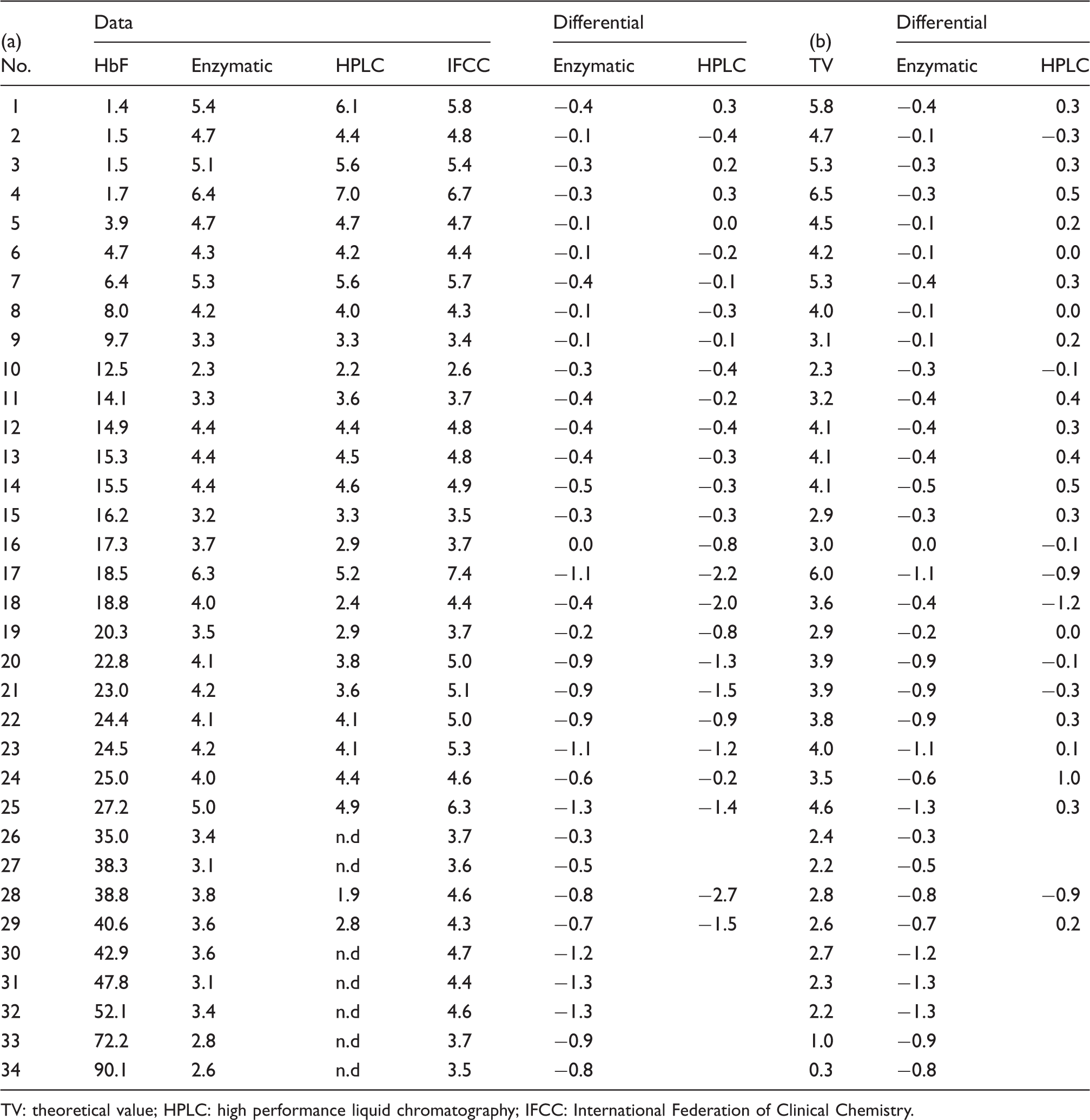
The IFCC value is calculated by the formula of HbA1c/(HbA0 + A1c) which is not affected by the Hbf concentration in principle, while the conventional HbA1c is calculated by HbA1c/Total Hb (HbA + HbF + another Hb). The results by the latter decrease when the amount of other hemoglobins increase, and consequently, the discrepancy against the IFCC results becomes wider. We, therefore, predicted the theoretical values (TVs) of HbA1c from IFCC values. The TV corresponds to the conventionally used formula for HbA1c value or HbA1c/Total Hb(HbA + HbF + another Hb). The TV is calculated by (100–HbF) × IFCC/100. Theoretically, if the IFCC value is correct, the enzymatic and HPLC concentrations conform to the TV (Table 1(b)). The differentials of [TV-enzymatic] or [TV-HPLC] versus HbF were plotted (Figure 3). The enzymatic method indicates gradual increase with HbF concentration (correlation being 0.0223), and HPLC has become unmeasureable on higher HbF concentration. When the HbF concentration is lower than 40%, HPLC method tends to be lower (correlation being −0.0116).
