Abstract
Introduction
Diabetes is one of the most common chronic diseases which describe a group of clinical syndromes characterized by continuously elevated serum glucose as the main clinical manifestation.1,2 Hyperglycemia can stimulate vascular endothelial hyperplasic growth and induce vascular stenosis, leading to micro vascular disease-induced organ dysfunction which has many clinical complications, and even dementia.2,3
At present, the morbidity of type 2 diabetes mellitus (T2DM) is increasing year by year with the changing lifestyles. 4 Diabetes not only increases blood glucose concentration in the body, but also has complications such as retinopathy, cardiovascular disease, cerebrovascular sclerosis and renal function changes.3,5 In recent years, studies have shown that end-stage renal disease (ESRD) caused by diabetic nephropathy (DN) is gradually increasing. 2 Related studies have shown that about 30–40% of patients with ESRD who require renal replacement therapy have a history of diabetes. 6 DN is a glomerular lesion with micro vascular disease characteristics. Early glomerular lesions mainly show increased glomerular volume with increased glomerular filtration rate, and gradually exhibit thickening of the glomerular basement membrane and an increase in extracellular matrix, resulting in proteinuria and decreased glomerular filtration rate. As the disease progresses, glomerular sclerosis and interstitial fibrosis appear, which are clinically characterized as chronic renal function in-sufficiency. 7
The mechanism of T2DM combined with microangiopathy is not completely understood. Several hypotheses, such as the theory of endothelial dysfunction, the theory of inflammatory response and the theory of oxidative stress have been postulated. 8 However, the theory of endothelial dysfunction is the most probable hypothesis which indicates that endothelium damage leads to metabolic disorders such as endothelial lipase, endothelin, nitric oxide and other active substances secreted by endothelial cells, resulting in thrombosis. The main pathological manifestations are increased levels of many growth factors, abnormal hemodynamics, hypoxic response, and weakened barrier function which collectively act to promote nascent blood vessels. 9
VEGF, hypoxia-inducible factor-1 alpha (HIF-1α) and endothelin-1(ET-1) are key factors in the theory of endothelial dysfunction. 10 Protein kinase C (PKC) is a member of cAMP-dependent protein kinases family with manifold isoforms, widely present in human tissues and cells, activated by hyperglycemia. Activated PKC does not only increase the release of ET-1 with strong vasoconstriction, but also promotes the expression of VEGF. 11 VEGF is known to be the strongest pro-angiogenic factor, which stimulates blood vessels, endothelial cell proliferation and up-regulates expression of plasma plasminogen activator, leading to degeneration of the extracellular matrix and induction of neovascularization. 12 ET-1 can strongly interact with blood vessels to stimulate proliferation of vascular smooth muscle cells, and induce a high-level expression of VEGF, forming proliferative lesions with neovascularization which eventually develop into DN.13,14
Early diagnosis and intervention of DN are particularly important. The value of HbA1c depends on the concentration of blood glucose, and the duration of hyperglycemia. Clinically, HbA1c is the gold standard bio-marker to determine levels of average blood glucose in patients over 2–3 months 15 VEGF and ET-1 are vaso-active substances that participate in the pathophysiological processes of various diseases. This study aimed to determine the relationship between serum changes in VEGF and ET-1 levels at different stages of DN and HbA1c, and to analyze the relationship between the damage to the vascular endothelium and progression of DN.
Material and methods
General information
This retrospective study included all patients with DN who were enrolled from Dept. of renal medicine, people’ s Hospital of Jilin in China from January 2019 to June 2021 for the study. Inclusion criteria: all patients who complied with the Diabetes Diagnostic Standards for T2DM established by WHO (1999) and had no other diseases causing kidney damage and tumors were included. 16 Exclusion criteria: patients with cardiac and hepatic diseases, another kidney disease were excluded. Besides, pregnant women combined with other endocrine diseases, and patients with infectious diseases were also ruled out. The study was performed after obtaining informed consent from all patients and after having obtained approval from the ethics committee of the hospital (formal ethical approval number: Protocol Number 2019–07–01).