Plots showing differentials of [TV-enzymatic] or [TV-HPLC] versus HbF. The enzymatic method indicates gradual increase with HbF concentration, and HPLC has become unmeasureable on higher HbF concentration. When the HbF concentration is lower than 40%, HPLC method tends to be lower.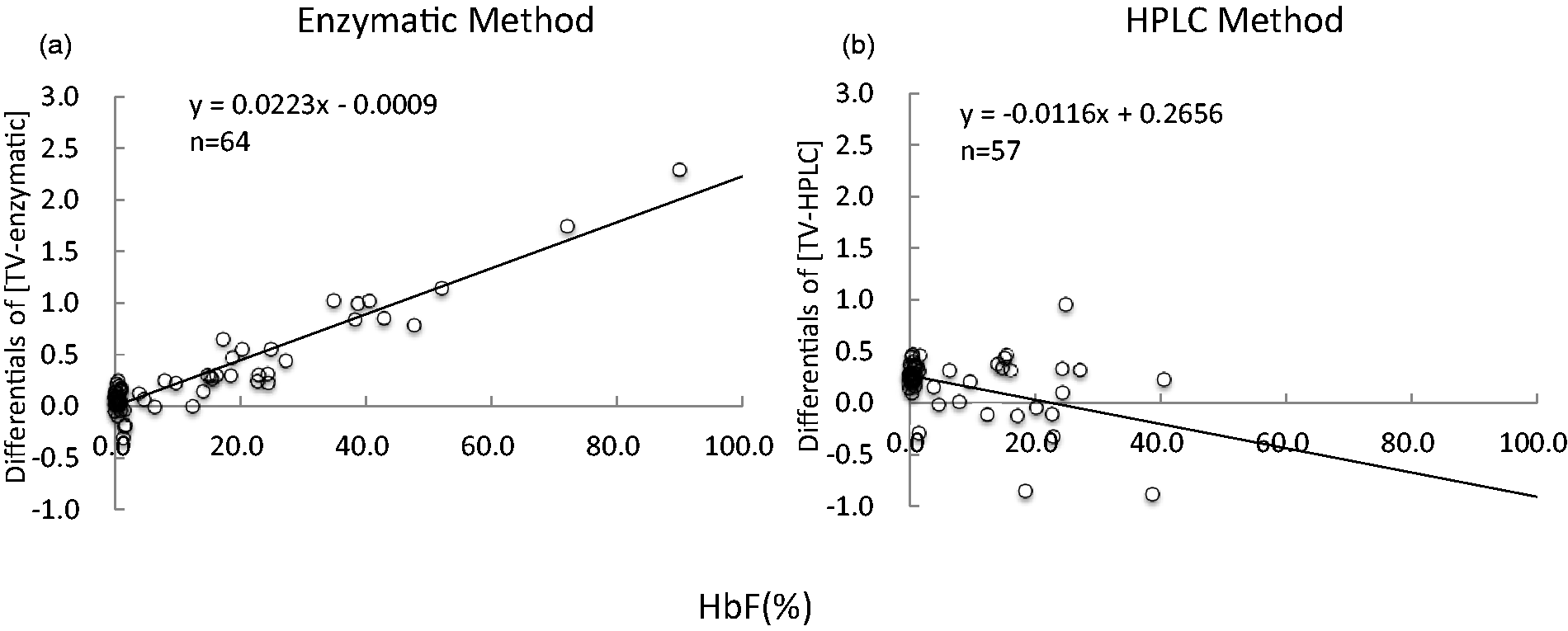
Discussion
The enzymatic method (BM Test HbA1c) revealed high positive correlation with HPLC method and IFCC reference method by r = 0.989 and r = 0.998, in the reference range of HbF (<1.0%). This correlation was indeed also observed in the high HbF group, but the distribution scatters and the correlation was not so high as the normal HbF group (Figure 1). Especially, the correlation between enzymatic method and HPLC in high HbF group was relatively low (0.763). The HPLC method is not measurable in the higher HbF (>35%) and appears to be affected largely by the increase of HbF. This can be ascribed to the incomplete separation of HbF from HbA1c.