Groups
A total of diabetic subjects were divided into three groups according to different levels of urinary albumin excretion rates (UAER). Patients with UAE <30 mg/d were classified as proteinuria-positive group (group I). Patients with 30 mg/d ≤ UAER≤300 mg/d were classified as microalbuminuria group (group II), patients with UAER higher than 300 mg/d were classified as a high proteinuria group (group III). 40 patients who received normal health check-ups in the same period at our hospital with normal blood glucose, normal blood routines and serum urea nitrogen, were selected as the control group.
Data collection
The main data were collected from the department of endocrinology and metabolism computerized database in our hospital. All information included patients gender, age, body mass index (BMI), fasting blood glucose (FBG), uric acid (UA), triglyceride (TG), glycated hemoglobin (HbAlc) and creatinine (Cr).
Serum levels of VEGF and ET-1 were detected by enzyme-linked immunosorbent assay (Bio Rad, Hercules, CA, USA). The ELISA kits were purchased from Beyotime Biotechnology Institute (Haimen China). The average of the two test results was then recorded as the final value.
Statistical methods
SPSS14.00 software was selected for analysis. The measurement data are expressed as the mean ± standard deviation and percentage, and the paired t test was used for comparison of experimental groups. Principal component analysis was also used to assess the HbA1c as a dependent variable association with VEGF and ET-1 as independent variables. Correlation analysis was conducted by Spearman’s analysis with p < 0.05 considered statistically significant.
Results
Clinical features of patients
Demographic characteristics of all subjects in the study.

Note: BMI: Body Mass Index; FBG: fasting blood glucose; UA:uric acid; TG:triglyceride.
HbAlc: glycated hemoglobin; Cr: creatinine.
*p < 0.05 indicates statistical significance; #p > 0.05 indicates no statistical significance compared to the control group determined by using the Student’s t-test.
Levels of HbAlc, VEGF and ET-1 in all groups.
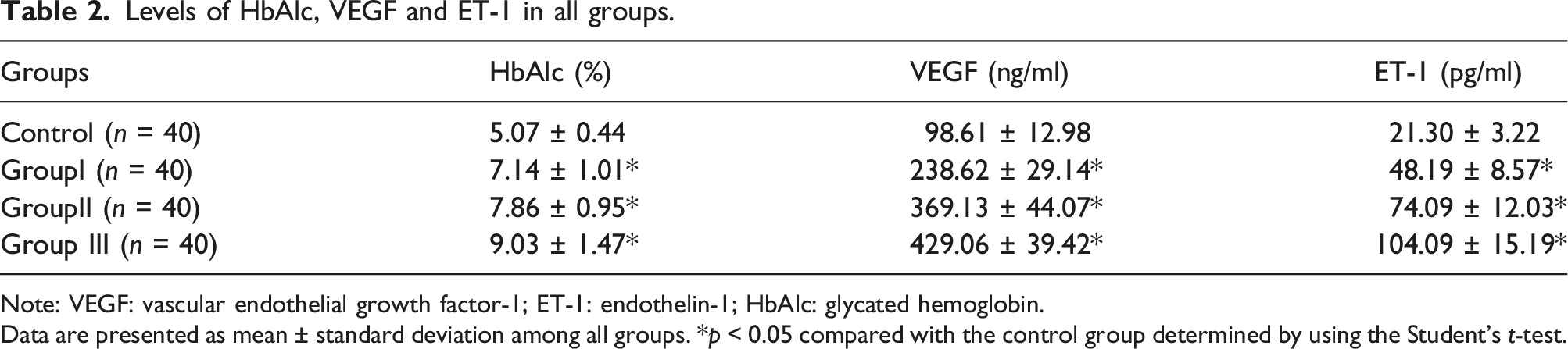
Note: VEGF: vascular endothelial growth factor-1; ET-1: endothelin-1; HbAlc: glycated hemoglobin.
Data are presented as mean ± standard deviation among all groups. *p < 0.05 compared with the control group determined by using the Student’s t-test.