The results of BM Test and HPLC HbA1c were lower than those of IFCC reference method when the HbF concentration was high. This is attributable to the difference in HbA1c calculation formula between the IFCC method [A1c/(A1c + A0)] and enzymatic/HPLC method [HbA1c/total Hb]. Theoretically, thus, HbA1c becomes lower with the increase in the HbF. In order to compare the data of each method, we calculated the TV. In theory, if the IFCC as a reference method is correct, or if it specifically measures only HbA1c, and enzymatic method and HPLC method are not affected by HbF, the difference between TV by IFCC versus enzymatic or HPLC methods will be nil or “0” for the correlation slopes.
Negative error was observed with the HPLC method, although this error was not confirmed in the high HbF group (HbF > 35%), whereas positive error was observed with the enzymatic method when the concentration of HbF increases (Figure 3).
The regression analysis indicates that HbA1c is 2.2% when HbF is 100%. Since no experimental data are available for the glycation of HbF, it is assumed to be the same as HbA. This assumption implies that 40% of HbF is calculated as HbA1c. The experimental study on the false calculation of glycated HbF as HbA1c indicated that when calculating 44% of HbF as HbA1c, the result showed the highest correlation (r = 0.9858) to the TV. The proteinase used in the BM Test HbA1c works in the first step as an endopeptidase to specifically hydrolyze between the His(β2) and Leu(β3) of the β-globin to make N-Val-His dipeptide. The fructosyl peptide oxidase in the second step of the BM Test HbA1c works specifically for the excised glycated dipeptide (Fru-Val-His).
9
The substrate specificity of these two enzymes may have caused 40% of HbF cleavage resulting in positive error (Figure 4).