Correlation analysis of HbAlc, VEGF and ET-1 levels
Compared with the control group, the concentrations of serum VEGF and ET-1 were significantly higher in group I, II and III. Spearma’s correlation analysis indicated that HbA1c was positively correlated with serum VEGF (r = 0.7941; p < 0. 0001) and ET-1 (r = 0.8504; p < 0.0001) in DN patients respectively (Table 2 and Figure 1). Principal component analysis demonstrated that HbA1c accounted for 15% of overall variance, whereas, VEGF and ET-1 accounted for 38% of overall variance, indicating that they had strong correlation in the predicting of progression of DN. Spearman’s correlation analysis of HbA1c in DN patients with serum VEGF and ET-1. Note: HbA1c: glycosylated hemoglobin; VEGF: vascular endothelial growth factor-1; ET-1: endothelin-1. A: Logarithm-transformed serum VEGF levels positively correlated with Logarithm-transformed HbA1c levels (r = 0.7941; p < 0.0001). B: Logarithm-transformed serum ET-1 levels positively correlated with Logarithm-transformed HbA1c levels (r = 0.8504; p < 0.0001).
Discussion
DN is one of the most frequent complications of diabetic microangiopathy. The occurrence of DN suggests the risk of serious vascular complications. At present, DN has become the primary cause of patients with end-stage renal disease in the world, bringing enormous economic burden to countries around the world. DN is irreversible, and the disease progresses rapidly owning to the early prediction and accurately diagnosis of DN is difficult. 17 There is therefore a critical unmet clinical need to develop an index for the early prediction of DN.
The initial symptoms of DN are not obvious and are characterized by polyuria only due to increased glomerular perfusion. However, when patients enter a state of high proteinuria, renal function can rapidly deteriorate, and further develop end-stage renal disease. Early diagnosis and intervention are therefore of great significance to DN patients. The clinical treatment of DN is mainly to control blood glucose, blood pressure and protein intake through usage of insulin or oral hypoglycemic drugs. HbA1c is an important index for monitoring blood glucose control status and may be related to the degree of renal damage. 18
In this study, HbA1c levels of patients in group II and III were significantly higher than those in group I, which indicated that worsening blood glucose control was associated with increased proteinuria and decreased renal function. The pathological basis of DN is micro vascular disease of the kidney. 19 For patients with long term type 1 diabetes, renal biopsy showed significant proliferation of microvessels in and around the glomerulus of the kidney. In addition, other studies have shown that the renal tissues of T2DM have the same characteristics. 20
VEGF is a key endothelial cell regulatory factor, which is responsible for a series of events leading to vasculogenesis, the process of de novo formation of vessels during physiological conditions in addition to angiogenesis during the progression of DN or pathological conditions within the body. 21 In some non-diabetic patients, as the disease progresses, VEGF expression is reduced, resulting in micro vascular loss, which in turn lead to renal function damage and fibrosis. VEGF can improve renal disease, indicating VEGF as an important regulator of renal physiology. However, VEGF is much increased in DN. Laboratory studies have shown that the renal tissue of STZ-induced diabetic rats was significantly increased, and that VEGF in renal tissue of endothelial cell nitric oxide synthase gene knockout rats was also increased. 22 In clinical trials, VEGF in patients with renal biopsy tissue and urine was significantly increased. VEGF-mediated angiogenic signalling pathways are critically involved in the induction of the concerned molecular damage. 23 To clarify the mechanism of action of VEGF in DN, studies have focused on two receptor functions of VEGF. Various isoforms of VEGF interact with VEGFR (VEGF receptors-VEGFR1 and VEGFR2) and induce their survival and over-expression, resulting in over-regulated functions of vessel outgrowth, endothelial cell differentiation, migration and tube formation during pathological angiogenesis. 24 Studies have shown that VEGFR1 can up-regulate the expression of nitric oxide (NO) in vascular endothelial cells, and VEGFR2 causes cell proliferation. 25 NO can down-regulate the function of VEFGR2. When the function of VEGFR1 is damaged in endothelial cells, cell proliferation is abnormal, leading to the abnormal proliferation of microvessels. In addition, oxidative stress owing to high blood glucose levels is responsible for inhibiting the physiologically performed survival-upregulating role of VEGF, while simultaneously leading its increased pathological events. Increased levels of oxidative stress altered PKC which further leads to occurrence of several redox mechanism depending events for cell survival, migration, development, differentiation and cell death. Besides that, high oxidative stress decreased sensitivity to insulin by impairing the signaling of insulin as well as activating pro-inflammatory proteins.26,27