BM Test HbA1c measurement principles. The proteinase used in the BM Test HbA1c works in the first step as an endopeptidase to specifically hydrolyze between the His(β2) and Leu(β3) of the β-globin to make N-Val-His dipeptide. The FPOX (Fructosyl Peptide Oxidase) in the second step of the BM Test HbA1c works specifically for the excised glycated dipeptide (Fru-Val-His). The substrate specificity of these two enzymes may have caused 40% of HbF cleavage resulting in positive error.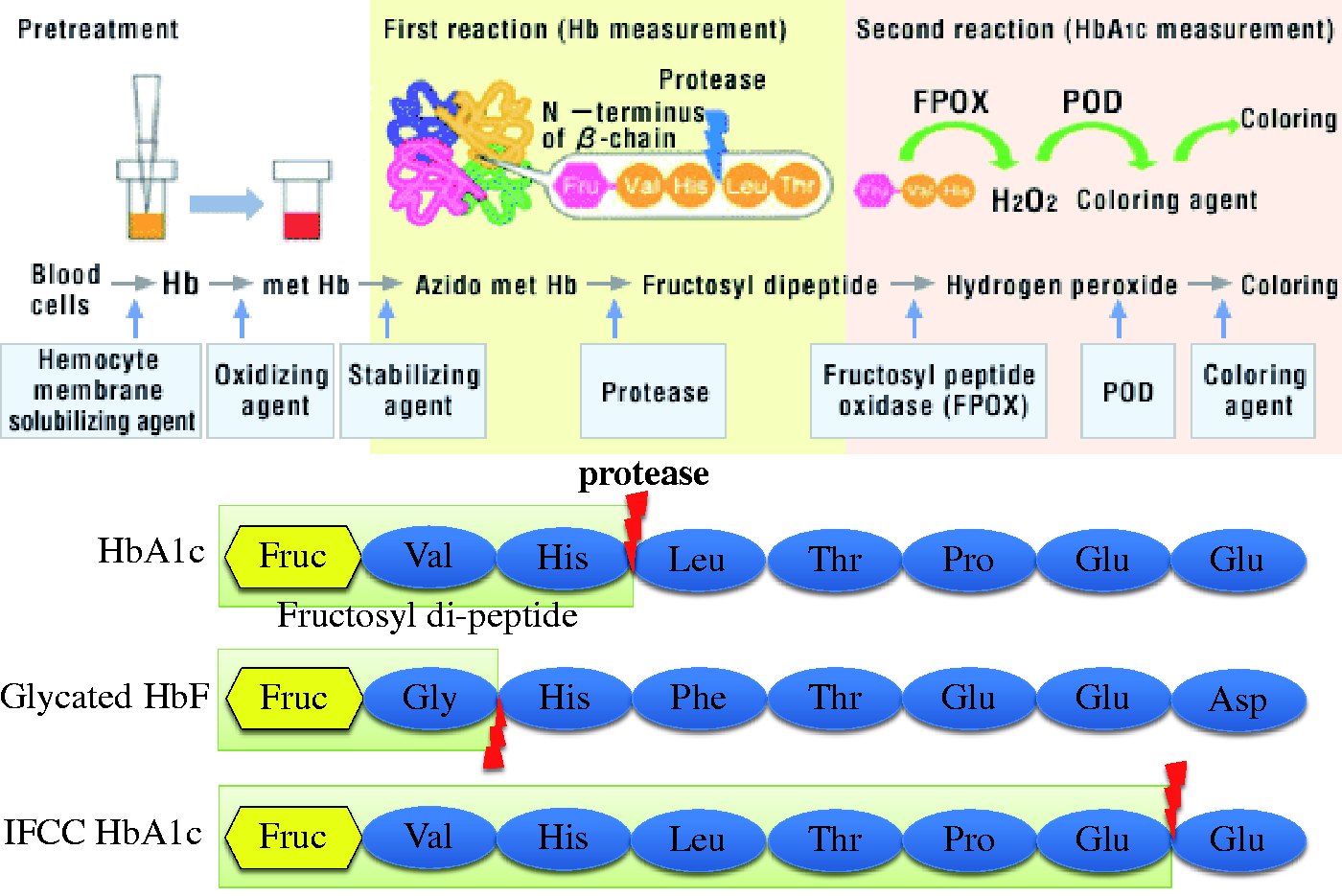
The typical diseases that present elevated HbF concentration are HPFH, δβ-thalassemia, and β-thalassemia major that includes homozygote or compound heterozygote for β-thalassemia genes. The β-thalassemia major, common in the endemic area of malaria produces hemolytic anemia that produces reticulocytosis. The reticulocytosis reduces the HbA1c concentration by the rapid turnover of the erythrocytes, which may cause the discrepancy between the clinical manifestation and HbA1c concentration. Nevertheless, the HPFH and δβ-thalassemia usually causes no or less hemolysis, therefore HbA1c can be used as the marker of diabetes mellitus even when the HbF concentration is high.
As for the effect of HbF on the enzymatic method, the HbA1c concentration tends to be lower than that of IFCC when the HbF concentration is increased, which can be reasonably explained from the difference of the calculation formula. However, it is noteworthy that the difference in the values between enzymatic method and IFCC method is only less than 0.5% when the HbF concentration is 25% (Figure 5(a)). This difference becomes even smaller (less than 0.3%) when the HbF is less than 14%.
The difference in the values between enzymatic method and IFCC method (a). The ratio of error for the enzymatic method against IFCC (b). (a) The difference in the values between enzymatic method and IFCC method is only less than 0.5% when the HbF concentration is 25% (Figure 5(a)). This difference becomes even smaller (less than 0.3%) when the HbF is less than 14%. (b) The HbF is less than 13%; the enzymatic method gives the results within 6% of allowable error range.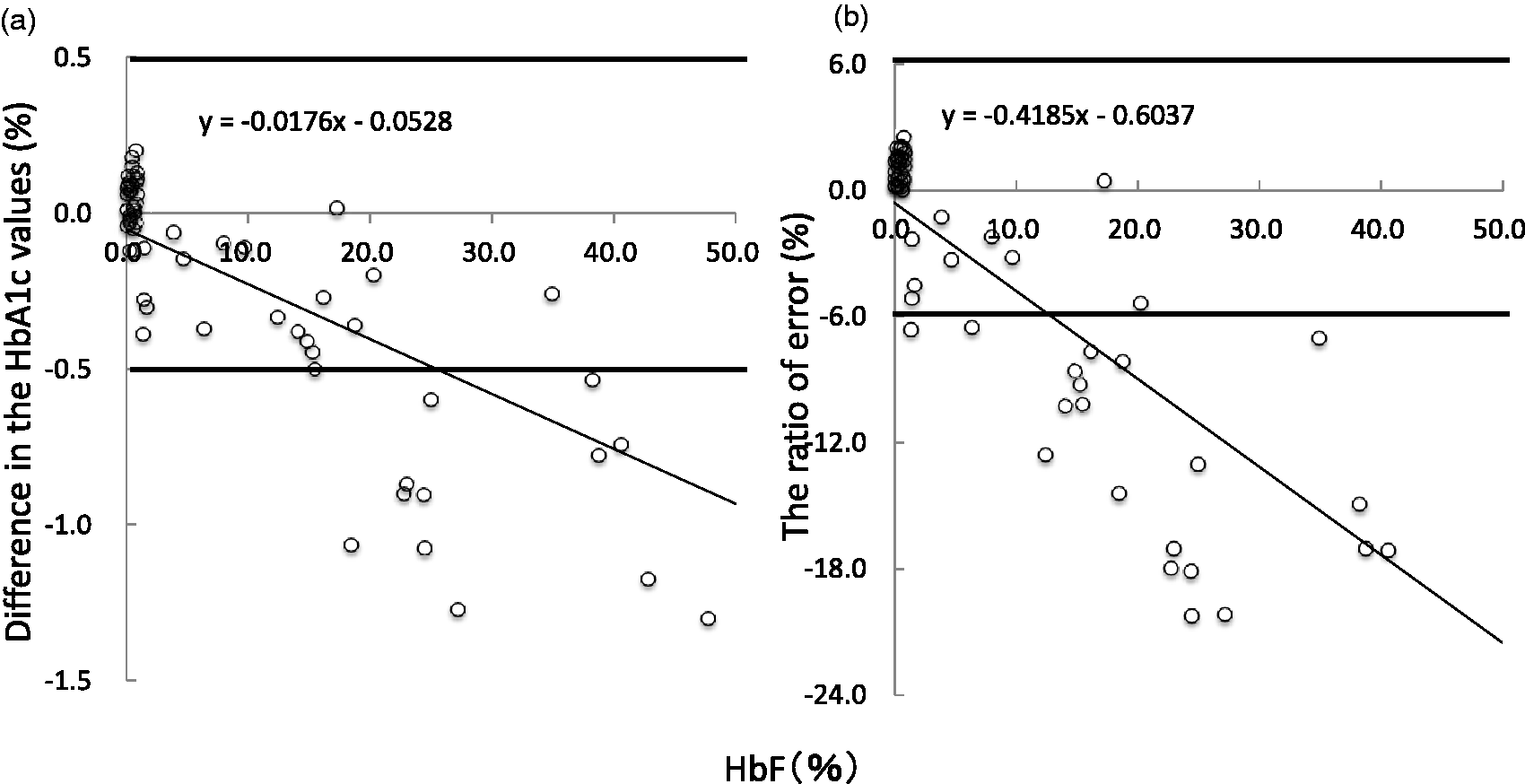