In this study, with the aggravation of proteinuria, the expression of VEGF increased significantly. Combined with other studies, it can be concluded that VEGF plays an important role in the progression of DN, and may indicate and mediate the aggravation of DN. 28
ET functions to control smooth muscle contraction, regulates nerve transmission, and controls cell proliferation. There are three known ET subtypes, and ET-1 plays a critical role in kidney diseases. Studies have demonstrated that ET-1 mainly binds to two ET receptors, namely ETAR and ETBR. 29 The combination of ET and ETAR can up-regulate the expression of extracellular matrix proteins in mesangial cells and aggravate fibrosis. By blocking the ET-1/ETAR pathway, the kidney can be effectively protected and proteinuria can be decreased. In STZ-induced DN mice, blocking ETAR alone can effectively reduce proteinuria. 30 However, when blocking the ET-1/ETAR/ETBR pathway, control of proteinuria is not so effective as blocking ETAR alone. Studies have found that ET-1 binding to ETAR can accelerate mesangial cell proliferation and extracellular matrix accumulation by up-regulating the RhoA/ROCK pathway. However, the NF-κB pathway, which can increase the expression of ET-1, was inhibited by ET-1/ETBR, and the expression of ET-1 was significantly increased in ETBR gene knockout DN mice.31,32 In this study, the ET-1 index of DN patients was significantly increased with the aggravation of proteinuria. This trend is in agreement with the above studies, indicating that ET-1 can reflect the severity of renal damage.
In patients with long term hyperglycemia, the renal micro vascular perfusion is poor, tissues become ischemic and hypoxic, and vascular endothelial cells are damaged.32,33 These processes lead to abnormal micro vascular proliferation, abnormal proliferation of mesangial cells and massive extracellular matrix deposition in the mesangial area, resulting in glomerular sclerosis eventually.34,35 As described in the above studies, VEGF and ET-1 play an important role in microangiogenesis and abnormal proliferation in the mesangial area, determining the extent of renal damage. HbA1c reflects the status of 2–3 months of glycemic control.
In this study, the results of statistical analysis showed a positive correlation between HbA1c, VEGF and ET-1 levels, indicating that long term high glucose status could damage vascular endothelial cells and aggravate the degree of renal damage. For determination of the VEGF and ET-1 factors using PCA, both were considered to be sufficient. The findings showed that the important predictors of both VEGF and ET-1 are consistent with previous studies, suggesting that VEGF and ET-1 increase along the progression of DN. Additionally, they are simple to obtain in clinical routine and are relative to the diagnostic markers of DN, microangiopathy and glomerular sclerosis. 36
There were several limitations to the study. The study was a cross-sectional in a single-center, and the sample size was small. The data were not population-based, and the numbers of DN patients were not sufficient to identify the predictive powers. Further a large multi-center cohort analysis was necessary in order to validate their values as multiple prognostic bio-markers for DN in the near future. Besides, we need follow-up study of the participants.
Conclusions
Serum VEGF and ET-1 may reflect the occurrence and progression of DN disease. We hypothesis that both factors may be significant indicators for early diagnosis and monitoring of DN efficacy.
Footnotes
Acknowledgements
We thank anonymous reviewers for excellent criticism of the article.
Author contribution
Wei Chu conceived, designed the experiments and wrote a draft manuscript. Binxian Li and Mingcheng Li analyzed, interpreted the results of the experiments and revised the manuscript. Lin-Lin Ma collected the clinical data and performed the experiments. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Developing Program in Jilin Province, China (20190905006SF) and Science and Technology Research Projects of the Department of Education of Jilin Province, China (JJKH20220076KJJ).
Ethics approval
Human experimental protocols to collect the clinical samples were approved by ethical committees of the hospital (formal ethical approval number: Protocol Number 2019–07–01).
Informed consent
The study was performed after obtaining informed consent from all participants in the study.