The allowable error range against true value for HbA1c proficiency tests performed by the College of American Pathologists has been reduced to ±6%, representing more strict requirements for clinical laboratories that measure HbA1c. 6
The ratio of error for the enzymatic method against IFCC is calculated (Figure 5(b)). If the HbF is less than 13%, the enzymatic method gives the results within 6% of allowable error range. Thus, interferences from HbF can be avoided in the HbA1c measurements by enzymatic method if the HbF value is less than 15%.
However, it should be noted that HbA1c value may be lower than true value when HbF concentration is more than 15%. The enzymatic method does not measure HbF directly. If there is a discrepancy between the clinical manifestation and HbA1c data which is lower than expected, the possibility of the elevated HbF concentration should be considered. The precision of the enzymatic method by duplicated measurements for the 17 samples with HbF concentration higher than 15% shows an excellent result of 0.016% (average error: Max = 0.06% on HbF 90.1%).
Conclusion
Considering the tendency toward lower result values with elevation of the HbF, the enzymatic method can be satisfactorily employed in the routine monitoring of blood glucose concentration. The enzymatic method with high sample throughput using a routine automated analyzer is a useful tool even for samples with higher HbF.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical approval for this work was approved by the Institution Review Board of Yamaguchi University Hospital (No. H25-39).
Guarantor
TN.
Contributorship
YY and YH collected samples. TE, MN, and JN performed research and analyzed data. TN, YY, and YH designed the study, analyzed and interpreted the data, and wrote the paper.
